Note: Descriptions are shown in the official language in which they were submitted.
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
1
INJECTION MOLDING OF STRUCTURAL
ZIRCONIA-BASED MATERIALS BY AN AQUEOUS PROCESS
BACKGROUND OF THE INVENTION
1. Field Of The Invention
This invention relates to a process for shaping ceramic parts from powder and
to
molding compositions used therein. More particularly, the invention is
directed to molding
processes and molding compositions that form high quality, net shape and near
netshape
complex parts of structural Zr02 based materials which can be fired to full
density and high
strength.
2. Description Of The Prior Art
Upon cooling from elevated, usually sintering, temperatures, Zr02 undergoes a
martensitic transformation from a tetragonal crystal structure to a monoclinic
crystal
structure. The transformation results in a volume and anisotropic shape
change. Under
controlled conditions, the tetragonal phase is maintained at room temperature,
and is only
transformed when a crack intersects with the grain. The subsequent
transformation puts a
closure force on the crack, thereby increasing the crack resistance of the
material.
The addition of small amounts of stabilizers, such as Y203, can have profound
effects on the
stability of the tetragonal phase. For instance, pure yttrium-stabilized
tetragonal
polycrystalline zirconia (Y-TZP) materials can be sintered to high strength or
fracture
toughness, depending on the Y203 concentration, grain size, and sintering
treatments (e.g.
pressureless vs. Hg''ing). A fine grain size in Y-TZP materials provides the
high strength
materials, while the instability of the tetragonal phase (depending on the
Y203 concentration)
determines the toughness. Such materials are disclosed by Masaki & Shingo in
US
4,742,030, Cassidy et al. in US 4, 866,014 and Ghoshid et al. in US 5,336,282.
One of the main drawbacks of Y-TZP materials is their environmental
degradation. Upon
exposure to especially humid environments and especially in the temperature
regime of 150-
300 °C, the tetragonal phase transforms spontaneously to monoclinic,
drastically reducing
the strength. A detailed review of this behavior is presented by S. Lawson in
the J. Europ.
Ceram. Soc., Vol. 15, pp. 485-502 (1995) titled Environmental Degradation of
Zirconia
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
2
Ceramics. The addition of alumina particuiates to Y-TZP increases both the
strength and the
environmental stability of Y-TZP materials. Similarly, a heat treatment at
elevated
temperatures also improves the environmental stability.
Applications for Zr02 based ceramics are widespread and include metal forming
tools, automotive applications, textile applications, and consumer
applications such as knifes,
scissors, golf clubs and the like. The ceramic components utilized in most of
these
applications are manufactured using powder pressing or slip cast forming
techniques.
One objective of any forming method is to produce articles in the unfired
state with a
certain density and particle packing (hereinafter called "green" parts,
forming, density, etc.)
which can be sintered to a shape that is reproducible to close dimensional
tolerances and is
free from defects. During green-forming and sintering, cracks, distortions and
other defects
can arise due to the shrinkage associated with the particle consolidation
processes. It is
generally recognized that these defect-producing processes are mitigated by
producing
homogeneous green bodies having adequate green strength
Another objective of shape-forming methods is to produce articles having net
shape,
eliminating or minimizing the need for downstream operations, such as
machining, to obtain
final part dimensions. Dry pressing involves compaction of powder in a die.
Among the
various shape-forming methods dry pressing, in particular, requires additional
downstream
processing in the form of machining and diamond grinding to attain intricate
shapes, non-
symmetrical geometrical formats and close tolerances. In slip casting a liquid
suspension of
ceramic powder is "de-watered" in a porous mold, producing a powder cake in
the shape
dictated by the mold. Although slip casting has the attribute of producing net
shape parts,
the method is considered to be relatively slow for the manufacture of complex
parts in high
volume.
