Note: Descriptions are shown in the official language in which they were submitted.
- ` ~2~7~
CAT~ETER FOR LASER ANGIOSURGERY
Description
I. Technical Field
This invention relates to devices in which
optical fibers are provided within a catheter and
laser radiation is directed through the fibers for
medical applications including diagnosis and removal
of arterial or vascular obstructions (angiosurgery).
MIT 3767
.
9~
--2--
II. Backqround Art
The term "laser" is an acronym for Light
Amplification by Stimulated Emission of Radiation.
As used herein, the term is meant to encompass a
device which utilizes the principle of amplification
of electromagnetic waves by stimulated emission of
radiation to produce coherent radiation in the
lnfrared, visible or ultraviolet region. Such
radiation has been used in external medical
applications, such as for cauterizing, for attaching
detached retinas and for removing various skin
cancers.
Likewise, optical fibers have been used in a
variety of medical applications. An optical fiber is
a clad plastic or glass tube wherein the cladding is
of a lower index of refraction than the core of the
tube. When a plurality of such tubes are combined, a
fiber op~ic bundle is produced. Optical fibers are
flexible and are therefore capable of guiding light
in a curved path defined by the placement of the
fiber.
Fiber optic scopes have been developed for
medical technology in order to enable illuminating
and viewing access by the medical practitioner to the
various interior parts of the body. In many medical
applications, fiber optic devices have been combined
with laser techniques to properly focus and apply
o.,~
laser radiation to interior parts of the body.
More recently, laser catheters have been
constructed in which flexible or rigid hollow tubular
devices (catheters~ containing optical fibers are
inserted into veins or arteries to illuminate
internal parts of the body for diagnostic and
surgical purposes. Such an application, in which
fiber optic bundles are contained within a flexible
catheter conduit, is described in U.S. Patent
~4,207,874 issued to ~.S.J. Choy on June 17, 1980.
This fiber optic catheter contains a combination of:
(1) a fiber optic viewing bundle; (2) a light source
bundle for illuminating the region to be viewed; (3)
a laser bundle for delivering laser light to the site
for removal of tissue; (4) an annular space around
the bundles for fluid supply or suction; and (S) a
proximal supply and a transparent reservoir connected
to the annular space. All of the above items
together constitute a "laser tunneling device". The
sole described use for the device is the removal of
thrombus in veins for applications in the circulatory
system.
The Choy device relies on visualizing the
thrombus obstruction in a a vein, via the viewing
bundle. It is therefore necessary to purge the blood.
As no means of blocking the blood flow is shown in
Choy, the Choy device can be used only when the vein
i5 already totally obstructed. As soon as the
obstruction is opened even a small amount, blood in
the transparent reservoir indicates ~he end point of
the procedure. A partial blockage which causes
inadequate flow cannot be visualized or treated by
the device. ~n the case of coronary arteries, an
important treatment area for the present invention,
complete blockage would cause death of the distal
tissue, and so restoring blood flow at such a late
state of disease would provide little clinical
benefit.
M. Bass, in U.S. Patents ~3,858,577 and
~4,146,019, describes a device which uses a
transparent window to proteet an optical fiber
carrying laser radiation into a body cavity (e.g. the
stomach). The window has a possible protective
function, that of preventing spattering of debris
from the laser tissue interaction back onto the
optical fiber in the gas-purged environment. The
cleanable or replacable window is in all examples
recessed into a metallic or non-optically transparent
holder. The design is such as to to avoid contact
between tissue and the window. In addition, the
cavity formed by the recess would tend to trap fluid,
such as blood, absorbing the laser radiation and
hindering it from reaching the target tissue.
In Bass, multiple fibers within the catheter
body are described, but only for the purpose of
replacement in case of fiber failure.
The Bass instrument also includes a flexible
fiber optic endoscope for viewing the body cavity as
> ~ ~
~¢~
an integral part of the device. Being a visual
device, the information which can be provided for
diagnosis by the endoscope is limited to what can be
seen. In addition, the endoscope is not contained
within the windowed enclosure, so the field of view
in front of the endoscope must be completely purged
of all non-transparent fluids, such as blood or blood
substitutes. Such a purge deprives distal tissues of
blood and oxygen. Therefore, the Bass instrument is
clearly an instrument not intended for use, and
cannot be used, in the vascular system.
J.H. Hett in U.S. Patent 4,072,147, describes a
device for viewing and for carrying therapeutic laser
radiation into a body cavity. This endoscopic device
contains a fiber optic bundle image transmitter
connec~ed to an eyepiece for viewing; a spotter light
path which indicates where the endoscope is aimed;
and optical fibers to deliver therapeutic radiation
(which need not be visible light~ to that visualized
spot. This instrument further may contain a
protective transparent cover over the distal en~ of
the instrument. It also may incorporate a manually
adjustable variable filter in the viewing path so as
to protect physicians eye. A servo system connected
to the manually adjusted filter can adjust
therapeutic laser power.
The Hett instrument is designed only for direct
visualization by eye and requires an optical image
transmitter coherent iber bundle. Since it is a
~2~
visual device, the information about the tissue
diagnosis is limited to what can be seen. Also,
because visualization is used, the path from the
distal end of the instrument to the tissue must be
clear, but no means of purging non-transparen~ fluids
(such as blood) is provided. The spotter beam, and
hence the therapeutic radiation, is delivered to a
single location in front on to the side of the dis~al
end of the d0vice: ~The image (therapeutic laser
beam) is located in a predetermined segment of the
field of view..n. The device must be physically
repositioned each time a different spot of tissue is
to be treated. In a blood vessel, treatment of a
lesion would be limited to one spot at a time. The
difficulty of maneuvering the long flexible catheter
to a new spot for each small piece of tissue removed,
and the likely damage to the delicate vessel wall
from repeated and prolonged manipulation of the
device would make its use impractical in such a
situation. Finally, since the control of the laser
power is connected to the position of the hand
opera~ed attenuating filter, such control is
essentially manual, and is therefore orders of
magnitude slower than an electronic control system.
It is inadequate for use in a blood vessel where
laser radiation can per~orate the wall in less than a
second. For all these reasons, the Hett instrument
is one which is not intended for an inadequate for,
use in the vascular system.
12799~
--7--
Hussein, et al, in V.S. Patent #4,445,8g2,
describes a vascular fiber-optic catheter with two
inflatable balloons which can seal off a segment of a
blood vessel, allowin~ it to be purged. Blood flow
is maintained past the distal end. A cyli-ndrical
window allows viewing and laser irradiation through
the side of the device. The balloons displace the
blood and protect the operating portion of the
instrument.
A significant lumen in a vessel must already
exist to allow insertion of the balloon distal to the
lesion, so the instrument could be used as described.
In the cases where the lumen is severely stenosed or
restricted, or totally occluded, forcable insertion
of the distal balloon may fail or cause serious
mechanical injury to the diseased vessel. This
instrument is least useful in the situation where the
need is greatest. Also as the therapeutic laser
radiation is angled to the side to avoid hitting the
distal balloon, perforation of the artery wall is
more likely than if it were aimed forward. Also, the
tube holding the distal balloon restricts the field
of view. As with ~ass and Hett, the device relies on
visualization and the diagnostic information is
limited as described. Electronic feedback control of
the laser ~wer is not included~
The application of laser catheters have been
documented in the literature [D.S.J. Choy, S.H.
Sterzer, H.Z. Rotterdam, N. Sharrock and I.P.
~7~9~
Kaminow, "Transluminal Laser Catheter Angioplasty"r
Am. J. Cardiol. 50, 1206-98 ~19a2); D.S.J. Choy, S.H.
Stertzer, ~.Z. Rotterdam and M~S. Bruno, "Laser
Coronary Artery Angioplasty: Experience with Nine
Cadaver Hearts~, Am. J. Cardiol. 50, 1209-11'(1982);
G.S. Abela, S. Normann, D. Cohen, R.L~ Feldman, E.A.
Geiser and C~R. Conti, "Effects of Carbon Dioxide,
Nd-YAG, and Argon ~aser Radiation on Coronary
Atheromatous Plaques", Am. J. Cardiol. 50, 1199-1205
(1982); G. Lee, R.M. Ikeda, R.M. Dyer,. H. Hussein,
P. Dietrich and D.T. Mason~ "Feasibility of
Intravascular Laser Irradiation for In Vivo
Visualization and Therapy of Cardiocirculatory
Diseases~, Am. Heart J. 103, 1076-77 (1982); R.
Ginsburg, D.S. Rim, D~ Guthaner, J. Toth and R.S.
Mitchell, "Salvage of an Ischemic Limb by Laser
Angioplasty; Description of a New Technique", Clin.
Cardiol. _, 54-58 (1984); and E. Armelin, R. Macruz,
M.P. Ribeiro, J.M.G. Brum, M.G.C. Madrigano, P.R.
Camargo, J. Mnitentag, P. Pileggi and G. Verginelli,
"Application of a Laser Beam in the Vessel Wall
Without Interruption of Blood Flow: n ~ Circulation 66
(abstract), II-136 (198~).
In all of these studies the optical f iber
conducting the laser light is placed in the artery in
an unprotected manner, in direct contact with the
blood. Reports in the literature enumerate severe
drawbacks in ths efficacy and safety of this simple
approach. At the t'ip of the fiber the reaction of
the emitted light with the intravascular target is
violent. ~ "crackling" sound during the irradiation
process, similar to that of bacon cooking, has been
described. The corrosive environment of the blood
vessel readily damages the delicate tip of the
Optical fiber. The light (particularly blue-green
argon laser radiation, which is most commonly used)
is strongly absorbed by any blood intervening between
the tip of the fiber and the tissue ~arget, with the
reaction forming debris and gas. There is evidence
that red blood cells are damaged, predisposing to the
formation of platelet aggregates. In addition to the
resultant problem of thrombosis, vascular perforation
is a major complication. The latter occurs because
of poor control of the laser radiation. Further,
even if perforation does not occur acutely, the
arterial wall may still be damaged, with the
resultant potential for long term aneurysm formation.
Modifications to reduce these complications have
been proposed. One approach has been to cover the
bare fiber with an absorbin~ metal tip which is
heated by the laser light, forming a hot probe.
lT.A. Sanborn, D.P. Faxon, C.C. Haudenschild and T.J.
Ryan, ~Laser Radiation of Atherosclerotic Lesions:
Decreased Incidence of Vessel E~erforation with Optic
Laser Heated Metallic Tip," J. Am. Coll. Cardiol.
~abstract) 3, ~90 (1984); G. Lee, R.M. Ikeda, M.C.
