Note: Descriptions are shown in the official language in which they were submitted.
CONTROLLED SYMPATHECTOMY AND MICRO-ABLATION SYSTEMS
AND METHODS
[00011
BACKGROUND
Technical Field
[0002] The present disclosure relates to the field of minimally invasive
sympathectomy. The disclosure relates to methods for locating, monitoring,
and/or
mapping nerve distributions before, during, and/or following an ablation
process facilitated
by way of catheterization procedures. The disclosure relates to systems and
methods for
monitoring the extent of an ablation process as it pertains to a surgical
goal, such as
denervation. The disclosure also relates to catheter systems specifically
designed for use
in vascular nerve monitoring and ablation.
Background
[0003] Congestive heart failure, hypertension, diabetes, and chronic renal
failure have
many different initial causes; however, all may include some form of renal
sympathetic
nerve hyperactivity. Renal sympathetic nerves communicate signals with
sympathetic
centers located in the spinal cord and brain via afferent renal nerve
activity, increasing
systemic sympathetic tone; meanwhile, through efferent activity, renal nerves
and arteries
participate in sympathetic hyperactivity in response to signals from the
brain, further
increasing systemic sympathetic tone.
- 1 -
CA 2862862 2019-04-17
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
[0004] Sympathetic activation can initially be beneficial but eventually
becomes
maladaptive. In a state of sympathetic hyperactivity, a number of pathological
events take
place: abnormalities of hormonal secretion such as increased catecholamine,
renine and
angiotensin II levels, increased blood pressure due to peripheral vascular
constriction
and/or water and sodium retention, renal failure due to impaired glomerular
filtration and
nephron loss, cardiac dysfunction and heart failure due to left ventricular
hypertrophy and
myocyte loss, stroke, and even diabetes. Therefore, modulation
(reduction/removal) of
this increased sympathetic activity can slow or prevent the progression of
these diseases.
[0005] Although ablation of such nerves can have positive effects on drug
resistant
hypertension and glucose metabolism abnormality current methodologies for
denervation
(e.g. ablation) are conducted without adequate feedback (with respect to the
site of a
denervation event, the extent of denervation, the effect of denervation on
local
physiology, etc.).
SUMMARY
[0006] One objective of this disclosure is to provide a microsurgical tool
for
monitoring, evaluating, mapping, and/or modulating electrophysiological
activity in the
vicinity of a lumen within a body. Another objective is to provide a system
and method
for evaluating the sympathetic tone of a subject. Yet another objective is to
provide a
system for neuromodulating an anatomical site in the vicinity of a lumen
within a body.
[0007] The above objectives are wholly or partially met by devices,
systems, and
methods according to the appended claims in accordance with the present
disclosure.
Features and aspects are set forth in the appended claims, in the following
description,
and in the annexed drawings in accordance with the present disclosure.
[0008] According to a first aspect there is provided, a microsurgical tool
for
monitoring electrophysiological activity within the vicinity of a lumen, the
microsurgical
tool including a microfinger in accordance with the present disclosure having
a
substantially elongate structure configured so as to bias a region thereof
against a wall of
the lumen upon deployment within the lumen, and a sensing tip in accordance
with the
present disclosure electrically and mechanically coupled to the microfinger in
the vicinity
-2-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
of the region, configured to interface with the wall of the lumen, the sensing
tip
configured to convey one or more electrophysiological signals associated with
the
activity.
[0009] In aspects, one or more of the electrophysiological signals may be
related to
one or more of water concentration, tone, evoked potential, remote stimulation
of nervous
activity, an electromyographic signal [EMG], a mechanomyo graphic signal
[MMG], a
local field potential, an electroacoustic event, vasodialation, vessel wall
stiffness, muscle
sympathetic nerve activity (MSNA), central sympathetic drive (e.g. bursts per
minute,
bursts per heartbeat, etc.), tissue tone, nerve traffic (e.g. post ganglionic
nerve traffic in
the peroneal nerve, celiac ganglion, superior mesenteric ganglion,
aorticorenal ganglion,
renal ganglion, and/or related nervous system structures), combinations
thereof, or the
like.
[0010] In aspects, one or more of the sensing tips may include one or more
electrodes, a needle electrode, a force sensor, mechanomyographic (MMG)
sensing
element, a strain sensor, a compliance sensor, a temperature sensor,
combinations thereof,
or the like each in accordance with the present disclosure. In aspects, one or
more
sensing tips may be electrically coupled with a microcircuit, the microcircuit
configured
to condition the signal.
[0011] In aspects, one or more of the microfingers may be configured so as
to
substantially embed the sensing tip into the wall of the lumen, to
substantially maintain
contact with the wall of the lumen while it is swept longitudinally down the
lumen and/or
circumferentially around the lumen, to substantially maintain a constant force
against the
wall of the lumen during relative movement there between, to substantially
electrically
isolate the sensing tip from a cavity of the lumen, to plunge the electrode
(particularly a
needle electrode) into the wall of the lumen upon deployment, combinations
thereof, and
the like.
[0012] In aspects, the microfinger may include an active material element
in
accordance with the present disclosure, configured to alter the contact force
between the
region or the sensing tip, and the wall upon receipt of a control signal.
-3-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[0013] In
aspects, the microfinger may be configured so as to be deployed from a
delivery catheter. In aspects, the delivery catheter may have a diameter less
than 3mm,
less than 2mm, less than lmm. In aspects, at least a portion of the delivery
catheter may
have a diameter of less than 0.75mm, less than 0.5mm, less than 0.25mm so as
to access a
miniature lumen within a body.
[0014] In
aspects, the microfinger may have a characteristic width of less than
150am, less than 100um, less than 75um, less than 50um, less than 25um, less
than
10um, less than Sum.
[0015] In
aspects, a plurality of microfingers may be configured to form a cage, a
mesh, or a stent-like structure, to independently maintain contact with the
wall during
relative movement there between, combinations thereof, or the like.
[0016] According
to aspects there is provided use of a microsurgical tool in
accordance with the present disclosure to monitor electrophysiological
activity in the
vicinity of a vessel, an artery, a vein, a renal artery, similar structures,
or the like.
[0017] According
to aspects there is provided use of a microsurgical tool in
accordance with the present disclosure to perform a surgical procedure.
[0018] According
to aspects there is provided, a system for neuromodulating an
anatomical site in the vicinity of a lumen, including a subsystem configured
to pedal in a
surgical procedure on the anatomical site, a microsurgical tool in accordance
with the
present disclosure, configured to monitor electrophysiological activity in the
vicinity of
the site; and a control unit configured to accept signals from the
microsurgical tool, and
to adjust the surgical procedure dependent upon the signals, to display the
signals, to
evaluate the surgical procedure dependent upon the signals, to plan a surgical
path
dependent upon the signal, to deteimine the extent of the procedure dependent
upon the
signals, combinations thereof, or the like.
[0019] In
aspects, the surgical procedure may include an ablation, an excision, a cut,
a burn, a radio frequency ablation, radiosurgery, an ultrasonic ablation, an
abrasion, a
biopsy, delivery of a substance, combinations thereof, or the like.
-4-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
[0020] In aspects, the system may include a stimulation and/or ablation
electrode
configured so as to convey a pulsatile and/or radio frequency signal to the
anatomical site
from the control unit, the microsurgical tool configured to convey one or more
feedback
signals related to the pulsatile and/or radio frequency signals back to the
control unit. In
aspects, the feedback signals may be related to an electrode impedance, a
bioimpedance,
a local electrical field, or an electrophysiological response to the pulsatile
and/or radio
frequency signal.
[0021] In aspects, the stimulation and/or ablation electrode may be
included within
the microsurgical tool, coupled to a microfmger, included in a sensing tip, or
the like.
[0022] In aspects, the control unit may be configured to sweep one or more
of the
sensing tips along the lumen wall, to use one or more of the
electrophysiological signals
to locate the anatomical site, to use one or more of the electrophysiological
signals to
exclude the anatomical site from a surgical procedure, combinations thereof,
or the like.
[0023] According to aspects there is provided, a method for determining an
afferent
electrophysiological activity and an efferent physiological activity in the
vicinity of a
lumen, including monitoring electrophysiological activity at a plurality of
sites within the
vicinity of the lumen in regions proximal and distal to a target region as
measured along a
length of the lumen, applying energy to a site within the target region to
form a
neurological block thereby, and extracting an afferent signal from activity in
the distal
region and an efferent signal from activity in the proximal region.
[0024] In aspects, the method may include comparing activity measured in
the
proximal region and the distal region to determine if the energy application
affected the
electrophysiological activity in the vicinity of the target region. In
aspects, the method
may include evaluating the coherence between activities measured in the
proximal region
and the distal region and/or using the coherence to evaluate the extent of the
neural block.
[0025] In aspects, the application of energy may be sufficient to form a
temporary
neuroblock (i.e. just sufficient to form a temporary block, controlled so as
to form a
temporary block, etc.). In aspects, the method may include comparing
activities from the
proximal region and the distal region during the temporary neuroblock and
diagnosing a
-5-.
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
neurological condition, evaluating a neurological state, deteimining if a
permanent
surgical procedure is required, combinations thereof, or the like.
[0026] According to aspects there is provided, a method for evaluating
sympathetic
tone of a subject, including inserting a microsurgical tool in accordance with
the present
disclosure into a lumen of the subject, recording the electrophysiological
signals
conveyed by the microsurgical tool from a wall of the lumen, removing the
microsurgical
tool from the lumen, and generating a metric relating to sympathetic tone from
the
recorded signals.
[0027] In aspects, the method may include monitoring another physiological
parameter remotely from the lumen to generate a corrective signal and using
the
corrective signal to remove movement artifacts from the cicctrophysiological
signals.
[0028] In aspects, the method may include stimulating one or more
anatomical sites
in the subject during the recording, and/or diagnosing a medical condition
based at least
in part upon the metric.
[0029] According to aspects there is provided, a method for monitoring
and/or
evaluating electrophysiological activity in the vicinity of a lumen, including
biasing an
electrode against a wall of the lumen; and recording one or more
electrophysiological
signals from the activity in the vicinity of the electrode.
[0030] In aspects, the method may include recording one or more of an
evoked
potential, remote stimulation of nervous activity, an electromyo graphic
signal [EMG], a
mechanomyographic signal [MMG], a local field potential, an electroacoustic
event,
vasodialation, vessel wall stiffness, muscle sympathetic nerve activity
(MSNA), central
sympathetic drive (e.g. bursts per minute, bursts per heartbeat, etc.), tissue
tone, nerve
traffic (e.g. post ganglionic nerve traffic in the peroneal nerve, celiac
ganglion, superior
mesenteric ganglion, aorticorenal ganglion, renal ganglion, and/or related
nervous system
structures) in the vicinity of the lumen.
[0031] The method may include electrically isolating the electrode from a
cavity of
the lumen, embedding the electrode into the wall of the lumen, sweeping the
electrode
along the wall of the lumen, generating a map of electrophysiological activity
from the
-6-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
recordings obtained during the sweep, recording electrophysiological activity
from a
plurality of electrodes, cancelling one or more movement artifacts from the
recordings,
combinations thereof, or the like.
[0032] In aspects, the method may include biasing a mechanomyographic (MMG)
sensing element against the wall of the lumen and recording a
mechanomyographic signal
(MMG) from the activity.
[0033] According to aspects there is provided, a method for perfoiming
controlled
neuromodulation in the vicinity of a lumen, including monitoring
electrophysiological
activity at one or more sites within the vicinity of the lumen to obtain a
first activity level,
applying energy to a treatment site within the vicinity of the lumen,
monitoring
electrophysiological activity at one or more sites within the vicinity of the
lumen to
obtain a second activity level, and comparing the first activity level and the
second
activity level to determine if the energy application affected the
electrophysiological
activity, if sufficient energy was applied, if further energy should be
applied,
combinations thereof, and the like.
[0034] In aspects, the electrophysiological activity may relate to one or
more of an
evoked potential, remote stimulation of nervous activity, an electromyographic
signal
[EMG], a mechanomyographic signal [MMG], a local field potential, an
electroacoustic
event, vasodialation, vessel wall stiffness, muscle sympathetic nerve activity
(MSNA),
central sympathetic drive (e.g. bursts per minute, bursts per heartbeat,
etc.), tissue tone,
nerve traffic (e.g. post ganglionic nerve traffic in the peroneal nerve,
celiac ganglion,
superior mesenteric ganglion, aorticorenal ganglion, renal ganglion, and/or
related
nervous system structures) as measured in the vicinity of the lumen.
[0035] In aspects, the method may include deteimining if sufficient energy
has been
applied to the treatment site based on the comparison, evaluating the first
activity level to
determine a suitable treatment site in the vicinity of the lumen, mapping
electrophysiological activity in the vicinity of the lumen using the first
activity level,
applying a stimulus in the vicinity of the lumen, recording
electrophysiological activity
before, during, and/or after the stimulus, or the like.
-7-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[0036] In aspects, the method may include recording electrophysiological
activity in
a proximal region and a distal region measured along the length of the lumen
as spaced
with respect to the treatment site, to determine if the energy application
affected the
electrophysiological activity in the vicinity of the treatment site,
determining if the
energy application was sufficient to foal' a neural block using the
comparison, applying
sufficient energy to the treatment site to faun a temporary block and
assessing if the
change in electrophysiological activity is desirable, if so, applying
sufficient energy to the
treatment site so as to form a substantially irreversible block, or the like.
[0037] In aspects, the energy may be provided in the form of a radio
frequency
current, an ultrasonic wave, theimal energy, a neuroblocking agent, radiation,
electromagnetic radiation, radiosurgically generated radiation, combinations
thereof, or
the like.
[0038] In aspects, one or more of the steps of a method in accordance with
the
present disclosure may be performed using a surgical tool in accordance with
the present
disclosure.
[0039] According to aspects there is provided a method for determining a
state of a
neurological connection along a neurological pathway between one or more
regions in a
body, including applying a pacing signal to a lumen in the vicinity of the
neurological
pathway, monitoring one or more of water concentration, tone, blood oxygen
saturation
of local tissues, evoked potential, stimulation/sensing of nervous activity,
electromyography, temperature, blood pressure, vasodialation, vessel wall
stiffness,
muscle sympathetic nerve activity (MSNA), central sympathetic drive (e.g.
bursts per
minute, bursts per heartbeat, etc.), tissue tone, blood flow (e.g. through an
artery, through
a renal artery), a blood flow differential signal (e.g. a significantly
abnormal and or
sudden change in blood flow within a structure of the body, a vessel, an
organ, etc.),
blood perfusion (e.g. to an organ, an eye, etc.), a blood analyte level (e.g.
a hormone
concentration, norepinephrine, catecholamine, renine, angiotensin H, an ion
concentration, a water level, an oxygen level, etc.), nerve traffic (e.g. post
ganglionic
nerve traffic in the peroneal nerve, celiac ganglion, superior mesenteric
ganglion,
aorticorenal ganglion, renal ganglion, and/or related nervous system
structures), or
-8-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
combinations thereof, or the like at one or more sites within the body to
generate one or
more physiological signals; and evaluating the influence of the pacing signal
on the
physiological signals and determining the state of neurological connection
therefrom.
[0040] In
aspects, the method may include applying energy in the vicinity of the
lumen so as to induce a neurological block along the neurological pathway,
pacing and
monitoring before and after induction of the neurological block, and/or
comparing the
physiological signals obtained before the neurological block to those obtained
during the
neurological block to determine the influence of the neurological block there
upon,
combinations thereof, and the like.
[0041] In
aspects, the method may include determining if the neurological block is
favorable in terms of treating an underlying disease state in the body, and/or
applying
energy in the vicinity of the lumen so as to induce a substantially permanent
neurological
block along the neurological pathway.
[0042] In
aspects, the method may include monitoring electrophysiological activity at
a plurality of sites within the vicinity of the lumen in regions proximal and
distal to the
pacing site and/or to the site of a suspected or known neurological block.
[0043] In
aspects, the method may include extracting an afferent signal from activity
in the distal region and an efferent signal from activity in the proximal
region and/or
comparing activity measured in the proximal region and the distal region to
determine if
the energy application affected the electrophysiological activity in the
vicinity of the
target region.
[0044] According
to aspects there is provided, use of a method in accordance with the
present disclosure for evaluation of the effectiveness of a neuromodulation
procedure
within a body.
BRIEF DESCRIPTION OF THE DRAWINGS
[0045] Several
aspects of the disclosure can be better understood with reference to
the following drawings. In the
drawings, like reference numerals designate
corresponding parts throughout the several views.
-9-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[0046] Figs. la-c show aspects of a surgical tool tip in accordance with
the present
disclosure in a delivery mode and a deployed mode.
[0047] Figs. 2a-c show aspects of deployed surgical tips in accordance with
the
present disclosure interacting with a local surgical site.
[0048] Fig. 3 shows aspects of a plurality of micro-tips configured for
monitoring
physiological response and/or stimulating local tissue during a surgical
procedure in
accordance with the present disclosure.
[0049] Figs. 4a-b show aspects of interactions between multiple micro-tips
and a
lumen wall in accordance with the present disclosure.
[0050] Figs. 5a-c show aspects of micro-tips in accordance with the present
disclosure.
[0051] Figs. 6a-b show aspects of a microfinger in accordance with the
present
disclosure.
[0052] Figs. 7a-b show aspects of a micro-tip including a mechanomyographic
(MMG) sensing element and a typical response in accordance with the present
disclosure.
[0053] Figs. 8a-b show aspects of a micro-tip in accordance with the
present
disclosure.
[0054] Fig. 9 shows aspects of a micro surgical tool deployed at a surgical
site in
accordance with the present disclosure.
[0055] Figs. 10a-d show aspects of non-limiting examples of monitoring
methods in
accordance with the present disclosure.
[0056] Figs. 1 1 a-g show aspects of non-limiting examples of ablation
patterns
applied to a lumen wall (i.e. an artery, a renal artery, etc.) in accordance
with the present
disclosure.
[0057] Figs. 12a-d show aspects of micro surgical tools in accordance with
the
present disclosure deployed at a surgical site.
-10-
CA 02862862 2014-07-25
WO 2013/112844 PCT[US2013/023157
[0058] Fig. 13 shows a schematic diagram of interaction between one or more
macroelectrodes and a micro surgical tool deployed at a surgical site in
accordance with
the present disclosure.
[0059] Fig. 14 shows aspects of a micro balloon catheter deployed at a
surgical site in
accordance with the present disclosure.
[0060] Figs 15a-b show aspects of an array of optical microsensing tips and
a
collective response therefrom in accordance with the present disclosure.
[0061] Fig. 16 shows aspects of a combination catheterization and
endoscopic
procedure on a renal artery in accordance with the present disclosure.
[0062] Figs. 17a-c show aspects of micro surgical tools in accordance with
the
present disclosure.
[0063] Figs. 18a-f show aspects of non-limiting examples of micro surgical
tools in
accordance with the present disclosure.
[0064] Figs. 19a-b show aspects of a tonal sensing micro-tip and sample
response in
accordance with the present disclosure.
[0065] Figs. 20a-b show aspects of surgical tools in accordance with the
present
disclosure.
[0066] Figs. 21 shows aspects of a system for performing a surgical
procedure in
accordance with the present disclosure.
DETAILED DESCRIPTION
[0067] Particular embodiments of the present disclosure are described
hereinbelow
with reference to the accompanying drawings; however, the disclosed
embodiments are
merely examples of the disclosure and may be embodied in various forms.
Therefore,
specific structural and functional details disclosed herein are not to be
interpreted as
limiting, but merely as a basis for the claims and as a representative basis
for teaching
one skilled in the art to variously employ the present disclosure in virtually
any
appropriately detailed structure. Like reference numerals may refer to similar
or identical
elements throughout the description of the figures.
