Note: Descriptions are shown in the official language in which they were submitted.
CA 02464457 2004-04-14
PHOTOSENSITIVE MEMBER HAVING NANO-SIZE FILLER
BACKGROUND OF THE INVENTION
The present invention is directed to photosensitive members
or photoconductors useful in electrostatographic apparatuses, including
printers, copiers, other reproductive devices, and digital apparatuses. In
specific embodiments, the present invention is directed to photosensitive
members having nano-size fillers dispersed or contained in one or more
layers of the photosensitive member. The nano-size fillers, in
embodiments, provide a photosensitive member with a transparent,
smooth, and less friction-prone surface. In addition, the nano-size fillers,
in
embodiments, provide a photosensitive member with longer life, and
io reduced marring, scratching, abrasion and wearing of the surface. Further,
the photoreceptor, in embodiments, has a reduced or eliminated deletion.
Moreover, the photoreceptor provides an improved filler, which has good
dispersion quality in the selected binder, and has reduced particle porosity.
Electrophotographic imaging members, including photoreceptors or
photoconductors, typically include a photoconductive layer formed on an
electrically conductive substrate or formed on layers between the substrate
and photoconductive layer. The photoconductive layer is an insulator in the
dark, so that electric charges are retained on its surface. Upon exposure to
light, the charge is dissipated, and an image can be formed thereon,
2o developed using a developer material, transferred to a copy substrate, and
fused thereto to form a copy or print.
Many advanced imaging systems are based on the use of small
diameter photoreceptor drums. The use of small diameter drums places a
premium on photoreceptor life. A major factor limiting photoreceptor life in
copiers and printers, is wear. The use of small diameter drum
photoreceptors exacerbates the wear problem because, for example, 3 to
10 revolutions are required to image a single letter size page. Multiple
CA 02464457 2004-04-14
revolutions of a small diameter drum photoreceptor to reproduce a single
letter size page can require up to 1 million cycles from the photoreceptor
drum to obtain 100,000 prints, a desirable goal for commercial systems.
For low volume copiers and printers, bias charging rolls (BCR) are
desirable because little or no ozone is produced during image cycling.
However, the microcorona generated by the BCR during charging,
damages the photoreceptor, resulting in rapid wear of the imaging surface,
for example, the exposed surface of the charge transport layer. More
specifically, wear rates can be as high as about 16 microns per 100,000
lo imaging cycles. Similar problems are encountered with bias transfer roll
(BTR) systems.
One approach to achieving longer photoreceptor drum life is to form
a protective overcoat on the imaging surface, for example, the charge
transporting layer of a photoreceptor. This overcoat layer must satisfy
many requirements, including transporting holes, resisting image deletion,
resisting wear, and avoidance of perturbation of underlying layers during
coating. One method of overcoating involves sol-gel silicone hardcoats.
Another approach to achieving longer life has been to reinforce the
transport layer of the photosensitive member by adding fillers. Fillers that
2o are known to have been used to increase wear include low surface energy
additives and cross-linked polymeric materials and metal oxides produced
both through sol-gel and gas phase hydrolytic chemistries.
Problems often arise with these materials since they are often
difficult to obtain in, or reduce to, the nano-size regime (less than 100
nanometers). Fillers with larger particle sizes very often are effective
scatterers of light, which can adversely affect device performance. Also,
dispersion in the selected binder then often becomes a problem. Even
with suitably sized material, particle porosity can be a major problem as
pores can act as traps for gases and ions produced by the charging
3o apparatus. When this occurs the electrical characteristics of the
2
CA 02464457 2004-04-14
photoreceptor are adversely affected. Of particular concern is the problem
of deletion, a phenomenon that causes fogging or blurring of the developed
image.