Injection molding is recognized as a premier forming method for complex,
ceramic
shapes. It ai~ords significant advantages over other forming methods, by being
capable of
rapidly producing net shape, complex parts in high volume. Initially,
injection molding
comprised the step of mixing ceramic powder with a dispersant and a
thermoplastic organic
binder of variable composition. The molten powder/binder mixture was heated
during the
injection molding process and injected into a relatively cold mold. After
solidification, the
part was ejected in a manner similar to plastic parts. Subsequently, the
binder was removed
and the part was densified by a high temperature heat treatment. There were a
number of
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
3
critical stages in this process, which included the initial mixing of the
powder and binder, the
injection of the mixture into the mold, and the removal of the organic matrix
material. One
of the main disadvantages of the initial powder injection molding (PIIvI)
process is the
removal of the organic vehicle. At present, with the PIM process the cross
section limit for
fine particle sizes is 0.5-0.75 inch. If the particle sizes exceed that Limit,
the binder removal
process will lead to defects, pinholes, cracks, blisters etc. Binder removal
takes place by
slow heat treatments that can take up to several weeks. During debinding at
elevated
temperatures, the binder becomes a liquid which can result in distortion of
the green part due
to capillary forces. Another disadvantage of the initial PIM process is the
tendency for the
relatively high molecular weight organic to decompose throughout the green
body, causing
internal or external defects. The use of solvent extraction, wherein a part of
the organic is
removed using an organic or supercritical liquid, sometimes minimizes defect
formation.
Solvent extraction encounters difficulties because the remainder still needs
to be removed at
elevated temperatures. However, the solvent extraction process allows for the
formation of
porosity throughout the part, with the result that removal of the remaining
organic is
facilitated. During binder removal, part slumping can pose problems,
especially for the larger
particle sizes if the green density/strength is not high enough.
As such, PIM offers certain advantages for high volume automation of net
shape,
high dimensional control and complex parts, but the limitation of part size
and the very long
binder removal times combined with their environmental impact has not resulted
in the
expected growth of the use of this technique.
Some improvements, such as the use of water based binder systems, have been
made
to the initial PIM process. Hens et at. developed a water leachable binder
system. [US Patent
5,332,537) The injection molding feed-stock is made with a tailored particle
size distribution
(to control the rheology), a PVA based majority binder, and a coating on each
of the binder
particles. During molding, these coatings form necks which give the part
rigidity. After
injection molding there is a water de-bind that lasts several hours. After the
remaining binder
is cross-linked by either UV or chemical methods, the part undergoes a thermal
de-bind,
which takes 8-12 hours for a part such as a golf club head. Other aqueous-
based binders
contain either polyethyelene glycols, PVA copolymers, or COOH-containing
polymers.
BASF has developed a polyacetal based system that is molded at moderately high
temperatures after which the binder is removed by a heat treatment with
gaseous formic or
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
4
nitric acid. The low temperature excludes the formation of a liquid phase and
thus distortion
of the green part due to viscous flow. The gaseous catalyst does not penetrate
the polymer
and the decomposition only takes place at the interface of the gas and binder,
thereby
preventing the formation of internal defects. These improvements are limited
by the
requirement for separate binder removal furnaces and times, depending on the
part size.
There remains a need in the powder injection molding art for ready moldable
feed-
stocks that contain the ceramic powders in correct proportion and the
necessary binder,
liquid earner and other additives in a form for immediate use in commercially
available
injection molding machines.
SUMMARY OF THE INVENTION
The present invention provides an aqueous, zirconia-based molding compound and
a
method for compounding its constituent materials into a homogeneous mixture
and format
that is useful for low cost manufacture of ceramic articles by injection
molding. As used
herein, the term "zirconia-based" means compositions containing 50 - 100 wt%
zirconium
oxide in the fired ceramic. The molding compounds of the present invention
advantageously
contain, as a homogeneous mixture, ingredients which (i) are essential for
shape-forming
parts by injection molding, and (ii) yield zirconia-based ceramic materials
after firing.
Generally stated, there is provided, in accordance with the invention, a
molding compound
comprising essentially the ceramic precursors, zirconium oxide, yttrium oxide
and alumina in
a form that is suitable for fabricating articles by injection molding.
Advantageously, the ready moldable zirconia-based compound of the invention
obviates the need for high molding pressures and special de-binding furnaces.