-11-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[0068] A controlled nerve ablation system may include the capability to
sense one or
more physiological parameters at one or more points around a surgical site,
and/or
include the capability to stimulate and/or ablate tissues at one or more of
the same points
and/or an alternative point around a surgical site. In aspects, the nerve
ablation system
may be configured so as to access a lumen, a vessel, very narrow vessels,
and/or surgical
sites in the body. The non-limiting examples disclosed herein are directed
towards such
configurations (e.g. so as to controllably ablate renal nerves along a renal
artery with a
catheterized procedure).
[0069] By lumen is meant a substantially hollow structure, with one or more
walls,
enclosing a cavity. In the context of the present disclosure, a lumen is
generally
considered elongate in shape, having a longitudinal direction running along
the length
thereof, a radial direction running substantially perpendicularly to a wall of
the lumen,
and a circumferential direction running substantially Perpendicular to the
longitudinal
direction along a wall of the lumen. In aspects, a lumen may include a branch
(a
bifurication), a bend, a tortuous pathway, a changing diameter (i.e. a
diameter that
changes along the length thereof), and the like. It is envisaged that a system
in
accordance with the present disclosure may be apt at navigating such
complicated
features, thus providing therapy to a range of challenging to reach locations.
[0070] The nerve ablation system may include one or more sensing tips (e.g.
as
located on a micro-tip, a wire, an electrode in a matrix, on a flexible
balloon, etc.). One
or more sensing tips may include a pressure sensor, a tonal sensor, a
temperature sensor,
an electrode (e.g. to interact with a local tissue site, provide a stimulus
thereto, measure a
potential therefrom, monitor current to/from the tissues, to measure a
bioimpedance,
measure an evoked potential, an electromyographic signal [EMG], an
electrocardiographic signal [ECG], a mechanomyographic signal [MMG], a local
field
potential, etc.), an acoustic sensor, an oxygen saturation sensor, or the
like.
[0071] The sensing tips may be configured to elucidate a range of key
physiological
aspects before, during, and/or after a procedure. The following description
outlines some
non-limiting approaches in this respect. Such sensing tips may be integrated
into one or
more microfingers, micro-tips, flexible circuits, stretchable substrates, etc.
-12-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[0072] In aspects, one or more sensing tips in accordance with the present
disclosure
may be configured to monitor bioimpedance between one or more sensing tips to
determine the degree of contact between the finger tips and the anatomical
site, and/or
potentially the bias force between the finger tips and the anatomical site.
Additionally,
alternatively, or in combination, bioimpedance measurements between one or
more
sensing tips may be useful in determining when adequate contact has been made
as well
as how much current should be applied to an anatomical site during an ablation
procedure. Furthermore, additionally, alternatively, or in combination
bioimpedance
between one or more sensing tips may be used to determine the status of tissue
positioned
there between. In one non-limiting example, the bioimpedance spectrum between
two or
more sensing tips may be used to map the local tissue impedance. Such
information may
be useful to elucidate where such tissue has been completely ablated, where
tissue has yet
to be ablated, etc.
[0073] In aspects, bioimpedance measurement between on or more sensing
tips, a
sensing tip and a separate electrode, etc. may be used to determine a state of
isolation
between one or more of the sensing tips and a local fluid (i.e. to determine a
state of
isolation between a sensing tip and fluid within a lumen, between a sensing
tip and blood,
etc.).
[0074] In aspects, one or more sensing tips in accordance with the present
disclosure
may be configured to obtain mechanomyographic information during a procedure
as
determined by slight changes in an associated strain measurement, tip
vibration, and/or
contact force measurement (e.g. via direct force measurement between the tip
and the
local anatomy, and/or via changes in the deformation of the microfinger as
measured by
an associated micro strain gage attached thereupon). Mechanomyographic
infoimation
may be related to local nervous activity either naturally occurring or in
response to a
stimulus (e.g. optionally applied by one or more sensory tips, locally,
remotely, during
and/or via a local RF pulse, etc.). In aspects, a sensing tip may include a
piezoresistive
strain gauge, a piezoelectric microtransducer, an interfacial pressure sensing
membrane
or the like to detect mechanomyographic signals. In one non-limiting example,
the
sensing tip may be coated with a micro or nano coating of a piezoresistive and
or
piezoelectric material (e.g. a piezoelectric polymer, an electret, a nano-
particulate filled
-13-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
elastomer, a conjugated polymer, etc.). In aspects, the mechanomyographic tip
may be
configured so as to measure one or more aspect of the tissue compliance of the
local
tissues (e.g. so as to identify calcified material, cancerous tissues, etc.).
[0075] In aspects, one or more sensing tips in accordance with the present
disclosure
may be configured to monitor an electrophysiological signal. Such
electrophysiological
monitoring at and/or between one or more sensing tips, may be used to map
nervous
response, electromyographic response (EMG), evoked potential, local field
potential,
extracellular field potentials, etc. along and/or within the wall of the local
anatomical site
(e.g. the wall of a lumen, a vessel wall, an artery wall, a venous wall, an
organ wall, etc.).
Such information may be advantageous for selecting tissues on which to perform
a
surgical procedure (e.g. an ablation procedure, a biopsy, etc.), to follow
and/or map a
nerve along the length of the surgical site (e.g. along the wall of an artery,
a vein, a
tubule, etc.), to determine the state of a surgical procedure, etc. In
aspects, one or more
sensing tips may be configured to monitor a local electromyographic (EMG)
signal
before, during and/or after a surgical procedure as a means for monitoring
local nervous
activity. In such aspects, the EMG signals may be used as feedback for
monitoring the
extent of a denervation procedure.
[0076] In aspects, one or more sensing tips in accordance with the present
disclosure
may be configured to monitor the tone of a tissue within a body. Monitoring
the tone
(e.g. mechanical properties, wall stiffness, elastic spectral response,
mechanical
impedance, physiological properties, etc.) of the adjacent tissues may be
determined by
combining strain and/or force measurement of the sensing tips while applying
movement
(optionally cyclical or oscillatory movement) to one or more sensor tips. Such
sensing
tips may be excited locally (e.g. such as by a local piezoelectric transducer,
a capacitive
transducer, an electrochemical transducer, a smart material, etc.) or globally
(e.g. such as
by oscillatory torsional oscillations, axial oscillations, linear oscillations
of the surgical
tool tip, the associated guide wire, catheter, etc.).
[0077] In aspects, one or more of the sensing tips may be interfaced
asymmetrically
with the associated tissues (i.e. with a bent tip, a micro finger, a wire-like
finger
configured substantially parallel to the tissue surface, oriented at an acute
angle thereto,
-14-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
etc.). By asymmetrically is meant such that the sensing tip approaches the
associated
tissue surface at an angle other than perpendicular thereto. To describe the
use of such a
tip to monitor local tissue tone and/or for providing a controlled interfacial
force before,
during and/or after a procedure, for purposes of discussion, a clockwise
torsion may be
used to advance the sensing tip along the surface of the local tissues and a
relatively small
counterclockwise torsion may be used to measure the tone of adjacent tissues.
By
relatively small is meant an excitation that is sufficiently small in
amplitude such that the
sensing tip may not appreciably slide along the tissue surface. In aspects,
one or more
sensory tips, in a structure attached thereto, and/or a system in accordance
with the
present disclosure may include a vibratory exciter may be configured to
generate the
excitation.
[0078] In aspects, such a tone monitor may be combined with interfacial
contact
sensing, electrophysiological measurement, and/or sensor tip strain
measurement in order
to generate a wealth of local tissue information before, during, and/or after
a surgical
procedure. In one non-limiting example, the local tissues may stiffen during
an ablation
procedure. By monitoring local tissue tone, a stiffness level may be used to
characterize
when a suitable degree of ablation has been applied so as to irreversibly
damage the
tissues. Monitoring of a local tissue tone, perhaps at a monitoring site
significantly
removed from the surgical site such that the surgical procedure does not
directly affect
tissues in the vicinity of the monitoring site (i.e. does not directly cut,
heat, ablate,
abrade, the tissues, etc.) may also be advantageous for determining an effect
of the
surgical procedure on one or more physiological parameters of a tissue (e.g. a
vessel wall
stiffness, change in nerve activity, change in blood perfusion, etc.) adjacent
to the
monitoring site.
[0079] In aspects, such tone measurement may be useful in determining the
local
stiffness of tissues (and/or overall wall stiffness of an adjacent vessel,
organ, etc.) in
contact with a sensing tip array (e.g. so as to determine the type of tissue
adjacent to one
or more sensing tips, locate plaque, locate a cancerous tumor, etc.). Tone
measurement
may further be used to characterize the type of tissue with which the tip is
interfacing
(e.g. muscle, nervous tissue, fat, plaque, cancerous tissue, etc.). In
aspects, such
information, possibly in combination with bioimpedance data,
electrophysiological
-1 5-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
monitoring, or the like, may be used to detelmine how much RE energy to apply
locally
during an RE ablation procedure.
[0080] In one non-limiting example of a method for RF ablating tissue, the
local
tissue tone may be measured before, during, between individual RE pulses,
and/or after a
train of RE pulses. As the local tissue tone changes during application of the
RE pulses,
the tonal changes may be used to determine the extent of the therapy. As the
RE ablation
process is applied to the adjacent tissues (perhaps via one or more sensing
tips), the tonal
measurements (as determined by one or more sensing tips, perhaps the same tip
through
which the RE signal may be applied) may be monitored as the tonal measurements
may
not be significantly affected by the local RE currents.
[0081] In aspects, clectrophysiological stimulation and/or sensing from one
or more
sensing tips in a sensing tip array, or a system in accordance with the
present disclosure
may be used to interface with, monitor and/or stimulate nervous function
within a local
anatomical structure (e.g. a lumen wall, a vessel wall, along a nerve, an
organ wall, a
duct, etc.). Such information may be used to hunt for target tissues (e.g.
nerves), select
tissues for a surgical procedure, to determine the degree of progression of a
surgical
procedure (e.g. a degree of ablation during RE surgery, etc.).
[0082] In aspects, an array of sensing tips may be configured to apply a
directional
stimulation and/or multi-site sensing so as to selectively treat/monitor only
nerves that are
configured to send signals in the preferred direction (e.g. to selectively
target primarily
efferent nerve bundles, afferent nerve bundles, etc.). Such a configuration
may be
advantageous for treating a neurological disorder with minimal impact to the
surrounding
anatomy and physiological function of the associated organs.
[0083] In aspects, one or more sensing tips in accordance with the present
disclosure
may include the capability to apply/receive an RE current to/from the
surrounding tissue.
The RE current may be provided locally between two of more sensing tips, or
alternatively between one or more sensing tips and a macroelectrode placed
elsewhere on
the body (e.g. on a large skin patch over the surgical site, as selected from
multiple
patches placed over the body, etc.). In a non-limiting example where current
is restricted
to being applied between sensing tips, the path for current flow may be well
controlled,
-16-
CA 02862862 2014-07-25
WO 2013/112844 PCT[US2013/023157
yet may be highly localized. Alternatively, in an example where RF current is
passed
between one or more sensing tips and one or more macroelectrodes, the
direction of
current flow may be more challenging to control, but may be used to access
tissues more
remote from the sensing tips (i.e. farther into the adjacent tissues, deeper
into an organ,
farther from a lumen wall, etc.).
[0084] In aspects, network impedance measurements between one or more
sensing
tips and one or more macroelectrodes (e.g. as attached to the body of the
patient), may be
monitored prior to and/or during application of an RF ablation current. Each
sensing tip
and/or macroelectrode may include an impedance control circuit that may be
adjustable
such that the overall current flow through the network formed from all the
elements is
controlled there through. Such a configuration may be advantageous to more
precisely
control the local ablation process, thus targeting the local tissues with more
accuracy,
precision, spatial discrimination, and confidence than less controlled
approaches.
[0085] In another non-limiting example, a plurality of sensing tips may be
engaged
with the flow of RF current during an ablation process. In aspects, the local
impedance
of each microfinger and/or sensing tip may be monitored and/or controlled so
as to better
optimize the current delivered thereto. Additionally, alternatively, or in
combination, the
local current flow through each sensing tip may be monitored so as to
determine the path
of the RF current flow, to ensure no leakage currents are detected, etc. Such
information
may be used to more precisely control the delivery of RF currents to the local
anatomy
during an ablation procedure.
[0086] Additionally, alternatively, or in combination, before, during
and/or after the
RF current is applied to the surrounding tissues, one or more sensing tips may
monitor a
physiological parameter (e.g. water concentration, tone, blood oxygen
saturation of local
tissues, evoked potential, stimulation/sensing of nervous activity, local
field potential,
extracellular activity, EMG, temperature, etc.) to detelinine the extent of
completion of
the intended surgical procedure.
[0087] In aspects, one or more sensing tips may include an optical
microsensor (e.g. a
micropackage including a light source and/or a CMOS photosensor) and/or a
fiber optic
-17-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
element. During a surgical procedure, the optical microsensor may be
positioned against
or_near to the local tissues for analysis before, during and/or after an
ablation procedure.
[0088] In aspects, an optically configured sensing tip (or group of tips)
may be
configured to locally assess blood perfusion and/or blood oxygenation in the
tissues
adjacent thereto. The system may be configured to automatically adjust and/or
halt the
surgical procedure based upon changes in this signal. Alternatively,
additionally, or in
combination, the system may alert a user (e.g. a surgeon, an attendant, etc.)
to a change in
this signal before, during, and/or after a surgical procedure. Such a
configuration may be
useful for assessing local tissue health before, during, and/or after a
surgical procedure,
the extent of a surgical procedure, etc.
[0089] In another non-limiting example, one or more optically configured
sensing
tips may be configured so as to be biased towards the tissues of a lumen, a
vessel, or the
like in the vicinity of the surgical site. The optical sensing tips may
include one or more
light sources (e.g. light emitting diodes, fiber optic tips, etc.) configured
to deliver
narrow, multiband, and/or wideband light to the adjacent tissues. In aspects,
one or more
of the optical sensing tips may include one or more photodetectors (e.g. a
photodetector,
a phototransistor, a fiber optic tip, etc.) to receive and/or analyze the
light reflected from
the adjacent tissues. The received light may be related to that emitted by one
or more of
the light sources, or may be received from an ambient light source, perhaps
located to the
exterior of the vessel, or the exterior of the subject's body.
[0090] The sources may be configured to emit light at predetermined
wavelengths
such that different absorption characteristics of the adjacent tissues,
perhaps dependent on
the wavelengths, may be observed during the surgical procedure. The
photodetectors
may be configured to receive at least a portion of this light, so as to assess
the absorption
characteristics with the system (perhaps via a pre-amplification system in
accordance
with the present disclosure, in an attached electronics unit, etc.). The
photodetected
signals may be used to determine an oximetry value or a signal related
thereto.
[0091] In one non-limiting example, the optically configured sensing tips
may be
biased towards a site on the vessel wall before, during, and/or after the
surgical
procedure. Alternatively or in combination, the optically configured sensing
tips may be
-18-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
substantially stationary with respect to the vessel wall (such as via being
attached to a
collar of known size, attached to a structure of known width, as part of a
structure that is
expanded to a known radius, etc.). In aspects, the magnitude of the bias may
be
controlled by sensors and actuators both accordance with the present
disclosure. Changes
in the optical signals detected by the photodetectors (perhaps due to changing
bias force)
before, during and/or after a surgical procedure may be related to changes in
the bias
force with which they are held against the vessel wall. Such a configuration
may be
advantageous for determining a change in sympathetic tone and/or vasodialation
before,
during and/or after a surgical procedure.
[0092] In one non-limiting example, the optically configured sensing tips
may be
coupled with one or more strain and/or interfacial force measurement methods,
perhaps
to give a more precise reading of the bias force between the sensing tip(s)
and the
adjacent tissues, to compensate for movement related artifacts, or the like.
[0093] In aspects, one or more of the optical sources may be selected such
that the
penetration of the light into the adjacent tissues may be controlled. In one
non-limiting
example, a blue wavelength and a red wavelength may be emitted into the
tissues. The
blue wavelength may provide information relating to the deformation and
absorption near
to the surface of the tissues, while the red wavelength may penetrate more
deeply into the
adjacent tissues, providing a signal that changes in response to deformation
of tissues
farther from the contact site(s) between the tip(s) and the tissue. The
photodetectors or
equivalent optical detection pathway may include filters, polarized windows,
or the like
to separately assess the different spectra during an analysis. Comparison
between
photodetccted signals in the blue spectrum with those obtained from the red
spectrum
may be used to deteimine tone and/or elastic modulus of the tissues of the
vessel in the
vicinity of the sensing tip(s). Such a configuration may be advantageous for
assessing
sympathetic tone (i.e. via muscular tension measurement), and/or
vasodialation, vessel
wall stiffness, and/or local tissue stiffness before, during and/or after a
surgical
procedure. Changes in such properties may be indicative of the degree of
completion of
the surgical procedure.
-19-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
[0094] In aspects, an externally placed (e.g. onto the body of the subject)
light source
(e.g. infrared, near infrared, visible, etc.) may be directed into the body
towards the
surgical site. The light source may optionally be modulated to provide a more
easily
detected signal within the subject. One or more sensing tips equipped with
optical
microsensors may sense light emitted from the light source. The mapping of
received
light may be used to locate and/or localize one or more anatomical features
such as
nerves near to one or more of the optical microsensor equipped sensing tips.
[0095] In aspects, one or more externally placed light sources may be used
to help
locate the anatomical sites of interest during the procedure. An external
light source may
include a narrow band light source, a broad band light source, light sources
spaced apart
from each other, and/or combinations thereof. The light sources may be
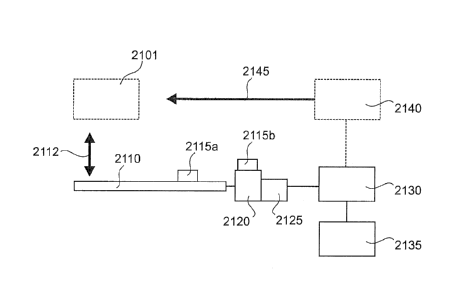
modulated so as
to be more easily detectable by sensors located on, in, or near to the anatomy
of interest.
In one non-limiting example, a plurality of light sources may be aimed at the
surgical site
from distinct vantage points within the body (i.e. as accessed via an
endoscopic
procedure, etc.) or externally to the body (i.e. as positioned at locations on
the body).
[0096] In another non-limiting example an endoscopic camera may be placed
near to
the anatomy, lumen wall, and/or surgical site during a procedure to observe
both the
anatomy, as well as placement of the surgical tools in the vicinity of the
anatomy. In one
non-limiting example, the endoscopic camera and/or light source may provide a
suitable
macroelectrode for RF ablation processes performed during the surgical
procedure.
[0097] In another non-limiting example, one or more sensing tips may be
equipped
with a corresponding micro-light source (e.g. an oLED, an LED, etc.). The
micro-light
source may be used to direct light into the adjacent tissues. One or more
sensing tips
equipped with optical microsensors may be configured to detect light emitted
from the
micro-light source as back scattered by the adjacent tissues. Such information
may be
used to detect anatomical features (e.g. nerves, tumors, etc.) in the adjacent
tissues.
[0098] Such optical configurations may be advantageous for mapping the
local
tissues before, during and/or after a surgical procedure. They may also be
advantageous
for implementation into a nerve detection system (e.g. perhaps as input to a
nerve hunting
algorithm, etc.).
-20-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[0099] In one
non-limiting example, the system may include a micro balloon catheter
for placement into a vessel (e.g. a renal artery, etc.). The micro balloon
catheter may be
coated with a thin layer of an indicator molecule. The indicator molecule may
be tagged
to attach to the target tissue of interest and/or tagged so as to change
chromatic properties
when bound to the target tissue (e.g. nervous tissue, etc.). The molecules may
be
delivered to the desired tissues during a balloon catheterization procedure.
During such a
procedure, the micro balloon catheter may be placed into the vessel of
interest and
inflated so as to kiss the walls of the vessel. While in contact with the
vessel walls, the
indicator molecules may attach and migrate/diffuse into the local tissues.
Such a
procedure may be performed as a first surgical step or as combined with other
aspects in
accordance with the present disclosure. In aspects, the balloon may also be
configured to
deliver a therapeutic agent (i.e. a neuroblocking agent, ethyl alcohol, botox,
etc.) to the
anatomy of interest.