Japan Patent No. P3286711 discloses a photoreceptor having a
surface protective layer containing at least 43 percent by weight but no
more than 60 percent by weight of the total weight of the surface protective
layer, of a conductive metal oxide micropowder. The micropowder has a
mean grain size of 0.5 micrometers or less, and a preferred size of 0.2
micrometers or less. Metal oxide micropowders disclosed are tin oxide,
io zinc oxide, titanium oxide, indium oxide, antimony-doped tin oxide, tin-
doped indium oxide, and the like.
U.S. Patent 6,492,081 B2 discloses an electrophotographic
photosensitive member having a protective layer having metal oxide
particles with a volume-average particle size of less than 0.3 micrometers,
or less than 0.1 micrometers.
U.S. Patent 6,503,674 B2 discloses a member for printer, fax or
copier or toner cartridge having a top layer with spherical particles having a
particle size of lower than 100 micrometers.
U.S. Patent Application 10/379,110, U.S. Publication No.
2o 20030077531 discloses an electrophotographic photoreceptor, image
forming method, image forming apparatus, and image forming apparatus
processing unit using same. Further, the reference discloses an
electroconductive substrate, the outermost surface layer of the
electroconductive substrate containing at least an inorganic filler, a binder
resin, and an aliphatic polyester, or, alternatively, the outermost surface
layer of the electroconductive substrate containing at least an inorganic
filler and a binder resin and the binder resin is a copolymer polyarylate
having an alkylene-arylcarboxylate structural unit.
U.S. Patent Application 09/985,347, U.S. Publication No.
3o 20030073015 Al, discloses an electrophotographic photoreceptor, and
3
CA 02464457 2006-11-27
image forming method and apparatus using the photoreceptor including an
electroconductive substrate, a photosensitive layer located overlying the
electroconductive substrate, and optionally a protective layer overlying the
photosensitive layer, wherein an outermost layer of the photoreceptor
includes a filler, a binder resin and an organic compound having an acid
value of from 10 to 700 mgKOH/g. The photosensitive layer can be the
outermost layer. A coating liquid for an outermost layer of a photoreceptor
including a filler, a binder resin, an organic compound having an acid value
of from 10 to 700 mgKOH/g and plural organic solvents.
Therefore, there exists a need in the art for an improved method of
increasing wear of a photosensitive member. In addition, there exists a
need for a photoreceptor surface with decreased susceptibility to marring,
scratching, micro-cracking, and abrasion. In addition, there exists a need
in the art for a photoreceptor with a transparent, smoother, and less
friction-prone surface. Further, there exists a need for a photoreceptor that
has reduced or eliminated deletion. Moreover, there is a need in the art for
an improved filler which has good dispersion quality in the selected binder,
and has reduced particle porosity.
SUMMARY OF THE INVENTION
In accordance with an aspect of the present invention, there is
provided n electrophotographic imaging member comprising:
a substrate;
a charge transport layer comprising charge transport materials
dispersed therein; and
an overcoat layer, wherein at least one of said charge transport
layer and overcoat layer comprise crystalline or spherical-shaped nano-
size fillers having a particle size of from about 1 to about 250 nanometers.
In accordance with another aspect of the present invention, there is
provided an electrophotographic imaging member comprising: a substrate;
4
CA 02464457 2006-11-27
a charge transport layer comprising charge transport materials dispersed
therein; and an overcoat layer, wherein said overcoat layer comprises
crystalline or spherical-shaped aluminum oxide nano-fillers having a
particle size of from about 1 to about 250 nanometers.
In accordance with a further aspect of the present invention, there is
provided an image forming apparatus for forming images on a recording
medium comprising:
a) a photoreceptor member having a charge retentive surface to
receive an electrostatic latent image thereon, wherein said photoreceptor
io member comprises a substrate, a charge transport layer comprising charge
transport materials therein, and an overcoat layer, wherein at least one of
said charge transport layer and said overcoat layer comprise crystalline or
spherical-shaped metal oxide nano-fillers having a particle size of from
about 1 to about 250 nanometers
b) a development component to apply a developer material to
said charge-retentive surface to develop said electrostatic latent image to
form a developed image on said charge-retentive surface;
c) a transfer component for transferring said developed image
from said charge-retentive surface to another member or a copy substrate;
2o and
d) a fusing member to fuse said developed image to said copy
substrate.
