Note: Descriptions are shown in the official language in which they were submitted.
206235n
BACKGROUND OF THE INVENTION
The invention pertains to a process for recovery of
nickel and magnesium from a naturally occurring material
Various hydrometalurgical processes for extracting
nickel from nickel bearing ores are known in the art.
For example, U.S. Patent No. 4,435,369 to Simpson
discloses a process whereby nickel bearing ores are
contacted in an extraction zone with an aqueous solution
having a concentration of chloride ions, metal ions and
sulfuric acid at an elevated temperature. This procedure
yields a nickel recovery of approximately 70%.
Unfortunately, the materials necessary for this
procedure, namely the sulfuric acid and metal and
chloride ions, are costly and thus limit the application
of this procedure to use on nickel bearing ores having a
sufficiently high portion of nickel to make the procedure
economically feasible. Further, the procedure disclosed
in U.S. Patent No. 4,435,369 requires the addition of
magnesium in the extraction zone to improve the results
of the procedure.
Several other methods of hydrometalurgical
processes, discussed in U.S. Patent No. 4,435,369,
similarly have poor recovery percentages which limit the
use of such processes to applications where the nickel
content of the nickel bearing ore must be sufficiently
high to make the procedure economically feasible.
'-A
20623s0
Magnesium is an additional metal which would
preferably be extracted during the process, rather than
being required as an additive to the process in order to
improve the recovery of nickel.
The present invention seeks to provide a process for
recovery of both nickel and magnesium from naturally
occurring materials whereby the cost of the procedure is
reduced to allow application of the process to nickel and
magnesium bearing materials having a much lower content
of these metals. Due to the reduced cost of the
extraction materials of the disclosed invention, as well
as the improved efficiency of the process, materials
having a low content of nickel and magnesium can be
treated in a commercially feasible manner to extract
these metals.
Hydroconversion treatments of heavy hydrocarbon oils
frequently result in production of spent catalysts which
have high sulfur content and which are also iron based.
Typical iron based catalysts used in these processes are
disclosed in U.S. Patent Nos. 3,936,371 to Ueda et al,
U.S. Patent No. 4,066,530 to Aldridge et al, and U.S.
Patent No. 4,591,426 to Krasuk et al. It would there-
fore also be desirable to provide a use for such spent
hydroconversion iron based catalysts which does not
involve costly procedures for its safe disposal.
The present invention also seeks to provide a use
for these spent hydroconversion catalysts in the
presently disclosed process for recovery of nickel and
magnesium.
~'A
2062350
SUMMARY OF THE INVENTION
The improved process for recovery of nickel and
magnesium from naturally occurring materials comprises
the steps of: providing a spent iron based catalyst
having a sulfur content of at least 10% by weight;
providing a naturally occurring material having a nickel
content of at least 0.1% by weight and a magnesium
. content of at least 0.1% by weight; admixing the catalyst
and the material in a ratio of catalyst to material in
the range of 1:2-8:1 by weight to form a mixture; forming
a suspension of the mixture in water having a pH no
greater than 4; subjecting the suspension to an elevated
temperature and an elevated pressure; and injecting an
oxidant into the suspension at a rate sufficient to
maintain a substantially homogeneous distribution of
catalyst, material and oxidant in the suspension, whereby
nickel and magnesium are extracted from the material into
the solution. Standard and known in the art techniques
can then be used to extract the nickel and magnesium from
the solution.
The spent catalyst and material are preferably
powdered prior to the operation to an average particle
. diameter of 200 microns or less. The catalyst may
suitably have an Fe2O3 content of 1 to 90%, by weight.
The preferable content of iron in the spent catalyst
falls in the range of 20-70% by weight as Fe, while it is
also preferable to have at least 15% by weight of sulfur
content.
,,.. ~
2062350
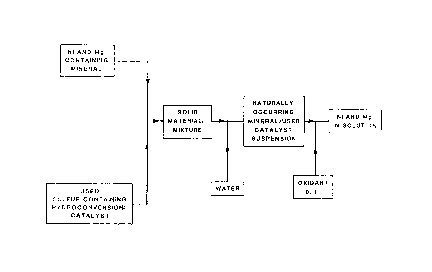
BRIEF DESCRIPTION OF THE DRAWINGS
A detailed description of the invention will now be
given, with reference to the accompanying Figure 1 which
shows a block diagram illustrating the process of the
present invention.
DETAILED DESCRIPTION
The drawing illustrates the several stages which
comprise the overall process for recovery of nickel and
magnesium from metal ores, according to the invention.
