Note: Descriptions are shown in the official language in which they were submitted.
CA 02372411 2002-02-25
A Process for the Separation and/or Isolation of
Plasma Proteins by Means of Annular Chromatography
The present invention relates to a process for the separation
and/or isolation of plasma proteins from a mixture containing
plasma proteins. .
This Application is a nivision of Canadian Patent Application
Serial No. 2,333,316, filed on May 27, 1999.;
Plasma proteins play an important role in many physiological
processes. Thus, for example, the vitamin K dependent plasma
glycoproteins. play a prominent part in the blood clotting
cascade.
Thus, the recovery of. plasma proteins is an important technical
process. The starting material for the preparation of. plasma
proteins is already relatively valuable, because it is ob-
tained, for example, from donated blood. Both for economical
and for ethical reasons, it is important to provide processes
which yield the desired plasma proteins with high recoveries
and in high activities. Today, in addition to recombinant
preparation methods,, plasma proteins are usually obtained, from
blood plasma by using conventional chromatographic methods.
It has been found that high molecular weight substances can be
separated and isolated within an order of magnitude by means of
annular chromatography. It has further been found that human'
plasma proteins can be obtained in purified form in a surpris-
ingly simple way even from complex rnixtures.~This is achieved
by a process comprising the following steps: the mixture con-
taining the plasma proteins, especially human~plasma proteins,
is applied to a separation.medium having~an annular design. The
CA 02372411 2002-02-25
- 2 -
separation medium having the annular design is rotated verti-
cally about an axis which is defined in the direction of flow
of the mixture through the separation medium having the annular
design. An eluent is passed through the separation medium
having the annular design, and fractions exiting at the end of
the separation medium having the annular design are collected.
The use of annular chromatography for the desalting of mixtures
containing bovine serum albumin has already been described by
K. Reissner~et al., Journal of Chromatography A, 763 (1997), 49
to 56. Reissner et al. held the opinion that the enormous
_ difference in size between the substances to be separated,
namely bovine serum albumin and low molecular weight salts, was
critical to the chromatographical purification's being success
ful. Bloomingburg et al. (Ind. Eng. Chem. Res. 30, 10_61-1067
(1991 have shown that a mixture~of the pure components, bovine
serum albumin and bovine hemoglobin, can be separated by means
of a cation exchanger having an annular design. Elution is
carried out with a single eluent under isocratic conditions. A
number of application means are employed for applying sample
,material.
For performing the process according to the invention, it~is W
preferred to use the device described in the publication men-
tioned: Figure 1 schematically shows the subject device. Figure
2 shows a typical. chromatogram of a complex of factor VIII and
von Willebrand factor, and Figure 3 shows a corresponding
chromatogram of factor TX. Figure 4 shows a separation of B5A
and IgG. Figure 5 shows the result of a conventional.separation .w
by column chromatography.
Preferably, blood plasma or mixtures containing virus-
inactivated plasma proteins are used as the source of the
plasma glycoproteins to be separated and/or isolated. Said
mixture containing plasma proteins is preliminarily processed,
e.g., in the usual way. For. virus inactivation, in .particular,
CA 02372411 2002-02-25
v
r
- 3 -
methods are employed which are known as solventldetergent
methods. Thus, such a method is described, in particular, in B.
Horowitz et al., "Blood" 79 (1992), 826 to 831.
Therefore, the mixture may also contain a detergent which may
also, in addition, suppress undesirable non-specific interac-
tions of the plasma proteins with the separation media. The
mixture employed can be obtained not only from blood plasma,
but it may also be provided as a fraction of a cell culture
containing the human plasma proteins which have been prepared
by genetic engineering.
The separation medium having the annular~.design preferably
consists of materials used far ,adsorption chromatography, such
as ion-exchange, gel permeation, molecular size exclwsion or
affinity chromatography or chromatography based on hydrophobic
interactions. The usual materials are employed.
It may also be preferred to use more than one separation~medium '
in one chromatographic configuration. Thus, a chromatographic
column may be packed with leyers of different separation media
to achieve a combined separation effect. Not only similar media
can be selected, but also very different ones, e.g.~, an anion
exchanger and a medium for hydrophobic interaction chromatogra- .
phy, or two anion exchangers of different strength, or an
adsorption material__and a material for gel permeation chroma-
tography. However, .care should be taken that an undesirable
mixing of. the materials does not occur and that the buffers
used are suitable for all the separation materials employed.
