Note: Descriptions are shown in the official language in which they were submitted.
`` 1328~86
-- 1
METHOD AND APPARATUS FOR LASER SURGERY
This invention relates to method and apparatus for
effecting surgery through the application of a laser beam.
The present method and apparatus are particularly
applicable to the delivery of laser energy for performing
various surgical procedures. These include eye surgery,
and more particularly cornea, cataract or vitreal surgery,
- and various dental procedures.
It is recognized that lasers have experienced
widespread use in the medical field, for such purposes as
cutting, cauterizing, melting and ablating tissue. In
order to affect the tissue to the greatest degree, it has
been found that one or more of three conditions must be
met:
(a) The laser must be delivered in high powered
pulses or with high continuous power;
(b) The laser must be tuned to the extreme violet
end of the spectrum where the photon absorption is high in
organic substance; or
(c) The laser must be tuned to the extreme
infrared end of the spectrum where water, a major
constituent of living tissue, is a strong absorber.
Condition (a) functions through non-linear optical
processes such as dielectric breakdown. See for example
"Laser Induced Electric Breakdown in Solids" by N.
~- ~V~
'~
-
---` 1328~8~
-- 2
Bloembergen, IESE. J. of Quantum Electronics, Volume QE10
Number 3, March 1974, pages 375 to 386. The non-linear
process creates a finely localized absorption site since
the resulting plasma is usually opaque to the laser beam.
The noted conditions are extreme conditions which have
resulted in the inability of optical components to handle
the laser energy delivered.
A particular application of the present invention is in
keratoplasty. There are various known keratoplasty
techniques which depend on a deposition of heat energy,
i.e., heat induced keratoplasty. The principle behind heat
induced keratoplasty is that of producing shrinkage in the
principal protein of the clear corneal medium, collagen.
When type I collagen is heated above 62 C, there is a
break in the collagen cross links, and contraction of the
collagen occurs. Collagen contraction, in turn, modifies
the corneal curvature. Original approaches to heat
delivery were dependent on direct contact of the corneal
surface with a heater. The clear disadvantage of this
technique is that unnecessary collateral heat damage of
anterior layers of the cornea takes place. Another
approach is by radio frequency radiation heating of the
mid-stromal layer by means of an antenna-like probe, as
described by J.D. Doss, et al U.S. Patent 4,326,529. The
method of Doss et al involves heating the central stroma
with a radio frequency electrode probe, at the same time
_,, . , , . _ _
,
--~ 132~486
-- 3 --
cooling the surface of the cornea with a saline solution.
A clear disadvantage of this approach is that the heat
deposition is not easily localized in the three-dimensional
space of the cornea, because of the use of the radio
frequency wavelengths required by this method. The
application of radio frequency radiation resulted in an
insufficiently localized distribution of the heat energy.
Another application of the present invention is in
cataract surgery. In one technique, the lens is removed by
suction, after having been emulsified by an ultrasonic
probe, known as a phacoemulsifier. The ultrasonic energy
causes fragmentation of the human lens nucleus, so that it
could then be aspirated through fine tubing and thereby
removed from the eye. There are, however, certain
limitations of this method of cataract surgery, which is at
present the prominent method used in the United States.
These limitations include the possibility that the high
ultrasonic frequency fragmentation may result in the
fragmentation of the nucleus of the lens in large multiple
pieces, thereby complicating the actual handling and
separation of the cortex. Other undesirable effects
include potential burns to the cornea and possible
battering effects of the nucleus against the corneal
endothelium, resulting in corneal endothelial decomposition
requiring penetrating keratoplasty. The cause of this
limitation is primarily a result of the difficulty of
_ _
.. .
;; ' .~ ~ `, - ~ ,'
- ... .
1328~8~
-- 4 --
controlling the energy delivery.
Another advance in ophthamolic surgery is in the field
of vitreoretinal surgery. There has been devised a
procedure in which a vitreous infusion suction cutter was
provided for the purpose of removal of vitreous from the
human eye through the pars plana. The vitreous infusion
suction cutter is basically comprised of various scissor-
like cutting devices which are combined with a suction
element for the removal of fragments in the vitreous.