BRIEF DESCRIPTION OF THE DRAWINGS
For a better understanding of the present invention, reference may
be had to the accompanying figures.
Figure 1 is an illustration of a general electrostatographic apparatus
using a photoreceptor member.
Figure 2 is an illustration of an embodiment of a photoreceptor
showing various layers and embodiments of filler dispersion.
5
CA 02464457 2006-11-27
DETAILED DESCRIPTION OF THE PRESENT INVENTION
The present invention relates to the use of nano-size fillers in a layer
or layers of a photosensitive member to increase wear resistance and
s promote longer life of the photosensitive member. In addition, in
embodiments, the nano-size filler provides a smoother, transparent, less
friction-prone surface. Moreover, the nano-size fillers provide, in
embodiments, decreased scratching, micro-cracking, marring and abrasion
of the photosensitive member. Further, the photoreceptor, in embodiments,
5a
CA 02464457 2004-04-14
has a reduced or eliminated deletion. Moreover, the photoreceptor
provides an improved filler which has good dispersion quality in the
selected binder, and has reduced particle porosity.
Referring to Figure 1, in a typical electrostatographic reproducing
apparatus, a light image of an original to be copied is recorded in the form
of an electrostatic latent image upon a photosensitive member and the
latent image is subsequently rendered visible by the application of
electroscopic thermoplastic resin particles which are commonly referred to
as toner. Specifically, photoreceptor 10 is charged on its surface by
io means of an electrical charger 12 to which a voltage has been supplied
from power supply 11. The photoreceptor is then imagewise exposed to
light from an optical system or an image input apparatus 13, such as a
laser and light emitting diode, to form an electrostatic latent image thereon.
Generally, the electrostatic latent image is developed by bringing a
is developer mixture from developer station 14 into contact therewith.
Development can be effected by use of a magnetic brush, powder cloud, or
other known development process.
After the toner particles have been deposited on the
photoconductive surface, in image configuration, they are transferred to a
20 copy sheet 16 by transfer means 15, which can be pressure transfer or
electrostatic transfer. In embodiments, the developed image can be
transferred to an intermediate transfer member and subsequently
transferred to a copy sheet.
After the transfer of the developed image is completed, copy sheet
25 16 advances to fusing station 19, depicted in Figure 1 as fusing and
pressure rolls, wherein the developed image is fused to copy sheet 16 by
passing copy sheet 16 between the fusing member 20 and pressure
member 21, thereby forming a permanent image. Fusing may be
accomplished by other fusing members such as a fusing belt in pressure
30 contact with a pressure roller, fusing roller in contact with a pressure
belt,
or other like systems. Photoreceptor 10, subsequent to transfer, advances
6
CA 02464457 2004-04-14
to cleaning station 17, wherein any toner left on photoreceptor 10 is
cleaned therefrom by use of a blade 22 (as shown in Figure 1), brush, or
other cleaning apparatus.
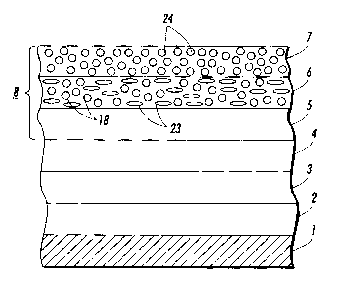
Electrophotographic imaging members are well known in the art.
Electrophotographic imaging members may be prepared by any suitable
technique. Referring to Figure 2, typically, a flexible or rigid substrate 1
is
provided with an electrically conductive surface or coating 2.
