Note: Descriptions are shown in the official language in which they were submitted.
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
PROCESS AND REACTOR FOR THE PRODUCTION OF OLEFINS BY THE
STEAM CRACKING REACTION OF LIQUID HYDROCARBON FEEDS IN THE
PRESENCE OF A CATALYST.
The present invention relates to a process and a reac-
for for the production of light olefins by steam cracking
reactions of liquid hydrocarbon feeds in the presence of a
suitable catalyst.
The most widely-used method for the production of
light olefins, in particular ethylene and propylene, is the
steam cracking process, in which a hydrocarbon feed is
heated, in the presence of water vapour, in. specific ovens
for producing a gaseous stream rich in olefins. Steam
cracking is a thermal process which, on. an industrial
scale, is carried out without catalysts. The setting up of
a catalytic system which allows increases in the yields to
the desired products, could lead to important advantages:
as a result of the large volumes of products in question,
(for example the world-wide production of ethylene reaches
100 Mtons/year) even small percentage increases in yield
- 1 -
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
would have a great impact on the process economy. The use
of catalysts for steam cracking reactions has not been
widely studied; since the 70s', however, various companies
and research groups have occasionally worked on this topic.
In some cases a process has been defined but as yet no in-
dustrial embodiments are known.
Among the most significant references, the following
can be mentioned, which have identified as the most active
materials for the cracking catalysis of naphtha, calcium-
aluminate compounds in which the prevalent phase is
12Ca0-7A1203 (mayenite):
- A.A. Lemonidou, I.A. Vasalos, Applied Catalysis, 54
(1989), 119-138;
- A.A. Lemonidou, I.A. Vasalos, Proc. 1987 AIChE Spring
National Meeting, Houston, March 29 - April 2, 1987;
- K. Kikuchi, T.Tomita, T. Sakamoto, T. Ishida, Chemi-
cal & Engineering Progress, 81 (1985) 6, 54;
- B. Basu, D. Kunzru, Industrial & Engineering Chemis-
try Res., 1992, 31, 146-155.
Another reference has also revealed the positive be-
haviour of materials consisting of mixtures of Ca-
aluminates:
- S. Nowak, G. Zimmermann, H. Gushel, K. Anders, in
"Catalysis in Petroleum Refining 1989" (D.L. Trimm et
al. Eds), Elsevier Science Publishers B.V., 1990.
- 2 -
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
Among the studies more related to industrial develop-
ment, Asahi Chemical can be mentioned, which claims a proc-
ess, close to commercialization, for steam cracking in cir-
culating bed, using a catalyst based on ZSM-5 and ZSM-11
zeolites, fed with metals such as Fe, Mg and/or Ib metals.
This process partially increases the yield to ethylene, but
above all directs the reaction towards the production of
propylene and aromatics. More recent developments (PERP Re-
port 96/97512, Chem Systems, September 1997) reveal how the
process still has various technological problems to be
solved, among which many aspects linked to the catalyst
(activity, regeneration, duration), before actually being
able to be commercialized. The same situation also applies
more or less for the process of the Russian of Vniios (Re-
search Institute for organic syntheses), which uses potas-
slum vanadate as catalyst, supported on corundum/mullite,
with the addition of promoters. Exxon has patented a proc-
ess which uses, as heat carrier, an inert solid or cata-
lysts based on mixed oxides of Mg, Ca, ~ Mn, Be, Sr, Ce, V,
Cs (W. Serrand et al., WO 97/31083). This process however
is preferentially projected for heavy feeds (for example b.
p. > 500°C) and comprises, in fact, a particular type of
horizontal moving bed reactor with two rotating screws
which help the movement of the feed.
A technology which seems closer to a possible indus-
- 3 -
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
trial application is the Pyrocat process, set up by Veba
Oel and Linde (M. Wyrosteck, M. Rupp, D. Kaufmann. H.
Zimmermann, Proc. 15th World Petroleum Congress, Beijing,
October 12-16, 1997). The technology involves the implemen-
tation of steam cracking plants without modifying the de-
sign of the ovens. The idea is based on the internal lining
of the cracking tubes with a layer of solid which has a
catalytic effect and inhibits the formation of coke, thus
prolonging the times between subsequent stoppages for de-
coking operations. The catalyst is based on A1~03/Ca0 and
contains, as gasification promoters, compounds of alkaline
metals. This technology can only be applied, however, to
conventionally designed cracking plants and operating with
conventional feeds.