A preferred separation medium i~s emphoyed as a compact block
material (monolith) which already has an annular shape suit-
able, e.g., for the separation column. This medium can be
combined, for example, with a loose packing of a.-chromato-
graphic material. The block-shaped media suitable as separation w ..
media are not only inorganic_or organic monoliths obtained by
" CA 02372411 2002-02-25
- 4 -
block polymerization which are disclosed, e.g., in EP-A-
0 32 0 023, incorporated herein by reference. Support materials
such as shaped membranes or membranes having a textile struc-
ture, such as based on cellulose, can also be employed for
annular chromatography. The monoliths.or support materials are
optionally surface-modified to obtain ligands which may be
required.. -
Another advantageous embodiment relates to the favorable appli-
cation of the sample material to the separation medium. It has
been found that an application medium for the well-aimed local
application of the sample material should preferably be used.
Any material can be employed which serves to shortly localize
the sample and which is not mixed with the separation medium.
This can be ensured by specifically selecting the material, in
which selection the density and the condition of the surface of
the material play a.role. However,~it is also possible to use a
device which provides a separation layer between the applica-
tion medium and the separation medium. Spherical particlesy
e.g., glass beads, are preferably employed as application
media, the particles optionally being treated to prevent non-
specific interactions.
It may be preferred to provide the spherical particles with a .
hydrophobic surface, if .they do not initially have a suffi- ~'
ciently hydrophobic, surface. For example, glass particles
hydrophobized with reagents such as silanization reagents can' -
be used. The particle size of the glass particles is preferably
in a range of from 20 to 500 ~.un. The spherical particles cover
the separation medium having the annular design and protect it
from mechanical impacts brought about by the application device
during the rotation of the device holding the separation medium
having the annular design. If surfaces are employed which are '
already relatively hydrophobic, such as those provided, for
example, by appropriate plastic particles, it is not necessary
to hydrophobize their surfaces.
CA 02372411 2002-02-25
- 5 -
The plastics of which the spherical particles consist may be,
in particular, polymethacrylates and polystyrene/divinylben-
zene.
The plasma proteins obtainable by the process according to the
invention include, in particular, inter-a-trypsin inhibitor,
immune globulins, such as IgG, human serum albumin or glycopro-
teins from the clotting cascade. Preferably, vitamin ~K depend-
ent factors of the blood clotting cascades such as factor IX,
othe r blood clotting factors, such as factors VIII, XI and
XIII, antithrombin III, al-antitrypsin and thrombin are ob-
tained from a plasma fraction. For example, factor VIII is
separated from accompanying proteins, such as fibrinogen,. in a
fraction of the cryoprecipitate by anion exchange chromatogra-
phy, optionally combined with molecular size exclusion chroma-
tography. Factor IX in admixture with vitamin K dependent
plasma proteins is also employed as a starting material,
wherein the accompanying' plasma proteins may be effectively
separated by means of a combination of anion-exchange and.
affinity chromatography or molecular size exclusion chromato-
graphy, but also by means of hydrophobic interaction chromato-
graphy.
One aspect of the present invention is the use of human pro-
teins which have themselves to be purified with great care. from
a complex mixture. of proteins. It-is to be noted that the human
proteins, in particular, must be activated or denatura.ted to as
low an extent as possible, and the mixture of human plasma
proteins is vdifficult to separate due to the similarity of the
physico-chemical properties between the human plasma proteins.
It has proven advantageous that the process according to the
invention can above all be. employed for separating at least .two
different human proteins without substantially affecting their
biological activity.
CA 02372411 2002-02-25
- 6 -
Due to the complexity of the starting materials, it is alsa
often necessary to apply and separate a large quantity of
proteins. A continuous procedure is highly advantageous since
the capacity of the column can be used in a virtually unlimited
way. With the annular design, a really continuous procedure can
be provided for the first time which enables not only the
application of the sample, the separation of the plasma pro-
teins and the fractioning to be performed simultaneously. The
separation medium can even be simultaneously regenerated and
equilibrated. While one region of the separation medium is
regenerated, another region can be equilibrated with a buffer,
which is then continuously employed for applying the sample.
Thus, the chromatographic plant can be used on a long-term
basis for days and weeks . The effective capacity of the column
can be increased thereby, depending on the duration of the
continuous chromatographic process. This is advantageous, above
a11, for gel permeation and, molecular size exclusion chroma-
tographies, since.these types of-chromatography are~limited for
the separation of proteins by their relatively low capacities
according to the prior art. This truly continuous procedure is
also fundamentally distinct from other quasi-continuous column-
chromatographic methods in which a series of physically sepa-
w~ rated compartments with separation media are' employed
("simulating moving bed"?~
The rotational speed is preferably selected within a range of
from 100 to 2000 degrees per hour, preferably from 600 to 2000 .
degrees per hour. It has-been established that at the higher
speeds, the fractioning is advantageous,~and the residence time'
of the human proteins in the separation medium .can be mini-
mized. Usually, flow rates within a range of from ~1 to
2000 cm/min, preferably from 15 to 300 cm/min, are selected for "
the separation.
The process according to the invention is suitable, above all,
for use on. an industrial scale for the preparative separation.