Limitations of this apparatus are significant and are
associated with the difficulty of actually moving the
vitreous bands and tissue into the cutting ports safely
without jeopardizing the adjacent retina. Also, there is
the potential for mechanically separating the retina,
producing holes by means of this mechanical cutting
device. The cutting of fine membrane attached to the
surface of the retina with such a large instrument,
typically 3 mm in diameter, makes the problem of
vitreoretinal membrane stripping, epiretinal membrane
mobilization and repair of difficult traction, retinal
detachments a long process involving many attendant
undesirable complications associated with such
instrumentation.
The prior art of surgery in the human body abounds with
demands for precise cutting, cauterizing and removal of
tissue of very small dimensions. Such surgical procedures
... .
' ~
, . -
:-. ~ - .
.,: ~ ~ . - . :
-
.: ~
-- 13~8~
- 5 -
in general require handheld surgical instruments used under
an operating microscope. Critical fields are neurological
and renal surgery where precise control of the cutting
instrument is required in order to avoid collateral damage
which could prove extremely harmful. The traditional
surgical blade has the limitation of limiting visibility of
the surgical site from lateral view, and with it there is
limited control of the depth of cut.
Among the laser apparatus used for surgery as
disclosed in prior art patents is Smith U.S. Patent
4,122,853 which discloses a carbon dioxide infrared laser
for use in photocoagulation or localized surgical
procedures. A probe is provided having a lens system which
includes first and second lenses remote from the distal end
of the probe, and which serve to receive a relatively large
diameter beam of collimated light and to reduce it to a
smaller diameter beam of collimated light: a lens at the
distal end or tip of the probe has either two planar
surfaces, or an external planar surface and an internal
curved surface which focus the beam on the window outer
surface. The probes disclosed are equipped to provide for
an endoscope, to view the site, and fluid passage for fluid
flow through the probe.
Choi U.S. Patent 4,~07,874 discloses an apparatus
using a laser beam, conducted through a fiberoptic bundle
to a thrombus to burn a tunnel through the thrombus.
Muncheryan U.S. Patent 3,382,343 and Muncheryan U.S.
~ '
.
~ ~3~8~
-- 6
Patent 3,383,491 teach laser beam apparatus with optical
systems for focussing the laser beam on a workpiece: the
lens position in this apparatus is fixed so that the plane
of the laser beam coincides with the surface of the
workpiece in contact with the instrument tip. In
Muncheryan U.S. Patent 3,381,510, a laser beam instrument
i8 provided in which there is a movable lens element, to
permit displacement of the focal plane to any desired
distance remote from the tip of the probe, so as to provide
a cutting operation on body tissue, for example, with the
tip of the probe remote from the body to avoid
contamination.
Goldenberg U.S. Patent 4,641,912 discloses a system
for delivering an excimer laser beam for angioplasty,
including an optical fiber with the distal end formed as a
lens to increase the density of the energy after it has
emerged from the fiber by reducing its cross-section: in
use, the end of the fiber is positioned at a distance from
the obstructing lesion, so that the focal plane is at the
surface thereof. Hett U.S. Patent 4,072,147 is another
: example of a laser beam focussed on an object for
therapeutic purposes.
In the field of dentistry, for decades the removal of
tooth material has been by use of rotary cutting
instruments, evolving from a motor driving the tool
through a belt or cord, to more recent developments in
which the tool is driven at higher speeds by an air
turbine. Although these instruments have been effective in
:
~: - ' . ` ' ,` '
-- 1328~86
-- 7 --
the removal of tooth material, they have experienced
certain disadvantages, such as the following:
(1) The instrument obscures the view of the site being
treated.
(2) The dimensions of the cutting components, such as
burrs, bits, etc. often are too large for the desired
precision of the procedure.
(3) The instruments are heavy and bulky.
r ( 4) The instruments require excessive amounts of
energy to operate them, a portion of which is often
delivered to the patient, making the patient very
uncomfortable as the excess energy is transmitted to non
local nerve endings.
There is provided a method and apparatus for
microsurgical and intra-oral dental purposes which are
particularly applicable to intrastromal keratomeleusis,
intrastromal keratotomy, and excimer radial keratotomy for
the correction of myopia, astigmatism, hyperopia, or
combinations of refractive errors. Laser energy of
. 20 predetermined characteristics is focussed to a focus spot
having a diameter of a few microns which is located a
predetermined distance, such as one millimeter, beyond a
lens at the distal end of a handpiece, the focus spot being
located, for example, in the intrastromal layer. The lens
at the distal end of the handpiece, of sapphire, is placed
against the cornea, and localized heating in the
- .. . ~ . .