Chan, J. Dukich, M.H. Lee, J.H. Theis, W.J. Bommer,
R.L. Reis, E. Hanna and D.T. Mason, "Dissolution of
--10--
Human Atheroschlerotic Disease by Fiberoptic
Laser-Heated Metal Cautery Cap", Am. ~ear-t J. 107,
777-78 (1984)]. This approach is unsatisfactory for
several reasons: (i) there is thermal damage to
surrounding tissue; (ii) only fatty plaques readily
melt away; (iii) the more advanced fiberous and
calcified plaques form char and debris; and (iv) the
hot tip tends to adhere to the tissue, so when it is
removed, the tissue is ruptured.
Despite the scope of the above efforts, a
need still exists for accurate control of high power
radiation delivered through optical fibers if
percutaneous intravascular laser treatment is to
reach its full potential.
`:
III. Summary of Disclosure of the Invention
In accordance with an embodiment of the
invention there is provided a laser catheter for
insertion into bodily cavities, ducts, vessels or
arteries for precise delivery of light onto tissue in
said cavities, ducts, vessels or arteries comprising:
a) a catheter with a proximal and distal
end;
b) a rigid optical shield on the distal end
of the catheter forming an optically transparent
2S enclosure over the end of the catheter -to transmit
laser radiation and displace bodily fluid such that
direct contact can be made by a distal surface of the
shiela with such tissue;
c) a fiber optic means disposed within the
catheter including a multiplicity of optical fibers
the ~distal ends of which are bound together in an
array and positioned at a fixed distance from the
distal surface of the shleld such that when
-lOa-
selectively coupled at their proximal ends to a laser
beam, a light spot pattern is formed on said shield
capable of covering a substantial region of the
distal surface of the shield with spots of controlled
size to apply a predetermined dose of radiation on
the distal surface of the shield for transmission to
said tissue; and
d) selector means at the proximal end of
the catheter for selecting which fibers are coupled
to said laser beam in order to provide said light
spot pattern.
Also in accordance with the invention there
is provided a laser catheter for insertion and
tunnelling into and removal of body tissue by
delivery of laser light of precise dosage to said
tissue, comprising:
a) a catheter with a proximal and a distal
end;
b) a multiplicity of optical fibers each
secured in a fixed position relative to the catheter
and having a distal end and a proximal end with the
proximal ends arranged in a first array and the
distal ends arrayed in a second array;
c) a rigid optical shield on the distal end
of the catheter providing a laser light transmitting
enclosure over the distal end of the catheter and a
distal surface for displacing fluid positioned at a
predetermined distance from the distal ends of the
fibers such that direct contact can be made by said
distal surface with the tissue; and
~ d) coupling means for coupling selected
proximal ends of said first array to a laser source
such that a laser beam of light is transmitted along
the:fibers to the distal ends of said second array
where said light lS transmitted -through said shield
!
-lOb-
to produce a pattern of spots of laser light of
predetermined dosage on a substantial region of the
distal surface of the shield sufficient to remove
controll.ed amounts of tissue adjacent the distal
surface of the shield in an amount and region large
enough to permit advancemen-t of said catheter further
into said tissue to remove additional amounts of the
tissue.
Also in accordance with the invention there
is provided a medical laser catheter comprising:
a plurality of optical fibers positioned
within a catheter having a longitudinal axis, a
proximal end and a distal end where the distal end
has a cross-sectional area orthogonal to the axis;
a laser beam coupled to any selected group
of the optical fibers at the proximal end of the
catheter;
an optical shield covering the cross-
sectional area on the distal end of the catheter to
transmit laser radiation emitted at the distal end by
the selected fibers; and
a distal surface on the shield comprised of
contiguous surface regions, each region correlated
with one of the fibers, where each fiber directs
laser radiation onto, and receives radiation from,
one region of the distal surface that is positioned
at a fixed distance from the distal end of the
correlated fiber and such that the regions in
combination overly :substantially the entire cross-
sectional area of the catheter on the distal surface
of the shield.
In accordance with the invention, an
optical fiber, or fibers, which can carry laserradiation is mounted in a flexible inert plastic
catheter materlal :with a transparent protective
:
:
.
~Z7990~
--lOc--
optical shield over the distal end. This assembly
constitutes the laser catheter. This catheter is
inser-ted into a blood vessel and the shield, at the
distal end, is brought into contact with the plaque.
This placement may be facilitated by use of a hollow
flexible guide or outer catheter. When the shield is
in contact with the plaque or o-ther obstruction site,
the intervening arterial blood is pushed away and
direct radiation
:
~`
~27~
for diagnosis and tissue removal is made possible.
The shield may be in the form of a glass, fused
silica, sapphire or other transparent member. The
shield may be flat, spherical or lens shaped. The
periphery of the shield is bonded to ~he end of ~he
catheter wall.
The enclosed protected region provided by the
shield can be used to mount or incorporate elements
of various kinds. Several fibers can be precisely
positioned at different locations within the shield.
Lenses or mirrors, and mechanical or optical aiming
and focusing devices can be mounted inside of the
shield. Light can be delivered to the tissue via one
fiber, and the reflected light returned by means of
the same or another "sensing" fiber for spectroscopic
or other forms of analysis. Other detection devices,
such as endoscopes, can also be mounted in the
shield. The fibers may be secured to each other with
an adhesive substance, and likewise may be bonded to
the optical shield.
The protective optical shield mechanically
displaces the blood and also protects the fiber(s)
from the intra-arterial contents. The fiber(s) are
anchored so that there is an appropriate distance
between the output end of the fiber(s) and the tip of
the shield. The catheter and shield are sealed
watertight, preventing blood from coming into contact
with the internal components. The intervening space
may be filled with fluid, or optical surfaces may be
-12-
optically contacted, or they may be anti-reflection
coated to reduce Fresnel reflections and maximize
transmitted light.
The optical shield overcomes disadvantages of
the bare fibers of the prior art and provïdes new
capabilitiesO By locally displacing blood, the
shield provides a clear field of view between the tip
of the fiber(s) and the tissue, without the need for
a purge or flush. Visualization via a viewing bundle
without flushing also becomes possible. As the blood
fluid is an excellent heat sink, this mechanical
displacement reduces the highly undesirable transfer
of heat from irradiated tissue to the blood.
The shield also provides greater control in
delivery of the laser light. The ratio of power to
intensity is no longer determined by the core
diameter of a fiber. The light emanating from the
tip of a fiber is in the form of a cone, the output
spot is smallest at the fiber tip and becomes larger
with increasing distance from the fiber tip.
Therefore, the distance between the end of a fiber
and the surface of the shield can be adjusted, for a
given application, to op~imize the light spot size on
the output surface of the optical shield, and
therefore on the tissue in contact with it. Spot
size can also be varied by means of lenses inserted
within the shield, or by mixing the modes of the
fibers to varying degrees.
When multiple optical fibers are used, the
O~
-13-
overall distribution of light from ~he laser catheter
may be controlled by disposing the fibers at
different positions and angles. Reducing the light
intensity minimizes excessive heating of the tissue
due to debris adherent to the tip of the device, a
complication which may occur in bare fiber tissue
removal. The optical shield also protects the
patient in case of fiber failure.
Most importantly, the optical shield of the
invention provides a means of delivering a precisely
controllable dose of photons to remove a specified
volume .of tissue. The rate and extent of tissue
removal is governed by three independent optical
parameters, which can be specified as incident laser
power, exposure time, and spot size. Other
parameters, which can be derived from these three
include the energy delivered (product of laser power
and exposure time), incident intensity (ratio of
power to spot area), and fluence ~product of laser
power and exposure time divided by spot area). The
ability to remove a specific amount of tissue
requires precise control of all three of these
parameters.
Spot size cannot be controlled in a bare fiber
laser catheter. If the fiber tip is in direct
contact with the tissue, the light spot diameter is
that of the fiber core (assuming the laser light
completely fills the core). With increasing spacing
between fiber and target the spot size increases
-14-
because of the spreading cone of light emanating from
the fiber~ But, experience shows that even under
direct visualization the spacing betwen the tip of
the fiber and the target lesion cannot be accurately
controlled. Furthermore, intervening blood and/or
debris then limits control of the incident laser
power. Hence, controlled tissue removal cannot be
achieved in a bare fiber laser catheter. In
contrast, experimental evidence presented below will
demons~rate that in the laser catheter invention
described herein tissue can be removed in a
controlled fashion.
The laser catheter can, of course, be used in
veins as well as in arteries. It can also be used in
other vessels, ducts or body cavities. It can be
used to penetrate most types of tissue. In all cases
the optical shield of the invention provides the
means for controlled delivery of light to the tissue
to be treated or removed. The tissue is
spectroscopically identified and the tissue is
removed as needed. Such removal enlarges an existing
lumen or creates a channel wherein solid tissue is
present. The laser catheter is advanced into the new
channel and the process is repeated. Mechanical
control devices built into the laser catheter body
can be used to bend or position the laser catheter
and so enlarge the channel internally, without
necessarily enlarging the opening through which it
was introduced. Thus, lesions larger than the laser
~2~
-15~
catheter and those with irregular shapes may be
removed from any tissue within the body.
IV. Brief Description of the ~rawinys
Fig. 1 is a broken longitudinal sectional view
of a laser catheter showing thé preferred embodiment
of the invention.
Fig. lA is a cross-sectional view of the distal
end of the laser catheter of Fig. 1 taken along line
I-I.
Fig. 2 is a longitudinal section of the distal
end of a seven fiber laser catheter.
Fig. 3 is a cross-sectional view of the distal
end of the laser catheter of Fig. 2 taken along line
III-III.
Fig. 4 is a sectional view of a laser catheter
embodiment with multiple optical fibers disposed in
an artery 30, showing the device in a typical
operating environment.
Fig. 4A is a sectional view of the laser
catheter of Fig. 4 disposed in tissue.
Fig. 5 is a sectional view of a mode mixer used
in conjunction with the laser catheter invention.
Fig. 6 is a cross-sectional view of an different
embodiment of a mode mixer.
Figs. 7A-F show alternate embodiments of the
optical shield of the laser catheter.
Fig. 8 iS a process diagram showing a method of
-16-
fabricating an optical shield in accordance with the
invention.
Fig, 9 is a cross-sectional view of alternate
embodiments of the optical shield~
Figs. lOA-G show various embodiments of~optical
fiber plugs for the laser catheter.
Figs. llA-B show alternate embodiments of the
optical fiber array o~ the proximal end of the laser
catheter.
Figs. 12A and 12B are side and top views,
respectively, of another alternate embodiment of the
optical fiber array of the proximal end of the laser
catheter, and the associated rotating mirror fiber
selector system.
Figs. 13 A-F show optical shield embodiments
with various types of optical elements incorporated.