[00100] In a method in accordance with the present disclosure, one or more
sensing
tips are inserted into a lumen with a wall within a body and biased towards
the wall of the
lumen, and one or more electrophysiological signals obtained therefrom. The
electrophysiological signals may be analyzed to locate one or more target
tissues for a
surgical procedure (i.e. one or more sympathetic nerves, parasympathetic
nerves, etc.). A
bolus of therapeutic agent, an RF current, a thermal energy source, and/or the
like may be
delivered to the identified tissues so as to perform the surgical procedure
thereupon. In
aspects, one or more post-procedural electrophysiological signals may be
analyzed to
determine the extent of the surgical procedure.
[00101] In aspects, the therapeutic agent may be provided via a micro balloon
catheter
in accordance with the present disclosure. In aspects, the therapeutic agent
may be
delivered via one or more microfingers in accordance with the present
disclosure.
[00102] In aspects, the micro balloon catheter may include one or more sensory
tips
(e.g. in the faun of functional elements attached to the balloon, attached to
a
superstructure surrounding the balloon, etc.) in accordance with the present
disclosure.
[00103] In aspects, the bioimpedance and/or electrophysiological signals
between one
or more sensing tips in the array and one or more sensing tips in the array,
an external
-21-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
electrode, a reference electrode, or the like may be used to determine changes
in the
structure of the adjacent tissues during an ablation procedure. Such
information may be
useful in determining the extent of the ablation procedure, char accumulation,
etc.
[00104] In aspects, bioimpedance measurements may be correlated with nerve
damage
data, perhaps obtained during prior surgeries, development of the procedure,
and/or
obtained during specific testing procedures, such that changes in local
bioimpedance data
may be used during a surgical procedure to determine the extent of the
ablation
procedure. Such a configuration may be advantageous in the case that the
surgical
procedure itself overwhelms the local electrophysiological activity to the
extent that
neurological monitoring may be hindered for a prolonged period of time after
the
procedure has been completed.
[00105] In aspects, one or more sensing tips may be configured to monitor
local
electrical fields during an ablation procedure in accordance with the present
disclosure in
order to better determine the current flow path through the adjacent anatomy,
perhaps
connected to a warning system to indicate to an operator when the ablation
field is
insufficient for achieving the intended goal. Such a configuration may be
advantageous
for avoiding unnecessary damage to the tissues during a misfired or
misdirected ablation
session.
[00106] In aspects, a system in accordance with the present disclosure may
include a
micro balloon catheter including one or more sensory tips (e.g. in the form of
functional
elements attached to the balloon, attached to a superstructure surrounding the
balloon,
etc.). The micro balloon catheter may be configured so as to bias the sensory
tips against
the adjacent vessel walls, thus providing a reliable interface from which
selective ablation
and detection processes may be performed. Such a micro balloon catheter may be
advantageous for single placement type surgical procedures in accordance with
the
present disclosure.
[00107] In aspects including a plurality of sensing tips (e.g. as placed onto
a micro
balloon catheter, a microfinger array, a microtool set, a flexible cage
assembly, etc.) the
sensing tips may be interconnected with each other, with signal processing
circuitry, a
local control circuit, and the like and/or combinations thereof. In order to
substantially
-22-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
reduce the number of signal wires that must be sent to the surgical site
during the
procedure, the networked array of sensing tips may be multiplexed together
with a locally
placed control circuit (e.g. an application specific integrated circuit,
distributed/interconnected circuit elements, a collection of flexible
semiconducting circuit
elements, etc.). The control circuit may be configured to communicate such
signals with
an extracorporeal system (e.g. a computer, a control system, an RF ablation
controller, a
data acquisition system, etc.). The control circuit may be configured to
communicate
with the extracorporeal system via analog and/or digital means and/or methods.
In one
non-limiting example, the communication may be of primarily digital means such
that the
control circuit may exchange data pertaining to any sensing tip in the array,
as well as
switch data, control data, RF pulse routing, etc.
[00108] In another non-limiting example, the networked array of sensing tips
may be
interconnected with distributed electronic elements and flexible electrical
interconnects
(e.g. as applied to a balloon wall, as provided by structural wires,
microfingers, wire
mesh elements, etc.). In aspects, one or more of the sensing tips,
microfingers, or the like
may be included upon a flexible or stretchable electronic substrate, the
electronic
substrate configured to interface the sensing tips with the anatomy as well as
to
electrically connect one or more sensing tips, or the like with a controller,
a control
system, an operator, a graphical user interface, a display, or the like.
[00109] A controlled nerve ablation system in accordance with the present
disclosure
may include one or more microfingers.
[00110] To this effect, a microfinger array microsurgical tool is disclosed
herein. Any
element in the microfinger array may include a sensing tip in accordance with
the present
disclosure to interact with the local anatomy during a surgical procedure.
[00111] The microfinger array may be advantageous for accessing very small
anatomical sites within a body, perhaps through tortuous vessels, deep into an
organ, etc.
[00112] A microfinger array may be arranged in a surgical tool in accordance
with the
present disclosure such that one or more of the microfingers may =
substantially
independently interface with the adjacent tissues. Thus if an array of
microfingers is
placed against a rough or otherwise uncontrolled surface, each microfinger may
be able
-23-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
to contact, maintain a controlled bias force against, substantially embed an
associated
sensing tip into, and/or substantially maintain contact with the surface
during use, even if
the microfinger array is dragged along the surface as part of a procedure,
during
movement of the surface, etc. Such independently adjustable microfingers may
be
advantageous so as to maintain a known interfacial pressure, especially while
monitoring,
stimulating and/or ablating the tissue with the microfingers. Such
independently
adjustable microfingers may be advantageous to substantially embed an
associated tip
(i.e. an associated sensory tip) into an adjacent tissue during a procedure.
[00113] By microfinger is meant a substantially curved fmger like member (i.e.
with
curvature at one or more points along the length thereof, with multi-axial
curvature, etc.).
Such microfingers may generally have a characteristic width (although may be
of any
cross sectional makeup). The microfingers may generally have characteristic
widths on
the order of approximately lmm, 0.5mm, 0.1mm, 0.05mm, 0.01mm, or the like. In
one
non-limiting example, one or more microfingers may include a Nitinol structure
(e.g. a
wire, a ribbon, etc.) with characteristic width of approximately 50um.
[00114] In aspects, one or more regions of a microfinger in accordance with
the
present disclosure may be selectively coated with an isolation layer (e.g. an
oxide layer, a
dielectric coating, a polymer layer, a lubricious layer, etc.). In aspects,
such an isolation
layer may be selectively applied to regions of the microfingers (i.e. so as to
create
isolated regions and sensitive regions thereof).
[00115] In aspects, the microfingers may be configured so as to deploy and/or
bias
against one or more adjacent tissues during a procedure and may be used to
contact ably
sweep the local anatomy, for purposes of sensing and/or ablating during a
surgical
procedure. In aspects, one or more microfinger dimensions and structure may be
designed so as to provide substantially uniform and predictable bias forces on
the
= adjacent tissues over a wide range of movements and dimensional
variation.
[00116] In aspects; an array of microfingers in accordance with the present
disclosure
may be configured so as to sufficiently collapse down into a delivery catheter
while
expanding radially outwards upon deployment so as to form a controllably
biased contact
within a tubular anatomical structure (e.g. an artery, a vein, an intestinal
wall, etc.).
-24-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00117] In aspects, one or more microfmgers in accordance with the present
disclosure
may be configured into the shape of a wire basket, a mesh-like structure, or
the like. In
aspects, one or more regions of such microfingers may be patterned with an
isolation
layer, so as to direct signals over the microfingers, towards associated
sensing tips, to
provide communication between associated sensing tips and control electronics,
to
control one or more mechanical properties thereof, or the like.
[00118] Such a configuration may be advantageous for accessing tight
anatomical
spaces of interest (e.g. small vessel walls), while also maintaining
consistent contact
forces at a surgical site during a procedure, substantially embedding one or
more sensory
tips into a lumen wall, substantially isolating one or more sensing tips from
an adjacent
fluid, or the like.
[00119] In aspects, a microfinger array in accordance with the present
disclosure may
include a plurality of fmgers, one or more such fingers configured to
interface with the
surrounding tissues and biased radially outwards from a deployment site (e.g.
a guide
wire, a catheter, etc.). In aspects, the microfinger array may be deployed via
longitudinal
retraction of a restraining shell (i.e. a restraining layer in the catheter),
via application of
heat or current (i.e. in the case of a shape memory microfinger, etc.), via
projection of the
microfinger array out of a delivery catheter (i.e. by advancing the
microfinger array
beyond the tip of the delivery catheter, etc.).
[00120] In aspects, one or more microfingers may include a spring-like wire
element
(e.g. Nitinol, spring steel, etc.) and/or may include composite structures
including a
spring-like element to provide a bias force so as to push the tip and/or one
or more
regions of the microfinger towards the wall of a vessel into which it is
placed (i.e.
towards a surface, a lumen wall, a vessel wall, etc.).
[00121] In aspects, a microfinger may include a Nitinol structure, optionally
configured for passage of current flow, to and from the surrounding tissues,
and/or
communication of electrophysiological infoimation between an associated
sensing tip
and a connected microcircuit. In aspects, the Nitinol structure may be
configured such
that, when an RF pulse is applied there through towards the surrounding
tissues, the
Nitinol structure may retreat from the tissues after a predeteitained amount
of energy has
-25-
CA 02862862 2014-07-25
WO 2013/112844 PCT[US2013/023157
passed there through, upon reaching a predeteimined temperature, or the like.
Thus the
Nitinol structure may provide an inherently controlled method for applying a
quantum of
RF energy to the surrounding tissues. Such a configuration may be adapted for
use
simultaneously, additionally, alternatively and/or in combination with one or
more of the
other aspects described in this disclosure.
[00122] In aspects, each finger in the array may move somewhat independently
of the
others such that all fingers may maintain contact with the vessel wall during
a procedure.
[00123] Such a configuration may be advantageous for maintaining robust
contact
with the walls of a tortuous anatomical site (e.g. a plaque filled artery, a
tortuous vein, a
damaged vessel, etc.) within the body. Such a configuration may be
advantageous for
maintaining robust contact with the walls of a lumen, surgical site, etc.
while performing
a procedure (i.e. scanning a surface with one or more microfingers, dragging a
microfinger along a surface, monitoring a tissue site, ablating a tissue site,
etc.) or during
periods of relative movement (i.e. in the presence of organ movement, perhaps
due to
physiological processes, stresses related to biorhythms, breathing, blood
pressure, etc.).
[00124] In aspects, at least a portion of the microfingers may be formed as
spirals such
that torsion applied at the operator end of the catheter may rotate the
microfingers about
the central axis of the lumen (i.e. blood vessel, etc.), thus allowing one to
sweep the
contact of the microfingers around the entirety of the vessel interior. Such
movements
may be advantageous for analyzing the adjacent tissues, selectively mapping
and ablating
the tissues, etc. In one non-limiting example, a microfinger array in
accordance with the
present disclosure may be swept circumferentially along the wall of a vessel,
optionally
starting and stopping so as to analyze the local tissues. If a suitable site
for ablation is
detected, the microfinger array may be used to ablate the tissues as well as
monitor the
ablation process to ensure controlled ablation is achieved before continuing
with the
sweeping procedure.
[00125] In aspects, the microfingers may be follned slightly off axis, such
that relative
axial movement of an overlying sheath may be used to retract the microfingers
into the
sheath or conversely to deploy them towards the anatomical site. Additionally,
alternatively, or in combination, off axis arrangements may provide the
capability to
-26-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
sweep the microfingers circumferentially along the anatomical site via
applying torsion to
the guide wire, delivery wire, and/or catheter to which they are attached.
[00126] Such a configuration may be advantageous for simultaneously mapping
and
selectively ablating an anatomical site during a surgical procedure.
[00127] Furthermore, such a configuration may be advantageous for working upon
an
anatomical site, while maintaining flow of fluid there through (i.e. as
opposed to an
occlusive tool, which may block flow during expansion thereof).
[00128] In aspects, one or more microfingers may be provided= with highly
miniaturized and flexible structure so as to more easily access highly
restricted
anatomical sites within the body.
[00129] In aspects, one or more microfingers may include one or more sensing
tips in
accordance with the present disclosure for capturing information from the
local surgical
site. Some non-limiting examples of sensing options include temperature
sensors,
electrodes, strain gauges, contact force sensors, combinations thereof, and
the like. For
purposes of discussion, a sensing tip may also be referred to as a
microsensor.
[00130] The sensing tips may be configured to elucidate a range of key
information
during a procedure. Some non-limiting examples are discussed in more detail
below.
[00131] Bioimpedance between one or more microfinger tips may be used to
determine the degree of contact between the finger tips and the anatomical
site, as well as
potentially the bias force between the finger tips and the anatomical site.
Such
information may be useful in determining when adequate contact and to gauge
how much
current should be applied to an anatomical site during an ablation procedure.
[00132] Mechanomyographic information may be obtained from fingertips during a
procedure as determined by slight changes in an associated strain measurement
and/or
contact force measurement (e.g. via direct force measurement between the tip
and the
local anatomy, and/or via changes in the deformation of the microfinger as
measured by
an associated micro strain gage attached thereupon).
[00133] Evoked potential monitoring at or between one or more finger tips, may
be
used to map nervous response, electromyographic response, extracellular
potentials, local
-27-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
field potentials, evoked potential, etc. along the wall of the local anatomy
(e.g. vessel
wall, organ wall, etc.). Such infonnation may be advantageous for selecting
tissues on
which to perform a surgical procedure (e.g. an ablation procedure, a biopsy, a
stimulation
procedure, etc.).
[00134] The tone of the adjacent tissues may be determined by combining strain
and/or force measurement of the microfingers while applying an excitation to
one or
more microfingers (e.g. optionally clockwise torsion to advance the
microfingers and
small counterclockwise torsion to measure the tone of adjacent tissues, a
vibratory exciter
in combination with contact and/or microfinger strain measurement, etc.).
[00135] Such tone measurement may be useful in determining the local stiffness
of
tissues in contact with the microfinger array (e.g. so as to determine the
type of tissue
adjacent to one or more microfingers, to locate plaque, to locate a cancerous
tumor, etc.).
[00136] Stimulation and sensing from one or more microfingers in the
microfinger
array may be used to elicit nervous function of local anatomy. Such
information may be
used to select tissues for a surgical procedure, to determine the degree of
progression of a
surgical procedure (e.g. a degree of ablation during RF surgery, etc.).
Directional
stimulation and sensing may be used to selectively treat only nerves that are
configured to
send signals in the preferred direction (i.e. via combination of stimulation
and/or sensing
from a plurality of sensing tips, sensing sites, etc.).
[00137] In aspects, one or more microfingers may include the capability to
apply/receive an RF current to/from the surrounding tissue.
[00138] Such RF currents may be applied between one microfinger in the array
and an
(optionally) distant counter electrode, between two or more microfingers in
the array, to a
extracorporeal patch on the body, etc.
[00139] In aspects pertaining to multiple microfinger RF current passage, the
local
impedance of each microfinger may be altered so as to control the current
delivered
thereto.
[00140] In aspects pertaining to multiple microfinger RF current passage, the
local
current flow through each microfinger may be monitored so as to determine the
path of
-28-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
the RF current flow, to ensure no leakage currents are detected, etc. Such
information
may be used to more precisely control the delivery of RF currents to the local
anatomy
during an ablation procedure.
[00141] In aspects, prior to, during, and/or after the RF current is
applied to the
surrounding tissues, one or more microfingers may be configured to monitor a
physiological parameter (e.g. water concentration, tone, blood oxygen
saturation of local
tissues, evoked potential, stimulation/sensing of nervous activity, emg,
temperature, etc.)
to determine the extent of completion of the intended surgical procedure.
[00142] In aspects, the bioimpedance between one or more microfingers in the
array
may be used to determine changes in the structure of the adjacent tissues
during an
ablation procedure. Such information may be useful in determining the extent
of the
ablation procedure, char accumulation, etc.
[00143] In aspects, bioimpedance measurements may be correlated with nerve
damage
data, perhaps obtained during prior surgeries or obtained during specific
testing
procedures, such that changes in local bioimpedance data may be used during a
surgical
procedure to determine the extent of the procedure. Such a configuration may
be
advantageous in the case that the surgical procedure itself overwhelms the
local
electrophysiological activity to the extent that neurological monitoring may
be hindered
for a prolonged period of time after the procedure has been completed.
[00144] In aspects, one or more microfingers may be configured to monitor
local
electrical fields during an ablation procedure in order to better determine
the current flow
path through the adjacent anatomy, perhaps connected to a warning system to
indicate to
an operator when the ablation field is insufficient for achieving the intended
goal, to
assist with the direction of energy towards the intended surgical site, to
conserve energy,
etc. Such a configuration may be advantageous for avoiding unnecessary damage
to the
tissues during a misfired ablation session.
[00145] A system may include an embolic net to capture char that may foal'
during the
ablation procedure. Such netting may be advantageous for preventing surgically
related
emboli from traveling throughout the body after the surgery.
-29-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
[00146] In asi3ects, the system and/or microfingers may include a coolant
delivery
system (e.g. a saline delivery system) in order to cool the microfingers
during and/or after
an ablation procedure. Such coolant delivery may be advantageous for
minimizing char
and excessive damage associated with an ablation procedure. Such coolant
delivery may
be part of a cryogenic surgical procedure, or the like.
[00147] In aspects, the system may include multiple microfinger arrays,
perhaps
located at specific radii from each other such that when sweeping a tubular
anatomical
site (e.g. a vessel), the bias forces may be reasonably maintained between the
microfingers and the tissue walls.
[00148] In aspects, one or more microfingers may include an exposed electrode
area
(i.e. as part of an electrode based sensing tip) that only touches the walls
of the adjacent
anatomy. Such a configuration may be advantageous for minimizing current flow
into
the adjacent fluids within the vessel, to better control RF current flow in
the vicinity of
the electrodes, minimize conductivity between the exposed area and the
surrounding
fluid, so as to substantially embed the exposed electrode area in to the wall
of the
adjacent anatomy, etc.
[00149] In aspects, one or more microfingers may include one or more active
material
elements. Control signals delivered to the active material element may help to
bias the
microfingers towards the intended surgical site, actively control the contact
forces
between finger tips and the surgical sites, etc. Some non-limiting examples of
active
materials that may be suitable for application to one or more microfingers
include shape
memory materials (e.g. shape memory alloys, polymers, combination thereof),
electroactive polymers (e.g. conjugated polymers, dielectric elastomers,
piezoelectric
polymers, electrets, liquid crystals, graft elastomers, etc.), piczoceramics
(e.g. amorphous
piezoceramics, single crystals, composites, etc.). In addition the active
material may be
used as a vibratory exciter and/or mechanical probe, for use in monitoring the
tone of the
adjacent tissues (see above), alternatively, in addition or in combination, to
cause
vibratory/ultrasonic ablation and/or local heating to the tissues. In such
aspects, the
active material may be included along the length and/or over a region of the
microfinget
-30-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
(i.e. so as to influence the shape of the microfinger during contraction or
expansion of the
active material).
[00150] In aspects, one or more microfingers may include an electrical shield
such that
the microfinger tips are effectively shielded from other currents flowing
through an
associated catheter, the body, etc. during a procedure.
[00151] In aspects, one or more elements of a microfinger based catheter may
include
a bidirection switching network, micro amplifier array, a sensory front end,
combinations
thereof, or the like in order to amplify sensed signals as close as possible
to the
anatomical interface, to switch the function of a microfinger tip between
sensory,
stimulatory, and/or ablative functions, perform combinations thereof, or the
like. In
aspects, the circuitry may be included in the delivery wire within the
catheter of the
system. In such aspects, the circuitry may be coupled to one or more
microfingers and/or
sensing tips each in accordance with the present disclosure, and a secondary
signal
acquisition circuit, a digital communication block, a controller, an RF signal
generator,
combinations thereof, and the like.
