Note: Descriptions are shown in the official language in which they were submitted.
5~WO93/17958 PCT/AU~3/00099
~ 1 3 0 ~
PROC~SS F~R PROD~CING A GASEO~S PRO~CT
The present invention relates to processes for producing a
gaseous product. In one particular application, ~he
invention relates to a process for the steam reforming of
hydrocarbons.
Steam reforming of hydrocarbons such as natural gas or
naphtha involves reaction with steam to produce a mixture of
carbon oxides, hydrogen and water vapour. This gas mixture,
common}y referred to as "syngas" in tha petrochemical
industry, is the starting point for numerous catalytic
synthesis products such as methanol, gssoline and wax. It is
also useful for metallurgical reduction processes, production
of eleFtrical energy from fuel cells and -production of
hydrogen for use in, for example, ammonia synthesis.
The reforming reaction is strongly endothermic and is
conYentionally performed at around 800-900C, at pressures of
10 to 30 bar in catalyst-filled, high-alloy tubes. Thes~
tubes are usually 100 to 150mm in diameter and around lOm in
length; heat transfer from hot flue gases on the outside of
the tubes to the reacting mixture within is a ma~or
rate-limiting step in the o~erall process. Thermal driving
forces for heat transfer are large and tube layout is
dictated, to a large degree, by the need for sufficient
radiation view-factors to avoid the formation of hot and
cold-spots on the tubes. This results in a need for large
furnace volumes and plan areas - a constraint more difficult
to accept in some applications than in others.
Numerous proposals to reduce the size of the steam reformer
have been developed. For example, US Patent No. 4888131
describes a process whereby a sharp reduction in furnace -~
volume is possible by introducing oxygen into the system.
~.
WO93/17958 PCT/AU93/00099
~9~ ~ 2 -
Partial oxidation schemes such as this are useful when oxygen
injection is feasible and does not substantially downgrade
the quality of the product gas. However, in other
application examples (eg ship-board gas conversion
operations) it may be undesirable to use oxygen for safety
and economic reasons and other means for reducing the volume
of the reformer must be considered.
Immersion of catalyst-filled tubes in a fluidized bed, as
de~cribed in, for example, British Patent No. 2126118, is an
alternative means for reducing the size of an otherwise
conventional reformer. In this case heat transfer to the
outside of the tubes is achiev~d via the hot par~icles that
comprise the fluidized bed. This results in a more even heat
distribu~ion and a significant reduction in the space needed
between tubes. However, this process does ha~e a number of
disadvan~ages including hot erosion of the high-alloy tubes
and unsuitability for ship-board applications, where wave
motion can affect the integrity of the tubes.
US Patent No. 3524819 describes a process in which a mixture
of hydrocarbons and steam is reformed in an internal-free
fluidized bed of fine-grained catalyst. Heat for the
reforming reaction is provided by continuously withdrawing a
portion of the catalyst and heating this catalyst by contact
wi~h hot flue gases before returning it to the reforming bed.
Howe~er, this process exposes the catalyst (in bulk) to high
temperatures which may cause the catalyst to sinter, thereby
losing catalytic activity and requiring rapid (and costly)
replacem~nt.
Hence, an ob~ect of the present invention is to provide an
alternative prscess utilising fluidized bed technology, which
may permit a reduction in the size of the steam reforming
plant.
U ~ ~ ~ O ~ ~ 9 9
~E~ 2 AU6 l~S3
3 ~ 1 3 0 .'3 .~ ~
Thus, according to a first aspect, the present invention .
provides a process for producing a gaseous product comprising
passing reactants into a fluidized bed operated under
conditions such that a reaction takes place to produce the
gaseous product, the fluidized bed comprising first particles
having a density and size distribution such that at least a
portion of the first particles are entrained by the gaseous
product leaving the fluidized bed and second particles having
a density and size distribution such that said second
particles substantially remain in the fluidized bed,
substantially removing the gaseous product and entrained
first particles from the fluidized bed, separating the first ~-
particles entrained in the gaseous product from the gaseous
product and returning the separated first particles to the
fluidized bed wherein at least a portion of the energy :~
required for the reaction is supplied by heating the
separated first particles during their return to the
fluidized bed. ~.
The separated first par~icles are preferably heated by
passing the particles through a chamber where fuel gas or the
like is combusted. The separated first particles may be :~
cat~lytic, but more preferably they are inert.
'.
'~
. . .
~CT~AU 9 3 / O O O 9 9
1 2 AUG 1993
. .
~ .
4~130g9 ~
It may be appreciated that in the process according to the
invention, a minor portion of second particles may escape the
fluidized bed by entrainment in the gaseous product. When
the second particles are catalytically active, such "escapes"
will result in a loss of catalyst. To avoid this loss, it is
preferable that following separation of the particles from
the gaseous product and prior to the first particles being
subjected to heating, any second particles are substantially
separated from the first particles and returned to the
fluidized bed. This arrangement also permits the fluidized
bed to be operated such that an amount of a mixture of first
and second particles i5 normally entrained in the gaseous
product.