Fig. 13A: lens Fig. 13B: mirror; Fig. 13C:
holographic element; Fig. 13D prism; and Fig. 13E:
multiple lenses; FigO 13F: acousto-optic deflector.
Figs. 13 G-J show methods of deflec~ing the
optical ~ibers in the optical shield o a laser
catheter. Fig. 13G: electromechanical device; Fig.
13H: control wires; Fig. 13I: balloons; Fig. 13J:
angled fibers.
Fig. 14 is a sectional view of the distal end of
a laser catheter disposed within a guide catheter.
Fig. 15 is a sectional view of the distal end of
a laser catheter which incorporates a guide wire in a
side lumen.
- 17 -
Fig. 16 is a sectional view of the distal
end of a laser catheter which incorporates a guide
wire in a central lumen.
Figs. 17A-D show several views of the
distal end of a laser catheter which incorporates
a guide wire and a deflecting wire.
Fig. 18 is a sectional view of the distal
end of a laser catheter which incorporates a
balloon on a rotary joint.
Fig. 19 is a block diagram of a linear
array fiber selector system in accordance with
the invention.
Fig. 20 is an alternate embodiment of
a linear array fiber selector system in accordance
with the invention.
Fig. 21 is a schematic diagram of a
method and apparatus wherein the same optical
fiber may be used for illuminating and collecting
return light for spectral analysis.
Fig. 22 is an alternate embodiment of
the apparatus of Fig. 21.
Fig~ 23 is a schematic of a multichannel
spectral detector which may be used in connection
with the system of the present invention.
Figs. 23A and 23B are plots of depth
and diameter, respectively, of holes formed by
laser ablation in samples o~ atherosclerotic
plaque with a 750 um spot size at various powers.
~9~
- 18 -
Fig. 24 is a block diagram of a typical
system in accordance with the invention for removal
of plaque in an artery.
Fig. 25 is a sectional view of a laser
catheter embodiment disposed in the bend of an
artery, showing the device in operation.
Figs. 26A and 26B show data of fluor-
escence intensity versus wavelength taken for
normal artery 27A and plaqued artery 27B, respect-
ively.
Fig. 27 shows the experimental arrange-
ment for which the data of Figs. 26A and 26B were
taken.
V. Best Mode of Carrying out the Invention
V.A. Components
V.A.l. Laser Catheter, Preferred Embodiment
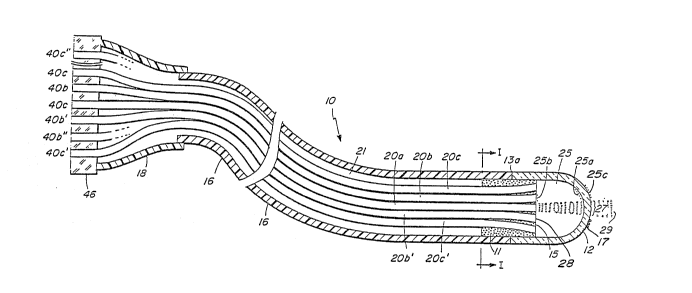
Fig. 1 shows the preferred embodiment
of the entire laser catheter 10 in broken longi-
tudinal section. It is terminated at the distal
end by the optical shield 12 and at the proximal
end by the fiber optic coupler 46. The flexible
catheter body 16, with lumen 21, is typically
1/2 to 2 meters long and is designed for insertion
into or to be in contact with the patient. A
protective enclosure 1~ which connects the catheter
body 16 to the coupler 46 is preferably short
but may be of any length. The optical shield
12 is a transparent enclosure made of fused silica,
glass, or sapphire or other optically
--19--
transparent material capable of withstanding heat,
steam and high laser power. Optical transparency may
include ultraviolet, visible and infrared light,
depending on the light and laser sources used.
The distal end o optical shield 1~ of Fig. 1 is
shown with a hemispherical cross section, but it may
also be rectangular, flat, l'ens-shaped or of any
other shape. The optical shield 12 may be secured to
catheter body 16 by a butt joint 13a, as in Fig. 1,
or by a tapered joint 13, as in Fig. 2. The joint
may be overlapping if desired. A bonding agent or
wrapping material may be used to secure the joints 13
or 13a.
Optical fibers 20a,b,b',c,c' are disposed within
the catheter body 16 and have a distal termination in
the vicinity of the optical shield 12. The
corresponding proximal ends 40a,b,b',c,c' of optical
fibers 20a,b,b',c,c' are secured by the fiber optic
coupler 46. The distal ends'of the optical fibers
20a-c' are secured in the material of plug 11. The
optical fibers 20a-c' may be angled with respect to
the axis of symmetry, as shown in Fig. 1, or they may
be straight and coaxial with the distal end of the
laser catheter 10, as shown 20a, b, b', in the
longitud~nal section Fig. 2.
The preferred embodiment of the laser catheter
10, shown in section in Fig. 1 and in cross section
in the plug at the distal en~ in Fig. lA, contains a
set of nineteen optical fibers, consisting of a
-20-
central optical fiber 20a, a ~irst ring of six
optical fibers represented by 20b,b', and a second
ring of twelve optical fibers 20c,c'. An al~ernate
embodiment, the distal end of which is shown in the
longitudinal section of Fig. 2 and the cross section
of Fig. 3, contains seven optical fibers, including a
central optical fiber 20a and a first ring of six
optical fibers 20b,b'. In either case each optical
fiber is composed of a core 22, a cladding 24 with
lower index material than the core 22, and a
protective buffer 26 which may or may not extend to
the distal end of the fiber. In the preferred
embodiment the core 22 and cladding 24 are fused
silica or glass or fluorite glass, so as to withstand
high laser power.
The preferred embodiment of the fiber cptic
coupler 46, a~ the proximal end of the laser catheter
10, is a flat linear array of the optical fiber ends
40a-c' of optical fibers 20a-c'. In addition,
optical fiber ends 40b" and 40c~ depict optical
fibers which do not appear in the sectional drawing
Fig. 1 of the distal end of the laser catheter. The
coupler 46 holds all nineteen optical fibers 40a-c~
in a linear array. An additional op~ical fiber,
shown as 20d in Fig. 19, may be incorporated if
desired, with one end disposed in the proximal linear
array and the other end connected to a laser power
monitor. Other geometries -for the coupler 46 may
also be used.
~9~
-21
.
The distal end of each of the optical fibers
20a-c' is terminated in a surface with an optical
finish 28 on the core and cladding. This surface may
be flush with or protrude from the securing plug 11.
In the preferred embodiment the distal ends of the
optical fibers 20a-c' are secured by a plug 11 formed
of epoxy which is molded around the optical fibers
20a-c'. The molded epoxy plug 11 has an optically
ground and polished surface 15, as shown in Figs. 1
and 2~ This plug 11 may be secured to the optical
shield 12, to the ca~heter body 16, or preferably to
both. Epoxy plug 11 adds strength to the optical
shield 12 and to the joint 13a or 13 with the
catheter body 16. As shown in Fig. 1, the optically
polished distal ends 28 of optical fibers 20a-c'
provide an exit surface for the laser beam or
diagnostic light beam 29. Fig. 1 shows a conical
beam of laser light 29 exiting optical fiber 20~
forming spot 27 on the ollter surace of optical
shield 12. Addition of an anti-reflection coating
25b to this optically polished surface 28~ and to one
or both surfaces 25a,c of the optical shield 12, will
reduce Fresnel reflections of the beam 29. Fresnel
raflections reduce delivered laser power, and the
reflected beams may damage the plug or irradiate
tissue in an unintended location.
The optical fibers 20a-c' must be able to accept
laser radiation at their proximal ends 40a-c', Fig.
1. In the embodiment of Fig. 1 the proximal ends of
-22-
-
-all the optical fibers are arranged in a linear array
and epoxied between a pair of glass slides to form
the proximal input end array 46. This assembly is
ground and optically polished as a unit. An
additional optical fiber 20d of Fig. 19 going to a
power monitor may also be included. Linear
translation of this array 46 past the laser beam or,
conversely, translation of the incident laser beam
past the array 46 will allow selec~i~n of the optical
fiber 20a-c' to be activated.
The laser catheter 10 permits delivery of high
power laser radiation from the coupler 46 at the
proximal end through the optical fibers 20a-c' and
through the optical shield 12 to the tissue to be
treated. The laser catheter 10 may also be employed
to deliver spectral diagnos~ic radiation, either from
a laser or a conventional light source. The
scattered or fluorescent light returning from the
tissue passes through the optical shield 12 and
re-enters the distal ends of the optical fibers
20a-c', and exits the proximal ends of the optical
fibers 40a-c' in the coupler 46, where it may be
analyzed. Returning scattered light or fluorescence
can also be collected and analyzed during high power
treatment irradiation, and can provide a signal for
feedback control.
Radio-opaque material may be incorporated in the
laser catheter 10 to aid visualization by
fluoroscopy. There are several locations at which
-23-
such ma~erial can be added, such as: incorporation in
the catheter body 16 material, in the buffer 26 of
the optical fiber 20, in the molded plug 11 material,
or within ~he silica or glass of the optical shield
12. A metal band or wire 13f,g,h, shown in Figs.
7A-D, may be placed around the laser catheter 10 near
the distal end, which can serve both as
radio-opaque marker and provide mechanical support to
the optical shield 12. A combination of these
radio-opaque indicators may be best for optimum
fluoroscopic observa~ion of the laser catheter 10
when it is used percutaneously.
Fig. 4 shows the laser catheter 10 in a typical
application, to remove plaque 34 from artery 30 which
is partially obstructing the normally blood filled lumen
36 formed by artery wall 32. In the embodiment of Fig. 4
the optical fibers 20a-c' are arrayed such that each
of the laser spots 27a-c' on the exterior surface of
the optical shield 12 formed by exitiny laser beams
29a-c' slightly overlap with adjacent spots. Assuming
the diameter of the hole or nibble produced in the
irradiated tissue to be the same as the spot size of
the impinging radiation, this overlap conditlon
insures that any and all plaque 34 in contact with
the distal end of the optical shield 12 can be
irradiated and removed by selecting the correct
optical fiber(s) 20a-c'. The overlap of spots 27a-c'
insures that laser radiation can be delivered through
all of the surface of the distal end of the optical
-24-
shield 12.
The above description assumes that the diameter
of the hole or nibble produced in the irradiated
tissue is the same as that of ~he spot of impinging
radiation. This is true when the fluence of the
impinging radiation is sufficiently high; otherwise
the hole diameter will be somewhat smaller than that
of the incident spot. In this case, in order to
insure that all plaque in contact with the distal end
of optical shield 12 can be removed, the optical
fibers 20a-c' in the plug must be arrayed so that the
degree of overlap of the laser spots on the exterior
surface of the optical shield is accordingly greater.