[00152] In aspects, a bidirectional switching network may be used to enable
bifunctional stimulation/sense capabilities in one or more microfingers, etc.
The
switching network may be included in a local amplifier array, as a flexible
circuit, or
silicon die interconnected to or placed upon one or more microfingers, etc.
Alternatively,
additionally, or in combination, an extracorporeal circuit element may be
coupled to the
switching network and/or microfingers, sensing tips, etc. and to a controller
included
within a surgical system including a microfinger array in accordance with the
present
disclosure.
[00153] In aspects, a micro amplifier array may be used to preamplify the
signals
obtained from one or more sensory aspects of the microfingers, so as to
improve the noise
signature, etc. during use. The microamplifier may be coupled to the catheter,
embedded
into the catheter, embedded into one or more microfingers, etc.
[00154] In aspects, one or more microfingers in accordance with the present
disclosure
may be provided such that they are sufficiently flexible so as to buckle, or
change
orientation during back travel, so as to prevent puncture of the local
anatomy. A
-31-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
configuration as outlined in this non-limiting example may be advantageous for
providing contact with the local anatomy without significant risk of damaging
the
adjacent anatomy (e.g. puncturing a vessel wall, etc) which may be a concern
with stiffer,
more traditional structures. Such microfingers may include a characteristic
width of less
than 200um, less than 100um, less than 50um, less than 25um, less than 10um.
[00155] In aspects, one or more microfingers in accordance with the present
disclosure
may include a substantially hyper elastic material (e.g. for _______ hied from
a memory alloy
material, a superelastic material, a spring steel, etc.) so as to effectively
deploy from a
very small deployment tube/catheter and expand outward to accommodate a large
range
of vessel diameters. Such a configuration may be advantageous in so far as a
small
number of unit sizes may be suitable for treating a wide range of anatomical
structures.
In addition, the designed curvature and form of a microfinger may be
substantially
chosen so as to further enable a wide deployable range of movement.
[00156] A
surgical tool including a plurality of microfinger arrays (i.e. clusters of
microfingers, fans of microfingers, etc.) may be employed so as to determine
physiological response more remotely from an intended surgical site than may
be
available within a single array. Aspects of the disclosed concepts may be
employed
along the same lines by extending interactions between microfingers within an
array, to
inter - array interactions. In aspects, a surgical tool including a plurality
of clustered
microfinger arrays may be advantageous to analyze one or more anatomical sites
simultaneously from a plurality of sites (macroscopically separated sites). In
one non-
limiting example, two microfinger arrays may be arranged along a catheter
based surgical
tool, so as to interface with the walls of a lumen, at two or more
longitudinally separated
distances. Physiological sensing from multiple microfingers may be
advantageous for
determining the extent of neurological traffic between the plurality of sites,
determine the
direction of traffic, determine if traffic from one direction or the other is
blocked (i.e.
after a surgical procedure, after RF current application, after a denervation
procedure,
etc.). Such configurations and methods for determining the state of a
plurality of
anatomical sites is further disclosed throughout the text and appended figures
of this
disclosure.
-32-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00157] In aspects, a system in accordance with the present disclosure may be
used to
monitor physiological activity associated with a surgical site prior to,
during and/or after
a surgical procedure is applied thereto. Some suitable examples of surgical
procedures
include an RE ablation, Argon plasma coagulation, laser ablation, ultrasonic
ablation,
cryoablation, microwave ablation, abrasion, biopsy, delivery of a substance
(e.g. a
chemical, a drug substance, an acid, a base, etc.), combinations thereof, and
the like. The
local physiological activity (e.g. nervous activity, blood perfusion, tonal
changes,
muscular sympathetic nerve activity, etc.) may be monitored with one more
sensors
(sensing tips, microfingers, etc.) and/or associated stimulators each in
accordance with
the present disclosure. Additionally, alternatively, or in combination, a
technique for
assessing one or more physiological properties and/or states of an associated
surgical site
may be employed. Such techniques include assessing values and/or trends in
bioimpedance, blood pressure, tissue oxygenation, tissue carbon dioxide
levels, local
temperatures and changes thereof, and the like.
[00158] In aspects, the system may include a substrate onto which the sensing
tips may
be placed. Such a substrate may be founed from a balloon wall, a mesh, an
interwoven
ribbon array, a cloth, etc. The substrate may include stretchable and/or
flexible electronic
materials.
[00159] Electrical interconnects may be formed from carbon nanotubes (e.g.
SWNTs,
MWNTs, etc.), nanowires, metallic wires, composites, conductive inks, and the
like.
[00160] In aspects, a portion, or all of the substrate and/or an associated
substrate
carrier film may be formed from polyurethane, a silicone, a general elastomer,
silk fibroin
materials, or the like and/or combinations thereof. Inclusion of microporous
or fibrous
substrates, may be advantageous to allow the substrate or substrate carrier
film to adhere
to the adjacent tissues via capillary effects (i.e. tendencies to wick fluid
from adjacent
tissues into the substrate). The thickness of films formed from the material
may be less
than 30um thick, less than 20um, less than 10um, less than 4um, less than lum.
Composites of somewhat stiffer materials (such as polyimide, PET, PEN, etc.)
and
somewhat softer materials (e.g. silicones, polyurethanes, thermoplastic
elastomers, etc.)
-33-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
maybe used to compromise between overall structural stiffness and conformal
capabilities of the substrate.
[00161] In aspects, patterned overcoats and/or composite layers may also be
used to
expose electrode materials and/or sensing tips to the surrounding tissues in
the vicinity of
measurement regions, etc.
[00162] In one non-limiting example, the substrate may be at least partially
formed
from a silk material (e.g. Bombyx mori cocoons). The material may be processed
to
remove sericin (which may cause undesirable immunological response) using
methods
known in the art, The resulting material can be solvent cast into shapes and
crystallized
to form self-supporting structures.
[00163] In aspects, adaptive temperature estimation may be used to better
control the
RF process. Such techniques may be supported by use of a surgical tool in
accordance
with the present disclosure, including one or more sensing tips configured
with
temperature and/or bioimpedance monitoring aspects. Modeling of changes in
local
bioimpedance may be related to local temperature changes during the ablation
process.
Such measurements as well as local theimoconductive properties, tissue
thermoconduction, etc. may also influence the rates at which a local ablation
process may
take place (i.e. as related to a thermal ablation process).
[00164] In aspects, a system in accordance with the present disclosure may
include
one or more microsensors for monitoring nervous activity and/or related
physiological
activity during the RF ablation process. Some examples of suitable monitoring
techniques include electromyography (EMG), muscle sympathetic nerve activity
(MSNA), mechanomyography (MMG), phonomyography (PMG), extraeellular
potentials, local field potentials, combinations thereof, and the like.
Mechanomyography
(MMG) measures the force created by local muscle contractions caused by
associated
neural activity. Phonomyography (PMG) measures low frequency sounds associated
with movement generated by associated neural activity. Traditionally,
techniques such as
MMG and PMG have been employed on externally accessible nervous and muscular
tissues. One advantage of such techniques is that they may not be as easily
affected by
-34-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
local electrical noise as EMG and the effects of the nervous activity may be
generally
sensed farther from the associated nerve than with clectromyographic
techniques.
[00165] Alternatively, additionally or in combination the ascribed sensing
techniques
may be combined with stimulation from local sources in accordance with the
present
disclosure. Such stimulation and sensing may be advantageous in determining
functionality of local nerves without the need to listen to complex
biologically generated
nervous activity. Furthermore, combined stimulation and sensing may be
advantageous
for determining functionality of a local nerve in real-time during a
denervation and/or
ablation procedure (e.g. the successive stimulation and sensing may be used to
deteimine
the degree of neurological block and/or neuromuscular block there between). In
aspects,
such functionality as well as directionality of the nerve signal propagation
(e.g. efferent,
afferent, etc.) may be more easily determined through use of combined local
stimulation
and sensing.
[00166] In aspects, one or more patterns of nerve stimulation may be used to
deteimine the function of the local nerve structures as well as one or more
aspects of
neurological block and/or neuromuscular block that may be caused by the
surgical
procedure (e.g. ablation), anesthesia, heating, chemical delivery, a related
condition, etc.
[00167] In aspects, a single stimulation may be applied to elicit maximal
response
from the associated nerve at frequencies of less than 10Hz, less than 1Hz,
less than
0.1Hz. The downstream response as measured by any of the described techniques
will
depend on the frequency with which the stimuli are applied. In aspects, in
order to allow
for complete recovery of the nerve between stimulations, a frequency of less
than or
equal to 0.1Hz may be advantageous.
[00168] During RF ablation of an associated nervous structure, the evoked
electrical
and/or muscular responses may be dramatically affected. Such changes in the
response
may be useful in determining the state of the denervation procedure. Thus they
may be
advantageous to determine the exact degree of RF energy that must be applied
to a given
structure in order to cause sufficient denervation as desired by a surgical
procedure. Such
an approach may be advantageous to limit damage to surrounding tissues caused
by the
denervation procedure, to ensure suitable denervation has been achieved, to
determine
-35-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
which nerves are affected by the procedure, to control the extent of a
denervation
procedure, etc.
[00169] Another technique for stimulation and sensing of the nervous response
includes applying a rapid succession of pulses followed by a period of
inactivity. Pulse
trains may be used to gradually force a nerve into a blocked state. The rate
at which a
nerve enters a blocked state and later recovers therefrom may be a suitable
indicator of
the overall health and functionality of the nerve (i.e. a suitable metric for
determining
how a procedure has affected that nerve).
[00170] In aspects, the sensing of the nervous response may not need to be
local to a
surgical site, but rather downstream (in the sense of the flow of an
associated nervous
signal) from the site. Such sensing of the nervous response may be
advantageous for
determining the progression of a particular faun of communication past a
surgical site
(i.e. afferent, efferent traffic, etc.).
[00171] In aspects, various mapping techniques may be applied to the surgical
site,
before, optionally during and after a surgical procedure. Some mapping
techniques as
used in cardiac interventions include pace mapping, activation mapping,
entrainment
mapping, and substrate mapping. It may be feasible to adapt such techniques
for use in
the intended application. In general, these techniques may complement each
other in
localizing where amongst a surgical site to target the ablation procedure.
[00172] In one non-limiting example, the micro fingers and/or associated
sensing tips
may be arranged in a polar configuration as an array of arches (i.e. an array
of thin, arch-
like elements each extending radially outwards from a central axis). The
arches may be
attached at each end, a first end connected to an axially oriented draw wire
and the other
end attached to a collar. The arches may be collapsed and/or expanded radially
by
extending and/or retracting the length of the draw wire between the first end
and the
collar respectively. The draw wire may extend through the surgical tool to the
operator
or a machine, where force on the draw wire may be used to achieve this
function. Thus
the arches may be provided in a substantially collapsed state (i.e. with small
overall
diameter) for easy delivery to the surgical site. Upon delivery to the
surgical site, the
draw wire may be retracted, perhaps automatically and/or with the help of an
operator
-36-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
and the arches may be extended radially outwards, so as to contact the
adjacent tissues of
the vessel. Such a procedure may be used to bias the array of sensing tips
and/or micro
fingers towards the walls of the vessel while maintaining blood flow there
through.
[00173] Alternatively, additionally, or in combination the arches may be
deployed at a
surgical site by removal of a restraining sheath (perhaps by retraction), by
dissolution of a
restraining element (e.g. an adhesive, an electrochemically destructible
member, etc.), via
thermal self-expansion of one or more elements of the arches, by combinations
thereof,
or the like.
[00174] Additionally, or in combination to the aspects described herein, the
surgical
system may be configured to monitor one or more physiological parameters at
one or
more locations in the body remote from the surgical site. Some non-limiting
examples
of what may be monitored include water concentration, tone, blood oxygen
saturation of
local tissues, evoked potential, stimulation/sensing of nervous activity,
electromyography, temperature, blood pressure, vasodialation, vessel wall
stiffness,
muscle sympathetic nerve activity (MSNA), central sympathetic drive (e.g.
bursts per
minute, bursts per heartbeat, etc.), tissue tone, blood flow (e.g. through an
artery, through
a renal artery), a blood flow differential signal (e.g. a significantly
abnotmal and or
sudden change in blood flow within a structure of the body, a vessel, an
organ, etc.),
blood perfusion (e.g. to an organ, an eye, etc.), a blood analyte level (e.g.
a hatmone
concentration, norcpinephrine, catecholamine, renine, angiotensin II, an ion
concentration, a water level, an oxygen level, etc.), nerve traffic (e.g. post
ganglionic
nerve traffic in the peroneal nerve, celiac ganglion, superior mesenteric
ganglion,
aorticorenal ganglion, renal ganglion, and/or related nervous system
structures),
combination thereof, and the like.
[00175] In aspects, a surgical system in accordance with the present
disclosure may
include one or more elements to monitor physiological activity and/or analyte
levels (e.g.
a haimone level), in and/or near to one or more portions of a gland, an
endocrine gland
(e.g. an adrenal gland, an adrenal medulla, etc.), etc.
[00176] In another non-limiting example, a multi catheter surgical system may
be
employed, each catheter in accordance with the present disclosure. In this non-
limiting
-37-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
example, one or more first catheters may be used to probe and/or ablate
tissues at a first
surgical site (e.g. an artery, a renal artery, a left renal artery, etc.)
while one or more
second catheters may be configured to monitor one or more physiological
parameters
elsewhere in the body (e.g. in an alternative artery, a vein, in an organ, at
a lymph node,
at a ganglion, etc.), perhaps to determine the effect of the surgical
procedure there upon.
In one non-limiting example, the catheters may be inserted into the same or
closely
positioned entry points into the body (e.g. the femoral artery, iliac artery,
radial artery,
femoral vein, etc.). Such a configuration may be advantageous for providing a
minimally
invasive surgical tool to perform the surgical procedure (e.g. a
sympathectomy, a renal
sympathectomy, etc.).
[00177] Figs. la-c show a surgical tool tip in accordance with the present
disclosure in
a delivery mode and a deployed mode. Fig. 1a shows a delivery catheter 110
with a
micro surgical tool 120 held within (e.g. in a retracted position). The micro
surgical tool
120 may include one or more microfingers 125 in accordance with the present
disclosure
for use in a surgical procedure (e.g. a denervation procedure, a biopsy, an
excision
procedure, etc.). The micro surgical tool 120 may be configured so as to
reversibly
collapse down into the delivery catheter 110 upon retraction. The
microsurgieal tool 120
and/or delivery catheter 110 may be connected 115 to a controller, a control
unit (e.g.
with a deployment control switch, etc.), an operator, a signal conditioning
circuit, etc.
[00178] Fig. lb shows a deployed micro surgical tool 120 with a plurality of
microfingers 125a-c (i.e. in this case, 3 microfingers are shown). In aspects,
the
microsurgical tool 120 may include any reasonable number of microfingers 125a-
c. Each
microfinger 125a-c may be generally spaced apart from the others such that if
the array is
biased towards a tissue site, they may form a pattern (e.g. a dotted line, a
diamond, a ring,
etc.). The microfingers 125a-c may be expand outward (e.g. radially, axially,
circumferentially, and/or combinations thereof) in one or more directions when
deployed
from the associated delivery catheter 120. Thus the microfingers 125a-c may
suitably
engage with a local tissue site be it to monitor the site, ablate the site, a
combination
thereof, or the like. One or more of the microfingers 125a-c may include one
or more
sensing tips 130a-d each in accordance with the present disclosure. As shown
in this
non-limiting example, each microfinger 125a-c includes a sensing tip 130a-c
located,
-38-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
primarily at the end of the microfinger 125a-c. In addition, one microfinger
125c
includes another sensing tip 130d (e.g. perhaps a temperature sensor, a
reference
electrode, a flow sensor, etc.) located near to the end of the microfinger
125c. In aspects,
a temperature sensor 130d in the flow may be advantageous to evaluate local
flow
changes (turbulence, micro heating, etc.) that may occur during an associated
surgical
process.
[00179] Fig. 1 c shows a deployed microtool 135 from a delivery catheter 111,
after
being placed within a lumen within a body (i.e. a vessel, an artery, a vein, a
tubule, etc.).
The microtool 135 includes a plurality of microfingers 140a-f each arranged
radially
around the axis of the lumen and biased agains the wall of the lumen 11. In
this non-
limiting example, the microfingers 140a-f are shown with electrode based
sensing tips
141a-f, The microfingers 140a-f are also shown with curled tips, configured so
as to
minimize stresses applied to the lumen wall 11 during deployment. The
electrode based
sensing tips 14 1 a-f may include one or more exposed electrically conducting
regions (i.e.
a metallic material, a conducting polymer, a conjugated polymer, a carbon
material,
combinations thereof, or the like) so as to interface electrically with the
adjacent lumen
wall 11. In aspects, the microfingers 140a-f may be coated with an insulating
layer in
accordance with the present disclosure, so as to minimize fluid contact there
along during
a monitoring, stimulation, and/or ablation process applied therewith.
[00180] Figs. 2a-b show deployed surgical microtools in accordance with the
present
disclosure interacting with a local surgical site. Fig. 2a shows a cross
section of a vessel
located at a surgical site. The vessel includes a vessel wall 12, which may
have one or
more anatomical features that are to be operated upon (i.e. nerves, tumor,
plaque, etc.).
An array of microfingers 210 in accordance with the present disclosure is
shown
interacting with the vessel wall 12. For purposes of discussion, an associated
delivery
catheter 215 is also shown in within the vessel wall 12. In the example shown,
the
microfinger array 210 may be swept along the vessel wall by counter clockwise
rotation
212 about the delivery catheter 215 (or clockwise, depending on the preference
of the
operator, design of the catheter, etc.). Such motion may be provided by an
operator (e.g.
by torsion of the micro surgical tool shaft), by a mechanism within the tool,
by a flexural
structure of the micro finger array 210 (e.g. a helical structure so as to
prefer rotational
-39-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
sweeping of the anatomy as the array is pulled through the vessel),
combinations thereof
or the like. The microfingers of the array 210 may include one or more sensing
tips each
in accordance with the present disclosure configured to interact with the
lumen wall 12.
In one aspect, shown, one or more of the sensing tips may be configured to
pass a current
214 locally between one or more of the sensing tips. Such current may be used
to
stimulate the local anatomy (i.e. as part of a stimulation/response monitoring
system), or
at higher intensities, as an RF source for ablation of local tissues (i.e. to
perform a
localized sympathectomy, etc.). In aspects, the microfinger array 210 may be
configured
to provide a combination of sensing and ablation (i.e. to perform a controlled
sympathectomy procedure, to vary the degree transmission of local signals, to
affect one
or more local anatomical sites, one or more receptors, etc.).
[00181] Fig. 2b shows a multi-array micro surgical tool including two arrays
of
microfingers 220a-b simultaneously interacting with substantially opposite
sides of the
vessel wall 13. One microfinger array 220b is shown ablating a local tissue
site, thus
folining an ablation zone 222. A hypothetical RF ablation current pathway 224
is shown
in the figure for purposes of discussion. The multi-array microsurgical tool
may be
rotated about the delivery catheter 225, in this example, in a counter
clockwise direction
230 with respect to the viewer. The microfingers 220a-b include one or more
sensing tips
226, in this case, the sensing tip 226 near the ablation site is configured to
deliver current
to and/or accept current from the local tissues. In a usage scenario, the
sensing tips 226,
may be configured to monitor local electrophysiological activity in the lumen
wall while
the surgical tool is rotated about the delivery catheter 225, when an
anatomical site 235 of
interest (i.e. a nerve plexus, a high traffic nerve plexus, a tumor, etc.) is
detected, the
ablation process may be initiated through select microfingers in the arrays
220a-b.
Current and/or sensing potentials may be provided between a first and one Of
more
sensing tips 226 in the arrays 220a-b, or between a sensing tip 226 and an
external
electrode (not explicitly shown), located in a direction 237 away from the
lumen wall 13.