The molding
compound of the invention uses water as the liquid earner and can be molded at
low machine
pressure below about 1,000 psi. Furthermore molded parts are dried before
sintering by
evaporation of the water, and the lengthy and complex de-binding step, typical
of polymer-
based molding systems, is eliminated. After firing, a ZrOz material is
obtained having full
density and high strength.
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
BRIEF DESCRIPTION OF THE DRAWINGS
The invention will be more fully understood and further advantages will become
apparent when reference is made to the following detailed description of the
preferred
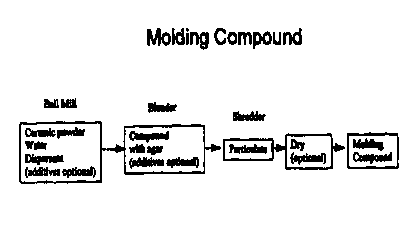
5 embodiments of the invention and the accompanying drawing, wherein Fig. 1 is
a schematic
representation depicting the basic steps of one embodiment of the invention.
DETAILED DESCRIPTION OFTHE INVENTION
According to the process of our invention, the ceramic powders are initially
mixed
with a gel-forming powder and a solvent for the gel-forming material. In
normal practice
zirconia ceramics require a stabilizing additive to prevent catastrophic
destruction of the
article due to the occurrence of a monoclinic phase transition upon cooling
from the sintering
temperature. Any of the stabilizers known to those skilled in the art of
fabricating zirconia
ceramics can be used in the process. Common stabilizers comprise oxides of the
elements Y,
Ce, Ca, and Mg or compounds such as carbonates, nitrates, oxylates and the
like, which
produce oxides of those elements during high temperature processing. The
amount of
stabilizer can be chosen to produce the tetragonal, cubic or monoclinic or a
mixture of
phases . Yttria is the preferred stabilizer. The presence of alumina produces
certain desired
effects, such as an improvement in the environmental stability. For the
purpose of densifying
the material to full density and obtaining high strengths, the average
particle size should be
below 1 pm. Preferably, the average particle size ranges from about 0.1-0.9
p.m, and more
preferably from about 0.3-0.5 p,m. As used herein, the term 'particle size"
means equivalent
spherical diameter.
The invention provides a ceramic molding compound consisting essentially of
zirconium oxide as the major phase with lesser amounts of other metal
inorganic compounds,
water, binder (selected from class of polysaccharides) and minor amounts of
other additives
that improve the processability of the molding feed-stocks. The invention
further provides a
method for producing a ready-moldable feed-stock from the constituent ceramic
powders,
binder, carrier and other processing aids. It is customary to represent the
ceramic
constituents of a Bred ceramic body in terms of the constituent metal oxide
compounds
irrespective of the actual phases present after firing. Using this convention
the ceramic
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
6
constituents of the molding compounds disclosed herein may be represented by
the formula
~Zr~2~a~y2~3~b~A12~3~c wherein a ranges from about 50 -95 wt and b ranges from
about 4 to
6 wt. % and c ranges from about 0-45 wt. %. In the present invention, one
preferred
molding compound in terms of the constituent metal oxides is composed of a =
about 85.8
wt %, and b = about 4.3 wt. % and c = about 14.3 wt. %. An example of a second
preferred
molding compound in terms of starting ceramic powders contains about 95 wt. %
zirconium
oxide and 5 wt. % yttrium oxide.
Generally, the amount of powder in the mixture is between about 50 and about
95
percent by weight of the mixture. Preferably, the powders constitute between
about 75 and
about 90 percent by weight of the mixture, and most preferably constitute
between about 83
and about 86 percent by weight of the mixture. The preferred and most
preferred amounts
are quite useful in producing net and near net shape injection molded parts.
