Note: Descriptions are shown in the official language in which they were submitted.
21 83 1 1 1
PRIMING SYSTEM
Technical Field
This invention relates to a device and method for venting and/or priming
fluid transfer and/or proce~cing systems. The present invention is particularly useful
S in the ~ isl~ation of parenteral fluids, e.g., drugs, nutrients, hydration solutions,
and/or transfusion products.
B~ oulld of the Invention
Many fluid tran~sfer sy~stems such as fluid proc~scin~ systems function more
effi< iently if they can be at least partially purged of air or ga~s before use. For
10 example, it may be desirable to prime a fluid transfer system, e.g., to displace or
remove air from devices and/or tubing in the system to allow them to be filled with
liquid before colllinuously operating the system. Since pockets of air or gas can
block or slow the flow of liquid, ...i.-i...i~;.-~ or elimin~ting the presence of air or gas
can allow the liquid to flow more quickly through the system.
Additionally, some ll~srer systems, e.g., for ~lminictering fluid to a patient,
should minimi7e or prevent air or gas from passing beyond a certain location in the
system. For example, some illll~Vellous (IV) systems in~ de a drip chamber or a
bubble trap to ca~ e air or gas so that it will not be ~iminictered to the patient.
The drip chamber can also be used to allow the operator of the system to monitor20 the rate at which the fluid is ~fli.~i~lered. For example, the operator may observe
the drip chamber to ensure that the patient is receiving the liquid at a given rate,
e.g., a rate of so many drops of liquid per minllte. If desired, the rate can bechanged and the new rate can be monitored by observing the rate at which the drops
of liquid pass through the chamber.
There are drawbacks to conventional fluid transfer systems. For example,
some systems for ~tiministering transfusion products require a labor intensive effort
by the operator of the system. Illustratively, when tr~n~ in~ blood or blood
products, especially when filtering and ~ ering blood at the patient's bedside,
the operator of the system may have to start the flow of blood from the source bag
with the drip chamber initially inverted until the drip chamber is about 2/3 full.
Then the partially filled drip chamber is placed in the upright position and the
21 831 1 1
system is primed. After the system is primed, blood can be ~clminictered, and the
rate monitored through the drip chamber.
The operator must careful monitor the initial filling of the inverted drip
chamber, since overfilling the chamber leads to difficulties in monitoring the drip
5 rate. For example, a high level of fluid in the chamber can provide insufficient
space for a drop to form before it contacts the fluid in the chamber, m~kin~ it
difficult to count drops. On the other hand, underfilling the chamber increases the
possibility that air will pass through the chamber, with possibly adverse consequences
for the patient.
Accordingly, there is a need in the art for a devioe and method that provides
for efflcient p,illling of a fluid l.roc~ system, e.g, that allows a drip chamber to
be filled to a desired level without a labor intensive effort. In particular, there is a
need in the art for a ~evel setting" drip chamber, that automatically vents air or gas,
without overfilling.
The present invention provides for ameliorating at least some of the
disadvantages of the prior art. These and other advantages of the present invention
will be apparent from the description as set forth below.
Summary of the Invention
In accordance with the presGnt invention, a device for use in a fluid ~ Çer
20 system is provided that vents gas, and seals after venting, without overfilling with
liquid. Illu~llalively, gas can be vented from a flow metering device such as a drip
chamber, and the drip chamber can be fîlled to a desired level of liquid, wherein the
amount of liquid in the chamber is less than the total liquid capacity of the chamber.
In an embodiment, once fluid flow toward the device is initiated by the operator of
25 the fluid transfer system, the device automatically vents gas and fills to a desired
level, without operator involvement. Moreover, the device can be filled with liquid,
and subsequently utilized during ~ ation of the liquid, while keeping the
device in an essentially upright position, and without allowing gas or air to enter the
device through the vent.
30 Brief Description of the D.~whlg~
Figure 1 is a side view an embodiment of a venting device according to the
present invention.
21~31 1 1
Figure 2 is a view of the device of Figure 1 along line A-A.
Figure 3 is a view of the device of Figure 2 along line B-B, showing a porous
medium for passing gas therethrough located in the device.