The substrate may be opaque or substantially transparent and may
comprise any suitable material having the required mechanical properties.
io Accordingly, the substrate may comprise a layer of an electrically non-
conductive or conductive material such as an inorganic or an organic
composition. As electrically non-conducting materials, there may be
employed various resins known for this purpose including polyesters,
polycarbonates, polyamides, polyurethanes, and the like which are flexible
as thin webs. An electrically conducting substrate may be any metal, for
example, aluminum, nickel, steel, copper, and the like or a polymeric
material, as described above, filled with an electrically conducting
substance, such as carbon, metallic powder, and the like or an organic
electrically conducting material. The electrically insulating or conductive
substrate may be in the form of an endless flexible belt, a web, a rigid
cylinder, a sheet and the like. The thickness of the substrate layer depends
on numerous factors, including strength desired and economical
considerations. Thus, for a drum, this layer may be of substantial thickness
of, for example, up to many centimeters or of a minimum thickness of less
than a millimeter. Similarly, a flexible belt may be of substantial thickness,
for example, about 250 micrometers, or of minimum thickness less than 50
micrometers, provided there are no adverse effects on the final
electrophotographic device.
In embodiments where the substrate layer is not conductive, the
surface thereof may be rendered electrically conductive by an electrically
conductive coating 2. The conductive coating may vary in thickness over
7
CA 02464457 2004-04-14
substantially wide ranges depending upon the optical transparency, degree
of flexibility desired, and economic factors. Accordingly, for a flexible
photoresponsive imaging device, the thickness of the conductive coating
may be between about 20 angstroms to about 750 angstroms, or from
about 100 angstroms to about 200 angstroms for an optimum combination
of electrical conductivity, flexibility and light transmission. The flexible
conductive coating may be an electrically conductive metal layer formed,
for example, on the substrate by any suitable coating technique, such as a
vacuum depositing technique or electrodeposition. Typical metals include
lo aluminum, zirconium, niobium, tantalum, vanadium and hafnium, titanium,
nickel, stainless steel, chromium, tungsten, molybdenum, and the like.
An optional hole blocking layer 3 may be applied to the substrate 1
or coatings. Any suitable and conventional blocking layer capable of
forming an electronic barrier to holes between the adjacent
photoconductive layer 8 (or electrophotographic imaging layer 8) and the
underlying conductive surface 2 of substrate I may be used.
An optional adhesive layer 4 may be applied to the hole-blocking
layer 3. Any suitable adhesive layer well known in the art may be used.
Typical adhesive layer materials include, for example, polyesters,
polyurethanes, and the like. Satisfactory results may be achieved with
adhesive layer thickness between about 0.05 micrometer (500 angstroms)
and about 0.3 micrometer (3,000 angstroms). Conventional techniques for
applying an adhesive layer coating mixture to the hole blocking layer
include spraying, dip coating, roll coating, wire wound rod coating, gravure
coating, Bird applicator coating, and the like. Drying of the deposited
coating may be effected by any suitable conventional technique such as
oven drying, infra red radiation drying, air drying and the like.
At least one electrophotographic imaging layer 8 is formed on the
adhesive layer 4, blocking layer 3 or substrate 1. The electrophotographic
imaging layer 8 may be a single layer (7 in Figure 2) that performs both
charge-generating and charge transport functions as is well known in the
8
CA 02464457 2004-04-14
art, or it may comprise multiple layers such as a charge generator layer 5
and charge transport layer 6 and overcoat 7.
The charge generating layer 5 can be applied to the electrically
conductive surface, or on other surfaces in between the substrate 1 and
charge generating layer 5. A charge blocking layer or hole-blocking layer 3
may optionally be applied to the electrically conductive surface prior to the
application of a charge generating layer 5. If desired, an adhesive layer 4
may be used between the charge blocking or hole-blocking layer 3 and the
charge generating layer 5. Usually, the charge generation layer 5 is applied
io onto the blocking layer 3 and a charge transport layer 6, is formed on the
charge generation layer 5. This structure may have the charge generation
layer 5 on top of or below the charge transport layer 6.