2~
-- 8 --
instrastromal layer modifies the corneal curvature to
effect keratoplasty or keratotomy. Laser energy is
delivered in the TEM(oo) mode, the laser beam having a
wavelength of approximately 1.0 to 2.0 microns, and in one
embodiment a repetitively Q-switched YAG laser emits at
1064 nanometer wavelength. The pulses have a duration of
about 70 nanoseconds, and pulsing is at the rate of
approximately 2000 per second. Alternatively, continuous
radiation having a wavelength in the range of 1400-1800
nanometers is generated by using a Neodymium YAG laser to
pump f-centers in sodium chloride crystals, or a YAG laser
is used to stimulate Raman scattering in a hydrogen gas
cell which emits at 1900 nanometers.
In a preferred embodiment, the handpiece contains an
optical wedge which is oscillated or rotated in order to
oscillate or rotate the focus spot while the handpiece is
stationary.
In another embodiment, a focussing lens of a handpiece
is spaced from the distal end, the end portion of the
handpiece narrowing or tapering towards the distal end,
there being a fluid inlet into the terminal portion of the
handpiece, with fluid flowing in the tapering terminal
portion and exiting through escape channels at the distal
end, the laser beam travelling through fluid flowing
through the handpiece terminal portion.
In a related embodiment, the terminal portion of a
; . . ;
,. .
- ~ -. ~ ' ' " .
- '
328486
handpiece is provided with a contact tip of glass, light
being delivered to the penetration point through a single
mode optical fiber and the contact tip, and an optical
fiber bundle receiving an image of the penetration point
from a reflective surface of the contact tip for passage of
it to a display. The contact tip is provided with escape
channels at its distal end for the escape of saline
solution passed through the terminal portion of the
handpiece.
In embodiments used for conducting cataract or vitreal
surgery in which photoablative laser radiation is used, a
probe has a quartz rod to which the radiation is coupled,
the probe containing an infusion passage and an aspirating
passage on either side of the beveled distal end surface of
the quartz rod, water flowing across the beveled distal end
surface of the quartz rod from the infusion passage to the
aspirating passage.
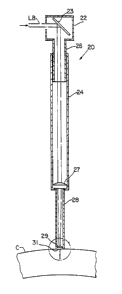
Fig. 1 is a perspective view showing a handpiece in
accordance with the present invention.
Fig. 2 is a longitudinal cross-sectional view of the
handpiece of Fig. 1 engaging a cornea.
Fig. 3 is an enlarged view of the portions of Fig. 2
within circle 3 of Fig. 2.
Fig. 4 is a chart showing results achievable with the
method and apparatus of the present invention.
Fig. 5 is a diagrammatic view of a laser system used in
- ,
1328~8~
-- 10 --
one embodiment of the present invention.
Fig. 6 is a schematic view of another embodiment of a
laser system used in and forming a part of the present
invention.
Fig. 7 is a view similar to Fig. 1 of a modification of
the handpiece of Fig. 2.
Fig. 7a is an enlarged view of the structure within
circle 7a of Fig. 7.
Fig. 8 is a diagrammatic view illustrating the
operation of the handpiece of Fig. 7.
Fig. 9 is a longitudinal cross-sectional view of a
modified handpiece terminal portion, and schematic
representations of other sources for generating photo-
ablative laser radiation.
Fig. 10 is a perspective view of the distal end of the
terminal portion of Fig. 9.
Fig. 11 is an elevational view, with parts in section,
of the terminal portion of a laser handpiece and
endoscope.
Fig. 12 is a cross-sectional view taken on line 12-12
of Fig. 11.
Fig. 13 is a view taken on line 13-13 of Fig. 11.
Fig. 14 is a longitudinal cross-sectional view of a
photoablative terminal probe for cataract surgery.
Fig. 15 is a view taken on line 15-15 of Fig. 14.
Fig. 16 is a cross-sectional view taken on line 16-16
.
.~ ' ' : :,
1328~8~ -
-- 11
of Fig. 14.
Fig. 17 is a longitudinal cross-sectional view of an
alternate embodiment of a photoablative probe for vitreal
surgery.
Fig. 18 is an end view of the probe of Fig. 17.