As previously discussed, the preferred source of
metal ions and sulfur is a spent or used sulfur
containing iron based hydroconversion catalyst. This
catalyst is preferably iron based, having a composition
of iron in the range of 20-70% by weight of Fe. The
spent catalyst also preferably includes a sulfur content
in the range of 10-40%. Prior to use, the spent catalyst
is preferably powdered to an average particle size of 200
microns or less, in order to insure a good reaction
during the contacting procedure. The material to be
treated may be a typical nickel and magnesium bearing
metal ore such as serpentine or olivine. The metal ore
preferably has a nickel content of at least 0.1% by
- weight, and also has a magnesium content of at least 0.1%
by weight. The content of nickel and magnesium in these
metal based ores more preferably falls in the range,
respectively, of 0.1-10.0% by weight and 0.1-30% by
weight.
A
2062350
Prior to being mixed with the spent catalyst, the
ore is also preferably powdered to an average particle
size of 200 microns or less in order to insure a good
reaction with the spent catalyst during the contacting
procedure. The spent catalyst and ore are then admixed
into a solid material mixture having a ratio by weight of
catalyst to ore of 1:2-8:1. The mixture ratio more pre-
ferably falls within the range of 1:1-5:1. At these
ratios, it has been discovered that the recovery percent-
age of nickel and magnesium greatly exceeds the recoverypercentages obtained by the prior art processes. The
solid material mixture is then added to a water solution
to form a suspension having a pH no greater than 4. At a
pH higher than 4, the desired chemical reaction during
the contacting procedure may be inhibited, and other
undesirable reactions take place which result in
production of a mix of solids.
An oxidant is then added to the suspension under
controlled conditions so as to avoid a "boiling" of the
particulate material and water contained in the
suspension. Such a "boiling" could entail the loss of
either particulate material or water, which could result
in variation of the pH of the reaction medium and adverse
effects on the contacting procedure. Further, the
25- injection rate of the oxidant should be sufficient to
maintain a substantial homogeneity between the spent
catalyst, ore and oxidant. During this reaction, the
temperature is elevated to a range of 50-350~C, and
~: A
2062350
the pressure is also elevated to a range of 200-600 psi.
More preferable ranges for the temperature and pressure,
respectively, are 150-300~C and 300-500 psi.
During the subjection of the mixture to the elevated
temperature and pressure, iron will displace the
magnesium and nickel, which will go into the liquid
solution. This nickel and magnesium can then be removed
from the solution by standard procedures which are known
in the art. Through such procedures, the expected
recovery is higher than 85% for the nickel content and
90% for the magnesium content of the ore.
The oxidant to be used in the process is preferably
either oxygen, air or a mixture of the two.
The composition of typical ores to be treated by
such a process is indicated below in Table I.
TABLE I
Mineral Percentaqe bY Weiqht
Ni 0.1-20
Fe 0.1-30
Si 0.1-40
Al 0.1-40
Mg 0.1-40
Compositions such as these are typically found in
silicates such as olivine, cordierite, pyroxene,
amphibole, kaolinite, serpentine, clays and the
aluminates.
A
...
~1-397
A typical procedure according to the ~ will be
illustrated in the example below.
Example:
For this example, the spent catalyst used was a
naturally occurring iron metal which was used during a
hydroconversion process of heavy oils. The chemical com-
position of this spent catalyst is given below in Table II.
The nickel and magnesium containing laterite ore of
serpentine type was obtained from the Loma de Hierro
reservoir in Western Venezuela. Its chemical composition is
also given below in Table II.
TABLE II
Catalyst Laterite
S21.3 wt% 0 wt%
C30.6 wt% 0 wt%
Ni1984 ppm 1.95 wt%
Fe54.5 wt% 6.57 wt%
V9968 ppm 0 ppm
Si4797 ppm 13.81 wt%
Al8141 ppm 4150 ppm
Mg0 wt% 17.6 wt~
Several mixtures of this spent catalyst and metal ore
were prepared and treated according to the procedure
described above. These samples were prepared at various
different ratios of catalyst to ore as shown below in Table
III.
TABLE III
Catalyst/Mineral RatioExtraction (%)
Ni Mg
0/1 0 0
2/1 73.783.2
3/1 81.987.5
4/1 87.392.0
2062350
A blank experiment, wherein the catalyst to ore
ratio was 0:1 indicated at 0% extraction of nickel and
magnesium. Further, it was apparent that as the ratio of
catalyst to ore increased from 2:1 to 4:1, the recovery
percentages of nickel and magnesium increased.
Specifically, at a catalyst to metal ratio of 4:1, nickel
was extracted at the rate of 87.3% and magnesium was
extracted at the rate of 92%. Clearly this compares
favorably with the recovery percentages obtained in the
processes disclosed in the prior art, and provides a
further desirable result in that the source of iron and
sulfur is a waste spent catalyst rather than materials
purchased specifically for the procedure, magnesium is
recovered rather than injected, and the extraction ratios
are greatly improved.
This invention may be embodied in other forms or
carried out in other ways without department from the
spirit or essential characteristics thereof. The present
embodiments are therefore to be considered as in all
respects illustrative and not restrictive, the scope of
the invention being indicated by the appended claims, and
all changes which come within the meaning and range of
equivalency are intended to be embrace therein.
f~ A
,. ~