Note: Descriptions are shown in the official language in which they were submitted.
1334070
This invention relates to prefilled syringes for
use in medical or veterinary treatment.
There has been an increasing trend in recent
years to the putting up of pharmaceuticals in dosage forms
so as to minimize the preparation required to administer a
medicament to a patient and to reduce the chances of dosage
errors or contamination. One dosage form which has been
gaining rapid acceptance is the prefilled disposable
syringe. Various difficulties are however associated with
the preparation and usage of such syringes, particularly
in the case of preparations which, in ready to use
condition, have a short shelf life. Numerous forms of
dual compartment syringe structure have been proposed for
the shipping of such preparations with components stored
in separate compartments for admixture immediately prior
to use. Although certain structures have met with some
degree of acceptance, they are commonly difficult to
manufacture and/or use because of difficulties in filling
the syringe with the components, and because they require
extensive manipulation immediately prior to use. Moreover
they are frequently substantially more bulky than
conventional syringes because in many cases they frequently
comprise components which effectively represent two
syringes in tandem.
- 2 - 1334 070
Problems in the manufacture of prefilled syringes
are not confined to two component systems and even with
single component systems the filling of syringes under
factory conditions is difficult to mechanize effectively
and requires expensive special purpose syringe filling
machinery. The same applies to related units prefilled
with liquids required for injection or infusion during
medical procedures.
Another approach where single component systems
are involved is exemplified by British Patent
Specifications Nos. 1,252,306 and 1,444,119, and U. S.
Patent No. 4,445,895, in which a prefilled cartridge having
a displaceable plug at one end, and a needle penetrable
closure at an opposite end, is inserted into the barrel of
a syringe for dispensing of its contents. Whilst such
cartridges and the equipment for filling them are known
and available, they are only really suitable for
preparations which can be stored in liquid form, and
require either a special or a modified syringe for their
use.
In a further arrangement disclosed in U. S.
Patent No. 3,845,763, a cartridge or vial is closed at its
bottom end by a slidable plug with a downwardly extending
stem, which cartridge or vial is inserted bottom end first
into a special holder which carries a double ended needle,
so that the stem is penetrated by the needle and the body
of the vial is converted into a plunger which can be
depressed to expel the contents of the vial through the
stem. The projecting stem means that the vial cannot be
filled utilizing conventional vial filling machinery.
The present invention seeks to provide a system
for the distribution of preparations required for injection
or infusion in liquid dosage form during medical
procedures, which has a wide range of utility both for
3 1334070
single component liquid preparations or for two component
systems of which one component may be a solid, which utilizes
a small number of components all suitable for mass production,
and which is simple to assemble and fill utilizing available
equipment.
The invention is based upon the use of vials
designed so that they may be filled utilizing conventional
filling machinery and techniques, yet also form the barrel of
a syringe in a prefilled syringe system which can be adapted
for the dispensing of single or two component systems,
including two component systems of the kind in which the solid
component is lyophilized in situ during manufacture of the
syringe.
In the context of the invention, it should be
understood that "vial" refers to a particular type of
container, having a rather squat cylindrical body whose height
compared to the diameter of its base is such that it may stand
stably on its base whilst being conveyed through a vial
filling machine and subsequently sealed and capped. A vial
has a neck with a large enough internal diameter to permit
filling from a vial filling machine: solid filling materials
will normally require a larger neck than liquids. Vials
should not be confused with cartridges, which are
comparatively long and slim, and cannot usually be filled
utilizing vial filling machinery since they are too tall to
rest stably on their bases.
Accordingly, the present invention provides a
prefilled syringe for administering a pharmaceutical
4 1334070
preparation, comprising a generally cylindrical syringe body
having a neck at one end, and a side wall terminating in a
bead finish at the other end, at least a component of the
pharmaceutical preparation filled into the body, an
elastomeric closure closing the body at the neck end and
secured by a cap, and an elastomeric piston closing the bead
finished end of the body and forming a hermetic seal with an
inside surface of said side wall, needle means for movement
relative to the cap to penetrate the elastomeric closure, and
plunger means for connection to an outer side of the piston,
characterised in that the syringe comprises an association of
components including said plunger and said needle with a
prefilled vial; and in that said vial comprises said body,
which is formed with a height to diameter ratio such that the
body is stable, and so that any outward extent of the bead
finish is insufficient to result in interference such as would
cause tipping, when the body is conveyed standing on said
other end through equipment for filling and capping
pharmaceutical vials with said elastomeric piston inserted
wholly within said other end of the body; said vial with the
piston in place having been filled through said neck with said
at least one component of the pharmaceutical preparation, and
said elastomeric closure and cap having been applied whilst it
was so conveyed.