Charge generator layers may comprise amorphous films of selenium
and alloys of selenium and arsenic, tellurium, germanium and the like,
hydrogenated amorphous silicon and compounds of silicon and
germanium, carbon, oxygen, nitrogen and the like fabricated by vacuum
evaporation or deposition. The charge-generator layers may also comprise
inorganic pigments of crystalline selenium and its alloys; Group II-VI
compounds; and organic pigments such as quinacridones, polycyclic
pigments such as dibromo anthanthrone pigments, perylene and perinone
diamines, polynuclear aromatic quinones, azo pigments including bis-, tris-
and tetrakis-azos; and the like dispersed in a film forming polymeric binder
and fabricated by solvent coating techniques.
Phthalocyanines have been employed as photogenerating materials
for use in laser printers using infrared exposure systems. Infrared
sensitivity is required for photoreceptors exposed to low-cost
semiconductor laser diode light exposure devices. The absorption
spectrum and photosensitivity of the phthalocyanines depend on the
central metal atom of the compound. Many metal phthalocyanines have
3o been reported and include, oxyvanadium phthalocyanine, chloroaluminum
phthalocyanine, copper phthalocyanine, oxytitanium phthalocyanine,
9
CA 02464457 2006-11-27
chlorogallium phthalocyanine, hydroxygallium phthalocyanine magnesium
phthalocyanine and metal-free phthalocyanine. The phthalocyanines exist
in many crystal forms, and have a strong influence on photogeneration.
Any suitable polymeric film forming binder material may be
employed as the matrix in the charge-generating (photogenerating) binder
layer. Typical polymeric film forming materials include those described, for
example, in U.S. Pat. No. 3,121,006. Thus, typical organic polymeric film
forming binders include thermoplastic and thermosetting resins such as
polycarbonates, polyesters, polyamides, polyurethanes, polystyrenes,
io polyarylethers, polyarylsulfones, polybutadienes, polysulfones,
polyethersulfones, polyethylenes, polypropylenes, polyimides,
polymethylpentenes, polyphenylene sulfides, polyvinyl acetate,
polysiloxanes, polyacrylates, polyvinyl acetals, polyamides, polyimides,
amino resins, phenylene oxide resins, terephthalic acid resins, phenoxy
resins, epoxy resins, phenolic resins, polystyrene and acrylonitrile
copolymers, polyvinylchloride, vinylchloride and vinyl acetate copolymers,
acrylate copolymers, alkyd resins, cellulosic film formers, poly(amideimide),
styrenebutadiene copolymers, vinylidenechloride-vinylchloride copolymers,
vinylacetate-vinylidenechloride copolymers, styrene-alkyd resins,
polyvinylcarbazole, and the like. These polymers may be block, random or
alternating copolymers.
The photogenerating composition or pigment is present in the
resinous binder composition in various amounts. Generally, however, from
about 5 percent by volume to about 90 percent by volume of the
photogenerating pigment is dispersed in about 10 percent by volume to
about 95 percent by volume of the resinous binder, or from about 20
percent by volume to about 30 percent by volume of the photogenerating
pigment is dispersed in about 70 percent by volume to about 80 percent by
volume of the resinous binder composition. In one embodiment, about 8
percent by volume of the photogenerating pigment is dispersed in about 92
CA 02464457 2006-11-27
percent by volume of the resinous binder composition. The photogenerator
layers can also fabricated by vacuum sublimation in which case there is no
binder.
Any suitable and conventional technique may be used to mix and
thereafter apply the photogenerating layer coating mixture. Typical
application techniques include spraying, dip coating, roll coating, wire
wound rod coating, vacuum sublimation and the like. For some
applications, the generator layer may be fabricated in a dot or line pattern.
Removing of the solvent of a solvent coated layer may be effected by any
io suitable conventional technique such as oven drying, infrared radiation
drying, air drying and the like.