Experimental information describing the relationship
between spot size and hole diameter is presented in
Sec. V.B.2 herein.
Referring again to the application example of
Fig. 4, the optical shield 12 of the laser catheter
10 is brought in contact with tissue such as plaque
to be removed. Laser light is applied fired
through optical fiber 20a, as indicated by the shaded
area, removing a "nibble" 35a of plaque 34. optical
fibers 20a,b,c are~ ~ired sequentially, removing
overlapping "nibbles~ 35a,b,c. Additional fibers,
not illustrated in the section drawing, aimed at
plaque are also fired~ Optical fibers 20b' aimed at
blood in lumen 36, and 20c' aimed at artery wall 32
are not fired. Removal of plaque nibbles 35a,b,c
allows the laser catheter 10 to be advanced.
-25-
VoA~ 2 Optical Fiber Mode Mixer
In the preferred embodiment an adjustable mode
mixer is used to mix the modes in the optical fibers
20a-c' and increase the angular divergencé of the
exiting laser beams. The pre~erred embodiment of the
mode mixer 120 is shown in Fig. 5; an alternate
embodiment is shown in Fig. 6.
Since the diameter or ~size~ of each spot
27a-c', Fig. 4, on the distal surface of the optical
shield 12 is dependent on the divergence angle of the
respective exiting laser beams 29a-c', this spot size
may be adjusted and overlap of adjacent spots
optimized by mixing the modes of the optical fibers
20a-c~. Microbends, which mix the modes, are made by
placing the optical fibers against small rod-shaped
structures 122 with bending surfaces, such as wires~
nylon manofila~ents molded projections, or the
like, and applying pressure by means of a movable pad
124.
Two or more bending surfaces 122 can be placed
in series by mounting the wires 122 on a support
block 126. The material of pad 124 at the surface at
which pressure is to be applied to the fiber(s)
should be chosen to be somewhat compliant, such as
rubber. Rod-shaped structures with bending surfaces
122 may be affixed to this surface. One or more
screws 128 press pad 124 against the rod-shaped
structures 122 with the optical fiber~s) 20 in
, 26-
between. The support block 126 may be made otransparent materials so as to view the optical
fibers 20a-c' as they are mixed. Excessive pressure
will cause excess light ~o be scattered from the
microbends in the optical fibers 20a c', and this can
be observed through the transparent block 126.
One or more mode mixers l20 may be placed in
permanant positions on optical fibers 20a-c' of the
laser catheter 10 near the proximal end. In this
case a removable pressure pad 127 presses the optical
fibers 20a-c' against a removable wire support or
molded microbend surface 123, and the assembly is
potted in epoxy and removed from the frame 125.
V.A.3. Laser Catheter, Alternate ~mbodiments
Yarious alternate embodiments of the laser
catheter 10 are possible. Beginning with the distal
end of the device, and referring to Fig. 7A-F, the
optical shield may have a square internal and
external surface shape 12a, a slightly rounded
external one 12b, or a combination of flat internal
and round external surface shape 12c. An
asymmetrical shape 12d, as well as fully rounded
shape 12 of Fig. 1, may be used if the device is to
be moved sideways or at an angle to the ~orward
direction.
A flexible balloon 12e may also be used as an
optical shield as shown in Fig. 7E. Pressurizing gas
or fluid to inflate the balloon optical shield 12e
-27-
may be supplied from the central lumen 21 of the
laser catheter body 16, or from an auxillary lumen
21a . The geometry of this optical shield can be
adjustabler and may partially conform to the tissue
it contacts. A deflated balloon 12e will be more
readily inserted and removed, and the expanded
balloon 12e may displace blood over an area larger
than the catheter body 16 diameter. The balloon
optical shield 12e may be made of translucent rubber
or soft plastic. The use of silicone rubber or
fluorocarbon polymers is suggested be to withstand
high laser power and heat. A rigid metal tube or
cannula 16 may be used with an optical shield that
is a transparent disc 12f. The ca~nula 16f may be
cut at an angle so as to aid in insertion into
tissue. The optical shield 12f may be angled as
shown or at right angles to the cannula.
There are also a variety of embodiments for the
joint 13 between the optical shield 12 and the
catheter body 16. Figs. 7A-D illustrate an overlap
joint 13a, an angled overlap 13b, a step joint 13c, a
butt jo;nt 13d reinforced by plug 11, and a tapered
joint 13 of Fig. 2, respectively. The taper 13 may
slope either way; the catheter body 16 may be bonded
to the inside of the optical shield 12 as well as to
the outside. In addition, joint reinforcing material
wire wrapping 13f, thread wrapping 139, and a metal
or plastic band 13h may be used. Bonding agents for
joints 13a-d include epoxy, cyanoacrylics, and
-28-
friction fit using shrinkable or compressible
catheter material. Any join~ and any reinforcing
material may be used in combination with any optical
shield design. As shown in Fig. 1, a thin coating
material 17 such as fluorocarbon, hydrocarbon, or
silicone polymer may be put on the exterior distal
end of the shield ~o minimize s~icking of the optical
shield 12 to tissue 34, to reduce adherent char, to
reduce biological interactions between blood 36 and
the optical shield 12, and to reduce Fresnel
reflections.
Various methods may be used to fabricate the
optical shield 12. The shield may be free formed
from tubing in a torch or other h0at source using
glassblowing techniques~ ~t may be formed over a
precision mandrell using heat. Or, as shown in Fig.
8 it may be assembled from two parts by fusing or
bonding the parts. In Fig. 8A a rod 600 or disc with
the polished end is inserted into a tube 602.
Typical dimensions are indicated but any size may be
made.
In Fig. 8B the tube 602 and 600 rod are fused
with a torch CO~ laser or other heat source. If a
rod is used, the excess rod is cut off as in Fig. 8C
and the assembly is polished on the distal end. The
assembly of Fig. 8D thus provides an optical surface
both inside and outside. It may be further shaped as
in Fig. 8E.
An alternate construction is shown in Fig. 9
-29-
wherein the rod or disc 600a is attached to the tube
602 using a butt joint as in Fig. 9A. Matching
conical surfaces for rod or disc 600b and tube 602
Fig. 9B will help keep the joint away from ~he laser
beam path 29c of Fig. 4. Alternatively a step
diameter in the rod and plug 600c, Fig. 9C, will help
to do the same. A metal band 606 can be used to bind
and strenghten the joint as in Fig. 9D.
Other embodiments of plug 11 holding optical
fibers 20 are shown in Figs. 10A-G. A piece of solid
material can be drilled with parallel holes to make a
straight array plug lla, or holes may be angled llb;
these holes are skewed so as not to intersect inside
the plug. Optical fibers 20 may be bonded with epoxy
or other bonding agent l9a,b in the holes in the
plug, with distal ends 28a,b optically finished.
When the optical fibers are finishedseparately from
the plug they may be recessed or protruding, or the
bonded optical fibers 20 and plug 11, lla, llb may be
optically finished as a unit. Optical fibers 20 may
be optically contacted or fused to transparent plug
llc, Fig. 10C, or embedded in the plug lld, Fig. 10D.
Preferably the transparent plug llc,d will have the
same thermal expansion properties as for the optical
fibers 20, to minimize stress at the ioint. Melting
temperatures need not be the same, however, as it may
be desirable to form the plug llc, lld around or in
contact with the optical fibers 20, without melting
the fibers. The junction 28c, 28d between optical
-30-
fiber 20 and transparent plug llc, lld must be of
good optical quality. Plug lle in Fig. 10E shows the
optical fibers fused into a block of glass and
optically polished 28e. This construc~ion is similar
to that of the molded epoxy plug 11 of Fig. 2.
Similarly, in Fig. 10F the optical ibers 20 may
themselves be fused together wi~h or without the
addition of a fusing or sintering material l9f to
form plug llf with optically polished surface 28f.
Fig. 10G shows optical fibers 20 fused to each other
with optional fusing material l9f, with their output
ends 28g fused, bonded or optically contacted
directly to optical shield 12g. In this case the
thickness of the distal end of the optical shield 12g
is adjusted to allow the proper divergence of the
emitted laser beam 299, to obtain the correct beam
spot size 27g on the distal face of the optical
shield 129. A similar arrangement can be used for
transparent plugs llc and lle in bonding or fusing
them directly to the optical shield 12g.
When, as in Fig. 1, there is an intervening
space 25, between the plug 11 and the optical shield
12, it may be filled with air. Alternate embodiments
may have this space 25 evacuated, gas filled, or
transparent~fluid 25d, Fig. 15 filled to reduce Fresnel
reflections. Optical fibers 20 and optical surfaces
adjacent to this space may be coated with an
anti-reflection coating 25a,b to reduce Fresnel
reflections.
-31-
Alternate embodiments of the catheter body
material l~ include plastic, plastic with "memory" or
an ability to retain deforma~ions, metal tubing,
corregated or spiral for flexibility, and coated
versions of the above for biological compatability.
A mesh or metal, glass, or plastic ~iber may be
incorporated into the cathe~er body 16 so as to
enhance control. For example, 7torque control" will
make the catheter body 16 ~lexible to a bending
motion, but it will resist twisting when torque is
applied. This allows ~he laser catheter 10 to follow
bends in a vessel, but the distal end may be rotated
by rotating the proximal end of the catheter body 16.
An alternate embodiment of proximal input array
46 is shown in section in Fig. llA. Optical fiber
ends 40a-c are arranged in a circular array 46a,
and selection is performed by rotating the array
46a with respect to the laser beam, about an axis
47. Another embodiment, shown in Fig. llB, has
the optical fibers at the proximal end 40a-c"
arranged in the same spatial array 46b as for the
distal end, with each optical fiber 20a-c' in
the same relative position at both ends. Central
fiber 40a is surrounded by first ring 40b-b" and
second ring 40c-c". Such an arrangement of optical
fibers is termed a "coherent bundle" by accepted
terminology. Square packing or other arrays may be
used in coherent bundles. Any number of rings may be
used. Another embodiment has optical fiber 20a-h ends
r
-32~
pointing inward on a ring 108, as shown in Fig.
12A,B.
Alternate embodiments of optical fibers 20
include any light conduit. The optical fiber
described previously has a core 22 which carries the
optical radiation, a cladding 24 of lower index of
refraction which confines the radiation, and a jacket
or buffer 26 which protects and strengthens the
optical fibers 20, Fig. 2. Alternate embodiments
include optical fibers 20 without buffer 26, and
without buffer 26 or cladding 24. ~In the case of
core only the surrounding air or gas functions as
lower index cladding.) Graded index optical fibers
may also be used. ~he core 22 need not be solid; a
fluid filled tube may also be considered an optical
fiber 20. A gas or air filled hollow waveguide tube
may also be used, and may be made of metal, glass or
plastic, with an optional reflective coating inside.