Fig. 19 shows probes in accordance with Figs. 14 a~d 17
in use.
Following is a description of the preferred
emdobiments.
Referring now to the drawings, wherein like or
corresponding reference numerals are used for like or
corresponding parts throughout the several views, there is
shown in Fig. 1 a handpiece 20 of generally cylindrical
form, and supported by an articulated arm system 21 which
contains prisms and hollow arms for transmitting and
refracting laser energy from a laser source S. The
handpiece 20 is shown being applied to the eye E of a
patient. Handpiece 20 depends from a suppor~ block 22
having a prism 23 therein for refracting light originating
from laser source S.
Referring to Fig. 2, the handpiece 20 includes an outer
tube 24 rotatably mounted on an inner tube 26 which is
connected to the support block 22. At the lower end of
tube 24 of handpiece 20, there is a plano-convex focussing
lens 27. Beyond lens 27, there is a terminal portion 28 of
smaller diameter, within which is a second lens 29, lens 29
', ~
~28~
- 12 -
also being a plano-convex lens. In a preferred embodiment,
the lens 27 is 6.3 mm diameter with a 30.7 mm focal length,
and is positioned 23 to 26 mm from the lens 29. Lens 29
is a sapphire lens 1.5 mm in diameter and having a 1.0 mm
focal length. In use, laser beam LB is reflected by prism
23 along the axis of the handpiece 20, and is focussed by
the objective lenses 27 and 29, passing through air along
its path until it exits from the lens 29. Lens 29 is held
with the plano surface 31 thereof against the cornea C of
the patient.
As shown in Fig. 3, with the plano surface 31 of lens
29 against the surface of cornea C, the residual fluid of
the corneal surface optically couples lens 29 to cornea C.
The laser beam radiation is in TEM(oo) mode and is
diffraction limited: it is focussed as a focus spot FS,
shown rotated 90 from its normal position for purposes of
clarity, the focus spot FS being of minimum size, of a few,
i.e., 3-5 microns diameter. The focus spot FS lies at a
predetermined distance into the cornea from the outer
surface thereof, as determined by the focal lengths of
lenses 27 and 29, which, in a preferred embodiment
provided, is one millimeter. Thus, the focus spot FS is
within the cornea C about 0.4 - 0.5 millimeter from the
surface thereof.
Because of the extremely fine focus of the laser
radiation, which is on the order of a few microns, the high
' ~ ' L
' ~ ' ' .' ,
'' ' ~ , '
1328~81~
- 13 -
field strength of the laser beam causes dielectric
breakdown and plasma formation. In a preferred embodiment,
the laser source S is a repetitively ~-switched CW lamp
pumped Neodymium YAG laser, such as the Quantronics Model
117. The pulses are at a high pulse rate of about 2000 per
second, and have a duration of about 70 nanoseconds or
less. This source emits a laser beam having a wavelength
of 1064 nanometer. The induced plasma shields the
posterior layers of the cornea C, and deposits highly
localized energy to the stromal fibers. This results in a
cutting process. However, the outer and inner layers of
the cornea C, the epithelium and the endothelium, are left
intact due to the localized or restricted region into which
the high energy focus spot FS is delivered. This permits
rapid healing of the treated site. Thus, the handpiece 20
may be used for intrastroma~ keratomeleusis for the purpose
of effecting and correcting myopia or myopic astigmatism.
The handpiece 20 can be freely passed along and over the
surface of the cornea C, so as to achieve a desired pattern
of treatment loci.
Fig. 4 is a schematic diagram showing the temperature
at various depths in the cornea due to different methods of
application of energy. Line A shows the temperature
distribution when a heated element is applied to the
surface of the cornea, there being a temperature of
approximately 75 C at the surface of the cornea, the
1328~8~
- 14 -
temperature dropping to approximately 40 C at the
endothelium layer. With the radiofrequency application
method as described in Doss et al 4,326,529, the
temperature distribution is shown by line B; there is
significant heating of Bowman's membrane and Descement's
membrane. The more finely localized line C shows the
energy distribution achieved with the present invention
apparatus and method. Thus, the significant heating is
confined to the central stroma, without undesirable heating
of ad~acent membranes.