Figure 4 is a side view of the upper chamber and the porous medium of the
5 device of Figure 1.
Figure 5 is a side view of the lower chamber of the device of Figure 1.
Figure 6 is an embodiment of a fluid processing system according to the
present invention, in~ ling a venting device, and a device for filtering biological
fluid.
Figure 7 is an embodiment of another fluid processing s,vstem according to
the present invention, inrlu<ling a v~ g device.
Figure 8 is another embodiment of a venling device accolding to the present
invention.
Specific Description of the Invention
In accordance with the present invention, a device for transferring fluid is
provided colll~ ,mg a housing com~lisillg a fluid reservoir for gas and liquid; a vent
communicating with the housing, the vent colll~lisillg a liquid sealable porous
me~ lm that allows gas in the housing to pass through the me~ m until the mediumis contacted by the liquid, the vent and the housing being cooperatively arranged to
20 allow the gas to be vented from the housing and for liquid to ffll the hou~illg to a
predetermined level that is less than the total liquid capacity of the housing. In one
embodiment, the device is a flow metering device such as a drip chamber.
The present invention also provides a drip chamber co~ g a housing
in~llltling a porous m~Aillm that allows liquid to fill the housing to a predetermined
25 level without completely filling the housing with liquid, the meflium allowing gas to
pass from the housing until the mellillm is contacted by the liquid, the medium
incl~l-ling a liquophilic element and a liquophobic element.
In accordance with the invention, a system for transferring fluid comprises a
coll~ainer suitable for holding a liquid to be transferred; a device in fluid
30 coml,lul~ication with the container, the device comprising a housing suitable for
holding gas, and liquid from the container; and a vent colllyli~ing a liquid sealable
porous medium that allows gas but not liquid in the housing to pass through the
medium until the medium is contacted by the liquid and gas flow stops, the device
21831 1 1
allowing a desired amount of liquid to held in the housing wherein the amount isless than the total liquid capacity of the housing. In an embodiment, the deviceco~ lises a drip chamber, and the system also includes a leukocyte filter interposed
between the drip chamber and the container.
The present invention also provides a method for transferring fluid
Co~ lisillg l,a~shlg a fluid into a device colll~lising a housing including a fluid
reservoir for gas and liquid and a vent co~ -micating with the housing, the ventCOlll~ illg a liquid sealable porous medium that allows gas in the housing to pass
through the m~lillm until the medium is cont~cte~l by the liquid; passing liquid into
the housing and passing gas through the porous me~ lm until the liquid cont~c the
porous ~ m and the housing is filled to a predetermined level that is less than
the total liquid capacity of the housing. In an embo~lim~nt, the fluid to be
transferred is blood or a blood component, and the method also includes
~(l."i";~lel~g the blood or blood component to a patient. In some embodiments,
the method includes filtering the blood or blood component, e.g., to remove
leukocytes from the blood or blood component, before ~tlminictering it to the
patient.
Emb~limentc of the present invention provide for efficient separation of gas
or air from a fluid flow path. More preferred embodiments provide for efficient
separation of gas or air from fluid delivery systems, e.g., systems involving
parenterally ~ ni~le~ed fluids, such as h~ v~llously ~ ered liquids ~for
example,~ drugs and nutrients); and biological fluids such as blood and blood
colllpollents. Thus, gas can be separated from the fluid deliveIy system, e.g., vented
to the atmosphere, or the gas can be directed to a desired location within the
system, e.g~ away from a desired liquid flow path.
Each of the components of the invention will now be described in more detail
below.
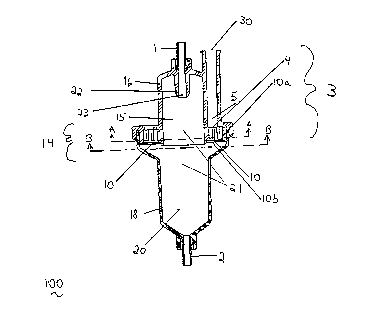
In the embodiment illustrated in Figures 1-5, device 100 includes a housing
14, having an inlet 1 and an outlet 2; a lumen 21 for liquid flow between the inlet
and the outlet; and a vent 3 providing a gas flow path. Vent 3 co.~l~rises a porous
me~ lm 10 that allows gas, but not liquid, to pass from the lumen 21 through a port
4 into a gas passageway 5 and through a port 30. Typically, porous medium 10 is
located in the housing 14, and has a surface 10a facing the inlet 1, and a surface 10b
facing the outlet 2. Preferably, porous medium 10 has a portion of the surface 10a
21831 1 1
near or adjacent to port 4 of gas passageway 5.