[00182] Fig. 2c shows a radially expanding micro surgical tool including an
array of
microfingers 250a-f (not all elements numbered due to clutter), each
microfinger
configured to bias against the lumen wall 14 upon deployment from the delivery
catheter
255. The microfingers 250a-f may include one or more sensing tips 260 (not all
-40-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
numbered due to clutter) each in accordance with the present disclosure,
configured to
simultaneously interact with localized regions of the vessel wall 13 upon
deployment. A
collection of microfingers 250a-c is shown in the process of ablating a local
tissue site,
thus forming an ablation zone 265. A hypothetical RF ablation current pathway
267 is
shown in the figure for purposes of discussion. In a usage scenario, the
microfingers
250a-e and associated sensing tips 260 may be configured to bias against the
lumen wall
14 and to monitor local electrophysiological activity in the lumen wall while
the surgical
tool is positioned therein, and/or drawn along the axis of the lumen during a
mapping
process, etc. In the given example, an anatomical site 269 of interest (i.e. a
nerve plexus,
a high traffic nerve plexus, a tumor, etc.) is detected near to sensing tip
260, and an
ablation process may be initiated through select microfingers in the arrays
250a-c. In
aspects, the anatomy of interest may be mapped, and/or the layout of the
anatomy
determined either via movement of the microtool along the length of the lumen,
via
coordinated sensing, and/or stimulation/sensing between related microfinger
arrays (not
explicitly shown), etc. via one or more methods in accordance with the present
disclosure. In aspects, two or more of the microfingers 250d-e may be
configured to pass
current 272 there between, to measure an electrophysiological signal there
between, etc.
In aspects, the system may include and/or be coupled to a controller, the
controller
configured to analyze the signals obtained from each microfinger and determine
the
location and/or state of an anatomical site of interest, determine the extent
of an ablation
procedure, etc.
[00183] Fig. 3 shows a plurality of micro-tips monitoring physiological
response
and/or stimulating local tissue during a surgical procedure. Three sensing
tips 310a-c are
shown abutted against a vessel wall 15. The sensing tips 310a-c may be
attached to
microfingers, substrates, balloons, or the like (each in accordance with the
present
disclosure).
[00184] In a first example 320, the first sensing tip 310a is used both to
stimulate the
local tissues (e.g. in order to deteimine proximity to a local nerve, to
determine one or
more aspects of local nerve function, etc.), and to ablate the local tissues
(e.g. as part of a
denervation event, to destroy cancerous tissue, to cauterize a tissue site,
etc.). The second
sensing tip 310b is configured to monitor local temperature variation of the
tissues with
-41-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
which it may be in contact during a surgical procedure. The third sensing tip
310c is
configured to sense an electrical response from the local tissues during a
surgical
procedure (e.g. evoked potential, EMG, microvoltage, current flow, etc.).
[00185] Fig. 3 also shows a time series of events for the first example 320,
shown
during an RF ablation procedure. During a testing period 316 one or more
stimulatory
pulses 312 may be applied to the first sensing tip 310a and monitored 322 by
one or more
of the other tips (in this case the third sensing tip 310c). Perhaps during
this period the
combination of stimulation and response satisfies a predetermined surgical
criterion for
initiating local ablation (i.e. local nerves identified, overactive
neurological traffic
detected, etc.). During the ablation period 317, an RF signal 323 is applied
to the tissues
via the first sensing tip 310a (e.g. perhaps with current flow to the third
sensing tip 310e,
to a remote macroclectrode, combinations thereof, etc.). The RE ablation may
be
performed sequentially or with a duty cycle so as to evaluate the progress
throughout. It
may also be performed in one sequence. The RF signal as measured by the third
sensing
tip 310c may be used to assist with determining bioimpedance of the local
tissues, the
state of the local tissues, etc. during the ablation process. In this non-
limiting example,
local tissue temperature 325 near to the ablation site (as monitored via the
second sensing
tip 310b) may also be used to estimate the extent of the ablation process,
perhaps in
combination with sensing via the third sensing tip 310c, and/or bioimpedance
measurements. When the temperature and/or ablation process reaches a setpoint,
the
ablation is stopped and the local tissues are allowed to recover. This
timeframe is shown
as a recovery period 318. In aspects, the recovery period 318 may be less than
2min,
lmin, 30s, 10s, 1 s, 0.1s. In an additional testing period 319, the first
sensing tip 310a
may stimulate the local tissues and the third sensing tip 310c may monitor for
a response.
In this case, an absent response indicates that the ablation procedure has
proceeded
sufficiently for the intended purposes and the microsurgical sensory tip array
may be
advanced to a new site or removed from the lumen.
[00186] Fig. 3 also shows a time series of events for a second example 330,
shown
during an RF ablation procedure. In this example, the first sensing tip 310a
and the third
sensing tip 310c are configured so as to monitor local electrophysiological
response of
the tissues (i.e. to monitor extracellular neurological activity, local field
potentials,
-42-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
electromyographic signals, etc.) and the second sensing tip 310b is configured
to apply an
RF current to the lumen wall 15 adjacent there to. During a testing period 331
electrophysiological responses are monitored at the first sensing tip 310a and
the third
sensing tip 330c. As can be seen from the a-site response curve 335 and the c-
site
response curve 337, prior to an ablation, the coherence between the sensed
signals is high
(i.e. closer to 1 than to 0). During the ablation period 332 an RF signal 336
is applied to
the tissues via the second sensing tip 310b (e.g. perhaps with current flow to
the first
sensing tip 310a, the third sensing tip 310c, or to a remote macroelectrode,
combinations
thereof, etc.). The RF ablation may be performed sequentially or with a duty
cycle so as
to evaluate the progress throughout. It may also be performed in one sequence.
The
associated RF signal as measured by the first sensing tip 310a and the third
sensing tip
310c may be used to assist with deteimining bioimpedance of the local tissues,
direction
of RF current flow from the second sensing tip 310b, the state of the local
tissues, etc.
during the ablation process. When the temperature and/or ablation process
reaches a
setpoint, the ablation is stopped and the local tissues are allowed to
recover. This
timeframe is shown as a recovery period 333. In aspects, the recovery period
333 may be
less than 10min, less than 5min, less than 2min, less than lmin, less than
30s, less than
10s, less than Is, less than 0.1s, or the like. During the
recovery period
electrophysiological responses are monitored at the first sensing tip 310a and
the third
sensing tip 330c. As can be seen from the a-site response curve 335 and the c-
site
response curve 337, after the ablation procedure 332, the coherence between
the sensed
signals has changed dramatically (i.e. it has decreased significantly). The
measure of
coherence between the signals before and after a surgical procedure, may be a
quantifiable indicator of a state of completion thereof, it may be a
quantifiable
measurement of the local percentage change in neurological activity, it may be
an
indicator of the ratio of afferent/efferent traffic in the vicinity of the
sensing tips 310a-c,
and the like. In this case, a markedly changed coherence between the a-site
signal 335
and c-site signal 337 indicates that the ablation procedure has proceeded
sufficiently for
the intended purposes and the microsurgical tool in accordance with the
present
disclosure, may advance to a new site or removed from the lumen. Such
coherence based
determination of procedural outcomes may be a suitable method for
automatically
-43-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
performing associated surgical procedures, for controlling the extent of such
surgical
procedures, and the like.
[00187] Figs. 4a-b show interactions between multiple micro-tips and the local
vasculature in accordance with the present disclosure. Fig. 4a shows a micro
surgical
tool in accordance with the present disclosure including three arrays of
microfingers
410a-c interacting with the vessel walls 16 of a local anatomical site. The
microfinger
arrays 410a-c are shown in a deployed state from a delivery catheter 415 in
accordance
with the present disclosure. In aspects, the microfinger arrays 410a-c may be
arranged so
as to sufficiently cover the lumen walls 16 after deployment. In aspects, the
microfinger
arrays 410a-c may be swept along the vessel walls via torsional action of the
micro
surgical tool or aspect thereof. The microfinger arrays 410a-c are shown as
swept in a
counter clockwise direction 420 in the Fig. 4a. A local contact site between a
microfinger array 410c and the vessel wall 16 is shown in more detail in
blowup B. One
or more of the microfingers may include one or more sensing tips in accordance
with the
present disclosure. Fig. 4b shows a magnified view of blowup B. Three micro-
tips 430a-
b included in the microfinger array 410c are shown pressed against a local
tissue site of
the lumen wall 16. Each microtip includes a sensing tip 435a-c in accordance
with the
present disclosure. In this case, the sensing tips shown may be electrodes,
MMG sensing
elements, force sensing elements, temperature sensors, any sensing tip in
accordance with
the present disclosure, combinations thereof, or the like. A single micro-tip
430b is
shown with a full outline for further discussion. During a procedure, the
micro-tip 430b
may be swept, oscillated, etc. the tip will interact locally with the tissue
(e.g. via
transverse movement, changing contact forces, etc.). Such movement may be
directed
towards/away from the tissue surface (i.e. in a direction normal 445 to the
tissue surface),
and/or along the surface of the tissue (i.e. in a direction parallel 440 to
the tissue surface).
Equipped with an associated deflection sensing tip and/or an interfacial
pressure sensing
tip, these movements may be used to elucidate local physiological properties
(e.g.
mechanical compliance, tone, etc.) of the tissues. Alternatively,
additionally, or in
combination a suitably equipped micro-tip 430b may be used to measure local
mechanomyographic response, perhaps due to electrophysiological activity in
the vicinity
-44-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
of, or upstream from the tip 430b. Such information may be used for several
intended
purposes as detailed throughout this disclosure.
[00188] In aspects, the microfinger may be equipped with a needle electrode
tip
(perhaps formed as a structural extension of the flexure, etc.). The needle
electrode tip
may be configured such that upon applied torsion in a given direction, the
needle may
pierce the local tissues so as to enhance the electrical interface between the
mierofinger
and the tissues. Such a needle electrode tip may be integrated into one or
more
microfingers and/or sensing tips in accordance with the present disclosure.
[00189] Figs. 5a-c show some non-limiting aspects of micro-tips and/or tips of
one or
more microfingers in accordance with the present disclosure. Fig. 5a shows
schematic
diagrams for the cross sections of four non-limiting examples of micro-tips in
accordance
with the present disclosure (i.e. in this case including one or more exposed
electrode
sensing tips). Fig. 4a shows a schematic of the tip of a micro-finger 510
including a core
flexure 512 (e.g. a superelastic spring-like material, optionally electrically
conducting, a
wire, a flex circuit, a micro interconnect, etc.), with an isolating layer 514
(e.g. an oxide,
a dielectric coating, a radio-opaque coating, etc.) applied selectively to
regions thereof.
At the tip of the micro-finger 510, a region 516 of uncoated core flexure is
exposed. This
region 516 may, provide an electrode property for interacting with local
tissues, provide a
site for attachment of a microsensor, etc. In aspects, the exposed region 516
may be
coated with one or more electrode materials (i.e. one or more metals, alloys,
conducting
polymers, composites, carbon materials, conjugated polymers, combinations
thereof, or
the like). In the example shown, the exposed region 516 is oriented to one
side of the
neutral axis of the core flexure 512. Such orientation may be advantageous for
maintaining contact with an adjacent tissue surface while sweeping or moving
the micro-
tip 510, while biasing the micro-tip 510 against a tissue surface, etc. In
aspects, the
micro-tip 510 may be configured with a curvature, oriented so as to ensure the
exposed
region 516 will face an approaching tissue surface during deployment.
[00190] Fig. 5a shows a schematic of a micro-tip 520 in accordance with the
present
disclosure. The micro-tip 520 is shown with a core flexure 522 and an
insulating layer
524, each in accordance with the present disclosure. The micro-tip 520 may
include an
-45-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
axially oriented exposed region 526, located at the tip thereof. The axially
oriented
exposed region 526 may be configured for electrically interfacing with an
adjacent tissue,
with a sensing tip (i.e. a sensing tip in accordance with the present
disclosure), or the like.
Such a configuration may be advantageous for the simplicity of manufacture,
etc. The
core flexure 522 may be shaped in the vicinity of the exposed region 526 so as
to
efficiently interface against an adjacent tissue surface.
[00191] Fig. 5a further shows a schematic of a microfinger 530 in accordance
with the
present disclosure. The microfinger 530 may include a core-flexure 532 and an
insulating layer 534 each in accordance with the present disclosure. The
microfinger 530
may include an exposed region 536 in accordance with the present disclosure.
The core-
flexure 532 may be shaped to a point and/or an edge within the vicinity of the
exposed
region 536. Such a configuration may be advantageous to cause the microfinger
530 to
grip and/or penetrate into a tissue surface when brought into contact
therewith.
[00192] Fig. 5a shows a schematic of a micro-tip 540 in accordance with the
present
disclosure. The micro-tip includes a plurality of core flexures 542a-b, each
in accordance
with the present disclosure and one or more regions covered with an insulating
layer 544
in accordance with the present disclosure. In the example shown, the micro-tip
540 may
include a plurality of exposed regions 546a-b oriented along the length or
near the tip
thereof. The exposed regions 546a-b may act as electrode based sensing tips in
accordance with the present disclosure, may be configured so as to accept one
or more
sensing tips in accordance with the present disclosure. Such a configuration
may
advantageous for monitoring local electrophysiological signals, bioimpedance,
impedance between the tip region 546b and the shank region 546a (i.e. so as to
determine
if the tip is in contact with an adjacent fluid, etc.).
[00193] In aspects, the core flexures 542a-b may include a flex circuit with a
plurality
of interconnects. The exposed regions 546a-b may include a plurality of
contacts for
interfacing between the core flexures 542a-b and one or more sensing tips
attached
thereto.
-46-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00194] In aspects, one or more of the microfingers 510, 520, 530, 540 may
include
one or more electrical interconnects arranged along the length thereof, one or
more
distributed integrated circuit elements, etc.
[00195] In aspects, the microtip 510, 520, 530, 540 may include a plated
electrode
structure, a mushroom like electrode (e.g. so as to increase the contact
surface area
between the microtip and the tissues), a bent tip, a loop formation, a foot-
like electrode
element, etc.
[00196] In aspects, the microtip 510, 520, 530, 540 may be equipped with a
needle
electrode tip (perhaps foimed as a structural extension of the flexure, etc.).
The needle
electrode tip may be configured such that upon applied torsion in a given
direction, the
needle may pierce the local tissues so as to enhance the electrical interface
between the
microtip and the tissues.
[00197] Fig. 5b shows a ribbon like microfinger 550 in accordance with the
present
disclosure. The ribbon microfinger 550 may include a substrate 552 in
accordance with
the present disclosure, a spring-like material, a flexible polymeric material,
or any
combination thereof. As shown, the ribbon microfinger 550 includes electrical
interconnects 554 coupled to the substrate 552 for communicating one or more
electrical
signals along the length thereof as well as regions 556a-b at the tip suitable
for interacting
with local tissues (i.e. a site suitable for a sensing tip in accordance with
the present
disclosure). The electrical interconnects 554 may be coupled to one or more of
the
regions 556a-b (i.e. coupled with one or more electrode based sensing tips,
coupled to
one or more sensing tip interconnects, etc.).
[00198] Fig. Sc shows a helical ribbon microfinger 560 in accordance with the
present
disclosure. The helical ribbon microfinger 560 may include a plurality of
sensing tips
566, each coupled to a substrate 562 and optionally to one or more
interconnects 566
each in accordance with the present disclosure. The substrate 562 may include
one or
more embedded microcircuits 568, coupled to the sensing tips 566 and/or the
interconnects 566, so as to provide a signal conditioning function, switching
function,
multiplexing functionality, or the like, in accordance with the present
disclosure.
-47..
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00199] A ribbon microfinger 550, 560 may be configured so as to take on a
particular
shape (i.e. a hook like shape 559 as shown in Fig. 5b, a helical shape 569 as
shown in
Fig. Sc, or the like) upon deployment, perhaps from a delivery catheter in
accordance
with the present disclosure.
[00200] Such a ribbon microfinger 550, 560 may be attachable to a micro
balloon
catheter, wound around a stent-like mesh, etc. so as to provide support
thereto and/or to
bias the ribbon microfinger into the adjacent= tissues for purposes of
monitoring,
stimulating, and/or perfoiming a procedure (i.e. heating, ablating, abrading,
etc.).
[00201] In aspects, the ribbon microfinger 550, 560 may include one or more
circuit
elements 568 (e.g. a switch, an amplifier, etc.) in order to control direction
of, perform a
conditioning function to, alter the impedance of, etc. a signal passed along
the
microfingcr (i.e. to or from the micro-tip).
[00202] Figs. 6a-b show a microfinger 610 in accordance with the present
disclosure.
Fig. 6a shows an axial view of a microfinger 610 demonstrating an optional
multi-axial
curvature thereof, as well as a sweeping action 620 that may be achieved
therewith
during a procedure with the microfinger 610 biased against a lumen wall 17.
Fig. 6b
shows a longitudinal view of the same microfinger 610, demonstrating
additional
curvature thereof as well as contact between the microfinger 610 and coupled
sensing tip
630 with a local anatomical surface (i.e. in this case a vessel wall 17). An
arrow 620 is
shown in Figs. 6a-b to demonstrate the sense of rotation of the microfinger
610 as it is
swept over a vessel wall 17. A lumen axis 18 is also shown so as to
demonstrate the
approach for the microfinger 610 after deployment from a delivery catheter
(not
explicitly shown).
[00203] Figs. 7a-b show a micro-tip 710 including a MMG sensing element and a
response in accordance with the present disclosure. The micro-tip 710 includes
an
interfacial force sensing element 720 (e.g. a nanomaterial coating, a
piezoresistive
coating, a piezoelectric coating, etc.) and a flexural sensing element 730
(e.g. a
nanomaterial coating, a piezoresistive coating, a piezoelectric coating,
etc.). Both
elements 720, 730 may be coupled to the substrate 715 of the microtip 710. The
micro-
tip 710 maybe subsequently connected 740 to a controller or microcircuit (not
explicitly
-48-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
shown) via one or more interconnects, included in the micro-tip (i.e. along a
substrate
715 and/or core flexure thereof). Such electrical elements may be embedded
into the
substrate 715, into a delivery catheter (not explicitly shown), coupled to one
or more
elements of an associated surgical tool, or the like. The interfacial force
sensing element
720 may be configured to measure a contact force between the microfinger 710
and an
adjacent tissue surface. The flexural sensing element 730 may be configured to
measure
flexure of the micro-tip 710 during such interaction. Thus, via monitoring
signals from
both sensing elements 720, 730 a local compliance of the adjacent tissues may
be
measured/inferred (i.e. via measurement of contact force, flexure, and/or some
combination thereof).
[00204] Fig. 7b shows a time series of conditioned signals received from a
flexural
sensor and an associated interfacial force sensor during a stimulation event
750 (e.g.
perhaps as excited by another sensing tip included in an associated micro
surgical tool,
etc.). The stimulation and associated response from each sensor 720, 730 is
shown on the
time series (i.e. force sensing 760 and strain sensing 770 respectively). In
aspects, the
stimulation may be caused by an electrical stimulation event, perhaps
elsewhere in the
body, in a related neurological circuit, or the like. The combination of force
sensing 760
and strain sensing 770 signals may be combined to form an MMG signal. The
resulting
MMG signal(s) may be sufficiently free from electrical noise that may be
present when
measuring via alternative measures.
[00205] Figs. 8a-b show a schematic of a micro-tip 810 in accordance with the
present
disclosure. Fig. 8a shows a micro-tip 810 with an integrated temperature
sensing tip 840.
The temperature sensing tip 840 may include a bimetallic configuration, a
silicon sensing
element, an infrared sensing microcircuit, etc. The micro-tip 810 includes a
plurality of
electrical interconnects directed between the temperature sensing tip 840 and
a controller
850 (e.g. a local control circuit, an analog to digital converter, a local
signal amplifier,
etc.). The micro-tip 810 may include a substrate and/or core flexure 820 along
which
such electrical interconnects may be coupled. The micro-tip 810 may include
one or
more insulating layers 830 in accordance with the present disclosure.