Various numbers of optical fibers may be ùsed. In
the preferred embodiment, nineteen optical fibers 20
form a symmetric hexagonal close packing array as
shown in Fig. lA~ This is likewise true for the
seven optical fiber 20 configuration shown in Fig. 3.
The sequence for larger numbers of optical ibers is
thirty-seven, sixty-one, etc., to form hexagonal
clo~e packing. The optical fibers need not all be
the same size or type in a laser catheter.
A fiber optic bundle, which consists o two or
more optical fibers mechanically bonded at each end,
.
~LZ799~
may be used instead of any of the individual optical
fibers 20. A coherent fiber-optic bundle, which is
a bundle of optical fibers with both ends bonded so
as to hold the optical fibers in identical or well
defined spatial arrays, may be used in place of any
or all optical fibers 20.
One alternative embodiment of the laser catheter
10, incorporates a single coherent bundle of optical
fibers composed of hundreds to tens of thousands or
more optical fibers. Illumination of the proximal
input end of the laser catheter 10 will then provide
corresponding output at the distal end. Changing the
number or area of optical fibers illuminated at the
input end will change the spot size at the output
end. Moving the input spot will shift the output
spot location accordingly. The output beam may be
smoothly moved across the target tissue. An output
beam which can provide complete overlap is thus
provided. Likewise, diagnostic radiation can be
supplied to any location, and the return fluoresCencQ
or scattered light from the tissue can be monitored
at any location. This embodiment is similar to the
preferred embodiment described, except that the few
optical fibers 20 are replaced by many optical
fibers, and the proximal input end has the coherent
bundle of optical fibers in a matching spatial array
46b, as shown in Fig. llB, rather than in a linear
array.
An alternate embodiment of the mode mixer is
` ' ~
-34-
shown in Fig. 6. Optical fiber(s) 20 is clamped
between two rigid blocks 136a, 136b, with one or both
blocks being transparent. An optional metal
reinforcing plate 137a,b may be used to increase
stiffness. Several screws 138 pass through plate
137a,b and/or blocks 136a, 136b. Compliant pads
134a,b, havingrods 132 or integral projectiQns 133 are dis-
posed between the blocks 136 a and b. These pro~jections
cause microbends in the optical fiber(s) 20 when the
screws are tightened. One of the pads 134a,b may be
transparent so as to view the light escaping from the
microbends in optical fiber(s) 20. Adjustment of
screws 138 enable variation of the amount of mode
mixing. The optical fi~er(s) 20 and (removable) pads
134a,b may be cast in epoxy or other bonding material
for permanent mode mixing.
Alternate embodiments of the distal end laser
catheter Figs. 13~-E use a lens 221, multiple lenses
222, holographic element, polarizer, or grating 223,
prism 224 or a mirror 225 to control the location and
divergence of the laser light and return
fluorescence or scattered light. These optical
elements shown in Figs. 13A-E may be fixed or capable
of translation, rotation, or tilt, so as to move the
position and direction of the existing laser beam.
Reflecting surface 225 is tilted, rotated, or
translated by control wires 225a. The plug 11 might
also be rotable~ Prism 224 is rotated or tilted by
one or more control wires 224a. Lens 221 is
~9~
translated in the axial direction by longitudinal
motion of control wires 221a, which changes the spot
size 27 and beam divergence 29 of Fig. 1. Rotary
motion of control wires 221a translates lens 221
perpendicular to the axial direction, and moves the
spot position 27 on the optical shield 12 Multiple
lens assembly 222 may be translated or rotated with
Control wires 222a~ Holographic element 223 may be
translated or rotated with one or more con~rol wires
223a. When the various optical elements are fixed,
control wires need not be included in the laser
catheter. The mirror 225 may also be controlled by
an electromechanical device 225b, affixed to the
mirror 225 and the optical shield 12, as shown in
Fig. 13B, or ~he mirror 225 may be affixed to the
plug 11. Devices 225b may comprise a piezoelectric,
electromagnetic, magnetostrictive, or bimetallic
thermal device. Power for device 225b is supplied by
wire 225c.
Laser beam deflection may also be achieved by an
electro-optic or acousto-optic device 229 as shown in
Fig. 13F. Light emerging from optical fiber(s~ 20
passes through one or more such devices 229 which are
powered by wires 229a; these devices affect the
direction of propagation of light when energized.
The direction of light emerging from the optical
fiber 20 may be changed by mechanically moving the
distal end of the optical. One or more control wires
226a, shown in Fig. 13H, are affixed near the distal
g~
-36-
end 226 of optical fiber 20. Longitudinal or rotary
motion of these control wires 226a will change the
position of the distal tip 226. Electromechanical
device5 227a, Figq 13G may also be used to deflect
the distal end 227 of the optical fiber 20, wires
227b supply electrial power to he ~evice 227a, which
can be secured in plug llg. PiezoelectriC,
electromagnetic, magnetostrictive, and thermal, such
as bimetallic devices may be utilized as the device
227a.
Mechanical motion as shown in Fig. 13I, of the
distal tip 228 of optical fiber 20 maybe accomplished
using one or more balloons 228a, pushing against the
fiber. Balloons 228a,b is inflated through one or more
lumens 228 c,d in central lumen 21. Central lumen 21
may also ~e used to inflate balloon(s) 228a,b. As
shown in Fig. 13I, balloon 228a is inflated more than
balloon 228b, deflecting fiber tip 228.
Fig. 13J shows aptical fibers arrayed
asymmetrically in plug llj with the distal ends 230
angled towards one side of the longitudinal axis of
the laser catheter 10. The plug llj may be fixed, or
may be allowed to rotate within rotary joint 230a.
Control wire 230b applies torque to plug llj. Rotary
joint 230a may be deleted, leaving the plug llj free
to both rotate and translate longitudinally.
The optical elements may occupy the space 25
between the optical fibers 20 and the optical shield
12, or they may be incorporated into the shield 12
r
~27~39~
such as by making it lens shaped, or they may ~e
incorporated into the optical fibers 20, such as by
means of graded index lenses at the distal ends, or
by physically shaping angled or lensed ends of the
optical fibers 20. The plug 11 may be polished in an
angled or nonplaner manner to refract the light. The
thickness of the optical shield"'l2, i.e. the distance
`measured in the axial direction, can be used to
control the amount of divergence of the laser light
when it reaches the output surface of the optical
shield 12. By making the surfaces curved instead of
flat ~he optical shield 12 may also be made to act as
a lens. The input surface curvature of the optical
shield 12 may or may not match the curvature of the
polished surface of the plug 11. In the case of
matching surfaces, both surfaces may be polished to
appropriate accuracy and optically contacted, as
shown by the con~act line 289 in Fig~ lOG. Such a
construction greatly reduces Fresnel reflections from
the optically bonded interface 2~9.
If not optically contacted, the intervening
space 25, Fig. 1, may be filled with a transparent
gas, liquid or solid material, as shown in Fig. 1 by
the numeral designation 25d. This transparent gas,
for axample, may comprise an inert gas, such as
argon, selected to withstand the high power of the
laser radiation traversing it. The filling material
may be chosen to match the index of refrac~ion of the
optical shield 12 and the optical fibes 20 held in
--38~
plug 11. The mating surfaces of the plug 11 or the
optical shield 12 may be curved, in which case a
filling rnaterial 25d with a non-matching index of
refraction can be provided. The fill material 25d in
such case forms a lens which will displace or, deviate
the laser radiation. The focal length of such lens
may be either positive or negative, depending on both
the curvatures of the surfaces and whether the index
of refraction of the filling material is higher or
lower than that of the fiber cores 22 and optical
shield 12.
The laser catheter 10 may be used in combination
with a guide catheter 140, Fig.14. The guide
catheter 140 is first inserted in the artery 30 of
Fig. 4, and brought near the lesion 34. Next, the
laser catheter lû is inserted coaxially within the
guide catheter 140 and brought in contact with the
lesion 34. A channel 142 (Fig. 14) may be
incorporated into the wall of the guide catheter 140.
This channel 142 may be used for purge and suction~
The annular space 144 between the guide catheter 140
and the laser catheter 10 may also be used ~or purge
and suction. A guide wire lS0 may be inserted into
the channel 142. The guide wire, which slides
independently of the guide catheter 140, helps to
position it in the artery 30. A second channel ~not
shown) adjacent and parallel to channel 142 allows
for both purge and suction and a guide wire at the
same time. A balloon 146 can be inflated through a
-39-
-
lumen (not shown), similar to lumen 142, to stabilize
the guide catheter.
A guide wire 150 incorporated into the laser
catheter 10, Fig. 1~, will aid in positioning the
catheter. Lumen 152 containing guide wire 150 i5
parallel to lumen 21 of the laser catheter body 16
which contains the optical fibers 20. The same lumen
152, or an additional adjacent lumen (not shown), can
be used for suetion and purge. When the guide wire
150 is in place, the laser catheter 10 may be both
advanced and rotated around the guide wire. If the
distal opening 154a for the guide wire lS0 is
proximal to the plug 11, rather than adjacent to it
as in Fig. lS then a more streamlined and smaller
catheter body 16 can be used. The guide wire lS0 may
also be hollow, for suction or purging, or may have
balloon 153 affixed to it. The guide wire 150 may
also pass through the modified optical shield 12g
through opening 154b, as shown in Fig. 16. This
opening 154b is sealed to plug 11 so that fluid
cannot enter annular space 25 between optical fibers
20b-b' and optical shield 12g. An optional inner
tube lSS affixed to plus 11 will provide a lumen 158
for the guide wire, separating it from the optical
fibers 20b-b'. An optional sliding seal 156 located
on plug 11 or modified optical shield 129 prevents
fluid from entering the catheter body 16. The
optical shield 12g may be solid eliminating space
25g. The guide wire 150 need not be centered. An
-40-
off-center guide wire 150 will allow rotary motion of
the laser catheter 10 as an aid in positioning it.
In the above embodiments an off-center guide
wire 150 allows improved flexibility in positioning
the catheter compared to a centered guide wire or no
guide wire. In another alternate embodiment the
laser catheter 10 is designed-primarily for rotary
motion and pivots about the guide wire 150, advancing
into the tissue in a helical motion. As such, the
rotating- laser catheter 10 of Figs. 17A-D is
asymmetrical and designed to cut tissue in one
direction. Laser light 29h emerges at an angle to
the axis of the lumen. The oblong optical shield
12h, shown in section in Fig. 17C, is the preferred
embodiment, but i~ is not restricted to this shape.