Typically, as illustrated in Figures 1, 3, and 4, housing 14 includes a first
portion 16, having an inlet 1, and a groove 29. The first portion 16 also includes a
nipple ~ having a port 23 leading to lumen 21. Preferably, the nipple 22 and port
23 allow fluid to enter the lumen 21 in the form of droplets.
As illustrated in Figures 1 and 5, housing 14 also includes a second portion 18
having an outlet 2, and a lip 19. In the embodiment shown in Figure 1, the firstportion 16 and the second portion 18 are sealed to form a first chamber 15 and asecond chamber 20 encolllyassillg lumen 21 and porous medium 10.
~l.,fe-ably, as illustrated in Figures 2 and 3, porouc medium 10 is ~nmll~r in
shape, and surface lOa faces a series of ribs 25, 26, 28, and/or f~nn.olc 24, 27 in the
first portion 16. Typically, in those embodiments having an ~nmll~r porous ...e-liul--
10, the ribs 25, 26, and 28, and the channels 24 are generally concentric. In the
illustrated embodiment, first portion 16 also includes radial channels 27.
Figures 6 and 7 illustrate embodiments of a typical system 500 ~ltili7ing device100 during the ~ .ation of at least one parenlelal fluid to a patient. Figure 6
also includes an optional filter 300 for filtering the parenteral fluid before
ering it to the patient. The illustrated embodiments include a colllai~er 200
for holding a parenteral fluid, which is in fluid conlll,ul~ication, via conduits such as
conduits 21Q 220 and 230, with the filter 300 (if present), the device 100, and the
patient. The system also inrludes at least one, and more prefe-~bly at least two,
flow control devices 400 such as clamps.
In another embodiment as illustrated in Figure 8, wherein like co-,-~onents
have like reference numbers, the porous medium 10 is located more exteriorly to
housing 14. For example, porous medium 10 can be disposed in a separate vent
housing 50 adjacent housing 14. Gas and liquid pass from lumen 21 through port 7and passageway 6 to porous medium 10 in vent housing 50. Porous medium 10
allows gas but not liquid to pass therelhrough to port 30.
In accordance with the invention, the vent 3 col~lises a porous medium 10
that allows gas, but not liquid, to pass therethrough. Porous medium 10, which is
preferably a membrane or film, preferably includes a liquophobic portion and a
liquophilic portion. Even more preferably, the porous medium 10 includes at least
one liquophobic porous element or layer and at least one liquophilic porous element
or layer. Typically, a liquophobic element is superimposed on a liquophilic element.
21831 1 1
Preferably, the liquophobic element or layer is hydrophobic, and the liquophilicelement or layer is hydrophilic.
Both the liquophobic element and the liquophilic element allow gas to pass
through. However, the liquophobic element is not wettable, or poorly wettable, by
5 the liquid being transferred in the system, and the liquophilic element is wettable by
the liquid. Once the liquophilic element is wetted by the liquid being ll~sÇerled,
gas flow through the vent 3 stops, and the metli~lm 10 is sealed. Once the medium
is sealed, gas or air external to the device 100 is also pr~vellled from flowingthrough the me-linm, even when the level of liquid in the device subsequently
10 decreases.