-49-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
[00206] Fig. 8b shows a time series measurement from the temperature sensing
tip 840
during a series of local RF ablation pulses 860. Local temperature rise 870 as
measured
by the temperature sensing tip 840 may be used to control the overall pulse
width of each
RF pulse, the overall RF energy delivery, the RF power, etc. In aspects, such
information
may be coupled with one or more signals obtained from an associated sensing
tip in
accordance with the present disclosure. Such information may be collectively
used to
deteimine the extent of an ablation process, in deciding to continue with an
ablation
procedure, or the like.
[00207] Fig. 9 shows a micro surgical tool 910 deployed at a surgical site 19,
20, 21 in
accordance with the present disclosure. The micro surgical tool 910 includes a
delivery
catheter 915 and a plurality of microfinger arrays 920a-b, the microfinger
arrays 920a-b
penetrating into the renal artery 21 of a subject. The micro surgical tool 910
includes a
guide wire 940 (alternatively a guiding arm, a control arm, etc.) coupled to
the
microfinger arrays 920a-b such that they may be controlled by an external
operator,
robot, etc. (i.e. coupled 950 to the micro surgical tool 910). In the
arrangement shown,
one of the microfinger arrays 920b is attached to a local signal conditioning
integrate
circuit 930, positioned so as to provide conditioning of signals sensed at the
microfinger
tips 920b, perhaps to convert the signals into digital forms, to provide a low
impedance
source, etc. The other microfinger array 920a is oriented adjacent to an
anatomical site
20 of interest (i.e. in this case nerve plexus). The presence/location of the
anatomical site
20 of interest may have been determined via monitoring of one or more sensing
tips
within the microfinger arrays 920a-b during a sweeping procedure, etc. Having
identified/located the anatomical site 20 of interest, the operator,
controller, etc. 950 may
perfortn a surgical procedure thereupon.
[00208] Figs. 10a-d show non-limiting examples of monitoring methods in
accordance
with the present disclosure.
[00209] Fig. 10a shows a lumen (i.e. a vessel, a vein, an artery, a renal
artery, etc.)
prior to a surgical process. Two sensing sites are shown, distal 1015 and
proximal 1010
to the intended surgical site. An ablation catheter tip 1020 (e.g. although
shown as a
separate unit, it may be included in an associated microfinger array as a
sensing tip in
-50-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
accordance with the present disclosure) including an ablation electrode 1025
is placed in
contact with the tissues between the sensing sites. One or more sensing tips
may be
placed at the sensing sites 1010, 1015, among others, as well as optionally at
the surgical
site (i.e. to perform a combination of sensing and procedures). Prior to
initiation of the
surgical procedure, nervous activity may be detected at both sensing sites
1010, 1015. In
aspects, the correlation between the electrophysiological signals (i.e.
neurological signals,
electromyographic signals, mechanical myographic signals, etc.) may be
relatively high
prior to initiation of a surgical procedure. In aspects, the correlation
between the
electrophysiological signals may include the step of extracting a portion of
each signal
that is substantially common to both signals for analysis.
[00210] Fig. 10b shows a lumen (i.e. a renal artery) after a surgical
ablation process.
Two sensing sites are shown, distal 1040 and proximal 1035 to the surgical
site. An
ablation catheter tip 1020 (e.g. although shown as a separate unit, it may be
included in
an associated microfinger array as a sensing tip in accordance with the
present disclosure)
equipped with an ablation electrode 1030 is placed in contact with the tissues
between the
sensing sites. The ablation catheter tip 1020 has been employed as part of a
surgical
procedure to form an ablation zone 1030, in this case shown substantially
around the
circumference of the arterial wall around the surgical site forming the
ablation zone 1030.
After completion of the ablation procedure, nervous activity may no longer be
detected at
one or more of the sensing sites 1035. In this example, the ablation procedure
has
substantially blocked afferent nerve traffic from proceeding through the
ablation zone
1030. In aspects, efferent nerve traffic may still be detectable at the
proximal sensing site
1035, and afferent nerve traffic may still be detectable at the distal sensing
site 1040. The
correlation between the resulting signals may be used to quantify the state of
the ablation
process, the extent of denervation, etc.
[00211] In aspects, the above method and variations thereof may be used to
extract the
afferent from the efferent nerve traffic in the vicinity of a surgical site of
interest. In
aspects, the surgical procedure may include the application of energy to the
surgical site
in a substantially low dosage so as to temporarily inhibit function of the
neurological
anatomy in the vicinity thereof In one non-limiting example, the energy may be
used to
heat the local tissues to a temperature of greater than 40C, 45C, 50C so as to
form the
-51-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
temporary block. Signals obtained by the distal and proximal sensing sites
1035, 1040
may be used to determine when the block has occurred, how the block has
affected the
traffic, and to distinguish, post block, infoiniation about the efferent and
afferent nerve
traffic in the vicinity of the surgical site.
[00212] In aspects, following a temporary block, if the procedure has
favorably altered
the neurological traffic, a more durable procedure may be completed (i.e. an
ablation
procedure, a chemical &nervation, a thermal ablation process, a radiation
based ablation,
etc.). Such an approach may be advantageous for safely determining the ideal
targets for
a surgical procedure, for minimizing damage to the surrounding tissues in
completing a
denervation procedure, and the like.
[00213] Fig. 10c shows a lumen (i.e. a renal artery) prior to a surgical
process. Two
sensing sites are shown, distal 1050 and proximal 1045 to the intended
surgical site and a
pacing site 1055 is shown located to one side of the intended surgical site.
An ablation
catheter tip 1020 (e.g. although shown as a separate unit, it may be included
in an
associated microfinger array as a sensing tip in accordance with the present
disclosure) is
placed in contact with the tissues between the sensing sites. One or more
sensing tips
may be placed at the sensing sites 1045, 1050, among others, as well as
optionally at the
surgical site (i.e. to perform a combination of sensing and procedures). Prior
to initiation
of the surgical procedure, both a pacing signal 1055 as well as associated
nervous activity
may be reliably detected at both sensing sites 1045, 1050. The pacing signal
1055 may
be used to detemiine a transmission velocity along the associated anatomy
between the
pacing site 1055 and each of the sensing sites 1045, 1050, may be used to
deteimine the
transmission characteristics of the anatomy between sites, etc. In aspects,
the coherence
between the electrophysiological signals (i.e. neurological signals,
electromyographic
signals, mechanical myographic signals, etc.) may be relatively high prior to
initiation of
a surgical procedure. In aspects, the coherence in combination with the pacing
signal
may be advantageous in extracting the relevant information to make an
assessment of
neurological function quickly and reliably, even in the presence in
considerable
background noise, movement, and physiologically relevant neurological
activity.
-52-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00214] In aspects, the step of evaluating the coherence between the
electrophysiological signals may include the step of extracting a portion of
each signal
that is substantially common to both signals for analysis.
[00215] Fig. 10d shows a renal artery after a surgical ablation process. Two
sensing
sites are shown, distal 1065 and proximal 1060 to the surgical site and a
pacing site 1070
is shown located to one side of the intended surgical site. An ablation
catheter tip 1020
(e.g. although shown as a separate unit, it may be included in an associated
microfinger
array as a sensing tip in accordance with the present disclosure) with an
ablation
electrode 1025 is placed in contact with the tissues between the sensing sites
1060, 1070.
The ablation catheter tip 1020 has been swept around the circumference of the
arterial
wall around the intended surgical site forming an ablation zone 1030. After
completion
of the ablation procedure, nervous activity may no longer be detected at one
or more of
the sensing sites 1060, 1065 even under the continued action of the pacing
signal 1070.
[00216] In aspects, one or more of the distal sensing 1015, 1040, 1050,
1065, proximal
sensing 1010, 1035, 1045, 1060, pacing 1055, 1070, and surgical procedure
(i.e.
fotination of a blocked region, an ablation zone 1030, etc.) may be completed
by one or
more sensing tips each in accordance with the present disclosure.
[00217] Figs. lla-f show some non-limiting examples of ablation patterns
applied to a
renal artery in accordance with the present disclosure.
[00218] Fig. ha shows a lumen 1105 (i.e. a tubule, a vessel, an artery, a
vein, a renal
artery, etc.) prior to the application of a surgical procedure thereto. As
outlined in the
Figure, a range of neurological structures (i.e. nerve plexuses) 1110, 1115,
1120, are
visible within the wall and surrounding adventitia of the lumen 1105. In
aspects, the
lumen 1105 may provide a conduit for flow of a fluid (i.e. blood, bile, lymph,
urine,
feces, etc.), and to interconnect one or more organs, and or aspects of an
organ (i.e. an
intra-organ vessel).
[00219] Fig. 1 lb shows the lumen (i.e. the renal artery) with a
circumferentially
ablated region 1125 generated by a micro surgical tool in accordance with the
present
disclosure. Sensing tips located to either side or within the ablation zone
1125 may be
used to confirm effective ablation, control the ablation process itself, for
decision making
-53-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
related to the size and placement of the ablation site, to limit the overall
amount of
damage caused by the ablation procedure, or the like. In this non-limiting
example, the
associated ablation zone 1125 may be produced by collective activation of a
plurality of
sensing tips, arranged around the circumference of the lumen 1105, by swept
motion of
one or more sensing tips during a procedure, or the like.
[00220] Fig. 11c shows the lumen (i.e. the renal artery) after a
selectively targeted
nerve bundle 1127 has been ablated by a micro surgical tool in accordance with
the
present disclosure. Sensing tips included in the micro surgical tool may have
been used
to locate target tissues for ablation, monitor the ablation process itself, to
avoid ablation
of nerve bundles that are not to be surgically treated, to confirm effective
ablation, and to
limit the overall amount of damage caused by the ablation procedure. In this
non-
limiting example, the nerve bundle 1127 is ablated at local sites 1130 along
the length
thereof so as to limit damage to the surrounding tissues. In aspects, such
ablation profiles
may be formed during collective activation of a plurality of sensing tips each
in
accordance with the present disclosure, via selective ablation of tips during
a longitudinal
sweeping process, via selective ablation of tips during a tracking process,
combinations
thereof, or the like.
[00221] Fig. 1 1 d shows the lumen (i.e. renal artery) after a group of
selectively
targeted nerve bundles 1132, 1134 have been ablated by a micro surgical tool
in
accordance with the present disclosure. In aspects, sensing tips included in
the micro
surgical tool may have been used to locate target tissues for ablation,
monitor the ablation
process itself, to avoid ablation of nerve bundles that are not to be
surgically treated, to
confirm effective ablation, and to limit the overall amount of damage caused
by the
ablation procedure. In this non-limiting example, the nerve bundles 1132, 1134
are
ablated at local sites 1135 along the length thereof so as to limit damage to
the
surrounding tissues. Local sites 1135 may be placed so as to minimize
potential damage
to nearby anatomical features, which may not be intended targets for the
surgical
procedure.
[00222] Fig. lie shows the lumen (i.e. renal artery) after a selectively
targeted nerve
bundle 1142 has been ablated by a micro surgical tool in accordance with the
present
-54-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
disclosure. In aspects, sensing tips included in the micro surgical tool may
have been
used to track the targeted tissues as the ablation process is occurring (so as
to establish an
ablation path along the target tissues), locate target tissues for ablation,
monitor the
ablation process itself, to avoid ablation of nerve bundles that are not to be
surgically
treated, to confinn effective ablation, and to limit the overall amount of
damage caused
by the ablation procedure. In this non-limiting example, the nerve bundle 1142
is ablated
along a continuous strip 1140 as followed with guidance from the sensing tips.
Such
long stretch of ablated tissue may be employed to limit the potential for
regeneration after
the surgical procedure has been completed. In aspects, feedback from signals
obtained
from one or more sensing tips may be used to guide the surgical hardware
during the
surgical procedure (i.e. ablation, chemical substance delivery, etc.).
[00223] Fig. 1 if shows the lumen (i.e. renal artery) after selectively
targeted nerve
bundles 1152, 1154, 1156 have been ablated by a micro surgical tool in
accordance with
the present disclosure. In aspects, the sensing tips included in the micro
surgical tool
may have been used to locate target tissues for ablation, to identify tissues
for ablation,
monitor the ablation process itself, to avoid ablation of nerve bundles that
are not to be
surgically treated, to confirm effective ablation, and to limit the overall
amount of
damage caused by the ablation procedure. A substantially helical tool path
1150 is
shown, as the micro surgical tool traced around the walls of the renal artery
during the
ablation procedure. In this non-limiting example, the nerve bundles 1152,
1154, 1156 are
ablated at a plurality of local sites 1145 along the length thereof so as to
limit damage to
the surrounding tissues. In aspects, the targeted neuroanatomical structures
1152, 1154,
1156 may be treated along the length thereof in order to control the post-
surgical
regrowth rate, or the like.
[00224] Fig. llf shows a lumen 1160 (i.e. a vessel, an artery, a renal
artery, a vein, a
tubule, etc.) after a selectively targeted treatment zones 1175, 1180, 1185
have been
formed around target anatomical structures 1065, 1067, 1069 by a micro
surgical tool in
accordance with the present disclosure. Target neurological structure 1165,
perhaps
connecting one or more structures 1170 in the vicinity of the lumen 1160 to
one or more
external organs, ganglia, or the like, that are somewhat removed from the
lumen 1160
may be targeted as well during such procedures. In aspects, sensing tips
included in the
-55-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
micro surgical tool may have been used to track the targeted tissues as the
surgical
process is occurring (so as to establish a treatment path in the vicinity of
the target
tissues), locate target tissues for treatment, monitor the treatment process
itself, to avoid
unintentional treatment of nerve bundles that are not to be surgically
treated, to confirm
effective treatment, and to limit the overall amount of damage caused by the
treatment
procedure. In this non-limiting example, the target anatomical structures
1165, 1167,
1169 are treated along one or more pathways 1177, 1182, 1187 as followed with
guidance from one or more of the sensing tips (or via collective local
treatment by a
collection of sensing tips). Such stretches and strategic placement of
treatment zones
1175, 1180, 1185 may be employed to limit the potential for regeneration after
the
surgical procedure has been completed. In aspects, feedback from signals
obtained from
one or more sensing tips may be used to guide the surgical hardware during the
surgical
procedure (i.e. ablation, chemical substance delivery, cryoablation, energy
delivery,
abrasion, etc.).
[00225] Fig. 12a shows a schematic diagram of a micro surgical tool 1210
deployed at
a surgical site in accordance with the present disclosure. The micro surgical
tool 1210 is
shown deployed into a renal artery 1202 of a subject having passed through a
superior or
inferior approach (brachial or femoral arteries), via aortic artery 1205 and
into the renal
artery 1202 (or to the mouth thereof). The micro surgical tool 1210 includes a
delivery
catheter 1224 and a microfinger array 1212 in accordance with the present
disclosure,
shown in contact with the walls of the renal artery 1203 (i.e. biased towards,
in controlled
contact with, penetrating into, etc.). Connected to the microfinger array 1212
via a
guiding aim 1215 is a local control circuit 1220 in accordance with the
present
disclosure. In aspects, the guiding arm 1215 may include one or more
electrical
interconnects, one or more structural elements, a conduit, or the like coupled
to the
microfinger array 1212 and/or the local control circuit 1220. The control
circuit 1220
may route signal traffic to and from the microfinger array 1212, etc. The
schematic
further depicts application of RF current 1221 applied locally between sensing
tips in the
microfinger array 1212 as well as an alternative RF current 1223 between one
or more
sensing tips in the microfinger array 1212 and an external electrode (not
explicitly
shown). The catheter 1224 may be coupled to an operator 1226, a controller, a
signal
-56-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
conditioning circuit, or the like for controlling the microfinger array 1212
during a
procedure. In aspects, the microfinger array 1212 may be advanced and/or
retracted
1227, along the lumen 1203 and/or rotated 1229 around the circumference of the
lumen
1203 during procedures related to searching for anatomical sites of interest,
performing
sensing, mapping, surgical treatments, ablation, or the like.
[00226] Fig. 12b shows a schematic diagram of a micro surgical tool 1230
deployed at
a surgical site in accordance with the present disclosure. The micro surgical
tool 1230 is
shown deployed into a renal artery 1202 of a subject having passed through a
superior or
inferior approach (brachial or femoral arteries), via aortic artery 1205 and
into the renal
artery 1202 (or to the mouth thereof). The micro surgical tool 1230 includes a
delivery
catheter 1232 and a microfinger array 1234 in accordance with the present
disclosure,
shown in contact with the walls of the renal artery 1203 (i.e. biased towards,
in controlled
contact with, penetrating into, etc.). In this, non-limiting example, the
microfinger array
1234 is configured as a longitudinal wire cage in accordance with the present
disclosure.
Such a configuration may be advantageous to maintain contact with the lumen
walls
during a procedure without inhibiting flow of fluids through lumen. Connected
to the
microfinger array 1234 via a guiding arm 1238 is a local control circuit 1236
in
accordance with the present disclosure. In aspects, the guiding arm 1238 may
include
one or more electrical interconnects, one or more structural elements, a
conduit, or the
like coupled to the microfinger array 1234 and/or the local control circuit
1236. The
micro-surgical tool 1230 is also configured to accommodate, or includes a
guide wire
1240 configured to assist with guiding the microfinger array 1234 to the
target
anatomical site. The microfinger array 1234 may be coupled to a distal ringlet
1241 or
equivalent feature, configured to accommodate the passage of the guide wire
1240 there
through during the procedure. In aspects, the control circuit 1236 may route
signal traffic
to and from the microfinger array 1234, etc. The schematic further depicts
application of
RF current 1243 applied locally between sensing tips in the microfinger array
1234 as
well as an alternative RF current 1245 between one or more sensing tips in the
microfinger array 1234 and an external electrode (not explicitly shown). The
catheter
1236 and/or guiding aim 1238 may be coupled to an operator 1226, a controller,
a signal
conditioning circuit, or the like for controlling the microfinger array 1234
during a
-57-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
procedure. In aspects, the microfinger array 1234 may be advanced and/or
retracted
1247, along the lumen 1203 and/or expanded/contracted 1238 as part of a
procedure, a
deployment, and/or a retraction procedure within the lumen 1203 during
procedures
related to searching for anatomical sites of interest, performing sensing,
mapping,
surgical treatments, ablation, or the like.
[00227] Fig. 12c shows a schematic diagram of a micro surgical tool 1250
deployed at
a surgical site in accordance with the present disclosure. The micro surgical
tool 1250 is
shown deployed into a renal artery 1202 of a subject having passed through a
superior or
inferior approach (brachial or femoral arteries), via aortic artery 1205 and
into the renal
artery 1202 (or to the mouth thereof). The micro surgical tool 1250 includes a
delivery
catheter 1258 and a plurality of microfinger arrays 1252, 1254 each in
accordance with
the present disclosure, shown in contact with the walls of the renal artery
1203 (i.e.
biased towards, in controlled contact with, penetrating into, etc.). In this,
non-limiting
example, the microfinger arrays 1252, 1254 are configured as a radially biased
flexural
springs, in accordance with the present disclosure. Such a configuration may
be
advantageous to maintain contact with the lumen walls during a procedure
without
inhibiting flow of fluids through lumen, to accommodate a wide range of
anatomical
features, to maintain a relatively constant bias force on the lumen walls 1203
during a
procedure, for simple deployment/retraction, combinations thereof, or the
like.
Connected to the microfinger arrays 1252, 1254 via one or more guiding arms
1256, 1257
is a local control circuit 1260 in accordance with the present disclosure. In
aspects, the
guiding arm(s) 1256, 1257 may include one or more electrical interconnects,
one or more
structural elements, a conduit, or the like coupled to the microfinger arrays
1252, 1254
and/or the local control circuit 1260. The micro-surgical tool 1250 may also
be
configured to accommodate, and/or include a guide wire (not explicitly shown)
configured to assist with guiding the microfinger arrays 1252, 1254 to the
target
anatomical site. In aspects, one or more of the guiding arms 1256, 1257 may be
configured so as to retract and or advance along the mierosurgical tool 1250
with respect
to the microfinger arrays 1252, 1254 so as to cover and/or expose one or more
of the
microfinger arrays 1252, 1254 during a retraction and/or deployment process.