The molding compound provides a binder which provides the mechanism for
allowing the fluid material to set in a mold and be removed as a self
supporting structure. In
the present invention this role is served by a compound derived from the
category of
polysaccharides known as agaroids. An agaroid has been defined as a gum
resembling agar
but not meeting all of the characteristics thereof (See H.H. Selby et al.,
"Agar", Industrial
Gums, Academic Press, New York, NY, 2nd ed., 1973, Chapter 3, p. 29). As used
herein,
however, agaroid not only refers to any gums resembling agar, but also to agar
and
derivatives thereof such as agarose. An agaroid is employed because it
exhibits rapid
gelation within a narrow temperature range, a factor which can dramatically
increase the rate
of production of articles. The preferred gel-forming materials are those which
are water
soluble and comprise agar, agarose, or carrageenan, and the most preferred gel-
forming
materials consist of agar, agarose, and mixtures thereof.
The gel-forming materials are present in an amount between 0.2 wt.% and about
6 wt.%
based upon the solids in the mixture. More than about 6 wt.% of the gel-
forming material
may be employed in the mixture. Higher amounts are not believed to have any
adverse
impact on the process, although such amounts may begin to reduced some of the
advantages
produced by our novel composition. Most preferably, the gel-forming material
comprises
between about 1 percent and about 4 percent by weight of solids in the
mixture.
The molding compound also provides a liquid carrier to facilitate transport of
the
molding compound along the barrel of an injection molding machine to a mold.
Water is the
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/186b3
7
most preferred liquid carrier in the molding compounds because it ideally
serves the dual
purpose of being a solvent for the gel forming binder and liquid carrier for
the solid
constituents in the mixture. In addition, because of its low boiling point,
water is easily
removed from the molded part prior to and/or during firing. The amount of
water is chosen
to confer the molding compounds with the essential rheological characteristics
for proper
behavior in the injection molding machine. The proper amount of water is
between about 10
wt. % and 30 wt. % of the mixture with amounts between about 15 wt. % and 20
wt.
being preferred.
The molding compound may also contain a variety of additives which can serve
any
number of useful purposes. Additives that have been found to be very useful in
the present
molding compounds comprise dispersants, pH control agents, biocides and gel
strength
enhancing agents (e.g., metal borate compounds such as calcium borate,
magnesium borate
and zinc borate). Biocides may be used to inhibit bacterial growth in the
molding
compounds, especially if they are to be stored for long periods of time.
It is well-known that use of dispersants and pH control can greatly improve
the
rheology and processabiliy of ceramic suspensions. In the present case
dispersants based on
polyacrylate and polymethylmethacrylate polymer backbones have been found
useful in
improving the processability of the aluminum oxide-based compositions, the
amount of
dispersant in the molding compound being about 0.2 wt. % to 1 wt. % and
preferably
0.2 wt. % to 0.8 wt. % based on the ceramic powders. Similarly,
tetramethylammonium
hydroxide has been found useful for controlling the pH of the suspensions, the
useful pH
range being about 8.8 to 11 and preferably 9.5 to 10.5.
The molding compounds of the present invention combine the ceramic powders,
liquid carrier, binder and processing aids in a ready-moldable form. A
preferred composition
in terms of the constituent compounds is 66.90 wt. % Zirconium oxide, 4 wt. %
yttrium
oxide, 11.7 wt. % aluminum oxide, 2.5 wt. % Agar, 0.33 wt. % dispersant, 0.53
wt.
tetramethylammonium hydroxide, 0.02 wt. % biocide and 14 wt. % water (where
the
dispersant is added as a 40 % aqueous solution and the TMA as a 25 % aqueous
solution).
The invention also provides a method for combining all of the various
constituents of the
molding compounds into a homogeneous mixture which will produce homogeneous
molded
bodies that can be fired free of cracks and other defects. Raw material
ceramic powders are
frequently highly agglomerated and require deagglomeration before they can be
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
8
manufactured into useful ceramic articles, free of cracks, distortions and
other defects. Of
the various available methods ball milling has been found convenient and
useful for
producing the aqueous-based molding compounds disclosed herein, the powders
being
simultaneously deagglomerated and homogenized in the aqueous medium. The
useful
concentration range for ball milling the ceramic powders is 50 wt. % to 85 wt.
%, the
preferable range being between 65 wt. % and 80 wt. %.