In the embodiment shown in Figure 1, porous m~li~lm 10 inchldes a first
surface 10a and a second surface 10b. ~efel~bly, porous me-lillm 10 inrllldes two
superimposed layers, with surface 10a representing the surface of liquophobic layer
that is not facing the liquophilic layer, and with surface 10b representing the surface
15 of liquophilic layer that is not facing the liquophobic layer. As illustrated in Figures
2-4, a portion of surface 10a is near or ~dj~ce.nt port 4 at the end of gas passageway
5. In another embodiment in~ ling superimposed layers, as illustrated in Figure 8,
a portion of surface 10b is near or ~dj~c~-nt port 7 at the end of passageway 6.In accordallce with the embodiment illustrated in Figures 1-3, the porous
20 meflinm 10 colllylises the liquophobic element and the liquophilic element arranged
in the housing 14 to vent gas lllrou~ll gas passageway 5 until the liquophilic elem~o-nt
is wetted with the liquid being transferred.
~ imil~rly, in accordallce with the embo liment illustrated in Figure 8, the
medium 10 colllylises the liquophobic element and the liquophilic element all~ged
25 near the housing 14 to vent gas from passageway 6 until the liquophilic element is
wetted with the liquid being transferred.
In the prefelled embodiments, gas flow autolllalically stops when the
liquophilic element is wetted, without liquid sl-bst~nti~lly y~SSillg through the
liquophobic element. Once the gas flow stops, the device 100 has been filled to the
30 desired level, without overfilling. Gas re.--~i..;.-g in the inteAor of the housing 14
will not be displaceable, and the liquid will stop rising in the houshlg. Furtherrnore,
once the liquophilic element is wetted, the medium 10 is sealed, preventing gas or
air from entering the device 100.
A variety of materials may be used, provided the requisite properties of the
21 831 1 1
porous medium are achieved. These properties include the n~cçcc~ry strength to
handle the differential pressures encountered in use and the ability to provide the
desired permeability without the application of excessive pressure. Suitable starting
materials are synthetic polymers inl~ltl-ling polyamides, polyesters, polyolefins,
S particularly polypropylene and polymell-ylpelllene, perfluorinated polyolefins, such as
polytetrafluoroethylene, polysulfones, polyvinylidene difluoride, polyacrylonitrile and
the like, and colul)alible ~ ules of polymers. Within the class of poly~midçc, the
prefellcd polymers include polyheY~methylene ~lip~mi~e, poly-~-caprol~ct tm,
polymethylene sebacamide, poly-7-~minoheptano~mide, polytetramethylene
3~lip~mi~e (nylon 46), or polyh~Y~methylene ~7~le~mi<le~ with polyhexamethylene
~lip tmi~e (nylon 66) being most preferred. Particularly preferred are skinless,subsPnti~lly alcohol-insoluble, l~drol~hilic polyamide ~el~anes, such as those
described in U. S. Patent 4,340,479.
In those embodiments in-~lu-ling ~ ;ni~lering fluid(s) to a patient, the
medium 10 is typically chosen so that the sterility of the system is not co~ioll~ised.
For example, meflillm should preferably have a bacterial blocking pore structure,
e.g., a pore size of about 0.2 micrometer or less, to preclude bacteria from entering
the system. In those embodiments having liquophobic and liquophilic layers, either
or both layers can have the bacterial blocking pore structure.
The porous medium 10 can include additional layers or elements, e.g., for use
as spacers and/or supports. An exemplary ~uppoll or spacer layer can be a mesh or
screen.
Exemplary porous media, in~ lin~ liquophobic and liquophilic elem~nt~, are
as disclosed in International Publication No. WO 91/17809 and U. S. Patent Nos.
5,126,054 and 5,451,321.
The housing 14 can be fabricated from any suitable rigid impervious materiaL
inçlll(ling any impervious thermoplastic material, which is compatible with the fluid
being procecce-l For example, the housing can be fabricated from a metaL such asstainlesc steel, or from a polymer. In a preferred embodiment, the housing is a
polymer, more preferably a trallspalenl or translucent polymer, such as an acrylic,
polypropylene, polystyrene, or a polycarbonated resin. Such a housing is easily and
economically fabricated, and allows observation of the passage of the liquid through
the housing.
In some embodiments, the housing may be constructed of
2 1 83 1 1 1
diethylhexylphthalate (DEHP) free and/or phth~l~te free material.
The surfaces of the housing cont~cting the fluid may be treated or untreated.