Referring to Figs. 5 and 6, there are shown alternate
sources S-2 and S-3 of laser beams. These emit preferably
a diffraction limited beam with a wavelength in the range
of 1400 to 1800 nanometers. Unlike the source S, the beam
can be continuous, rather than pulsed as is the case with
source S. This is possible because the radiation would be
attenuated by a factor of 100 after travelling through only
about one millimeter of the corneal or other tissue, due to
water absorption. Since the absorption is linear and the
energy deposited per unit volume varies as the reciprocal
of the cubic power of the distance traversed into the
corneal media, the linear energy deposition per unit
volume, hence temperature, rises very sharply at the focus
site. Thus, the melting process can be affected at the
focus point, modifying the stromal tissue fibers in the
desired localized manner, and without disturbing adjacent
'
' ~ .
. - ,-" . ~' ,
'~
1~2~86
- 15 -
portions of the cornea.
One apparatus for achieving the desired wavelength
generation wlth a continuous beam source S-2 shown in
Fig. 5, in which a laser beam LB is generated by a
Neodymium YAG laser 32, the laser beam LB having a
wavelength of 1064 nanometers. The laser beam LB passes
through a lens 33 and strikes a sodium chloride crystal 34
having f-centers, which are pumped in conventional manner,
the resulting beam LB' being a tuneable near infrared beam
having a wavelength range of 1400 to 1800 nanometers.
Reference is made to the work of C. Pollack, 1985. In
Fig. 6, the source S-3 includes the Neodymium YAG laser 32,
the laser beam LB of which passes through a focussing lens
- 36 and into a pressurized hydrogen gas cell 37. The laser
beam causes stimulated Raman scattering in the pressurized
hydrogen gas cell 37 so that the incoming laser beam LB is
Raman shifted from 1064 nanometer wavelength to 1900
nanometer. As will be understood, the sources S-2 and S-3
may be used instead of the source S, shown in Fig. 1, to
provide continuous as well as a pulsed, laser beam
application to the cornea, shifted toward the absorption
bands in water. However, although reference has been made
specifically to corneal treatment, the apparatus and method
are not limited to treatment of corneas, but may be used
for other general surgical purposes.
Fig. 7 discloses an alternate embodiment of a handpiece
.
132~g6
- 16 -
40 for laser surgery, having, as in the handpiece 20, an
outer tube 24 rotatably supported by an inner tube 26, with
a terminal portion 28 having at the distal end thereof a
lens 29 preceded by lens 27. An optical wedge 41 is
rotatably supported in the tube 24, and caused to rotate
or oscillate by any suitable apparatus. There is shown, by
way of example, a miniature motor 42 which drives through
gears 43a and 43b a shaft 44, on the lower end of which is
a gear 45. As shown in Fig. 7a, the optical wedge 41 may
be supported in an annular holder 46, having an annular
gear 47 thereon which is in mesh with the drive gear 45
through an opening 49 in tube 24. The carrier 46 is
rotatably supported by ball bearings 48 which engage the
interior wall of the tube 24.
Referring to Fig. 8, there is shown the laser beam LB
refracted by the plano-convex sapphire objective lens 29,
to focus the laser beam LB at the focus spot FS which is
within the cornea C. Due to the oscillatory or rotational
movement of the optical wedge 41, the focus spot FS wil1
move relative to the axis of handpiece 40 while the
handpiece 40 is held stationary. Thus, the focus spot FS
will move in an arc of a circle, or in a complete circular
path, due to the oscillatory or rotary movement of optical
wedge 41. Motion of the focus spot FS at the target site
over loci in very close proximity causes a marked reduction
of the plasma induction threshold; this facilitates the
-` 132~
- 17 -
cutting action and increases the energy localization many-
fold. This reduction of the plasma induction threshold is
caused by the interception of migrating plasma-induced
ions. The handpiece 40 may also be used in the dental
field, where the finely focussed and moving focus spot FS
enables very hard tooth enamel to be cut without either
charring the enamel or heat build-up. In either surgery or
dental applications, the oscillatory movement of the focus
Y spot FS so as to reach progressively further loci along the
arc is an effective method for facilitating the cutting
action.