This is likewise true for the linear array of optical
fibers 20 held by plug llh . The optical shield 12h
contains a reflective or refractive element to change
the angle of the light beam 29h emerging from the
optical ~ibers 20. In the preferred embodiment a
prismatic surface 164 has an air space 16S so that
the laser light 29h suffers total internal reflection
and exits at an angle through the distal end of the
optical shield 12h. The surface 164 may also be a
mirror.
Fig. 17A shows a section through the narrow
dimension of the optical shield 12h; Fig. 17B shows a
section through the wide dimension o~ the optical
shield 12h; Figs. 17C, 17D shows a cross section.
-41-
The location of the guide wire 150 is indicated on
all four figures. ~ bending Wirl3 162 controls the
separation between the optica~ shield 12h and the
guide wire 150. Increasing this distance allows the
laser catheter 10 to reach tissue farther from the
QUide wire. Decreasing this distance to a minimum,
as shown in section in Fig. 14D, makes the laser
catheter more compact for easier insertion. Control
wire 162 may be straight for pulling, bent with a
rotary joint 167 at the distal end, or a wire,
spring, and tube combination which bends when the
wire is pulled within the tube. In this embodiment
the laser beam 29h emerges through the side of the
optical shield 12h, preferably close to ~he distal
end, as shown by laser beam spot positions 27h on
Fig. 17B. Thus the tissue removal will be almo~t
entirely on one side. When such diseased tissue is
removed the laser catheter 10 is rotated about the
guide wire in the direction Indicated by the arrow
168 in Fig. 17C until it contacts more tissue to be
removed. The optical shield 12h is advanced in this
helical path as tissue is removed. The radius of the
new lumen formed in the tissue depends on the
distance between the optical shield 12h and the guide
wire lS0 pivot point. The guide wire 150 need not be
the only means of pivoting the laser catheter.
An alternate embodiment uses a balloon 16S Fig.
17 A and B, to provide an anchor point. If the
balloon 166 is proximal to the optical shield 12,
~x~
-42-
then the laser catheter 10 can be positioned using
deflecting wire 162, without using a guide wire 150.
The balloon 146 can be incorporated into the guide
catheter 142, Fig. 14, and the laser catheter 10 rotated
inside it. Similarly, a balloon 146 which is
inflated asymmetrically affixed to either the guide
catheter 142, or the laser catheter 10, will position
it. A balloon 176 on a rotary joint, shown in Fig.
18, will allow rotation of the laser catheter 10
without it shifting. An annulus 173 is cut into
catheter body 16, and sleeve 174 in one or two parts
is installed in the groove. Balloon 176 is attached
to the sleeve 174, which has a ring of openings 175
to allow fluid to flow from lumen 172 to inflate the
balloon. The fluid will also lubricate che annular
space 173 between the sleeve 174 and the catheter
body 16, allowing easy ro~ary motion. An alternate
embodiment of this laser catheter 10 for helical
cutting uses an asymmetrical optical shield 12d shown
in Fig. 7D.
An alternate embodiment of this laser catheter
10, not shown, designed for rotary cutting, utilizes
two or more identical optical shields 12h spaced
symmetrically around the guide wire 150. The
proximal end of the rotating ca~heter may be mounted
on a rotary stage so as to avoid accumulated twist in
the catheter body 16.
V.A.4. Optical Input System
~2~9~3~
-43-
.
Figs. 12A, 12B, 19, and 20, illustrate fiber
selector system devices useful in connection with the
invention, Fig. 19 is a system in which the
positions of the laser 92 and a shutter 44 and lens
41 are all fixed, and the optical fibers 20,
typically an arrangement of 7 or 19 of them, are
mounted on a holder 46 in a linear array 46 and
translated by mechanical translator 200 in one
dimension in front of the focal point of the lens 41.
The optical fibers 20 are mounted in the array either
by a mechanical clamp, which may be provided with a
set of groves to seat the fibers, or they may be
glued between two glass slides and this assembly then
clamped as a unit on top o~ the translator 200. The
translator system consists of two small hand-operated
mechanical stages 202 which bring the fiber array to the
correct elevational position and focus in front of
the focal point of the laser. The third
translational stage is electrically operated by a
motor 204 and computer 80. This stage translates the
array of fibers 46 as in Fig. 19 along the horizontal
dimension such that one fiber after another is
brought to the focal point of the laser.
Such mechanical translators may be obtained from
Klinger Scientific, New York~ Electrically operated
translators are available from Aerotech Corporation,
Pittsburg, PA.
Prior to actual use, the stopping positions of
-44-
the translator ~oo must be properly set. The procedure
used is to translate the array of fibers, with the
laser set to low power operation, to determine if the
fibers are properly aligned. The motor driven
translation stage ~00 has an optical encoder which allows
the computer to monitor the position of the stage at
all times. Once properly aligned, the computer 80
stores the location of each particular optical fiber
end 40, and then each fiber can be assigned a position
and the computer can rapidly access any one of these
fibers, or it can be instructed to repeatedly step from
one fiber to another in any desired sequence and for
any desired time duration.
The operation of shutter 44 controlled by
computer 80 determines the amount of time each fiber
20 conducts the optical radiation to the tissue. In
actual use it is essential that the laser be turned
off when the fibers are being moved, because if high
power laser radiation falls on the fiber cladding or
any other part of the system damage may result.
Thus, or example, 100 um core optical fiber must be
brought into proper position with an accuracy of
approximately 20 micrometers before the shutter is
opened and the laser light is allowed to enter the
fiber.
A photodiode 45 mounted near the input ends 40
of the optical fibers 20 can detect scattered light,
either from the input ends or from the cladding 24.
Minimum scattered light indicates the best alignment.
-45-
This signal from the photodiode 45 can be coupled to
the computer 80 to cause the computer to command
motor 204 to reposition the fibers to optimize the
coupling of laser light 94 into the input ends 40.
It can also serve as a continuous monito~ during
operation, providing feedback to computer 80 for
small corrections in optical fiber array 46 position.
In this case all three axes of translation stage 200
should be motorized. A laser power meter 45a can
also be used for automa~ic alignment. It is placed
at the distal end of the laser catheter 10 and
connected to the computer 80, or it can monitor extra
optical fiber 20d, not incorporated into catheter
body 16. The computer 80 adjusts ~he input ends 40
for maximum power transmission.
A photodiode or photomultiplier 64 can monitor
scattered return light at the laser wavelength, as
shown in Fig. 21. Failure of optical fiber 20 or
optical shield 12 will scatter high power laser light
94, Fig. 19, or illuminating light 95, which returns
54 through optical fiber 20 and is detected by de-
tector 64 connected to the computer 80, which closes
shutter 44, Fig. 19.
The shutter 44 is a mechanical shutter similar
to that used on a camera, except that it is electric-
ally ~riven and is activated by the computer. When
a rapid sequence of exposures is desired, such as
on a millisecond time scale, the computer 80 closes
the shutter 44 and causes the motor 204 to
move the translater 200 to a new position, bringing a
-46-
-
new fiber into alignment, The shutter is then opened
by the computer, allowing laser light to enter the
selected fiber. The shutter exposure time is
predetermined and programmed in the computer. An
attenuator 47 is placed in the path of the high power
laser beam 94. This contains Fresnel reflection
plates with adjustable angles which vary the amount
of attenuation of the laser beam. An alternate
embodiment uses a half-wave plater followed by a
polarizer, as an attenuator 47. Rotation of the
half-wave plate rotates the polarization and changes
the amount of laser light 94 which passes through the
polarizer.
Fig. 20 shows an al~ernate embodiment for
coupling laser light into the optical fibers. In
this case the light beam is moved by the transla~or
200, rather than the fibers 20. In this way the
fibers, which are still in a linear array, are
selected by moving a mirror 48 which is mechanically
fixed in holder 206 in spaced relation to lens 41'.
Shutter 44 turns off the laser, as in Fig. 19, while
the alignment is being made. Also, the shutter
operation and translation of the holder are
controlled by a computer driven motorized system. An
alternate embodiment shown in Fig. 12A and 12B, uses
a rotary system for positioning the laser beam 94
instead of a linear translator. In this system the
optical fibers 20a-h are arranged in a circular array
on a circular holder 108 by clamps 206. The holder
9~
-47-
is concentric to the motor shaft 104 of a
galvonometer scanner, motor, or stepper motor 102. A
mirror 98 is mounted on this shaft at a 45 or other
angle to the shaft axis. The optical radiation from
laser 92 is focused by lens 41 on mirror 98. As the
shaft 104 is rotated the beam of converging light is
reflected onto different points on the circle and
enters different optical fibers 20, depending on the
rotational angle of the shaft 104. Although the
optical fibers 20 are shown evenly spaced all around
the holder 108, they may also be closely spaced on an
arc, in an array similar to linear array 46, of Fig. 20,
An alternate embodiment uses an acousto-optic
or electro-optic deflector 49 of Fig. 19 to steer the
laser beam to the input ends 40 of the optical fibers
20. Optical fiber holder 46 remains stationary and
the computer controlled acousto-optic deflector 49
directs the laser beam 94 to the appropriate optical
fiber 200 A two dimensional acousto-optic deflector
49 will allow input ends 40 to be arranged in a two
dimensional array.
V.A.S SPectral Detection System
While catheter techniques have been used for
many years to access vascular atherosclerotic sites,
diagnosis of these lesions has remained indirect, ~he
standard method being x-ray visualization of the
vessel lumen with radio-opaque dye. By employing
optical fiber catheters, in accordance with the
~79~
-48-
invention, spectroscopic methods to diagnose in situ
plaque deposits is possible. Percutaneous methods
for evaluting atherosclerotic lesions are of
considerable interest; and are particularly valuable
for therapies employing laser ablation.
A generalized spectral system is shown
schematically in Figs. 21 and 22. Excitation iight
95, Fig. 21, from a laser or conventional light
source is sent into a selected optical fiber 20. The
excitation light 95 should be of sufficiently low
power so as not to injure the tissue 34 of Fig. 4 to
be analyzed. This light passes throuqh a beam
splitter 52 wllich may have anti-reflective surface 52a
or a mirror with- a hole 50, Fig. 22. It is focussed
onto the input end 40 by a lens 41. The light exits
the distal end of the optical fiber 20, passes through
the optical shield 12, and impinges on the tissue 34
of Fig. 4. The fluorescence and scattered light
is returned via the same or a different optical fiber
20 -to the proximal end 40 of the optical fiber 20.
This return light 54 is separated by the beam splitter
52, which may be of the polarizing or dichronic type
coating 52b, or by the mirror 50 with hole 51 (Fig. 22).
This return fluorescent or scattered light 54 enters
a spectrum analyzer 60 and detector 64.