Thus, in a second aspect, the present invention provides a
process for producing a gaseous product comprising passing
reactants into a fluidized bed operated under conditions such
that a reaction takes: place to produce a gaseous product, the
fluidized bed comprising f irst and second particles having
density and size distributions such that at least a portion
of both first and second particles are entrained by the
gaseous product leaving the fluidized bed, removing the
gaseous product and entrained first and second particles from
the fluidized bed, separating the first and second particles
~entrained in the gaseous product from the gaseous product,
substantially separating the first and second particles, and
thereafter separately returning the separated first particles
and separated second particles to the fluidized bed wherein
at least a portion of the energy required for the reaction is
supplied by heating the separated first particles during
their return to the fluidized bed.
ILEAlsu~TuTF SH~
~ W093/1795~ LJ P~T/AU93/00099
. ~
-- 5
The surface area of the first particles is not critical~
However, the first particles are preferably catalytically
inert. Examples of suitable materials include but are not
limited to dense alumina, zirconia, rutile, metals such as -
nicksl or mixtures thereof. The second particles are
preferably catalytieally active.
The first and second particles may be separated by a
fluidized bed segregator. :~
~.
I~ will be appreciated that the fluidized bed reactor and
a~30c~ated equipment to facilitate the separation and return
of particles entrained in the gaseous product comprise a
circulating fluidized bed system.
The prbcess according to the inven~ion may be suitable for a
number of h~at driven reactions including pyrolysis :~;
reactions, nap~ha cracking, ethane con~ersion to ethane and
maleic anhydride synthesis. H~we~er, the proce~ses are
particularly ~uitable for steam reforming of hydrocarbons to
produce a gaseous product containing hydrogen and carbon
oxides. Accordingly, the invention is hereinafter described
in relation to this application.
':
In this application of the inYention, the reactants are
hydrocarbons and steam and the fluidized bed is operated ;:
under conditions such that a steam reforming reaction ~akes
place. r~
The flsidized bed reformer may be operated at a temperature
from 750 to 950C preferably in the range of 800 to 900C
and at a presBure in the range of 5 to 80 bar, praferably 10
to 30 bar.
: .
The first particles are preferably catalytically inert and
haYe a maximum particle size of less than about 0.5mm with an
WO93/17958 PCT/AU~3/000~9 :-
-- 6 --
~ 1 3~9!~
average particle size preferably lying in the range of about
O.03 to 0.3mm. ~-
The second particles are preferably catalytically active and
may have a function and chemical composition typical of known
steam reforming catalysts (e.g. ~i or Ru supported on
alumina, calcium alumnate or ~irconia~. The~e particles may
be porous and display high catalytic activity for the steam
reforming reaction. Average par~icle size preferably lies
in the range of 0.7 to 7mm with little or no material smaller ~:
in size than 0.Smm. ;~
The suspension density of the reformer vessel may be about
200 to 2000 kg/m3, preferably about 1000 to 1800kg/m3.
The separated particles are preferably heated to a
tempersture of about 950 to 1200C, preferably about 1000 to
1100C during their retuxn to the fluidized bed reformer.
The invention is further described by way of the following
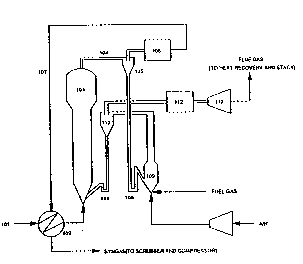
non-limiting examples and with ref~rence to Figures 1 and 2. --
Figure~ 1 and 2 provide diagrammatic repre~entations of
apparatus suitable for performing the process of the present ~.^
inYention. : :~
EXAMPLE 1
A mixture of hydrocarbons (natural gas or higher hydrocarbons
such as ethane or naphtha) and steam 101 is pre-heat~d in -
heat exchanger 102 before being fed into the bottom of the
fluidized bed reformsr 103. Unit 103 contains a dense-phase
fluidized bed with two types of particle present:
i) a component which is catalytically acti~e, similar in
structure and pu~pose to con~entional alumina-supported
Ni catalysts le.g. ICI 57-s~ries catalysts). These
` W093/17958 PCT/AU93/00099
,
_ 7 _ ~1 3 0~
particles may be porous and display high catalytic
activity for the steam reforming reaction. A~erage
particle size lies in the range 0.7 to 7mm, with little
or no material smaller in size than 0.5mm.
ii) A component (e.g. fused alumina) which has low specific
surface area and is catalytically inert. This component
of the population is smaller in size than the
catalytically active component, all particles being
smaller than 0~5mm and average size for the inert
component lying in the range 0.03 to 0.3mm.