In aspects,
the control circuit 1260 may route signal traffic to and from one or more of
the
-58-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
microfinger arrays 1252, 1254, etc. The schematic further depicts application
of RF
current 1261 applied locally between sensing tips in the microfinger arrays
1252, 1254 as
well as an alternative RF current 1263 between one or more sensing tips in the
microfinger arrays 1252, 1254 and an external electrode (not explicitly
shown). The
catheter 1260 and/or guiding arm(s) 1256, 1257 may be coupled to an operator
1226, a
controller, a signal conditioning circuit, or the like for controlling the
microfinger arrays
1252, 1254 during a procedure. In aspects, the microfinger arrays 1252, 1254
may be
advanced and/or retracted 1265, along the lumen 1203 and/or deployed or
retracted by
movement 1267 of one or more guiding arms 1256, 1257 during deployment, and/or
a
retraction procedure within the lumen 1203 during procedures related to
searching for
anatomical sites of interest, perfoiming sensing, mapping, surgical
treatments, ablation,
or the like. In aspects, the microsurgical tool 1250 may be advanced after
deployment of
the microfinger arrays 1252, 1254 so as to strongly bias and/or penetrate one
or more
sensing tips in the microfinger arrays 1252, 1254 into the wall 1203 of the
lumen 1205
during a procedure.
[00228] Fig. 12d shows a schematic diagram of a micro surgical tool 1270
deployed at
a surgical site in accordance with the present disclosure. The micro surgical
tool 1270 is
shown deployed into a renal artery 1202 of a subject having passed through a
superior or
inferior approach (brachial or femoral arteries), via aortic artery 1205 and
into the renal
artery 1202 (or to the mouth thereof). The micro surgical tool 1270 includes a
delivery
catheter 1272 and a plurality of sensing tips 1274 arranged over a balloon
1275 each in
accordance with the present disclosure, shown in contact with the walls of the
renal artery
1203 (i.e. biased towards, in controlled contact with, penetrating into,
etc.). In this, non-
limiting example, one or more of the sensing tips 1274 maybe arranged along
the ballon
1275 walls so as to contact the lumen wall 1203 during and/or after
deployment. Such a
configuration may be advantageous for isolating one or more of the sensing
tips 1274
from the fluid which would notmally flow through the lumen 1202. Connected to
the
balloon 1275 and one or more of the sensing tips 1274, via a guiding arm 1277
is a local
control circuit 1280 in accordance with the present disclosure. In aspects,
the guiding
arm 1277 may include one or more electrical interconnects, one or more
structural
elements, a conduit for delivery/removal of fluid to/from the balloon 1275, or
the like
-59..
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
coupled to the sensing tips 1274 and/or the local control circuit 1280. The
micro-surgical
tool 1270 may also be configured to accommodate, and/or include a guide wire
(not
explicitly shown) configured to assist with guiding the balloon 1275 to the
target
anatomical site. In aspects, the balloon 1275 and/or guiding arm 1277 may be
coupled to
a distal ringlet 1282 or equivalent feature, configured to fasten the balloon
to the guiding
arm 1277 and/or to accommodate the passage of the guide wire 1240 there
through
during the procedure. In aspects, the guiding aim 1277 may be configured so as
to retract
and or advance along the microsurgical tool 1270 with respect to the balloon
1275 so as
to cover and/or expose one or more of the sensing tips 1274 during a
retraction and/or
deployment process. In aspects, the control circuit 1280 may route signal
traffic to and
from one or more of the sensing tips 1274, etc. The schematic further depicts
application
of RF current 1281 applied locally between sensing tips 1274 as well as an
alternative RF
current 1283 between one or more sensing tips 1274 and an external electrode
(not
explicitly shown). The catheter 1272 and/or guiding arm 1277 may be coupled to
an
operator 1226, a controller, a signal conditioning circuit, or the like for
controlling the
sensing tips 1274 during a procedure. In aspects, the balloon 1275 may be
repositioned
1285 along the lumen 1203 and/or expanded or contracted 1287 during
deployment,
and/or a retraction procedure within the lumen 1203 during procedures related
to
searching for anatomical sites of interest, performing sensing, mapping,
surgical
treatments, ablation, or the like.
[00229] Fig. 13 shows a schematic diagram of interaction between one or more
macroelectrodes 1302, 1304, 1306 (i.e. not limited to three, could be a range
of
possibilities) and a micro surgical tool 1310 deployed at a surgical site in
accordance with
the present disclosure. The abdomen 1301 of a subject is shown with an
internally placed
micro surgical tool 1310 (dotted line) located with the tip 1315 in a renal
artery of the
subject. 'When a suitable target site for an ablation process is determined,
the electrical
impedance (e.g. DC impedance, AC impedance, real, imaginary, complex impedance
spectra, etc.) between elements of the network formed by one or more sensing
tips 1315
included in the micro surgical tool 1310, one or more macroelectrodes 1302,
1304, 1306
placed on the body of the patient, pushed up against the patient, located
along the catheter
wall of the micro surgical tool, placed within the patient (perhaps
endoscopically, via a
-60-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
catheterization procedure, etc.) may be monitored. Such
relational impedance
measurements are depicted diagrammatically with arrows 1321, 1323, 1325, 1331,
1333,
1335 between elements 1302, 1304, 1306, 1310, 1315 of the network in Fig. 13.
[00230] Based upon the impedances in the associated network, an RF ablation
current
may be applied between two or more elements 1301, 1304, 1306, 1310, 1315
thereof. In
one non-limiting example, each element 1301, 1304, 1306, 1310, 1315 of the
network
may include a controllable impedance circuit. The impedance control circuits
may be
used to draw a portion of the RF current into/out of the associated element
1301, 1304,
1306, 1310, 1315. In this sense, local control of the RF current at the
sensing tips 1315
may be more precisely controlled. Electric field strengths, current flow, etc.
may be
monitored at any element 1301, 1304, 1306, 1310, 1315 of the network so as to
determine the RF current flow path into the local tissues of the target
anatomy (i.e. into a
wall of a lumen, a renal artery, etc.).
[00231] Fig. 14 shows a micro balloon catheter 1410 deployed at a surgical
site in
accordance with the present disclosure. In this non-limiting example, the
micro balloon
catheter 1410 is shown deployed into a renal artery 1402 of a subject having
passed
through a superior or inferior approach (brachial or femoral arteries), via
aortic artery
1405 and into the renal artery 1402 (or to the mouth thereof). The micro
balloon catheter
1410 is shown with a layer of indicating agents 1415 and/or contrast agent
coated onto
the balloon 1420 thereof. The micro balloon catheter 1410 is shown as placed
within the
renal artery 1402 of a subject, in an inflated state. In this state, the
indicating agents 1415
and/or contrast agents are released 1419 (i.e. via diffusion, active
transport, etc.) into the
surrounding tissues for later use during a surgical procedure. In aspects, the
micro
balloon catheter 1410 may include one or more sensory tips, a delivery
catheter 1422, a
guiding arm 1424, coupled to the balloon 1420 in accordance with the present
disclosure.
The catheter 1422 may be coupled to an operator 1426, a controller, a signal
conditioning
circuit, or the like for controlling the balloon 1420 during a procedure. In
aspects, the
indicating agent 1415 may be configured so as to change chromatic and/or
photochemical
properties in the presence, when bound to, or when incorporated into the
target anatomy.
-61-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00232] Figs 15a-b show aspects of non-limiting examples of optical
microsensing tips
1510 and a collective response therefrom in accordance with the present
disclosure. Fig.
15a shows an array of optical microsensing tips 1510 biased towards the wall
1501 of a
vessel. The optical microsensing tips 1510 are configured to receive from
and/or emit
energy into the adjacent tissues of the wall 1501. In aspects, an external
light source
1515 may also provide light towards the surgical site (i.e. vessel walls
1501). In aspects,
energy 1520 passing through an anatomical site of interest 1503 may be
accepted by one
or more of the optical microsensing tips 1510, each configured to generate a
signal
therefrom. In aspects, the microsensing tips 1510 may include a fiber optic
element
coupled to a remote light source and/or photodetector. Such a configuration
may be
coupled with the indicating agents described in Fig. 15 (i.e. so as to locate
the target
anatomy as part of a surgical procedure). In aspects, the indicating agent
1415 may be
configured so as to change chromatic and/or photochemical properties in the
presence,
when bound to, or when incorporated into the target anatomy 1503, thus being
detectable
by one or more optical microsensing tips 1510.
[00233] Fig. 15b shows a spectral response of the light received by the
optical
microsensing tips 1510 and that emitted by an external light source. The
detected signals
1535, 1540 may be used to determine the location of target tissues in the
vessel wall. In
aspects, the optical microsensing tips 1510 may include one or more electrode
elements
so as to selectively and locally ablate target tissues based on the response
of the sensed
signals 1535, 1540.
[00234] Fig. 16 shows a combination catheterization and endoscopic procedure
on a
renal artery in accordance with the present disclosure. A micro surgical tool
1610 in
accordance with the present disclosure is shown as placed into the renal
artery of a
subject. One or more endoscopically placed light sources 1615, 1617 may be
shone at
the renal artery. In aspects, the light sources 1615, 1617 may be multi-band
sources,
broadband sources, narrow band sources, modulated, or any combination thereof.
In
aspects, the micro surgical 1610 tool may include one or more optical
microscnsors to
receive such light, the processed signals used to deteimine the location of
target tissues in
the renal artery. In aspects, the microsurgical tool 1610 may include one or
more sensing
tips in accordance with the present disclosure to selectively treat target
anatomy based on
-62-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
the determined locations thereof. An optional endoscopically placed camera
1620 is also
shown. In aspects, the camera 1620 may include a light source. The camera 1620
may be
used as part of a feedback mechanism to control placement of the micro
surgical tool
1610 in the renal artery. In aspects, the camera 1620 may use a range of light
sources to
elicit placement information of target anatomy (perhaps in combination with
indicating/contrast agents in accordance with the present disclosure),
placement of the
micro surgical tool 1610 within the renal artery, and/or monitoring of the
surgical
procedure (i.e. ablation procedure, chemical denervation, chemical deployment,
etc.).
Such a feedback mechanism may be used to precisely guide the micro surgical
tool 1610
during a surgical procedure (i.e. ablation procedure, etc.). In one non-
limiting example
the camera 1620 and/or a light source 1615, 1617 may include a macroelectrode
in
accordance with the present disclosure.
[00235] Fig. 17a shows a schematic diagram of aspects of a micro surgical tool
in
accordance with the present disclosure. The micro surgical tool includes a
plurality of
microfingers 1710 equipped with sensing tips 1715 in accordance with the
present
disclosure, a local control circuit 1720 (optionally located near to the tip
of the micro
surgical tool) in accordance with the present disclosure, and a guiding arm
1726 and a
delivery catheter 1724 to connect, both mechanically and electrically the tip
of the tool to
an operator, robot, etc. In aspects, the local control circuit 1720 may be
located at the
operating end of the catheter 1724 (i.e outside the body of a subject during
use).
Alternatively, additionally, or in combination a control circuit 1720 may be
coupled to
the microfingers 1710 directly, or via the guiding aim 1726 in order to
communicate
signals to or from the sensing tips 1715 during a procedure. In aspects, the
microfingers
1710 may be configured so as to bend within the body of a subject (i.e. after
a
deployment process, etc.) so as to bias toward the walls of the anatomy of
interest, etc. In
aspects, the microfingers 1710 may be directed (i.e. like a pencil) towards
the wall of an
organ, and/or anatomical feature so as to form a plurality of microcontacts at
each
sensing tips, for the purposes of mapping, sensing, performing a treatment,
etc.
thereupon.
[00236] Fig. 17b shows a schematic diagram of aspects of a micro surgical tool
in
accordance with the present disclosure. The micro surgical tool includes a
plurality of
-63-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
microfinger arrays 1730a-b equipped with sensing tips 173la-b, a guiding aim
1732, and
a delivery catheter 1734 each in accordance with the present disclosure. In
aspects, the
guiding arm 1732, one or more microfingers 1730a-b, and/or the catheter 1734
may
include a control circuit in accordance with the present disclosure. In
aspects, a local
control circuit 1720 in accordance with the present disclosure may be located
at the
operating end of the catheter 1734 (i.e outside the body of a subject during
use).
Alternatively, additionally, or in combination a control circuit may be
coupled to one or
more of the microfingers 1730a-b directly, or via the guiding arm 1732 in
order to
communicate signals to or from the sensing tips 1731a-b during a procedure. In
aspects,
the microfinger arrays 1730a-b may be configured so as to bend within the body
of a
subject (i.e. after a deployment process, etc.) so as to bias toward the walls
of the
anatomy of interest, etc. In aspects, one or more of the microfingers 1730a-b
may be
configured so as to bend when heated to body temperature (i.e. so as to self-
deploy
during a procedure). In aspects, the guiding atm 1732 and/or the catheter 1734
(and/or a
sleeve thereupon) may be retracted 1736 to initiate a deployment process so as
to expose
one or more of the microfingers 1730a-b and bring them into contact with the
intended
anatomy. In aspects, one or more sensing tips 173l a-b may be equipped with
one or
more electrodes for electrophysiological sensing, stimulation, and/or RE
current delivery
to the surrounding tissues. Thus signals may be monitored between 1738 sensing
tips
173l a-b in different microfinger arrays 1730a-b or within 1739 the same
microfinger
array 1730a, 1730b. In aspects, the guiding arm 1732 and/or catheter 1736 may
be
adjustable so as to adjust the distance between microfinger arrays 1730a-b in
the micro
surgical tool tip.
[00237] Fig. 17c shows a schematic diagram of aspects of a micro surgical tool
in
accordance with the present disclosure. The micro surgical tool includes a
longitudinal
wire cage including a plurality of microfingers 1740a-b, with regions coupled
to sensing
tips 1742a-f in accordance with the present disclosure. Such a configuration
may be
advantageous to maintain contact between one or more sensing tips 1742a-f with
the
lumen walls during a procedure without inhibiting flow of fluids through
lumen. In
aspects, the micro-surgical tool may be configured to accommodate, or to
include a guide
wire (not explicitly shown) configured to assist with guiding wire cage to the
target
-64-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
anatomical site. In aspects, the wire cage may be coupled to a distal ringlet
1750 or
equivalent feature, configured to accommodate the passage of a guide wire
there through
during the procedure. The schematic further depicts application of RF current
1752
applied locally between sensing tips 1742e,f in the wire cage. The wire cage
may be
coupled to a delivery catheter 1754, perhaps coupled as a sleeve that can
extend over the
wire cage so as to force collapse thereof In aspects, retraction 1760 of the
delivery
catheter 1754 may be used to deploy the wire cage during a procedure. The
catheter
1754 and/or an enclosed guiding aim (not explicitly shown) may be coupled to
an
operator, a controller, a signal conditioning circuit, or the like for
controlling the sensing
tips 1742a-f, and/or the wire cage during a procedure. In aspects, the wire
cage may be
advanced and/or retracted, along a lumen (not explicitly shown) and/or
expanded/contracted as part of a procedure, a deployment, and/or a retraction
procedure
within the lumen during procedures related to searching for anatomical sites
of interest,
performing sensing, mapping, surgical treatments, ablation, or the like.
[00238] In aspects, one or more sensing tips 1742a-f may be equipped with
one or
more electrodes for eleetrophysiological sensing, stimulation, and/or RF
current delivery
to the surrounding tissues. Thus signals may be monitored between sensing tips
1742a-f,
between a sensing tip 1742a-f and an external electrode, etc.
[00239] In aspects, one or more sensing tips 1742a-f may be arranged
longitudinally
along the axis of the microsurgical tip, such that the sensing tips 1742a-f
are biased
against a lumen wall at site spaced along the longitudinal direction thereof
upon
deployment.
[00240] Figs. 18a-f show aspects of non-limiting examples of micro surgical
tools in
accordance with the present disclosure.
[00241] Fig. 18a shows a microfinger array in accordance with the present
disclosure.
The array includes five microfingers 1810 each equipped with one or more
sensory tips
1815 in accordance with the present disclosure. The microfingers 1810 and
associated
sensing tips 1815 are shown biased against a tissue surface 1801. Interaction
between
two of the sensing tips 1815 is depicted with an arrow 1820 in the diagram.
-65-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00242] Fig. 18b shows a time series of data collected by several sensing tips
1815 in
the microfinger array 1810. The neurological activity 1825 of a local tissue
site is
monitored 1827. At a ablation start time 1826, an ablation current 1829 is
sent through
one or more sensing tips 1815 and an altered neurological activity 1831 is
confirmed
afterwards.
[00243] Fig. 18c shows an aspect of a mesh-like array 1840 of interwoven wires
1842
(i.e. microfingers in accordance with the present disclosure), with associated
sensing tips
1844. The sensing tips 1844 may be arranged such that they contact the local
tissues
1802 when the mesh 1840 is biased against the tissue 1802. In one non-limiting
example,
the mesh-like array 1840 may be formed from an interwoven group of
superelastic wires
(e.g. Nitinol wires, spring steel wires, etc.). The mesh 1840 may be formed as
a sock,
webbing, an arched structure, a donut, a net, etc. Upon deployment to the
surgical site,
the mesh 1840 may expand so as to contact the local tissues 1802 of interest.
Electrical
interconnects for the sensing tips 1844 may be provided via the wires 1842,
routed along
the wires 1842, etc. In on non-limiting example, substrates in accordance with
the
present disclosure may be interwoven instead of the wires 1842 as shown. Such
substrates may be used to form a deployable mesh like structure complete with
electrical
interconnects, sensing tips 1844, distributed integrated circuits, etc.
[00244] Fig. 18d shows aspects of a net like micro surgical tool in accordance
with the
present disclosure. The net like structure 1850 may be formed from one or more
fibers,
wires, ribbons, etc. Additionally, alternatively, or in combination the one or
more net
like structures 1850 may include a substrate in accordance with the present
disclosure
(e.g. a porous substrate material such as a silk structure, an elastomer,
polymer, netting,
fabric, fiber composite, etc.). In one non-limiting example, a silk-
flexcircuit composite
may form the net like structure 1850. In this example, the flexcircuit may be
formed
from materials as known to those skilled in the art, the flex circuit may be
constructed
such that substantial material, not occupied by electrical interconnects is
removed (thus
forming a loosely connected webbing of flexcircuit elements). The flexcircuit
may thus
be formed in an excessively thin form (e.g. less than 25um, less than 10um,
less than
4um, less than lum thick). A supporting material such as silk may be used to
complete
the substrate and form a functional, robust net like structure 1850 included
in the micro
-66-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
surgical tool. The net like structure may be interconnected 1852 to a delivery
catheter, an
operator, a controller, one or more control circuits, etc. each in accordance
with the
present disclosure.
[00245] The micro surgical tool may include or be coupled with a micro
balloon, the
micro balloon configured so as to bias the net up against the local tissues
1803.
[00246] Fig. 18e shows a stent-like deployable micro surgical tool in
accordance with
the present disclosure. The stent-like micro tool 1862 may include a plurality
of sensing
tips 1860 electrically interconnected with the remainder of the micro surgical
tool. The
sensing tips 1860 may be positioned throughout the stent-like micro tool 1862.
In one
non-limiting example, the sensing tips 1860 may be generally positioned
towards the end
of the tool. The stent-like micro tool may be interconnected to a guiding aim
1864 for
connection 1865 to an operator (not explicitly shown), controller, etc.
[00247] The stent-like micro tool may be inserted into a lumen 1804 past the
intended
surgical site. It may then be deployed so as to expand outwards and make
contact with
the lumen walls 1804. The micro-tool may then be dragged 1865 forward,
sweeping
along the walls of the vessel. In one non-limiting example, the sensing tips
1860 may be
configured to monitor physiological parameters during this initial sweep (e.g.
so as to
map the local tissue properties). After the first sweep, the tool 1862 may be
retracted and
once again placed beyond the intended surgical site. It may then be deployed
so as to
expand outwards and make contact with the surgical site. The tool 1862 may
then be
dragged forward, sweeping along the walls of the vessel for a second time.