The differences between such a vial and conventional
vial do not prevent it from being filled and capped in
conventional vial fllling and capping machinery; indeed, apart
4a 1334070
from the replacement of the bottom wall of the vial by a
piston as specified, it is a conventional vial, and can be
handled normally by the machinery during filling with either
liquid or solid material. Furthermore, liquid filled vials
may be lyophilized utilizing special stoppers either as known
in the art or as described below.
Such a vial in accordance with the invention may be
converted into a syringe by the addition of a plunger coupled
to the piston and an outer cap which acts as a needle carrier.
More specifically, the syringe includes as well as the vial a
plunger connected to said piston, and an outer cap engaged
over the cap of said vial, the outer cap having a hollow
needle projecting axially within the cap and a coupling -for
engagement with injection means and communicating wi~h said
hollow needle, the outer cap being axially movable relative to
said cap of the vial from a position in which the needle ends
short of the cap of the vial to a position in which it
penetrates the cap of the vial, and both the plunger and the
outer cap being provided with radially extending flanges for
sustaining actuating forces applied to the syringe.
A
_ ~ 5 ~ 1 334 070
In a syringe for a two component medicament, it
is necessary to provide for packaging of the second
component and its admixture with the first component in
the vial prior to dispensing. The invention thus further
extends to a capsule assembly comprising a generally
cylindrical sealed capsule having walls formed of a
flexible needle penetrable material, a generally
cylindrical neck defined by said walls at one end of the
capsule, said neck having axially spaced inner and outer
peripheral ridges, and a generally cylindrical cap applied
to said neck so that a detent within the cap engages the
outer peripheral ridge on the neck, a double ended hollow
needle passing through said cap so that an inner end within
the cap ends short of the neck of the capsule and an outer
end extends outwardly of the cap, the cap being
displaceable relative to the capsule to a position in
which the detent rides over the inner ridge and the inner
end of the needle penetrates the neck of the capsule, the
cap and capsule being of a diameter such that they can
enter the tubular plunger to a position in which the outer
end of the needle on the cap of the capsule penetrates the
septum of the piston when the plunger is engaged with the
latter.
Thus the injection system comprises a sequence
of components of which various subsequences can be combined
to form injection systems for preparations requiring ship
and storage as two separate components, certain
subsequences themselves having utility re~pectively as
injection systems for single component liquid preparations.
HInjection" is utilized broadly to cover hypodermic,
intramuscular and intravenous injection, gravity and
m~chAnical infusion, and injection into other vessels
utilized in medical treatment or testing. For the purposes
of description, the Hfront" of an injection system will be
considered the end of the system from which a liquid
preparation is so injected.
1334070
The arrangement including the capsule assembly
has a number of advantages in the manufacture and use of
prefilled syringes for two component systems; furthermore,
without the third cap and the sealed capsule containing the
S second component the remaining components provide,
according to a further feature of the invention, advantages
in the manufacture and use of prefilled syringes for single
component systems. The third cap and sealed capsule
provide, according to yet a further feature of the
invention, an advantageous subsystem for various
applications in which a sealed sterile source of a liquid
is required for injection, or dropwise introduction into
other containers used in medical procedures. With
prefilled syringes for two components systems, either the
capsule or the capsule and the third cap, may be sold, or
shipped separately. This enables different diluents or
sizes of capsule to be selected, or a common set of diluent
capsules to be utilized with syringe assemblies containing
different first components, thus simplifying inventory
control.
Further features of the invention will become
apparent from the following description of a preferred
embodiment thereof with reference to the accompanying
drawings.
In the drawings:
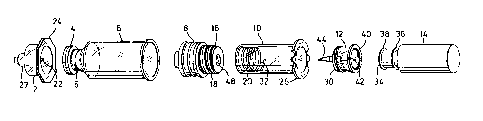
Figure 1 is a perspective exploded view of the
mechanical components of a syringe system including a vial
in accordance with the invention;
Figure 2 is a partially longitll~in~lly sectioned,
partially exploded view of the syringe components showing
some further details of their construction;
_ _ 7 _ 1 334 070
Figures 3, 4 and S illustrate preparation of the
syringe system to provide a syringe ready for use;
Figures 6, 7 and 8 illustrate exemplary
applications of the syringe; and
Figures 9 and lO illustrate an optional feature
of a vial in accordance with the invention.