For example, the surfaces of the housing cont~cting the fluid may be rendered
liquophilic for better p~ lillg. Methods for treating the surface of the housingS include but are not limited to radiation grafting and gas plasma treatment.
The housing may be configured for ease of use. For example, the housing
may inrl~de a bracket or hook for ease of mounting or ~7Ul)l)Oll during
ion of the parenteral fluid.
The housing can be sealed as is known in the art. For example, first portion
16 and second portion 18 can be welded together, e.g., after fitting lip 19 into groove
29.
Additionally, the porous medium 10 can be sealed wit_in the housing 14 to achieve
coll~cnience of use, rapid priming, and efflcient air clearance. Suitable methods for
sealing, fitting, and/or bonding the medium within or to the hollcing are known in
15 the art. For example, the porous me~ lm 10 can be con~ression sealed or
interference fit within the housing, or bonded to the housing. Preferably, the porous
medium is welded to the housing, e.g., heat welded or ultrasonically welded.
The housing 14 can inrlude an all~l~l,lent of ribs, walls or projectionc, or
may conll),ise one or more channels, grooves, conduits, passages, or the like which
20 may be se~ell~ e, paralleL curved, radial, spiraL or have a variety of other
co~l~;uldlions~ Typically, at least one projection faces the first surface 10a and/or
faces the second surface 1ûb of the porous me-linm lQ The ribs, walls or
projections may be uced to define the ch~nn~lc, grooves, con-hlitc, or passages.Typically, the housing 14 includes at least one rib, wall, or projection to support
25 and/or position the porous medium 10 within the housing. In some embo~lim~ntc,
the housing in-~hl(lec at least one rib, wall or projection to i~ rove the efficiency of
gas flow, e.g., by SU~Ol ~ing or positioning the porous medium 10 within the housing
while providing clearance between portions of the first surface 10a and the housing.
Typically, as illustrated in Figure 2 first portion 16 includes ribs 25 and
30 rh~nnel~ 24, which are preferably concentric ribs and channels, to allow moreefficient flow of gas to the gas passageway 5. The first portion can also have radial
channels 27, and at least one, more preferably, at least two, additional ribs such as
concentric ribs 26 and 28. Figures 2 and 3 also illustrate rib 26, that preferably
positions the medium 10 with respect to first portion 16.
21831 1 1
In the illustrated embodiments, the first portion 16 and the second portion 18
are sealed to form a first chamber 15 and a second chamber 20, encomp~c~in~
lumen 21. The first and second chambers can be similar in volume, or one chambercan be larger in volume than the other. For example, in some embodiments
5 accor(ii"g to Figure 1, the ratio of the volume of the first chamber to the second
chamber can be in the range of about 1:1 to about 3:1.
In some embodiments, e.g., as illustrated in Figure 8, the porous me(~ m 10
can be located in an additional housing, such as vent housing 50. Exemplary
housings incl~lcle those disclosed in International Publication No. WO 91/17809 and
U. S. Patent Nos. 5,126,054 and 5,451,321.
In some embodiments, the device 100 can also inrlnde a cap or closure, e.g.,
for co~/eli~g or sealing port 30.
The device 100 may be incorporated into a variety of fluid transfer and/or
procç~sin~ systems or sets. Embodiments of exemplary systen~s 500, which are
15 preferably parenteral fluid ~ lion sets, are illustrated in Figures 6 and 7.
Typically, the system inrlnd~ at least one c~n-~in~r for holding the fluid to
be lral~srelled, e.g., ~ ered, and one or more conduits that provide fluid
cou.lllullication between the container, the device 100, and the ~lestin~tion of the
fluid, e.g., a patient receiving the ~ lered fluid. Preferably, as illustrated in
20 Figure 7, the system also includes at least one filter for filtering the fluid to be
lministered.
The container 200 which can be used in the system can be co~llucted of any
material col~tible with the fluid to be l-~llsf~;lled. A wide variety of suitable
conlainers are already known in the art. Typically, cont~iner 200 may be colllposed
25 of a flexible material, for example, polyvinyl chloride (PVC). An exemplary
conlainer is a flexible bag, e.g., as is used for holding blood or blood coll",onents.