Compounding of the ceramic suspension with the binder can be done in any
number of
efficient mixers, e.g., a sigma mixer or planetary-type mixer. The biocide may
be blended
into the composition at the compounding stage of the process or optionally
near the end of
the ball milling cycle. During compounding the blend is heated in the range
75°C to 95°C
and preferably between 80°C and 90°C for a period of about 1 S
min to 120 min and
preferably between 30 min and 60 min.
The molding compound must be in a suitable form for charging an injection
molding
machine. In the present invention the compounded, homogeneous mixture is
allowed to cool
below the gel point of the gel-forming agent (<37°C) and removed from
the blender.
Thereafter it is shredded into a particulate format using a rotating cutter
blade typically used
in food processing. The shredded format can be fed directly into the hopper of
an injection
molding machine. The shredded feed-stock may be dried to a particular molding
solids by
evaporation, by exposure of the material to the atmosphere, until the desired
moisture level is
obtained. The useful solids levels in the molding compounds are in the range
75 wt. % to 88
wt% and preferably between 83 wt. % and 86 wt. %.
A very wide range of molding pressures may be employed. Generally, the molding
pressure is between 20 psi and about 3500 psi. Most preferably, the molding
pressure is in
the range of 40 psi to about 1 S00 psi. The mold temperature must of course be
at below the
gel point of the gel forming material in order to produce a self supporting
body. The
appropriate mold temperature can be achieved before, during or after the
mixture is supplied
to the mold. Ordinarily, the mold temperature is maintained at less than
40°C, and preferably
between about 15 °C an about 25 °C.
After the part is molded and cooled to a temperature below the gel point of
the gel-
forming material, the body is removed from the mold. The green body is
typically sufficiently
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
9
self supporting that it requires no special handling during removal from the
mold. After
removal from the mold, the part is dried. Similar to the drying of slip-cast
parts, care needs
to be taken to control the drying behavior. Depending on part size and
complexity, fast
drying may result in cracking. In such a case, the part may be dried in a
controlled humidity
environment.
After the part is are dried, the body is sintered at an elevated temperature
to produce
the final product. The sintering time and temperature is regulated according
to the powdered
material employed to form the part. Preferably, the elevated temperature at
which the body
is sintered is at least 1250°C, and more preferably ranges from 1300 to
1550 °C, and most
preferably ranges from 1350 °C to 1500 °C. Preferably, the
sintering time at maximum
temperature is less than 4 hrs., more preferably from 1-3 hrs, and most
preferably from
1-2 hrs.
The present invention can thus be used to form complex and thick net-shape or
near
net-shape bodies of zirconia based materials which have excellent strength
properties and
environmental stability. The physical properties of the densified ceramic from
one preferred
molding compound containing 20 vol.% alumina, referred to AS280, have been
found
excellent for a variety of structural applications, as summarized in Table I.
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
Table I. Injection Molded AS280 Alumina-Zirconia Properties
PROPERTY UHITS TEST VALUE
Color - - OFF-WHITE
~
Density glcm~ ASTM C20-83 5.63
Flexural strengthMPA (ksi) 3-point 970 (140)
Flexural strengthMPA (ksi) 4-point 820 (118)
Hardness kg/mm' Knoop (100 g.) 1518
Fractwe ToughnessMPa.m"' Indentation 5.6
Elastic ModulusGPA (10 psi) ASTM C623 239 (34.7)
Shear Modulus GPA (10 psi) ASTM C623 92 (13.4)
Poisson's Ratio- ASTM C623 0.3
CTE
50 C ppm/C ASTM E 228 8.9
250 C 9.42
500 C (Theta dilatometer)9.98
750 C 10.31
1000 C 10.5
5 The following examples are presented to provide a more complete
understanding of the
invention. The specifc techniques, conditions, materials, proportions and
reported data set
forth to illustrate the principles and practice of the invention are exemplary
and should not be
construed as limiting the scope of the invention.