Referring to Figures l and 2, a syringe system
for the injection of a liquid preparation stored as two
components comprises seven primary mechanical components,
apart from the components of the preparation, which latter
are shown in Figure 2 but not Figure 1. The components of
the preparation typically comprise a first component A
which may be in any physical state suitable for storage in
vial, and a second liquid component B, typically but not
necess~rily sterile water. The liquid component B is
stored in a sealed capsule 14 of flexible material,
manufactured using conventional techniques from a material,
usually synthetic plastic, which is compatible with the
contents of the capsule. The first component is stored in
a cylindrical vial 6, typically of glass, and capped by an
annular cap 4 which retains a conventional needle
penetrable sealing member accessible through a central
opening in the cap. By a vial i8 meant a cylindrical
vessel which can assume a stable upright position supported
by its base, the overall height of the vessel preferably
not exceeding 2.5 times the external diameter of the rim
of its base so that it remains stable when passing through
conventional vial filling and capping equipment utilized
to fill and cap the vial. A neck at the upper end of the
vial 6, which is capped by the cap 4, has a relatively
internal diameter characteristic of such vessels, usually
not less than about 7.5 mm for liquid or lO mm for solids,
so that filling either liquids or solids can be readily
achieved. The cap 4 is formed by an aluminum sleeve,
- - 8 _ 1 334 0 70
having a flange retaining a sealing member formed by a
soft rubber disc or plug 5 over or in the front end
opening, and tightly crimped onto a neck at the front end
of the vial so as to seal the latter. A major difference
from conventional vials is that the conventional bottom
wall of the vial is replaced by an axially movable piston
8 wholly within the vial and in sealing contact with the
vial walls. When received within the vial 6, this piston
in no way interferes with the handling of the vial using
conventional machinery, and in particular permits the vial
to be stood on its base with its neck (which forms the
front end of the vial when in use) upwards as it passes
through the filling and capping equipment.
The filled vial 6 may be converted into a
prefilled syringe by applying an outer cap 2 over the cap
4 and positively attaching a cylindrical plunger sleeve 10
to the piston 8. The piston 8, typically formed of rubber,
is moulded with a rearward extension 16 with an external
thread 18, whilst the interior of the front end of the
plunger sleeve 10 is formed with a complementary internal
thread 20 so that it may be screwed onto the piston 8.
The outer cap 2 fits over the inner cap 4 so that a hollow
needle 22 formed within the cap 2 does not reach the
penetrable zone of the cap 4. On the front of the cap 2
and in communication with the hollow needle 2 is a coupling
adapter 27, for example similar to those sold under the
trade mark LUER-LOR, for connection of the syringe to a
needle 28 or other instrumentality (see Figures 6-8). The
rear ends of both cap 2 and the sleeve 10 are formed with
radially extending flanges 24 and 26 respectively which
form finger grips for operation of the syringe. Thus if a
user grips the syringe by the flanges as shown in Figure 6
and presses them towards each other, the cap 2 is pulled
rearwardly onto the cap 4 so that the needle 22 penetrates
the cap and the contents of the syringe can be expelled
through the needle 22 and the needle 28. It will be noted
~ - 9 - 1334070
that the rear end of the vial 6 is formed with only a
relatively slight external flange 7 rather than the wide
finger flange commonly found on the barrels of conventional
syringes. In the present arrangement, the flange 24
provides the function of such a finger flange, enabling
the flange 7 to be reduced to a size which will avoid such
interference between the flanges of adjacent vials as
would cause tipping when the vials are conveyed in a
vertical attitude through filling and capping equipment.
A prefilled syringe so constructed has
significant advantages over conventional prefilled syringes
in that the vial may be filled using conventional vial
filling equipment, and yet may be utilized directly instead
of requiring its contents to be transferred to a syringe
prior to use as has been conventional in the use of vials.
The vial may also be charged with material which
is not directly injectable, such as solids which must be
dissolved or suspended in a liquid medium prior to
injection. In this case the liquid medium is sealed as
already described in a flexible capsule 14. A third cap
12 is either applied to the capsule as shown in Figure 2,
or inserted into the plunger sleeve 10 so that a screw
thread 30 on the exterior of the cap engages the screw
thread 20 within the sleeve.