Fig. 9 is a view of an alternate terminal portion for a
- handpiece such as the handpiece 20, there being shown the
- outer tube 24 having a plano-convex lens 27 sealed in the
distal end thereof. Objective lens 27 has a 30 mm focal
; length, and is of 4 mm diameter, in a preferred embodiment,
providing a spot size of about ten to thirty microns. A
terminal portion 50 of the handpiece has an enlarged
cylinder 51 into which the end part of the outer tube 24
extends, cylinder 51 at its upper end being hermetically
secured to the outer surface of outer tube 24. An inlet
conduit 52 is connected to the cylinder 51 for the
introduction of a fluid. Terminal portion 50 below the
cylinder 51 has a tapered connecting portion 53 and below
that is a conical portion 54. The distal end of tapered
portion 54, as shown in Fig. 10, is provided with radially
,, , , . _
'~;
'~ . - ' , ':
132~486
- 18 -
extending channels 56 to enable the escape of fluid from
within the terminal portion 50.
In use, the laser beam LB is delivered form sources
described below. Normal saline solution is introduced into
the terminal portion 50 through the inlet conduit 52, and
substantially fills it, as shown. The radiation, after
passing through the lens 27, is focussed through the column
of normal saline solution. The distal end of the terminal
' portion 50 is engaged with the outer surface of the cornea
C, and saline solution is caused to flow into terminal
portion 50, across the exposure site and out through the
channels 56. The escape of the saline solution aids in the
removal of debris from the exposure site, preserving the
optical clarity and homogeneity. The apparatus of Figs. 9
and 10 is particularly useful where a cutting is required,
such as the making of radial keratotomy cuts for the
purpose of effecting correction of myopia and myopic
astigmatism, although the apparatus may be used in general
surgical practice. The cutting action results from the
r 20 photoablative property of the excimer laser pulses from
source S-4, provided by excimer laser 55: these pulses have
a wavelength of 248 nanometers for a krypton fluoride laser
55, 193 nanometers for an argon fluoride laser 55, and 308
nanometers for an Xenon chloride laser 55. As will be
understood, these lasers provide far UV excimer
laser radiation in the TEM(oo) mode. Alternatively, a
_ _ _
.
, ~ ~
1~2~86
-- 19 --
source S-5 may be utilized, comprising a Q-switched/mode-
locked Neodymium YAG laser 56, which generates, as above
noted, a laser beam having a wavelength of 1064 nanometers.
This is delivered to a second harmonic crystal 57, which
doubles the radiation to 532 nanometers: the crystal 57 is
preferably potassium titanium phosphate. The radiation
from the crystal 57 is delivered to a second crystal 58,
where it is doubled, the crystal 58 being a fourth harmonic
crystal, and may be beta barium borate. The resulting
ultraviolet wavelength is at 266 nanometers. The
radiation from the source S-5 will be recognized as being
TEM(oo) ultraviolet radiation which has been up-converted
by successive doubling caused by the second and fourth
harmonic crystals 57 and 58, respectively.
There is shown in Fig. 11 a terminal portion 60 which
includes a tapered portion 64, shown filled with a liquid
such as a normal saline solution, and having a laser beam
LB passing axially thereof, focussing beyond the distal end
and in a cornea C. The lower end of the tapered portion 64
is inserted into a socket formed by sleeve 66 (see also
Fig. 12) of a glass contact tip 65. The sleeve 66 extends
upwardly from a solid internally reflective conical glass
element 67 having internal reflection at surface 68. The
distal end of the conical element 67 is provided, as shown
in Fig. 13, with channels 69 for the escape of the saline
solution. Between the channels 69 are contact surfaces 61
:
: ~ .
-- 1328~81~
- 20 -
which are transverse of the axis of the contact tip 65 and
are polished optically clear, as is the cylindrical surface
of conical element 67. A single multimods fiber 71
adjacent tube 64 receives illumination from a light source
72, the light being transmitted into the glass contact tip
65 and illuminating the penetration point P of the ablative
laser radiation provlded by laser beam LB. The image of
the penetration point P is transmitted to the reflecting
surface 68, and thence through the coupling lens 62 to a
fine coherent fiberoptic bundle 63. The fiberoptic bundle
63 conveys the image to any receiver R desired, such as to
monitoring and display elements and penetration control
components (not shown). Since the inner core or passage 73
- of the contact tip 65 is filled with fluid, such as the
normal saline solution, index matching occurs at the
interfaces and an image of the penetration point P is
readily obtained by the contact tip 65, coupling lens 62
and fiberoptic bundle 63.
In Figs. 14-16, there is shown an embodiment of a
terminal portion 80 forming a probe useful for conducting
~ ophthalmic intraocular, in particular, cataract surgery.