During this
second sweep, the sensing tips 1860 may be activated to locally ablate tissue
at
predetet mined locations determined by the initial sweep. Sensory tips 1860
may further
be monitored during ablation processes to ensure that the processes are
sufficiently
completed before further sweeping the stcnt-like micro tool 1862 though the
vessel.
[00248] In another non-limiting example the stent-like micro tool 1862 may be
inserted past the intended surgical site. It may then be deployed so as to
expand outwards
and make contact with the lumen wall 1804. The micro-tool 1862 may then be
dragged
forward, sweeping along the walls of the vessel 1804. The sensing tips 1860
may, in
concert, monitor the local physiological properties of the tissues and
selectively activated
-67-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
to locally ablate tissues. Thus both the functions of monitoring and ablation
may be
completed in a single sweep.
[00249] The stent-like micro tool 1862 may include any features described
herein as
they pertain to a microfinger in accordance with the present disclosure.
[00250] Fig. 18f shows a two dimensional graph, indicating the ablation
profile of 5
sensing tips located in a stent-likc micro tool in accordance with the present
disclosure.
The desired ablation profile may be predetettnined (e.g. as determined by an
initial
sweep), or determined in concert during a sensing + ablation sweep. As can be
seen in
the example shown, two of the sensing tips did not pass any target tissue in
need of
ablation during this sweep, thus an ablation procedure may be directed towards
other
sensing tips in the array so as to minimize damage to local tissues during the
procedure.
[00251] Figs. 19a-b show a tonal sensing tip and sample response in accordance
with
the present disclosure. Fig. 19a shows a close up of an associated microfinger
1910 in
accordance with the present disclosure, The microfinger 1910 includes an
interfacial
pressure sensor (at the tip, in accordance with the present disclosure) and/or
a flexural
sensor (along the length thereof, in accordance with the present disclosure).
An
excitation 1915, 1920, 1925 applied to the microfinger 1910 may be used to
generate
variable contact forces and contact deflections at the point of contact
between the
microfinger and a local tissue surface 1901. Signals obtained from the
flexural sensor
may be representative of the contact deflections that occur during the
excitation period.
Signals obtained from the interfacial pressure sensor may be representative of
the contact
forces that occur during the excitation period. The simultaneous monitoring of
both
signals, perhaps in combination with a compliance model for the microfinger
1810 may
be useful for determining the local mechanical properties of the tissue in the
vicinity of
the contact point.
[00252] Fig. 19b shows a deflection force curve, generated by the microfinger
1810
described in Fig. 19a during an excitation session. The deflection/force
relationships
(e.g, mean relationships, hysteresis, frequency dependence, creep, strain
hardening, etc.)
may be used to detetmine the type of tissues 1801 in which the microfinger is
in contact.
As can be seen in the figure, a particularly soft relationship 1930 (low
modulus of
-68-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
elasticity) may be associated with a potential tumor tissue. Healthy tissue
may exhibit a
modulus of elasticity within a known "good" range 1935, and the trend 1950 in
elastic
modulus that occurs as the tissue is ablated by a surgical procedure, may be
followed to
determine the extent of the ablation process. A successful ablation process
may be
qualified by a range of elastic modulus change 1940, as observed during the
ablation
process.
[00253] Figs. 20a-b show aspects of surgical tools in accordance with the
present
disclosure. Fig. 20a shows a surgical tool including a delivery catheter 2005
in
accordance with the present disclosure including an array of microfingers
2010, the
microfingers 2010 connected 2011 through the catheter 2005 to an operating
fixture,
control circuit, signal conditioning circuit, hand held control unit, surgical
robot, a
coupling, or the like. The microfingers 2010 in accordance with the present
disclosure
are arranged along the inside of the delivery catheter 2005 and are arranged
with a pre-
biased shape, such that upon retraction 2021 of the catheter 2005, the
microfingers 2010
may be deployed radially 2019 towards an anatomical site of interest (i.e. a
surgical site,
a tissue surface, a lumen wall, etc.). One or more of the microfingers 2010
may include
one or more sensing tips 2015 in accordance with the present disclosure. In
the non-
limiting example shown, each sensing tip includes an electrode configured to
interface
with an anatomical site of interest. The catheter 2005 is configured to slide
over an
associated guide wire 2030, so as to be easily directed to a surgical site
during an
insertion procedure. In the non-limiting example shown, the microfingers 2010
include
shaped tips (upon which the sensing tips 2015 are arranged). Such shaped tips
may be
advantageous to control the bias pressure against an anatomical site of
interest (i.e. so as
to prevent penetration, etc.). In aspects, one or more of the microfmgers 2010
may
terminate at a microneedle sensing tip in accordance with the present
disclosure. Such a
configuration may be advantageous to allow for controlled penetration of one
or more
sensing tips 2015 into the wall of a surgical site. In aspects, after
deployment, the entire
microfinger array 2010 may be drawn 2023 along the length of a lumen, so as to
map the
lumen, sweep monitor and ablate the lumen, assess the state of anatomy after a
surgical
procedure, combinations thereof, or the like.
-69-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
[00254] Fig. 20b shows catheter 2005 in accordance with the present disclosure
including a longitudinal wire cage including an array of microfingers 2010,
the
microfingers 2050 connected 2053 through a delivery catheter 2054 to an
operating
fixture, control circuit, signal conditioning circuit, hand held control unit,
surgical robot,
a coupling, or the like. The microfingers 2050 in accordance with the present
disclosure
are arranged along the inside of the delivery catheter 2054 with a range of
pre-biased
shapes, such that upon retraction 2056 of the delivery catheter 2054 or an
over sheath
coupled thereto, the microfingers 2050 may be deployed radially 2057 towards
an
anatomical site of interest (i.e. a surgical site, a tissue surface, a lumen
wall, etc.) to form
the wire cage. One or more of the microfingers 2050 may include one or more
sensing
tips 2051 in accordance with the present disclosure. In the non-limiting
example shown,
each sensing tip includes an electrode configured to interface with an
anatomical site of
interest. The catheter 2054 is configured to slide over an associated guide
wire 2060, so
as to be easily directed to a surgical site during an insertion procedure. In
aspects, the
wire cage may be coupled to a distal ringlet 2062 or equivalent feature,
configured to
accommodate the passage of a guide wire there through during the procedure.
[00255] In aspects, the microfingers 2050 may be arranged such the sensing
tips 2051
are arranged so as to contact the lumen wall upon deployment.
[00256] Such a configuration may be advantageous to maintain contact between
one or
more sensing tips 2051 with the lumen walls during a procedure without
inhibiting flow
of fluids through lumen. In aspects, the wire cage may be advanced and/or
retracted,
along a lumen (not explicitly shown) and/or expanded/contracted as part of a
procedure, a
deployment, and/or a retraction procedure within the lumen during procedures
related to
searching for anatomical sites of interest, perfoiiiiing sensing, mapping,
surgical
treatments, ablation, or the like.
[00257] In aspects, one or more sensing tips 2051 may be equipped with one
or more
electrodes for electrophysiological sensing, stimulation, and/or RF current
delivery to the
surrounding tissues.
-70-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00258] In aspects, one or more sensing tips 2051 may be arranged
longitudinally
along the axis of the microsurgical tip, such that the sensing tips 2051 are
biased against a
lumen wall at site spaced along the longitudinal direction thereof upon
deployment.
[00259] Figs. 21 shows aspects of a system for perfotining a surgical
procedure in
accordance with the present disclosure. The system is shown interfacing with a
surgical
site 2101 within a body, a subject, a patient, etc. The system includes a
microsurgical
tool 2110 in accordance with the present disclosure. During use, the
microsurgical tool
2110 is configured to interact 2112 with the surgical site 2101 in accordance
with the
present disclosure. In aspects, the microsurgical tool 2110 may he coupled to
a connector
2120, the connector providing a mechanical and electrical interface between
the
microsurgical tool 2110 and one or more other modules of the system. In
aspects, the
microsurgical tool may include an embedded local control circuit 2115a (a
microcircuit, a
switch network, a signal conditioning circuit, etc.) in accordance with the
present
disclosure. In aspects, the connector 2120 may include a local control circuit
2115b in
accordance with the present disclosure. In aspects, the connector 2120 may be
coupled to
an operator input device 2125 (i.e. a foot pedal, an advancing slider, a
torqueing
mechanism, a recording button, an ablation button, etc.). In aspects, the
connector 2120
may be coupled to a control unit 2130 configured to accept one or more signals
from the
microsurgical tool 2110, communicate one or more control signals= thereto,
send one or
more pulsatile and/or radio frequency signals to the microcontroller, record
one or more
electrophysiological signals from the microsurgical tool, or the like.
1002601 In aspects, the control unit 2130 may be connected to a display 2135
configured to present one or more aspects of the recorded signals from the
microsurgical
tool to an operator, to present a map, at least partially dependent on the
recorded signals,
etc.
[00261] In aspects, the control unit 2130 may be coupled to a surgical
subsystem 2140,
the surgical subsystem 2140 configured to perform a surgical procedure 2145 to
the
surgical site 2101. Some non-limiting examples of suitable surgical procedures
include
an ablation, an excision, a cut, a bum, a radio frequency ablation,
radiosurgery, an
ultrasonic ablation, an abrasion, a biopsy, and delivery of a substance. The
control unit
-71-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
2130 may be configured to influence, direct, control, and/or provide feedback
for one or
more aspects of the surgical procedure 2140, based upon one or more of the
electrophysiological signals conveyed by the microsurgical tool 2110.
[00262] Some non-limiting methods for performing a surgical procedure in
accordance
with the present disclosure are discussed herein.
[00263] In one non-limiting example, a method for addressing a surgical site
within a
vessel (e.g. an artery, a vein, a renal artery, a micro-vessel, etc.) is
considered. The
method includes, monitoring one or more local physiological signals (e.g. an
evoked
potential, a neurological activity, MSNA, EMG, MMG, extracellular signal,
sympathetic
tonal change, etc.) in accordance with the present disclosure at one or more
measurement
locations within the vessel to determine one or more reference signals;
performing at
least a portion of a surgical procedure (e.g. an ablation, an excision, a cut,
a burn, an RF
ablation, an abrasion, radiosurgery, an ultrasonic ablation, a biopsy,
delivery of a
substance, etc.) in accordance with the present disclosure at or near to one
or more
surgical locations (e.g. proximal, distal, remotely therefrom, and/or
collocated with one
or more of the measurement locations); monitoring one or more local
physiological
signals at one or more of the measurement locations to deteimine one or more
updated
signals; and comparing one or more reference signals with one or more updated
signals to
determine an extent of completion for the surgical procedure.
[00264] In aspects, the extent of completion may include a change, reduction
and/or
substantial elimination of at least a portion of one or more of the local
physiological
signals (e.g. reduction in amplitude of a frequency band, reduction in
responsiveness, a
change in a lag between measurement locations, a change in cross-talk between
measurement locations, substantial elimination of the signal, etc.)
[00265] In aspects, the extent of completion may include measuring a change in
coherence between two or more signals obtained from sites affected by the
surgical
procedure (i.e. from a first site distal to where the surgical procedure was
performed, and
from a second site proximal to where the surgical procedure was performed).
[00266] In aspects, the procedure may be to perform a temporary neurological
block.
In this aspect, the method may be used to separate afferent and efferent
traffic from either
-72-
CA 02862862 2014-07-25
WO 2013/112844 PCMJS2013/023157
side of the temporary block, for further analysis, diagnosis of disease,
evaluation of
neurological activity, or the like. In aspects, a temporary block may be
followed by a
more permanent block if the analysis demonstrates that such a substantially
permanent
block would be warranted.
[00267] The step of monitoring to determine an updated signal may be performed
before, during, and/or after the step of performing at least a portion of the
surgical
procedure. In aspects, monitoring, stimulation, and ablation may be performed
in
succession and/or in parallel.
[00268] In aspects, the method may include sweeping one or more electrodes
over the
lumen wall while monitoring, stimulating, and/or ablating the surface thereof.
In aspects,
simultaneous monitoring and sweeping may be used to generate a map of
neurological
activity along the lumen wall.
[00269] The step of performing at least a portion of the surgical procedure
may be
repeated. Thus the method may be incrementally applied, so as to head towards
completion in a stepwise process without excessive application of the surgical
procedure.
[00270] The method may include waiting after performing at least a portion of
the
surgical procedure. Monitoring may be performed during the waiting procedure,
perhaps
so as to determine a recovery period for the local physiological signal (i.e.
a time period
over which the local physiological signal recovers). Such a recovery period
may be an
indication of the extent of completion.
[00271] In aspects, the method may include stimulating one or more stimulation
locations (proximal, distal, remotely therefrom, and/or collocated with one or
more of the
measurement locations and/or the surgical locations). The step of stimulating
may be
coordinated with the step of performing at least a portion of the surgical
procedure,
and/or with the step of monitoring to determine a reference and/or updated
signal. The
stimulation may be provided in any form in accordance with the present
disclosure. In
one non-limiting example, the stimulation may include one or more current
pulses, one or
more voltage pulses, combinations thereof, or the like. The step of
stimulation may be
advantageous for assessing the updated signal at one or more measurement
locations
-73-.
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
and/or between two or more measurement locations in the presence of background
noise
and/or local physiological activity.
[00272] In aspects, the method may include monitoring one or more remote
physiological parameters in accordance with the present disclosure at a remote
location
(e.g. an alternative vessel, an organ, a ganglion, a nerve, etc.)
substantially removed from
the immediate vicinity of the vessel to determine an updated remote
physiological signal
and/or reference remote physiological signal.
[00273] Some non-limiting examples of remote physiological parameters that may
be
monitored include water concentration, tone, blood oxygen saturation of local
tissues,
evoked potential, stimulation/sensing of nervous activity, electromyography,
temperature,
blood pressure, vasodialation, vessel wall stiffness, muscle sympathetic nerve
activity
(MSNA), central sympathetic drive (e.g. bursts per minute, bursts per
heartbeat, etc.),
tissue tone, blood flow (e.g. through an artery, through a renal artery), a
blood flow
differential signal (e.g. a significantly abnormal and or sudden change in
blood flow
within a structure of the body, a vessel, an organ, etc.), blood perfusion
(e.g. to an organ,
an eye, etc.), a blood analyte level (e.g. a hormone concentration,
norepinephrine,
catecholaminc, refine, angiotensin II, an ion concentration, a water level, an
oxygen
level, etc.), nerve traffic (e.g. post ganglionic nerve traffic in the
peroneal nerve, celiac
ganglion, superior mesenteric ganglion, aorticorenal ganglion, renal ganglion,
and/or
related nervous system structures), combinations thereof, and the like.
[00274] The updated remote physiological signal and/or reference remote
physiological signal may be combined and/or compared with one or more
reference
signals, and/or one or more updated signals in order to determine the extent
of
completion, as part of a decision making process, and/or as part of a surgical
control
system (i.e. so as to determine whether to continue with, stop, or alter the
surgical
procedure).
[00275] The method may include selecting a surgical location. The step of
selection
may depend upon one or more monitoring steps, proximity to an alternative
surgical
location (i.e. perhaps a previously treated surgical location, etc.).
-74-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00276] In aspects, the method may include sweeping the lumen while monitoring
in
order to localize one or more anatomical sites of interest, one or more
regions of
abnormal activity, etc.
[00277] In aspects, the steps of monitoring may be completed sequentially.
Alternatively, additionally, or in combination, the steps of monitoring may be
effectively
continuously applied through the procedure. The comparison may be made using
one or
more data points obtained from one or more steps of monitoring. The comparison
may
be made via algorithmic combination of one or more measurements.
[00278] In aspects, the step of monitoring may be used to extract one or more
electrophysiological parameters during a first period and monitoring an
applied field (i.e.
as caused by a stimulation and/or ablation event) during a second period.
[00279] In aspects, the method may include generating a topographical map from
the
one or more measurements (e.g. from one or more of the signals). The method
may
include determining a topographical map of physiological functionality in the
vicinity of
the surgical site derived from one or more of the physiological signals. The
method may
include updating the topographical map after the step of performing at least a
portion of
the surgical procedure. The method may include generating the map during a
sweeping
process (i.e. a longitudinal sweep, a circumferential sweep, a helical sweep,
etc.).
[00280] In aspects, the method may include placement of a plurality of
surgical tools,
one or more surgical tools (i.e. a procedural tool) placed so as to access one
or more of
the surgical locations, and one or more surgical tools (i.e. a monitoring
tool) placed so as
to access one or more of the monitoring locations. In one non-limiting
example, a
procedural tool may be placed in a first vessel (e.g. a renal artery, a left
renal artery, etc.)
and a monitoring tool may be placed into a second vessel (e.g. an opposing
renal artery, a
right renal artery, a femoral artery, an iliac artery, etc.). Thus, the
monitoring tool may be
used to monitor one or more of the measurement locations in the second vessel.
The
procedural tool may be used to surgically treat one or more surgical locations
in the first
vessel. Additionally, alternatively, or in combination, the procedural tool
may monitor
one or more monitoring locations in the first vessel, perhaps in combination
with
monitoring perfotmed in the second vessel by the monitoring tool.
-75-.
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
[00281] In aspects, the method may be performed with one or more surgical
tools in
accordance with the present disclosure.
[00282] One or more steps of monitoring may be performed with one or more
sensing
tips in accordance with the present disclosure.
[00283] One or more steps of performing at least a portion of the surgical
procedure
may be performed with one or more sensing tips in accordance with the present
disclosure.
[00284] In one non-limiting example of a method for RF ablating tissue, the
local
tissue tone may be measured before, during, between individual RF pulses,
and/or after a
train of RF pulses. As the local tissue tone changes during application of the
RI' pulses,
the tonal changes may be used to detennine the extent of the therapy. As the
RF ablation
process is applied to the adjacent tissues (perhaps via one or more sensing
tips), the tonal
measurements (as determined by one or more sensing tips, perhaps the same tip
through
which the RF signal may be applied) may be monitored to determine an extent of
completion of the procedure. Such an approach may be advantageous as the tonal
measurement techniques may not be significantly affected by the local RF
currents
associated with the RF ablation procedure.
[00285] In aspects, an interventionalist/proceduralist may insert a catheter
in
accordance with the present disclosure into the aorta from either the superior
or inferior
approach (brachial or femoral arteries) and selectively cannulate the renal
artery. In
aspects, a guiding catheter may be used for this purpose. In aspects, a
microsurgical tool
in accordance with the present disclosure may be placed through the guiding
catheter. In
aspects, one or more regions of the microsurgical tool may be deployed thus
allowing one
or more electrodes included therein to bias against the lumen of the renal
artery. Such a
configuration may be advantageous to establish excellent mechanical and
electrical
contact with the walls of the renal artery.
[00286] In aspects, the electrodes may be made to puncture the vessel wall
from the
lumen side. The electrodes may be expandable and/or retractable, exiting in a
stable
pattern of 1 to 6, or more microfingers that permit stability and counter-
opposition force
to cause penetration of one or more of the electrodes into the intima, media,
or adventitia
-76-
CA 02862862 2014-07-25
WO 2013/112844 PCT/US2013/023157
of the lumen (i.e. artery, vein, etc.) to be measured. In aspects, one or more
electrodes
may be configured for microscopic or macroscopic spatial recording. Following
a
suitable period of recording, the device may be withdrawn into the guiding
catheter and
removed from the body.
1002871 It will be appreciated that additional advantages and modifications
will readily
occur to those skilled in the art. Therefore, the disclosures presented herein
and broader
aspects thereof are not limited to the specific details and representative
embodiments
shown and described herein. Accordingly, many modifications, equivalents, and
improvements may be included without departing from the spirit or scope of the
general
inventive concept as defined by the appended claims and their equivalents.
-77-