10 Example 1
2314.27 g. of HSY-3 zirconia powder and 384.74 g. of Alcan C-901 alumina
powder
were weighed in a 1.6 gallon Abbethane ball-mill jar. 10.6 kg of 3/8" zirconia
media was
added. A mixture was made by weighing oif 889.2 g. of deionized water, 10.8 g.
of Darvan
821 A ammonium polyacrylate (40 % solution Vanderbilt Laboratories) and 17.5
g. of TMA
(25 wt. % solution, Alfa Inorganics). The slip was ball-milled for 24 hrs. and
3200 g. was
recovered and transferred to a sigma mixer. During agitation in the sigma
mixer, 72 g. of
Agar (S-100, Frutarom Meer Corp.), 0.62 g. methyl-p-hydroxy benzoate (Penta
Mfg) and
0.45 g. propyl-p-hydroxy benzoate (Penta Mfg.) were added incrementally. The
sigma mixer
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18653
11
was heated to 190 F for 45 min, after which the temperature was reduced to 170
F and
mixing continued for another 45 min. After the material was allowed to cool to
_ room
temperature, it was shredded using a food processor (Kitchen Aid KSM90) and
sieved using
a #5 sieve to remove any large and fine shards.
Before being molded, the shredded feed-stock was dried to a desired solids
level by
exposing a loose bed of material to the atmosphere. Solids loadings were
determined using a
moisture balance (Ohaus Corp.).
Plates were molded on both a Boy 15s and 22M. The plates were dried slowly on
the bench for several hours aRer which they were dried in a vacuum oven @
100°C. After
the plates were dry, they were densified @ 1450°C for 2 hrs. Standard 3-
and 4-point bars
were cut (Military type B) and the bending strength was determined to be 0.97
and 0.82
GPa, respectively.
Eaample 2
A molding feed-stock was prepared as in Example 1, and was used to mold a
variety
of shapes, such as "3-hole sensors". The fired parts were cylindrical in
shape, nominally
0.85" in length with 3 holes, nominally 0.1" in diameter running lengthwise. A
step divided
each part into a larger diameter shoulder, 0.45" OD x 0.35" length and a
smaller diameter
shoulder, 0.35" OD x 0.5" length. Molding was performed at 85 wt. %, after
which the
parts were dried under ambient conditions and fired at 1450°C for 2
hrs. After firing, the
average density was 5.59 f 0.012 g/cm3. Their average dimensions were 0.407" +
0.001"
for the larger diameter, 0.358" + 0.0011" for the smaller diameter and 0.7404
t 0.002" for
the length. The average shrinkages for the three dimensions were 21.6 t 0.2%,
22.2 ~ 0.2%,
and 19.7 f 0.2 %, respectively.
Another batch of parts, hereinafter called "half shell" parts were molded. The
parts
were half cylindrical in shape with several steps and grooves on the flat
side. Molding was
performed at 86 wt. %, after which the parts were dried under ambient
conditions and fired
at 1450°C for 2 hrs. After firing, the average density of the parts was
5.6 t 0.01 glcm3. The
fired length was nominally 0.9" with a width of 0.4". Within a sample of 84
green parts, the
average length was 0.949 t 0.005" and the average diameter was 0.496 + 0.003".
After
firing, the average shrinkage was 21.1 t 0.5 % and 21.4 ~ 0.5 %, for the
length and diameter
respectively.
CA 02303036 2000-03-10
WO 99/12864 PCT/US98/18663
12
Egamule 3
This example represents a scale-up of the molding compound preparation
described
in Example 1. A slip was prepared from 38.4 kg HSY-3 zirconia/ytrria, 6.24 kg
aluminum
oxide, 14.62 kg D.I. water, 0.179 kg ammonium polyacrylate and adjusted to pH
11 with
TMA. After ball milling, 55 kg of slip was transferred to a planetary type
blender where it
was blended ( in three separate runs) with 1.24 kg agar, 0.011 kg methyl-p-
hydroxy benzoate
and 0.0077 kg propyl-p-benzoate while being agitated and heated. Mixing was
continued for
lh after the blender reached a final temperature of 95°C. The material
was put into feed-
stock form by shredding.
Having thus described the invention in rather fill detail it will be
understood that such
detail need not be strictly adhered to but that various changes and
modifications may suggest
themselves to one skilled in the art, all falling within the scope of the
invention as defined by
the subjoined claims.