CA 02372411 2002-02-25
of human plasma proteins. Thus, depending on the specific
activity of the proteins, recoveries of from at least 40 to
almost 100% can be obtained. If prepurified proteins are used
as the starting material, a recovery of at least 900, prefera-
bly at least 950, rnay even be achieved in the further purifica-
tion, as "polishing". Thus, pharmaceutical preparations con-
taining plasma proteins, especially human plasma proteins, in a
highly purified form can be provided in an economical way.
In addition, annular chromatography has the advantage that it
can be performed continuously.
The separation of human plasma proteins is achieved particu-
larly well by adsorption chromatography using a step gradient,
using at least two different elution solutions of different
eluting strength. It has also proven advantageous to provide
only one device for applying the sample material. lest the
separation medium should be overloaded by the samplew material
having the high protein concentration.
For monitoring the fractioning process and for purposefully
selecting .the fractions to be collected, a monitor is advanta-
geously used which continuously monitors the proteiw concentra-
tion in the eluates . As said monitor, a suitable detector, for
example, a photometer, may be employed.
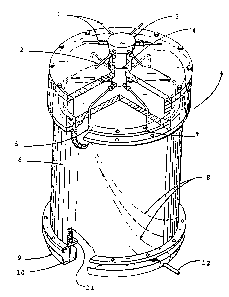
Figure 1 schematically. shows a preferred device fox performing
the process according to the invention. The support material
having an annular design is provided between two concentric
cylinders..The outer cylinder is closed with a flange at the
top end. Preferably, the cylinder itself is made of a steel
material. The inner cylinder is preferably made of a durable
material, and shorter than the outer one, so that a space
results at the top end which allows to distribute the eluent
uniformly on the whole separation medium having the annular
design. At the head of the..chromatographic unit, the sample
CA 02372411 2002-02-25
application device and the eluent application device are pro-
vided. At the bottom of the unit, both cylinders are connected
with a second flange. This flange contains a number of equidis-
tantly spaced holes, the holes being preferably distributed
uniformly in intervals of angles from at least 2° to 180°,
preferably from 4 to 36°. Preferably, flexible capillaries are
inserted in these holes which capillaries lead to a fraction
collector, for example.
In detail, Figure 1 shows an eluent inlet 1, a sample inlet 2,
a stationary inlet distributor 3, an inlet pressure closure 4,
a continuous sample flow S,, a rotating annular chromatography
unit 6, an eluent flow 7, a separated sample 8, a stationary
collector means for the waste eluent 9, an eluent outlet 1D, a
support material having an annular design 11, and a product
outlet 12.
Figure 2 shows an annular chromatogram of factor VIII/von
Willebrand factor. .
Figure 3 shows an annular chromatogram of factor IX.
Figure 4 shows an annular chromatogram of immune globulin G and
bovine serum albumin (BSA), wherein immune globulin G elutes
first.
Figure 5 shows a size exclusion chromatogram of a factor VIII
concentrate on a conventional discontinuous axial col:umn~ for
comparison.
The invention is further illustrated by means of the following
Examples.
CA 02372411 2002-02-25
_ g _
Example 1
An annular chromatography device according to Journal of Chro-
matography A, 563 (1997), 49 to 56, was packed with 2 1 of
Fractogel BioSec EMD 650 (S). The duration of the application
of the mixture to be separated depends on the elution flow
rate. After passing the bottom of the support medium hawing the
annular design, the sample application was continued for an-
other hour to achieve equilibrium. Thereafter, the collection
of fractions was,begun. The apparatus used in the Examples had
exits at the bottom of the chromatographic unit at a distance
of 2° each,. The outlet holes were respectively combined in
pairs to give one fraction.
Separation of human polyclonal IgG and BSA
Mixture to be separated: 2.5 mg/ml of IgG and 5 mg/ml of BSA in
aqua injectabilia.
Buffer: 27.5 mM disodium hydrogenphosphate dehydrate
12.5 mM sodium dihydrogenphosphate dehydrate
0.2 mM sodium chloride
pH = 7.2
The result of the separation is shown in Figure 4.
Example 2
Separation of factor IX concentrate
Sample solution: Factor IX lyophilizates with 500 IU (Octanyne,
Octapharma GmbH) were dissolved and adjus~.ed to a concentration
of from 20 to 500 IU/ml.
CA 02372411 2002-02-25
- 10 -
Buffer: 20 mM sodium citrate
0.2 M sodium chloride
2 mM calcium chloride
pH = 7.4
The chromatogram of the separation is shown in Figure 3.
Example 3
Separation of factor VIII concentrate
Mixture to be separated: Factor VIII lyophilizate with 500 IU
(Emoclot) was dissolved and adjusted to a concentration of from
20 to 500 IU/ml.
Buffer: 20 mM sodium citrate
0.2 M sodium chloride
2 mM calcium chloride
pH = 7.4
The chromatogram of the separation is shown in Figure 2.