A neck 34 of the capsule 14 has two peripheral
ridges 36 and 28. If the cap 12 is applied to the capsule,
a detent 40 within the cap i8 pushed over only the outer
ridge 38 so that a rear end portion 42 of a hollow needle
mounted in the cap stops short of the end of the capsule.
By forcing the detent 40 rearwardly over the ridge 36, the
needle portion 42 can be forced rearwardly so as to
penetrate the capsule. A forward end portion 44 of the
hollow needle has a length such that when the cap 12 is
screwed into the sleeve 10, and the sleeve 10 is s~Lewed
- - lO- 1334070
onto the piston 8, the needle portion 44 penetrates a
resilient septum 50 normally separating axial passages 46
and 48 formed in the front and rear of the piston.
In use, if the capsule 14 and cap 12 are shipped
as a separate unit, this unit is screwed into the sleeve
10 (see Figure 3), and the sleeve 10 is pushed into the
rear of the vial 6 so that the needle portion 44 penetrates
the septum 50 of the piston 8 and the thread 20 is screwed
onto the thread 18 of the piston (see Figure 4). This
action also substantially unscrews the cap 12 from the
thread 20. The capsule 14 is then pressed forward onto
the needle portion 42, and the liquid contents of the
capsule can then be squeezed through the needle and into
admixture with the first component in front of the piston
8. Thereafter the capsule 14 and cap 12 may be pulled as
a unit from the sleeve 10 and ~isc~rded (see Figure 5).
The septum 50 reseals as the needle portion 44 is
withdrawn, leaving a syringe ready for use as illustrated
in Figures 6 - 8. Alternatively, if the cap 12 is
prefitted to the sleeve, the sleeve 10 may be screwed onto
the piston 8, and the capsule 14 pressed into the sleeve
10 and the cap 12 so as to establish communication between
the capsule and the space forward of the piston, the
procedure thereafter being the same.
Rather than being used conventionally with a
needle as shown in Figure 6, the prepared syringe may be
used for gravitational or mechanical infusion as shown in
Figures 7 and 8. In Figure 7, the adapter 27 is fitted to
a complementary coupling on a gravity infuser 52 to provide
a drip feed, the sleeve 10 having been unscrewed and
discarded, together with the cap 12 and capsule 14, if
used. In Figure 8, the syringe is mounted in a mech~nica
infuser 54 such as that sold under the trade mark BARD,
the latter being equipped with clamps 56, 58, 60 suited
for engagement with the syringe.
- - 11 - 133~070
By basing the system on an open-bottomed vial 6
closed at its bottom end by a piston 8 equipped with means
such as the screw thread 18 for coupling it to a plunger
of sleeve form, and with a needle penetrable septum 50, in
optional conjunction with sealed flexible capsules of
diluent, great flexibility in application can be obtained,
using components which are easy to fill, compact to ship,
and easy to make ready for use.
Referring now to Figures 9 and 10, the rubber
disk or plug retained by the cap 4 on the vial 6 may be
replaced by a modified plug 60 as shown in perspective
from beneath and one side in Figure 8, and partially
installed on a vial 6 in Figure 9. Use of such a plug 60
is advantageous when the solid component of a medicament
is to be prepared in situ in the vial by lyophilization.
The vial is filled with a liquid preparation to be
lyophilized, and plug 60 inserted to the position shown in
Figure 9, so that the interior of the vial communicates
with its environment through a central passageway 61 and
radial bores 62, the passageway and the bores being no
larger than needed for the removal of water vapour during
lyophilization. The plug i8 split at 63 to facilitate
moulding. After filling the contents of the vial are
rapidly frozen and vacuum dried to leave a solid residue
in the vial which can be reconstituted immediately before
use. The plug 60 is then moved to the full extent
permitted by a flange 64 into the neck of the vial 6 and
secured by a cap 4. Whilst a conventional lyophilization
stopper could be utilized in place of the plug 60, the
latter has the advantage of minimizing the amount of liquid
trapped within the stopper during use of the syringe. For
the same reason, the head of the piston 8 is shaped so as
to minimize dead space in the neck of the vial when the
contents of the vial are expelled during use of the
syringe.