:- As will be understood, the terminal portion 80 replaces the
terminal portion of the handpiece 20 beyond the lens 27.
There are provided slightly diverging lens 81 which
receives the laser beam LB, the spreading beam then passing
into a quartz rod 82 which is cylindrical, and has a
. . ~ . .
- ,
1328~8~
- 21 -
beveled distal end surface 83. The quarts rod 82 conveys
the laser radiation by means of total internal reflection
to the beveled end surface 83, where the radiation leaves
the instrument and is sharply defined into a circular or
semi-circular radiation distribution area A (shown rotated
for clarity). The quartz rod 82 is housed within a
generally elliptical tube 84 which is preferably a tight
sealing rubber silicon sheath having an inlet conduit 85
connection to an internal passage 86 lying between the wall
of the tube 84 and the quartz rod 82, the outlet end of
passage 86 having a shape defined by two curved walls, as
shown in Fig. 15. Preferably, a discharge tube 87 is
inserted part way into the tube 84 from the distal end
thereof, to define the discharge port of the passage 86.
~pposite the passage 86, there is an aspiration passage 88
having an aspiration conduit 89 connected to its end
opposite the distal end, there being provided at the distal
end of aspiration passage 88 a tubular extension 91. As
shown in Fig. 15, the ends of the discharge tube 87 and the
tubular extension 91 are beveled, and lie in the same plane
as the beveled end surface 83 of quartz rod 82. The
relative locations of the passage 86, beveled end surface
83 and the entry port of aspiration passage 88 are shown in
Fig. 16.
The beveled end surface 83 causes the radiation
distribution area A to lie displaced from the geometric
---` 132~6
axis of quartz rod 82 and at or towards the geometric axis
of the passage 88. Tissue fragments TF which are drawn
towards the aspiration passage 88 are subjected to the
energy in radiation distribution area A, and are cut by the
ablative radiation. Saline solution which enters the
passage 86 is discharged adjacent to and flows across the
beveled end surface 83 toward the entry port of aspiration
passage 88, to remove debris.
Fig. 17 discloses an alternate embodiment of probe, in
which the silicon tube or sheath 94 has the quartz rod 82
in it, with, as shown in Fig. 18, the longitudinal axis of
the cylindrical quartz rod 82 and the major longitudinal
axis of elliptical sheath 94 coinciding. There are thereby
provided an infusion passage 96, and an aspiration passage
98 in the equal-area spaces between the quartz rod 82 and
sheath 94. The sheath 94 has the distal end thereof
beveled, as shown in Fig. 17, and the end face 83 of quartz
rod 82 lies in the plane of the beveled end of sheath 94.
The longer end portion 97 of sheath 94 which forms the
entrance to the aspiration passage 98 may have other shapes
than as shown in Fig. 17, i.e. it may be angled as shown in
Fig. 19.
Fig. 19 shows the application of the tips of Figs. 14
and 17 to cataract surgery and to vitreal strand removal or
vitrectomy, respectively. For cataract surgery, there is
inserted through the cornea C and into the cataractous lens
... .
- , .
: , . ,,, - .
.
1328~86
- 23 -
CL the terminal portion 80 of a handpiece. Photoablative
` laser energy, as above described, is delivered to the
cataractous lens CL, to cause its disintegration, and the
ablated lens is withdrawn through the aspiration conduit
89. Because of the localized character of the
photoablative laser energy delivered to the lens, there are
avoided such undesirable effects as the possibility of
burning of the cornea and the possible shattering effect of
.- the nucleus of the corneal endothelium resulting in corneal
endothelial decomposition, hazards present in the currently
used techniques of cataract extraction.
For vitrectomy, the terminal portion 95 of the probe
shown in Figs. 17 and 18 is inserted through the pars plana
and is used to cut vitreal strands VS. Preferably, the end
97 of the sheath 94 is bent at an angle, as shown in
Fig. 19, so as to enhance the lateral flow of vitreous
strands across the radiation distribution area A of the
cutting ablative radiation source which is emitted from the
end surface of the quartz rod 82.
20- Tt will be obvious to those skilled in the art that
various changes may be made in the herein disclosed method
and apparatus. However, the invention is not limited to
what is set forth in the specification or described in the
drawings, but only as defined in the claims appended
hereto.
,~