Alternatively, the containers may be composed of a non-flexible material, for
example, polypropylene, acrylonitrile butadiene styrene (ABS), polycarbonate, orstainless steel. It is intended that the invention should not be limited by the type or
30 composition of the container being employed.
As with the containers, the conduits 210, 22Q and 230 can be constructed of
any material that is compatible with the fluid to be transferred. A plefelled
material is PVC. As used herein, the conduits are any tubing or means which
provide fluid comlllullication between the various components of the ~lmini~tration
21831 1 ~ -
set. At least one flow control device 400 such as a clamp (inrlll~ling, for example, a
roller clarnp), seaL stopcock, valve, transfer leg closure, or the like, may be
associated with at least one of the conduits in order to facilitate a desired function,
i.e., to establish a desired flow path for parenteral fluid and/or gas.
S Typically, as illustrated in Figure 7, the system also includes at least one filter
300, preferably upsllealll of device 100, to filter the fluid to be ~-imini~tered. A
variety of filters are suitable for use in the system, e.g., IV filters, e.g., for
;clering drugs and/or nutrients; and blood filters, in~ ling leukocyte filters.
In some embo li~ , the leukocyte filter comprises a synthetic polyrneric leukocyte
depletion medium, more preferably a synthetic, polymeric, fibrous me~ lm In an
embo~liment the leukocyte depletion me lillm has a Critical Wetting Surface
Tension (CWST) of about 55 dynes/cm or greater, e.g., about 60 dynes/cm or
greater. For example, the CWST can be in the range of about 62 dynes/cm to
about 100 dynes/cm. Exen~pla,~ leukocyte filters include those disclosed in, forexample, U.S. Patent Nos. 4,925,572; 4,880,548; and 5,217,627.
It is int~nde~l that the present invention is not to be limited by the above
listed components of the ~ ation set. For example, the parenteral fluid
lini~ ation set may have components such as, but not limited to, additional
containers, and injection ports.
Embodiments of a method accordillg to the invention include locating the
device in a fluid flow path between a source of fluid, e.g., a cont~iner, and the
inten-led destin~tion of the fluid, e.g~ a patient~ as illustrated in Figures 6 and 7.
Initially, fluid flow control devices 400 such as clamps up~lle~ll and dow,~leam of
the device 100 are typically closed, and then the flow control device up~leam ofdevice 100 is opened, to allow fluid to enter the housing. Since the flow control
device dowl,sl,earn of the device 100 remains closed, gas (e.g., gas displaced into the
housing by liquid passing through the conduit(s)) passes through the vent 3 as the
device fills with liquid. Preferably, device 100 remains in an upright position during
filling and subsequent use. Accordingly, once the operator of the fluid transfersystem, e.g., a nurse, opens the upstream flow control device, the device can
automatically vent and fill to a desired level, without operator involvement. As a
result, there is no need for the operator to monitor the system during priming, and
the operator can perform other tasks in the me~ntime. Since the device
automatically fills to a predetermined level, there is no need for the operator to rush
21 ~31 1 1
back to the system to avoid overfilling.
Using Figures 1, 2, and 7 for reference, fluid is passed, preferably by gravity,from conlailler 200 through at least one conduit to device 100. Liquid and gas enter
the housing 14 via inlet 1. Preferably, device 100 includes a nipple ~ and a port 23,
5 so that fluid passes into lumen 21 in the form of droplets. Since the fluid flow path
dow~ ea, l of the device 100 remains closed, the device acts as a reservoir, as
second chamber 20 fills with liquid. As the second chamber 20 fills with liquid, gas
is displaced. Some of this gas is vented from the device, i.e., the gas passes from the
interior of the housing along a gas flow path through porous medium 10 via surface
10b and surface 10a, and through port 4 and gas passageway 5 to the exterior of the
housing. In those embo lim-ontc wherein the housing incl~des ribs and rh~nn.o!c, e.g.,
ribs 25 and channels 24 and 27 as illustrated in Figure 2, gas passes along channels
24 and 27 through port 4 to gas passageway 5 to more efficiently vent the device.