The charge transport layer 6 may comprise a charge transporting
small molecule 23 dissolved or molecularly dispersed in a film forming
electrically inert polymer such as a polycarbonate. The term "dissolved" as
employed herein is defined herein as forming a solution in which the small
molecule is dissolved in the polymer to form a homogeneous phase. The
expression "molecularly dispersed" is used herein is defined as a charge
transporting small molecule dispersed in the polymer, the small molecules
being dispersed in the polymer on a molecular scale. Any suitable charge
transporting or electrically active small molecule may be employed in the
charge transport layer of this invention. The expression charge transporting
"small molecule" is defined herein as a monomer that allows the free
charge photogenerated in the charge transport layer to be transported across
the transport layer. Typical charge transporting small molecules include, for
example, pyrazolines such as 1-phenyl-3-(4'-diethylamino styryl)-5-(4"-
diethylamino phenyl)pyrazoline, diamines such as N,N'-diphenyl-N,N'-bis(3-
methylphenyl)-(1,1'-biphenyl)-4,4'-diamine, hydrazones such as N-phenyl-
N-methyl-3-(9-ethyl)carbazyl hydrazone and 4-diethyl amino
benzaldehyde-1,2-diphenyl hydrazone, and oxadiazoles such as 2,5-bis (4-
3o N,N'-diethylaminophenyl)-1,2,4-oxadiazole, stilbenes and the like.
However, to avoid cycle-up in machines with high throughput, the charge
ii
CA 02464457 2006-11-27
transport layer should be substantially free (less than about two percent) of
di or triamino-triphenyl methane. As indicated above, suitable electrically
active small molecule charge transporting compounds are dissolved or
molecularly dispersed in electrically inactive polymeric film forming
materials. A small molecule charge transporting compound that permits
injection of holes from the pigment into the charge transport layer with high
efficiency and transports them across the charge transport layer with very
short transit times is N,N'-diphenyl-N,N'-bis(3-methylphenyl)-(1,1'-
biphenyl)-4,4'-diamine. If desired, the charge transport material in the
io charge transport layer may comprise a polymeric charge transport material
or a combination of a small molecule charge transport material and a
polymeric charge transport material.
Any suitable electrically inactive resin binder insoluble in the alcohol
solvent used to apply the overcoat layer 7 may be employed in the charge
transport layer of this invention. Typical inactive resin binders include
polycarbonate resin, polyester, polyarylate, polyacrylate, polyether,
polysulfone, and the like. Molecular weights can vary, for example, from
about 20,000 to about 150,000. Examples of binders include
polycarbonates such as poly(4,4'-isopropylidene-diphenylene)carbonate
(also referred to as bisphenol-A-polycarbonate, poly(4,4'-
cyclohexylidinediphenylene) carbonate (referred to as bisphenol-Z
polycarbonate), poly(4,4'-isopropylidene-3,3'-dimethyl-diphenyl)carbonate
(also referred to as bisphenol-C-polycarbonate) and the like. Any suitable
charge transporting polymer may also be used in the charge transporting
layer of this invention. The charge transporting polymer should be insoluble
in the alcohol solvent employed to apply the overcoat layer of this
invention. These electrically active charge transporting polymeric materials
should be capable of supporting the injection of photogenerated holes from
the charge generation material and be capable of allowing the transport of
these holes there-through.
12
CA 02464457 2004-04-14
Any suitable and conventional technique may be used to mix and
thereafter apply the charge transport layer coating mixture to the charge
generating layer. Typical application techniques include spraying, dip
coating, roll coating, wire wound rod coating, and the like. Drying of the
deposited coating may be effected by any suitable conventional technique
such as oven drying, infra red radiation drying, air drying and the like.
Generally, the thickness of the charge transport layer is between
about 10 and about 50 micrometers, but thicknesses outside this range
can also be used. The hole transport layer should be an insulator to the
io extent that the electrostatic charge placed on the hole transport layer is
not
conducted in the absence of illumination at a rate sufficient to prevent
formation and retention of an electrostatic latent image thereon. In general,
the ratio of the thickness of the hole transport layer to the charge generator
layers can be maintained from about 2:1 to 200:1 and in some instances
as great as 400:1. The charge transport layer, is substantially non-
absorbing to visible light or radiation in the region of intended use but is
electrically "active" in that it allows the injection of photogenerated holes
from the photoconductive layer, i.e., charge generation layer, and allows
these holes to be transported through itself to selectively discharge a
surface charge on the surface of the active layer.
