Note: Descriptions are shown in the official language in which they were submitted.
20227~9 ~ 1-
.
Method of reconditioning foundry sand
.
S .
.;:
10 The invention relates to a method for reconditioning foundry sand.
The wet, mechanical, thermal or combined regeneration of foundry sand such
as quartz sand, olivine, zircon, chromite sand and the like having organic or
inorganic binders is known. The goal of such regeneration processes is to
15 provide a reconditioned sand which can be re-used instead of or as new
sand. At the same time, substances in the foundry sand hazardous to the
environement are to be elirninated by regeneration. Sand bound organically
- usually only needs to be regenerated thermally, if it does not contain basic,
acidic or other disturbing components, which do not combust or vaporize.
Mono-sand or mixed sand bound inorganically, in particular sand originally ;
treated with bentonite must be thermally and mechanically regenerated to
produce a~sand which can be re-used. ;
A disadvantage of these known methods, for example for organic binder `
systems ~ with ~basic or acidic components which are difficult or impossible to
eliminate and in particular ` for mono-sand or rnixed sand with inorganic
bindersj is that the regenerate sand has properties which deviate from new `;; ~ -
sand, for example the pH-value,~ the electrical conductivity, the degree of
~;~; 30 o~iithization, the sludge content and ~ the like. The properties of such re- ;~ ~ -
; `gènerate `sand ~ are ~more ,or~ ~less ~disadvantageous especially !for reuse in
making~ cores. The poorer properties of such regenerate sand apply particu-
larly in it~ use ~as core sand with~ a synthetic resin binder and cause reduced
rigidity, reduced processing time of the sand mixtures, increased consumption
35 ~ of binder and the like.
:
The object of the present invention is to provide a process of the above
20227~9 . 2 ~
described type, with which thermally and/or mechanically regenerated sand
can be treated so that subsequently it is like new sand or similar to it.
This object is solved according to the invention in that sand containing an
S organic binder after thermal regeneration or sand containing inorganic binder
after a thermal and mechanical regeneration is neutralized with a suited base
or acid depending on its pH-value determined after regeneration by a
measuring procedure, for example by titration, so that subsequently it has a
pH-value between 6 and 8.
Further embodiments of the process are given in the subclaims.
The essence of the invention is then to additionally wet-neutralize and
purify the sand grains such as quartz grains for example originally treated
with organic and/or inorganic binders (bentonite) after a thermal treatment
and a subsequent mechanical purification, in particular to bring its pH-value
and its electrical conductivity to the corresponding values of new sand.
The organic binder is extensively combusted or the inorganic binder, for
example clay or bentonite, is dead-burned by the thermal and mechanical
treatment and substantially extracted and separated from the sand grains by
the mechanical purification. According to the invention, the regenerate sand
is subsequently neutralized in the wet state in containers and purified. The
mi~ture is stirred or turned in the treatment vessel to support and accele-
rate the process.
The necessary amounts of neutralization additives can be advantageously
deterrnined by titration of 50 to 100 g of a sand sample, namely when the
sand is in its thermal or possibly its mechanical regeneration state.
~ , ! i ,! ~ ,
Experiments have shown surprisingly that a certain turbidity of the aqueous
solution arises when adding a suitable acid as the neutralizer to regenerate `~
sand which was bound inorganically. Apparently a slight separation of dirt or
sludge or binder still adhearing to the sand grains is caused by the neutra-
lization Thus a possible electrostatic binding and adhesion of the grains is
also eliminated.
,:
2~22~9 - 3 -
After the neutralization and optionally a washing and subsequent drying, the
treated sand when observed under the m~croscope already has a clearly
cleaner appearance than before and has properties which are the same or
S similar to new sand as is shown in the following examples I and II.
Example I
Quartz sand regenerate: AFS 60.3
Proper~,r Initial Neutral~zation with Comparable new sand
regenerate 10 ml conc.
sand H2S04 per kg
. . :
pH-value 9.6 7.4 ca. 7.0
Electrical
sonductivity
in ,uS/cm 172 4 ca. 0
Red heat
loss (%) 0.17 0.14 ca. 0
Sludge
content ~o 1.04 0.34 ca. 0.3
De~ree of
oollthization 0.93 0.79 ca. 0
.