From literature, it therefore emerges that catalysts
based on calcium aluminate can be used in steam cracking
reactions for the production of ethylene and propylene. The
calcium aluminates which can be formed are the following,
in an increasing order of calcium content:
Ca0~6A1203, Ca0~2A1203, 3CaO~5A1203, Ca0~A1203, 5CaO~3A1203,
12Ca0~7A1~03, 2Ca0~A1~03 and 3Ca0~A1203, but it cannot be de-
duced from literature which is the preferred crystalline
phase for the steam cracking reaction. According to
Lemonidou in fact (A. A. Lemonidou, I.A. Vasalos, Applied
Catalysis, 54 (1989), 119-138) the most effective catalyst
_ 4 _
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
is a mixture of calcium-aluminates in which the prevalent
compound is mayenite ( 12Ca0~7A1~03 ) ; S . Nowak, on the other
hand, has patented (DD-243 647 of 1987) a catalyst in which
the preferred phases have a lower content of calcium oxide:
Ca0~A1203 and Ca0~2A1~03.
From laboratory data and scientific knowledge, it can
be deduced that steam cracking reactions are conveniently
carried out with low residence times (~t) of the reagents in
contact with the catalyst at the reaction temperature. A
decrease in the residence time in fact leads to higher
yields in the desired products (ethylene and propylene).
The evolution in industrial cracking ovens moves in this
direction: furnaces are preferably produced with tubes hav-
ing an. ever-decreasing diameter to reduce 2, with the con-
straint that diameters which are too small cause high pres-
sure drops and this can jeopardize a correct running of the
plant. Among technologies for cracking furnaces with a low
residence time, Millisecond of Kellogg B&R and Pyrocrack of
Linde can be mentioned.
The fact that a low ~ is a favourable condition for
the reaction is also true for catalytic steam cracking and
in particular for the cracking of heavier feeds (for exam-
pie: vacuum gas oil), or for feeds which, under high tem-
perature conditions, have a greater tendency to form coke.
Figure 1 compares coke formation data for the catalytic
_ 5 -
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
steam cracking of vacuum gas oil in the presence of an in-
ert product (quartz) and catalyst (mayenite), at a tempera-
ture of 750°C. It can be observed how the presence of the
catalyst causes a decrease in the coke formation, which in
any case decreases at a lower ~.
In order to carry out the reaction at low residence
times, it is necessary to operate with high flows of feed
and vapour. High flows of reagents cause high pressure
drops, above all when operating in a reactor containing a
fixed catalyst bed. High pressure drops not only jeopard-
ized the reaction, but also create problems relating to the
running of the plant, as mentioned above. For a favorable
running of the process, it is consequently desirable to op-
erate under such conditions as to maintain a low ~ without
excessively high pressure drops.
If extremely low residence times are used (30-50 ms),
optimal performances are obtained in the cracking of heavy
feeds. The following table indicates the yields to ethyl-
ene+propylene, with a varying ~ for the steam cracking of
partially hydrogenated vacuum gas oil (HVGO), in the pres-
ence of a catalyst in granules (mayenite), at 750°C and a
ratio H20/feed (b) of 0.8 w/w.
i=0.05s ~=0.07s ~=0.1
s
Ethylene+propylene yield34.8 32.6 30.0
(w%)
- 6 -
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
These results were obtained using specific laboratory
reactors. In order to transfer the process to an industrial
scale, a reactor with particular characteristics must be
developed, which allows the process to be effected as close
as possible to the optimal operating conditions.
For an effective running of the process, the following
points should be considered:
~ the residence time of the gas in the catalytic bed should
be minimum, preferably less than 0.1 s;
~ the pressure drops must be limited;
~ the tubular element of the reactor should not be sub-
j ected to a quantity of heat per surface unit higher than
25,000 Btu/h/ft2;
~ the operating temperature of the wall of the reaction
tube is preferably lower than 1050°C.
In order to obtain a low residence time, it is neces-
sary to use a reactor with a limited length and diameter.
This, however, is in contrast with the necessity of having
a sufficiently radiated surface to be able to supply the
necessary heat for the reaction, as it is not possible to
exceed the quantity of heat per surface unit specified
above.