Crosslinking agents can be used in combination with the overcoat to
promote crosslinking of the polymer, thereby providing a strong bond.
Examples of suitable crosslinking agents include oxalic acid, p-toluene
sulfonic acid, phosphoric acid, sulfuric acid, and the like, and mixtures
thereof. The crosslinking agent can be used in an amount of from about 1
to about 20 percent, or from about 5 to about 10 percent, or about 8 to
about 9 percent by weight of total polymer content.
The thickness of the continuous overcoat layer selected depends
upon the abrasiveness of the charging (e.g., bias charging roll), cleaning
(e.g., blade or web), development (e.g., brush), transfer (e.g., bias transfer
roll), etc., in the system employed and can range up to about 10
13
CA 02464457 2006-11-27
micrometers. In embodiments, the thickness is from about 1 micrometer
and about 5 micrometers. Any suitable and conventional technique may be
used to mix and thereafter apply the overcoat layer coating mixture to the
charge transporting. Typical application techniques include spraying, dip
coating, roll coating, wire wound rod coating, and the like. Drying of the
deposited coating may be effected by any suitable conventional technique
such as oven drying, infrared radiation drying, air drying, and the like. The
dried overcoating of this invention should transport holes during imaging
and should not have too high a free carrier concentration. Free carrier
1o concentration in the overcoat increases the dark decay. In embodiments,
the dark decay of the overcoated layer should be about the same as that of
the unovercoated device.
A nano-size filler can be added to a layer or layers in the
photosensitive member. In embodiments, the nano-size filler is added to
the charge transport layer 6 as filler 18, or the overcoat layer 7 as filler
24.
In embodiments, the nano-size filler is relatively simple to disperse,
has extremely high surface area to unit volume ratio, has a larger
interaction zone with dispersing medium, is non-porous, and/or chemically
pure. Further, in embodiments, the nano-size filler is highly crystalline,
spherical, and/or has a high surface area.
In embodiments, the nano-size filler is spherical or crystalline-
shaped. The nano-size filler is prepared via plasma synthesis or vapor
phase synthesis, in embodiments. This synthesis distinguishes these
particulate fillers from those prepared by other methods (particularly
hydroiytic methods), in that the fillers prepared by vapor phase synthesis
are non-porous as evidenced by their relatively low BET values. An
example of an advantage of such prepared fillers is that the spherical-
shaped or crystalline-shaped nano-size fillers are less likely to absorb and
trap gaseous corona effluents.
14
CA 02464457 2004-04-14
In embodiments, the nano-size filler has a surface area of from
about 0.1 to about 75, or from about 20 to about 40, or about 42 m2/g.
In embodiments, the nano-size filler is added to the layer or layers of
the photosensistive member in an amount of from about 0.1 to about 30
percent, from about 3 to about 15 percent, or from about 5 to about 10
percent by weight of total solids.
Examples of nano-size fillers include fillers having an average
particle size of from about 1 to about 250 nanometers, or from about I to
about 199 nanometers, or from about 1 to about 195 nanometers, or from
io about 1 to about 175 nanometers, or from about 1 to about 150
nanometers, or from about I to about 100 nanometers, or from about 1 to
about 50 nanometers.
Examples of suitable nano-size fillers include nano-size fillers
prepared by vapor phase synthesis or plasma reaction. Specific examples
of nano-size fillers include metal oxides such as silicon oxide, aluminum
oxide, chromium oxide, zirconium oxide, zinc oxide, tin oxide, iron oxide,
magnesium oxide, manganese oxide, nickel oxide, copper oxide,
conductive antimony pentoxide and indium tin oxide, and the like, and
mixtures thereof.