Fig. 23 is a schematic of one type of spectral
detector 65 which may be desirable to use with this
system and which can detect many different wavelengths
simultaneously. A diffraction grating 68 which
disperses the return light from a target. The
<,
~7g~1
-49-
dispersed light is projected onto a multichannel
detector 70 which has many detectors, each one of
which corresponds to a single wavelength of light
leaving the grating 68. In this manner the entire
spectrum of the return light may be obtained in a
very brief time beca~se all wavelengths are collected
simultaneously.
A typical type of detector 70 is an array of
photodiodes. An optical image intensifier may be
provided in front of the array, if the light signals
are weak. The photodiodes convert light photon
signals into electrical currents. The array of
diodes may be coupled to an automatic electronic
scanning device that samples each of these diodes
briefly. This spectral information is then fed into
computer 80 and can ~hen be shown in a display ~6 of
the intensity of light at each waveiength or by
comparison to some previously s~ored spectrum.
The correlation with a previously stored
spectrum may be used to determine whether the
spectrum of the return light is similar to the
spectrum of plaque or of arterial wall or of some
other type of tissue, and the resulting comparison
may be displayed on a numerical display 84.
Alternatively, detector 70 may comprise a
plurality of colored glass or interference filter
elements, rather than a diffrac~ion grating. The
filters are selected to correspond to particular
wavelengths where a large discrepancy is observed
~7~
-50-
between the light from healthy arterial wall tissue
compared to that of plaque. A more detailed
description of a preferred embodiment o~ a computer
controlled spectral detection system will be given
later in connection with Fig. 24.
V. B . Control Syst em
V~B.1. Spectral Diagnostics
V . B . l ( a ~ . Gener al Methods
-
Visual diagnosis of suspected atheromateous
arterial lesions, both inside the body and in excised
tissue, usually requires histological confirmation.
Normal and diseased tissue are often rather similar
to the eye, and visual clinical judgements are based
on subtle impressions from texture, color and other
factors which are often difficult to quantify.
Visual diagnosis through the narrow lumen o~ a blood
vessel using a fiber optic imaging bundle is
particularly difficult, since the field of view is
greatly restricted and visual impressions can be
distorted.
The method of spectroscopic visualization, which
is the subject of this par~ of the invention, is a
powerful new adjunct to visual methods. As
previously indicated, this method is based on the
fact that normal and diseased tissue of a ~iven type,
-51-
as well as tissues of different types, all exhibit
distinct spectroscopic features~ These
characteristics can often differ dramatically,
enabling a diagnosis to be performed rapidly and
accurately.
Spectroscopic characteristics are obtained by
illuminating the portion of the tissue to be
diagnosed with optical radiation, either from a
conventional source or a laser, and collecting and
analyzing the returning spectroscopic signal. Either
pulsed or continuous optical radiation can be used.
Any of a number of characteristics can provide
spectroscopic signals useful for distinguishing
tissue type or tissue condition. The spectroscopic
phenomena of reflection, elastic and inelastic
scattering including Raman scattering, absorption and
fluorescence can all be used to diagnose tissue. A
pulsed source can yield fluorescence, the decay time
(lifetime) of which can be different for various
tissue types or conditions. Short optical pulses can
also provide distance (ranging) information. Pulses
of acoustical radiation, propagating along an
appropriate fiber conduit, can also be used for
ranging. Further, selective staining of tissue
plaque can be used to enhance spectral distinctions
betwen different types of tissue. The detection
system can remain in operation while the intense
laser beam removes tissue, providing real-time
diagnostics. Continuous monitoring enables rapid
```"`^
~79
-52-
detection of changes such as would occur when a
plaque-arterial wall boundary is encountered. After
the diseased tissue is removed, the system can
perform diagnostics on the excised area. Although
the above example illustrates use of this method for
atherosclerosis, it should be of general
applicability for in vivo medical use.
Various types of illumination may be applied to
the area to be diagnosed. Light from conventional
sources may be used broadband, or it may be filtered
or dispersed before being directed into the fiber.
Likewise, light from tunable or fixed frequency
lasers, either pulsed or continuous may be used. A
beam splitter arrangement or the equivalent can be
employed if the same fiber is used both to deliver
intense light and collect returning light for
analysis. The returning light can be filtered
dispersed, or detected broadband. Solid state light
detectors such as photodiodes can be used to detect
the returning light. If high sensitivity is required
a photomultiplier detector can be used. Detectors
and light sources can be coupled to the optical
fibers by mechanical, electro-mechanical, or
acousto-optical or other means.
A vidicon, diode array or imaging detector with
a dispersing spectrometer can be used to
simultaneously collect a broad range of spectral
information from a sigle fiber and, as discussed
earlier, this data can be used to differentiate types
~2~39~
-53-
of tissue. Spectra can be displayed, or preferably
analyzed by computer, providing a rapid de~ermination
of whether the material being irradiated is plaque or
healthy tissue. Using this information, the firing
of the intense laser can be controlled either by the
surgeon or by the computer. When the diseased tissue
is removed from this site a nearby fiber will be
selected and the process repeated. The same type of
computer control should be feasible with many other
detection schemes.
Multiple fibers connected to individual diodes,
or to a vidicon or imaging detector, can be used to
map the distribution of plaque and other tissue at
the tip of the catheter. Likewise a single movable
fiber can provide similar information. This
information map can be stored in memory and the image
displayed. The surgeon then has the option either to
select the portion of the "field of view" to be
irradiated by the intense laser and fire it, or else
to let the computer automatically fire the laser at
the diseased points on the map. In either case the
plaque is selectively removed, leaving the healthy
artery wall intact. As tissue is removed the map is
continually updated, and the catheter tip then
advanced.
The detection systems and fiber optic systems
described above are of general applicability to in
vivo diagnostics of all types. The application to
atherosclerotic plaque is an illustration of a
~2~
-54-
particularly attractive use of a miniaturized fiber
optic spectroscopic analysis system.
V.B.l(b). Fluorescence Diagnostic
Experiments in our laboratory establish that
fluorescence is a specific spectral diagnostic that
can be used ~o distinguish fiberous plaque from
healthy arterial wallO The studies were made on
human cadaver carotid artery samples, obtained and
examined within 24 hours of extraction. Using
standard pathologic classification, all samples used
were determined to be fiberous plaque or early
atherosclerotic plaque.
Whole arterial wall samples were placed in
quartz cuvettes, immersed in saline solution. The
lumen side of each sample was secured flush against a
face of the cuvette, thus providing a well defined
suface from which to observe fluorescence. Sample
cuvettes were placed in a Perkin Elmer
spectrofluorimeter of standard type. Filters with
cutoffs at wavelengths longer than the excitation
wavelength were used to suppress backgroUnd light
scattered from the incident beam. Incident power was
less than 100 uW, and the beam irradiated an area of
3 mm x 5 mm.
Immediately after spectroscopic examination,
samples were fixed in formalin. The irradiated area
was then isolated and several histological sections
.. .
~2 ~9 9
-55-
made. Standard hematoxilin and eosin staining was
performed. The presence or absence of plaque was
established from the resulting slides, and plaque
thickness was measured for each sample in at least 15
different locations in the irradiated area, ~nd then
averaged.
The wavelength 480 nm was found to be a peak for
exciting fluorescence, with an excitation width of
about 50 nm. Excitation at this wavelength resulted
in spectra displaying pronounced differences between
normal arterial wall and artery wall with plaque.
Typical fluorescence spectra are shown in Fig. 26.
Normal artery samples displayed distinct spectral
peaks of approximately equal size at 550 and 600 nm
(Fig. 26A). Fiberous plagued artery samples
exhibited peaks at the same two wavelengths, but with
the 600 nm peak always smaller than the one at 550 nm
~Fig. 26B).
We devised a simple procedure to quantify the
fluorescence lineshape differences. Using the fact
that the height of the 600 nm peak relative to the
valley at 580 is much greater for normal than for
plaqued artery, we defined the contrast ratio, R =
I(600)/I~580), with I(~) the fluorescence intensity
at wavelength . Contrast ratios were obtained for
six histologically determined samples. The values
for three normal samples ranged from 1.72 to 2.00,
whereas the values for three samples with plaque
thickness greater than 0.5 mm ranged from 1.03 to
` ~ '~
~27~
-56-
1.09. A test comparing these two groups yielded a P
value o less than 0.01, confirming that fluorescence
can distinguish between normal artery and artery with
a plaque which is 0.5 mm or more in thickness. We
thus conclude that fluorescence induced by 480 nm
exciting light is a an effective spectral diagnostic
for the laser catheter invention described herein.
V.B.2. Ti~sue Removal Dosimetry
Laser catheters designed in accordance with the
principles of ~he invention have been constructed in
our laboratory and used to study various features of
the invention, including determining the degree of
control in the tissue removal process. Bo~h single
fiber and multiple fiber laser catheters have been
constructed.
FigO 27 depicts one prototype studied. In this
prototype a single optical ~iber 20 with a carefully
cleaved or polished output tip 28 was rigidly
centered inside a transparent optical shield 12. The
fiber 20 had a 133 um core diameter and a numerical
aperture of 0.21. The optical shield 12 was formed
by a length of 0.5 mm thick quartz tubing of 3 mm
outer diameter, closed at one end with a torch to
form a hemispherical output surface. The laser beam
29 emerging from the distal end 2~ oE the optical
fiber 20 produced a distribution of light in the form
of a circular spot 27 on the outer surface of the
~` c
~L2~99~3
-57-
optical shield. The spot size, defined as the
diameter at which the intensity of the spot decreased
by half, was adjusted by choosing the appropriate
distance between the ~ip 28 of optical fiber 20 and
the outer surface of the optical shield 12. Reticon
measurements showed the beam profile to be
approximately uniform across the spot, falling
rapidly to ~ero at the edges. Spot size
determinations were accurate to +25 um~
The experiments used blue-green light from a
Coherent I-20 argon ion laser. Data were taken in
reshly excised sections of human cadaver carotid
artery with fiberous plaque, cut open lengthwise to
expose the luminal surface. Samples, typically 1 mm
thick, exhibited relatively acellular intimal
fibroplasia 34, often infiltrated by lipid and foam
cells, overlaying media 32. The sample was placed in a
petri dish 37 and immersed in either blood or saline
solution 36a. As ~hown in Fig. 27, the tip of the
optical shield 12 was brought into contact
perpendicular to the luminal surface of the sample,
displacing the intervening fluid, and pressed against
it with a constant force of one ounce ~28 kdynes).
Dimensions of holes 35 produced in blood and saline were
the same with experimental variability, and so saline
was used in most studies. Laser power was measured
at the output surface of the device with an accuracy
of +50 mW. Exposure times, controlled by an
electronic shutter placed in the laser beam, were
~L~7~
accurate to + msec.