Example Il ~ ~
Quartz sand regenerate: AFS 45 ~ -
:: :
1 ~oper~r Initial Neutralization with 12 ml ~- - -
regenerate sand NH40H (25 ~o) per kg sand ~ ~
~ ~ .
pH-valué 3.6 ` 7.2
;`
Electrical
conductivity in
,uS/cm 189 12
`: . ',` '~
2 ~2 27 ~ 9 4
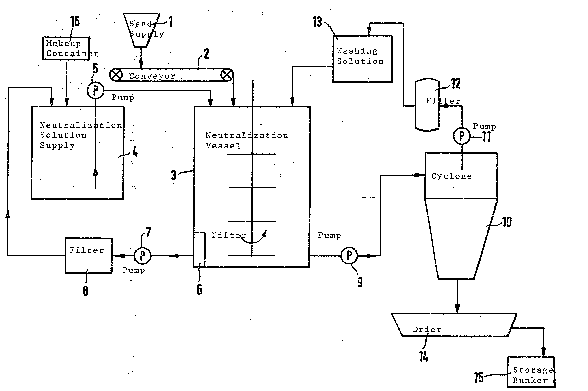
A neutralization of regenerated sand is illustrated in the attached drawing.
The sand already regenerated is fed to a neutralization vessel 3 from a sand
supply 1 over a belt weigher for adjusting the weight per charge. ~long with
5 the supply of sand, a neutralization solution is pumped out of a storage
container 4 by a pump 5 into the neutralization vessel 3 which is controlled
by a fill level display (not shown). A stirring or turning of the material
takes place in the vessel 3 by schematically illustrated stirring means during
the filling process and during neutralization. After neutralization, the
10 solution is drawn through a filter plate 6, a pump 7 and a filter 8 and fed
back to the storage container 4. A refreshing of the used neutralization
solution with wash or fresh water and the addition of new acid or base
takes place by means of a container 16. After neutralization, a washing and
purification of the neutralized regenerate takes place by introducing a
15 washing solution from the storage container 13 into the vessel 3 when the
stirring process is continued. The sand-wash water mL~Lture is then pumped
out of the vessel 3 by a pump 9 into a cyclone 10. The sand and sludge-con-
taining water separate in the cyclone 10. The sludge water is purified in a
filter 12 and fed back to the storage container 13 by a pump 11. Fresh
20 water is added here to the used washing water. The separated, purified and
neutralized sand from the cyclone 10 is dried by means of a swing drier 14
and then transported to a storage bunker 15.
The neutralization and washing or purification take place alternatively in two
25 respectively closed circulation systems. To adjust the neutrality of the
washing solution, bases can also be added for example when neutralizing with
an acid or also vice versa. The belt weigher can also fill several neutrali~
zation vessels by means of a distributor belt, where also several cyclons can
be employed-
The neutralization and purification processes are performed in closed cycleswith the least possible amount of water consumption for environmental
reasons. The neutralization solution after the completed reaction is drawn
out of the vessel together with the extracted sludge fraction and subse-
35 quently passed through a filter to separate the sludge material. When neces-
sary, the filtered and used neutralization solution is refreshed with new
;, ~. ,~
2~227~9 5-
acid or base and with fresh water or wash water and is always fed back in
the cyclic process for the next treatment, for example by means of an inter-
mediate container.
S A further exarnple is shown in the following of a mixed sand bound inor-
ganically, where the initial old sand is thermally and mechanically prepared
to give regenerate sand and it is subsequently neutralized and purified.
A strength test is made on particularly sensitive cold-box core sand mixtures
10 at various stages of treatment compared to new sand.
~ 2022759
Example III
Mesh Bentonite Therm.-mech. regen. Neutral. with 10 ml conc.
analysis old sand sand from I H2S04 per kg sand from I
s
II III
_ _
% % %
0.71 mm 0.2 0 0.1
0.5 mm 0.5 0.7 0-9
0.355 mm 3.6 6.1 5.7
0.25 rnm 25.1 22.3 21~5
0.18 mm 52.B 48.0 50.2
0.125 mm 14.2 21.0 20.0
0.09 mm 3.3 1.8 1.5
0.063 mm 0.3 0.1 0.1 -
0.02 rnm 0 0 0
20.02 mm 0 0 0
-
pH-value 9.8 9.7 7.1 ~ -
Electr. con- -
ductivity
in,uS/cm 976 165 3 -~
Sludge % 8.26 0.93 0.32 ~ ;-
Red heat ~ ~
loss % 2.57 0.17 0.14 ~ ~;
Oolith. degree 2.3 0.89 0.77
Comparison of flexural strength inl N~cm~ on cold-box sand samples~
` ` sand mixture: 30 ~o new sand, AFS about 60
70 ~o therm.-mech. regenerate
35 0.8 % synthetic resin 352 T 14
0.8 % synthetic resin 652 TEA 700
~` 2~22769 7-
Test time Sand IISalld III 100 new sand
S :
150 260 230
1/2 h lgS 390 340
h 190 420 410
2 h 192 440 420 ~ ~ -
24 h 225 450 460
~ ~
; ~:
-.
.