Fluidized bed reformer 103 is operated surh that ths fine,
inert component of the particle population is preferentially
entrained from the top of ~he vessel while the coarse,
catalytic component remains in the ve sel. Steam reforming
takes place within vessel 103 at a temperature of about 850C
and a pressure in the range 10 ~o 30 bar. Effluent gas 104,
along with entrained fine solids, en~ers cyclone 105. ~ulk
disengagement occurs and the cyclone oYerhead stream enters
hot gas cleaner 106. In this unit the bulk of the remaining
solid is removed by, for example, a hot multicyclone system.
Cleaned gas 107 leaves the cleaning unit 106 and passes to
heat exchanger 102 before being scrubbed and passed on for
use downstream.
Solids from the underflow of cyclone 10~ are fed, Yia
pressure recovery system such as loopseal 108, to chamber 103
where fuel gas ~nd air are combusted at a pressure similar ~o
tha~ in vessel 103. Solids are heated from 800-850 C to
around 1000-1100 C and are subsequently remo~ed from the
hot flue gases in cyclo~e 110. From here the hot solids are
returned to the base of the reforming vessel 103 via pressure
recovery system 111 to pro~ided the heat necessary for the
reforming reaction. The bottom inlet system for hot solids is
designed to ensure a larg~ mass flux of bed material is
WO93/17958 PCT~AU93/0009~ .
~9~ 8 -
available to rapidly dilute the hot solids stream, thereby
avoiding the exposure of catalytically active particles to
(destructive) high temperatures.
.
Hot, pressurised flue gas from the overflow of cyclone 110 is
used for energy recovery purposes. Hot gas cleaning is
carried out in unit 112, the precise form of which is not
critical. Partial cooling (to 800C for example) followed by
a micro-cyclone system may be used, or a ceramic
barrier-filter system similar to those currently under
dPvelopment for pressuxised fluidized bed combustion and
gasification. The cleaned gas is subsequPntly expanded
~hrough turbine 113. From here it is subjected to further
heat recovery and finally vented.
:
EX~NPLE 2
.
A mixture of hydrocarbons ~natural gas or higher hydrocarbons
such as ethane or naptha) and steam 20} is pr~-heated in heat
exchanger 202 before being fed into the bottom of a fluidized
bed reformer 203. Unit 203 contains a dense-phase fluidized
bed with two types of particles as described in Example 1~
Steam reforming takes place within vessel 203 again at a
temperature of about 850 C and a pressure in the range 10
to 30 bar. However, in this case the fluidized bed reformer
203 is operated such that a mixture of bed solids are
entrained in gas leaving the vessel. Effluent gas 204, along
with entrained solids, enters cyclone 205. Bulk
di~engagement occurs and the cyclone overhead stream !enters
hot gas clean~r 206 where the bulk of the remaining solid is
removed by, for example, a hot multicyclone system. Cleaned
gas 207 leaves the cleaning unit 206 and passes to heat
exchanger 202 before being scrubbed and passed on for use
downstream
~WO93/17958 l1 3 Q g g -~ PCT/AU93/00099
Solids from cyclone 205 are fed to fluidized.bed segregator
208. Unit 208 contains a low velocity bubbling fluid-zed bed
within which the coarse ~catalytically active) particles
preferentially sink to the bottom and are returned, along
with some fine heat-carrier solids, to unit 203. The
fluidizing gas for unit 20~ may be any convenient gas, but is
preferentially steam or fuel gas.
Fine heat-carrier solids, substantially free of coarse
(catalytically active) solids, leave the top of unit 208 and
enter unit 20g where fuel gas and air are combusted at a
pressure similar to ~hat in vessel 203. Solids are heated
from 800 -850 C to around 1000 -1100 C and are
subsequently removed from the hot ~lue gases in cyclone 210.
From here the hot solids are returned to the base of
reforming vessel 203 ~ia pressure recovery system 211 to
pro~ide the heat necessary for the reforming reaction.
Hot, pressurised flue gas from the overflow of cyclone 210 i3
us~d for energy recovery purposes. ~ot gas cleaning is
carried out in unit 212. Partial cooling (to 800 C for
example) followed by a micro-cyclone system or a ceramic
barrier-~ilter system may be used. The cleaned gas is
subsequently expanded through turbine 213, after which it may
be subjected to further heat recovery and finally vented.
The present invention offers a compact system, that may be
opexated free of the need for oxygen, for application to
processing in situations such as shipboard reforming. The
reforming vessel itself is free of expensive,
high-maintenance tubes of high-alloy metal and associated
maintenance and safety issues arising from wave motion. It
may be configured to avoid substantial exposure of catalyst
particles to high temperatures which would otherwise promote
sintering, thereby achieving good catalyst utilisation.
W093/17958 PCT/AU93/00099
~ 13~99 ~ lO
Those skilled in the art will appreciate that the invention ~
described herein is susceptible to variations and ~:
modifications other than those specifically descxibed. It is
therefore to be understood that the invention includes all
such variations and modifications which fall within its
spirit and scope.