Holes 35 formed by laser ablation were
roughtly cylindrical with rounded bottoms. Hole
diameter was measured at the luminal surface
of the tissue using a dissecting microscope with
eyepiece reticle. Hole depth was measured by
using a histological microscope with a calibrated
fine adjustment focussing knob, bringing first
the tissue surface and then the hole bottom into
focus. Measured hole dimensions were accurate
to +25um.
Each data point was averaged from at
least 7 individual holes. Since the full range
of variations will be encountered in clinical
use, error bars were drawn to encompass all values
observed.
Figures 23A and 23B plot diameter and
depth, respectively, of holes produced using a
750 um spot size at powers of 2.5, 5, 7.5 and
10 W, versus exposure time. There are several
important trends. Consider first the curves of
hole diameter versus exposure time, Fig. 23A:
As exposure time increases hole diameter approaches
the spot size. Also, as intensity increases hole
diameter approahces spot size more rapidly. As
discussed in Sec. V.A.l, knowledge of hole diameter
as a function of exposure time for given values
of laser power and spot size can be used in the
design of laser catheters with multiple fibers
to provide a set of spots on the distal end of
optical shield 12 of Fig. 4 with sufficient
overlap for complete coverage in the
I(
` `
~799~9L
-59-
tissue removal process.
Exposure times at which 90% of the limiting hole
size is reached are 25, 25, 200 and ~lO00 msec for
10, 7.5, 5 and 2.5 W laser powers, respectively. The
90~ diameter is useful because sample-to-sample hole
size variations are much reduced at this exposure
time. It thus represents a practical threshold for
producing reproducible holes. Exposure times for
which the first perceptible spot of tisue is removed
("absolute" threshold) were found to vary greatly
from tissue sample to sample, and are therefore of
little clinical value.
The hole depth measurements, Fig. 28B, show that
in each case depth increases linearly with exposure
time up to the point of perforation. This indicates
that the removal rates ~or fiberous plaque and normal
arterial wall are similar. The slope of each curve
is the penetration velocity (mm/sec). Note that
penetration velocity increases with increasing
intensity. The observed penetration velocities are
2.56, 2.43, 1.42 and 1.05 mm/sec Eor lO, 7.5, 5 and
2.5 W laser powers, respectively.
The data show that by vary,ing laser power and
exposure time in an optical shield laser catheter of
chosen spot size, one can predictably control hole
depth and diameter in the tissue ablation process.
Selection of the proper penetration velocity, which
can be achieved by choosing appropriate spot size and
power, is crucial. An uncontrollable penetration
31,~ 0~
-60-
velocity is undesirable because in many cases there
is only a small range between the absolute threshold
photon dose and the dose leading to perforation of
the thin arterial wall. The laser ca~heter invention
describqd herein provides the needed control for
clinical use.
V~B.3. System Operation and Control
Fig. 24 is a block diagram of the entire
operating system for removal of plaque in an artery.
First, the laser catheter 10 with optical shield 12
is inserted and the shield is brought into contact
with the lesion. Next, a determination is made as to
the type of tissue at which each optical fiber 20a-c'
is aimed. Optical fibers aimed at diseased tissue
are s~lected to deliver high power laser radiation,
removing the tissue, whereas those aimed at healthy
tissue or blood are not so activated. Thus,
selective tissue removal is obtained. The
aforementioned spectral diagnostics are used to
diagnose the tissue in front of each fiber.
A light source, which may be a laser or
conventional source, 98, is applied to the fibers.
In the case of a conventinal source, wavelengths
should be selected by an optional monochronometer or
filters l00. This diagnostic light 95 is sent through
the optical fiber selector 74 to the fiber of choice.
The fibers are placed on a mechanical translator previously
described in connection with Fig. l9. The translator
-
~99~
-61-
is controlled by a computer 80, so that the correct
fiber is moved into position in ~ront of the light
emergîng from the fiber bundle. AlternatiVely, a
rotating mirror 98 as in Fig 12 or acousto-optic
beam deflector 49 as in Fig. 19 may be used to align
the light source with the fibers.
The diagnostic light exits-the distal end of the
selected optical fiber 20a-c', passes through the
optical shield 12, and falls on the tissue~ The
tissue scatters and absorbs the light, and in the
latter case re-emits some fraction of this light,
usually of a longer wavelength. This light re-enters
the distal ends of the various optical fibers 20.
The return light may come through the same or a
different fiber and is then coupled out by the
selector system 74 using, for example, a beam
splitter. This light goes to either a
monochromometer or a filter system 76 and then is
detected by a photodiode, photomultiplier or other
detector 64. A rapid scan control 90 moves the
grating, or the prism, or whatever spectrum-selective
element is used in monochromometer 76, so that it
selects one wavelength after another, sequentially.
In this way the 0ntire spectral signal from the
selected fiber(s) is converted into a time varying
signal, which is coupled to computer 80 via the
detector 64. Alternately, a multichannel analyzer 65
as shown in Fig. 23 may be used, collecting the
entire spectrum simultaneously and coupling it to the
- .
-62-
computer 80.
The computer stores the information as a
spectrum, which is a graph of light intensity vs.
wavelength. This can be displayed immediately on the
video display 82. Alternately, the spectrum can be
compared to an existing spectrum already stored in
the computer, and the difference displayed on the
spectral display 86. Corrections can be made for the
wavelength dependent sensitivity of the instrument.
In the case of a pulsed laser source, instead of a
continuous light source, a temporal display of the
return light can be shown on display 88. In~ormation
from either the temporal or spectral display may be
compared to standard spectra stored in the computer
80. The comparative data may then be read onto a
numerical display 84 to provide a quantative measure
of how well the spectral or the temperal behavior of
the return light compares to that emitted from
plaque, or conversely, healthy arterial wall tissue.
It is quite feasible that fluorescence intensities
measured at only a few wavelengths can provide
adequate information. In this case, an entire
spectrum need not be collected.
In the preferred embodiment, the light source 98
is 476 nm radiation from an argon ion laser. The
1uorescent light is monitored at peaks 550 and 600
nm, and valley 580 nm when the 600 nm peak is
comparable to the 550 nm peak and the 600 nm peak to
580 nm valley ratio is much larger than one, this
-
~2~9~
-63-
indicates healthy artery wall. When the 600 nm peak
is much smaller than the 550 nm peak and the peak to
valley ratio is near unity, this indicates the
presence of plaque.
With a multichannel detector and a reasonably
fast computer, or with appropriate multiple filters
and detectors, it is feasible to gather this
information in a fraction of a second. Thus, within
a fraction of a second after the low power spectral
light source 98 is turned on, either a spectral or
numerical display is provided which indicates the
type of tissue at which the fiber of interest is
aimed. If the tissue is plaque, and it is to be
removed, then the fiber selector 74 will align this
fiber wi~h the output beam of the high power laser
92. Then the high power laser 92 is turned on ~or it
may be already on), an appropriate level of power is
selected by attenuator 47, and shutter 94 is open for
a predetermined amount of time to remove a certain
amount of this diseased tissue. Once this event has
occurred, the shutter is closed and the high power
laser radiation is stopped.
Next, the procedure is repeated for a different
optical fiber 20. If the split-second spectral
diagnostic again indicates diseased tissue is
present, it is quickly removed by the high power
laser radiation. If, however, the spectral
diagnostic indicates healthy tissue or blood, the
high power laser is not sent through that particular
~2~9~
-64
optical fiber 20. This procedure is repeated until
all the diseased tissue in contact with the distal
end of the optical shield 12 is removed. Then the
laser catheter 16 is advanced (typically 0.3-2 mm) or
repositioned so as to again be in contact with the
remaining lesion 34. The above steps are repeated
and the laser catheter 10 nibbles away at the
diseased tissue, leaving the healthy artery wall 32
intact. In cases in which significant amounts of
plaque 34 have diffused into the artery wall 32, the
computer 80 criteria are set so that this less
diseased tissue is left intact. The laser catheter
follows the trail of the plaque, tunneling through
it, leaving the artery wall 32 intact.
If the artery 30 makes a bend 31 as shown in
Fig. 25, the laser catheter 10 will tend to make
contact with the artery wall 32 at the outside wall
of the bend. In the illustrated case of an artery
totally obstructed by plaque 34, the optical fibers
20a, b, c, b' aimed at the plaque are fired in turn,
removing "nibbles" of plaque 35a, b, c, and b'.
Optical fiber 20c' aimed at artery wall 32, is not
fired. The lesion is removed asymmetrically.
Previously, in the straight section of the artery 30,
all optical fibers 20a-c' had been fired, making
straight lumen 39. But the asymmetric removal causes
the lumen to turn and as the laser catheter 10 is
advanced, it follows the new lumen 39a around the
bend. The artery wall 32 is not irradiated, and so
-
-65-
-
is not perforated.
Fig. 4 represents the laser catheter 10
removing tissue 34 in an artery 30. However, this
laser catheter and control system may be used for
removing lesions or obstructions, veins, or in any
tube, duct, vessel, or body cavity. It may also
tunnel through various types of tissue as in Fig. 4A.
The laser catheter 10 is in tissue 334, having made
or been introduced through, lumen 333. Previous
laser firings of optical fibers 20a-c' have removed
tissue forming cavity 335. Control wires 338, or
electromechanical devices similar to those described
in conjunction with Figs. 13A-J, are used to position
the optical shield 12 against tissue to be ablated.
As shown, in Fig. 4A, optical fiber 20a has just been
fired along beam path 29a, removing nibble 335a from
tissue 334. Optical fiber 20b is being fired along
the indicated path 29b, removing nibble 335b.
Optical fiber 20c is to be fired next, removing more
of tissue 334, enlarging cavity 335. Spectroscopic
analysis may be performed on each nibble as needed,
prior to removal. Optical shield 12 design provides
control over spot size 27b and therefore the size of
the nibble 335b. It displaces fluids which may
accumulate in cavity 335, which can absorb or scatter
the laser light. The laser catheter 10 is rotated,
advanced or distal end bent by control wires 338 or
needed to bring it into contact with additional
tissue 334 which is examined and removed as needed.
~27~
-66-
A cavity 335 of removed tissue may be substantially
larger than lumen 333 through which the laser
catheter is introduced. An alternative design as in
Fig. 7F uses semi-rigid tube or cannula 16f with an
optical shield 12f. The cannula may be straight or
preformed into a curved shape. This may be
mechanically inserted to the site of a lesion in any
tissue before proceeding with laser treatment or removal
of tissue. Such a device is most likely to be useful
for smaller lesions.
VI . EQUIYALENTS
This completes the description of the preferred
and alternate embodiments of the invention. Those
skilled in the art may recognize other equivalents to
the specific embodiments described herein, which
equivalents are intended to be encompassed by the
claims attached hereto.