In embodiments, the nano-size filler can be prepared by plasma
reaction of the filler, or by vapour phase synthesis, resulting in very high
purity and very low porosity. In embodiments, a filler is prepared by
plasma reaction of the nano-size filler. In this method, in a high vacuum
flow reactor, a metal rod or wire is irradiated to produce intense heating
creating plasma-like conditions. Metal atoms are boiled off and carried
downstream where they are quenched and quickly cooled by a reactant
gas, most notably oxygen, to produce spherical low porosity nano-sized
metal oxides. Particle properties and size are controlled by the
temperature profiles in the reactor as well as the concentration of the
3o quench gas.
CA 02464457 2006-11-27
In embodiments, the nano-size fillers are surface treated to enable
them to be more easily dispersed. The metal oxide nanoparticles are
dispersed in an inert solvent by high power sonication for a suitable length
of time. A surface-active agent or agents (such as organochlorosilanes,
organosilane esters or their titanium analogs) is then added, and the
mixture is heated to allow reaction with and passivation of the metal oxide
surface. Removal of solvent then affords the surface-treated particle. The
amount of surface treatment obtained can be ascertained by thermal
gravimetric analysis. Generally, a 1 to 10% weight increase is observed
1o indicating successful surface treatment.
The following Examples further define and describe embodiments of
the present invention. Unless otherwise indicated, all parts and
percentages are by weight.
16
CA 02464457 2004-04-14
EXAMPLES
xample I
Preparation and Testing of Photoreceptor having Nano-Size Filler
Dispersed in Charge Trans op rt Layer
Electrophotographic imaging members were prepared by dip-
coating aluminum drums with charge transport layers of a polycarbonate
binder (PcZ400) and m-TBD (N,N'-diphenyl-N,N'-bis(3-methylphenyl)-(1,1'-
biphenyl)-4,4'diamine) in monochlorobenzene. Various amounts of nano-
size aluminum oxide fillers having an average particle diameter of 39
io nanometers and a specific surface area (BET) of 42 m2/g were added. The
amounts of nano-size fillers were 0 percent (control), 5 weight percent, and
weight percent by weight of total solids. The nano-size fillers were
added to the charge transport layer (25 micron).
A 25 micron transport layer was tested. The devices were tested
is using a surrogate wear fixture, a device which simulates wear by
cascading single component developer over a rotating drum with
subsequent removal of the toner by means of a blade cleaner. This fixture
has been shown to be intemally consistent and allows a ranking of
potential candidates against one another.
The wear results are shown below in Table 1. These results show
good wear results by use of the nano-size filler.
TABLE 1
Percentage A120, in Transport Layer Wear results
10 weight percent AI203 7.2 nm/kilocycle (2.0 nm/kilocycle
standard deviation)
5 weight percent A1203 16.8 nm/kilocycle (2.0 nm/kilocycle
standard deviation)
0 weight percent A1203 43 nm/kilocycle (6.5 nm/kilocycle
standard deviation)
17
CA 02464457 2004-04-14
Example 2
Preparation of Testing Photoreceptor having Nano-Size Filler Dispersed in
Overcoat La_ver
The above procedure in Example 1 was repeated, except that the
nano-size aluminum oxide was added to a 5 micron overcoat layer. Exactly
as the previous example, polycarbonate, m-TBD hole transport small
molecule and aluminum oxide were used.
Table 2 below shows the results of the testing. The results clearly
show increased wear by use of the nano-size filler.
TABLE 2
Percentage AIz0$ in overcoat Wear results
10 weight percent A1203 7.9 nm/kilocycle (1.5 nm/kilocycle
standard deviation)
5 weight percent A1203 12.1 nm/kilocycle (2.0 nm/kilocycle
standard deviation)
0 weight percent A1203 42 nm/kilocycle (4 nm/kilocycle
standard deviation)
While the invention has been described in detail with reference to
specific embodiments, it will be appreciated that various modifications and
1s variations will be apparent to the artisan. All such modifications and
embodiments as may readily occur to one skilled in the art are intended to
be within the scope of the appended claims.
18