Second chamber 20 continues to fill with liquid, and eventually, the level of
15 liquid will reach surface 10b of porous medium 10. Once liquid wets the porous
me-lium, gas flow through the vent 3 stops, without liquid subst~nti~lly passingthrough the medium 10. Once the gas flow stops, the device 100 is filled to the
desired level, as gas re...~ in the interior of the housing 14 will not be
displaceable, and the liquid will stop rising in the housing. Furthermore, since vent
20 3 is sealed, gas or air exterior to the device is pl~vt;ll~ed from entering the device,
even if the liquid level in second chamber 20 decreases.
In yrefe.,ed embo.1i...e--l~, gas flow stops when the device 100 is about 2/3
filled ~-vith liquid. Illusllalively~ when passing a red blood cell~ont~ g fluid such
as packed red cells (PRC) or whole blood into the device, the device has a totalcapacity of, for example, about 15 cc, and gas flow stops when the device contains
about 10 cc of liquid. In another embodiment, when passing a platelet-co.~l~i.-i.-g
fluid such as platelet concentrate (PC) into the device, the device has a total
capacity of, for example, about 9 cc, and gas flow stops when the device contains
about 6 cc of liquid.
Once the device is filled, the liquid can be delivered as desired. For example,
using Figure 7 for reference, the flow control device 400 dow~ eam of device 100can be opened, and the liquid can be ~lmini~tered to a patient.
In some embodiments, e.g., as illustrated in Figure 6, the system 500 also
includes a filter 300 such as a leukocyte depletion filter, and the container 200
2 1 83 1 1 1
contains leukocyte-co,.~ g biological fluid such as blood or a blood component.
Typically, flow control devices 400 are initially closed, and the flow control device
between container 200 and filter 300 is opened. If desired, container 200 may besqueezed to more efficiently prime the filter 300. Fluid exiting the filter 300 enters
S the device 100, which vents gas and fills with liquid as described with respect to
Figure 7. After the device au~olllalically vents and fills to a desired leveL the flow
control device 400 dowl~lleam of device 100 can be opened, and the liquid can beered to the patient. In some embodiments, the leukocyte-cont~ining
biological fluid is depleted of about 99.9% of the leukocytes or more as the fluid
passes through the leukocyte depletion filter.
In a variation of the embodiment illustrated in Figure 1, porous medium 10
col~lises a liquophobic element having surface 10a and surface 10b, and porous
medium 10 lacks a liquophilic element. Since s~ ces 10a and 10b are both
liquophobic, both surfaces can resist the passage of liquid(s) therell~ough. Forexample, a liquid can be placed in gas passageway 5 to provide a column of liquid in
the passageway. Since this liquid in the passageway 5 is typically discarded along
with the device after the device is used, it is more cco~ollPical to utilize a drug and
nutrient free liquid such as water in the passageway, in conlrasl with the parenteral
fluid to be passed through the device and into the patient.
Using Figures 1 and 7 for reference, as parenteral fluid passes from container
200 into the device 100, liquid and gas enter the housing via inlet 1. As the device
fills with liquid, gas is displaced, and passes through surfaces 10b and 10a, through
the column of liquid in passageway 5, and through port 30. Once the level of fluid
in second chamber 20 reaches surface 10b, gas flow through medium 10 stops,
without liquid yassing through the medium 10. Moreover, the liquid in passageway 5
prevell~ gas or air exterior to the device from entering the device, even if the liquid
level in second chamber 20 decreases. Howt;ver, after the fluid level reaches surface
10b, additional gas entering the housing via inlet 1 can be vented through surfaces
10a and 10b, through the column of liquid in passageway 5, and through port 30.
All of the references cited herein, including publications, patents, and patent
applications, are hereby incorporated in their entireties by reference.
While the invention has been described in some detail by way of illustration
and example, it should be understood that the invention is susceptible to various
modifications and alternative forms, and is not restricted to the specific
12
21 831 1 1
embodiments set forth. It should be understood that these specific embodiments are
not intended to limit the invention but, on the contrary, the intention is to cover all
modifications, equivalents, and alternatives falling within the spirit and scope of the
nvention.