We have found that by effecting the steam cracking
process in the presence of a suitable catalyst with a cer-
taro reactor which allows the reaction to be carried out
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
with a low contact time, better results can be obtained in
terms of yield to light olefins in the field of steam
cracking reactions of naphtha and heavier feeds.
In order to produce tubes with acceptable dimensions,
respecting the restrictions imposed by pressure losses and,
above all, by the low contact times necessary for maintain-
ing high selectivities, a solution was found by filling
part of the tube with a cylinder made of a material imper-
meable to the reagent gases, having a lower radius than
that of the tube itself. In this way, the catalyst is dis-
tributed on a thin circular crown, the residence times are
kept low, whereas the exchange surface increases as longer
tubes with greater diameters can be used (without exces-
lively increasing the internal section, due to the presence
of the fully concentric tube).
The presence of the internal cylindrical element al-
lows a lower contact time and limits pressure drops.
The process for the production of light olefins, ob
ject of the present invention, by the steam cracking reac
tion of liquid hydrocarbon feeds, preferably selected from
naphtha, kerosene, atmospheric gas oil, vacuum gas oil and
oil residues, alone or mixed with each other, in the pres-
ence of a suitable catalyst, is characterized in that it is
carried out at a reaction temperature ranging from 700 to
850°C, at a pressure ranging from 1.1 to 4.5 absolute Atm
_ g _
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
and for a contact time ranging from 0.02 to 0.3 sec, pref-
erably from 0.05 to 0.15 sec, in one or more reaction zones
whose transversal section is in the form of a circular
crown, the ratio between the area of said circular crown
and the circular section ranging from 0.35 to 0.99, pref-
erably from 0.75 to 0.95.
The catalyst used can be uniformly distributed along
the reaction zone or it can be deposited, preferably by
means of laying, along the internal concave wall with a
higher radius and/or along the internal convex wall with a
lower radius.
The results can be improved, allowing even more re-
duced ~ to be adopted with respect to the basic case or
with tubes having a greater length, if the catalyst is
formed in such a way as to reduce the pressure drops.
The catalyst can therefore be in the form of spheres
with suitable dimensions or in particular in extruded form
suitably shaped to give the least possible resistance to
the flow of the feed (rings, saddles, trilobates, etc.) or
structured (of the honeycomb type or similar), which offer
a high exposed surface with low pressure drops.
The catalytic material is suitable for being processed
to produce extruded or structured products.
The structured products can entirely consist of calcium-
aluminate catalyst or the catalyst can be deposited on a
- 9 -
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
metallic carrier.
In particular, it is advisable that the catalyst con-
sist of one or more crystalline calcium aluminates and mo-
lybdenum and/or vanadium oxides.
The fact that structured products, under the same con-
ditions, produce much lower pressure drops with respect to
a packed bed is demonstrated both by theoretical calcula-
Lions and experimental data. For example, for use in a re-
actor for the Selective Catalytic Reduction of nitrogen ox-
ides in gaseous streams, a reduction of 2-3 orders of mag-
nitude is indicated, with the same linear gas rate, by the
use of monolithic catalysts (structured) instead of a bed
packed with spherical particles (E. Tronconi., A. Beretta,
Catalysis Today, 52 (1999), 249).
Hot recovery fumes can be optionally sent along the
tubular-shaped area obtained, enclosed by the reaction
zone, to contribute to the heating of the process itself.
A further object of the present patent application re
lates to the reactor for effecting steam cracking reac
tions, which is characterized in that it comprises one or
more cylindrical tubes, with the same or different section,
consisting of a tubular core and a reaction zone whose
transversal section is in the form of a circular crown, the
ratio between the area of said circular crown and the cir-
cular section ranging from 0.35 to 0.99, preferably from
- 10 -
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
0.75 to 0.95.
The catalyst can be uniformly distributed in the reac-
for along the reaction zone or it can be deposited, option-
ally by means of laying, along the internal concave wall
with a higher radius and/or along the internal convex wall
with a lower radius.
The tubular core can be made of inert material or it
can be hollow using a hollow tube, in which hot recovery
fumes from the cracking process flow, thus contributing to
the heating (or diminishing the thermal dispersions) of the
process.
Some embodiments of the invention are provided here-
under with the help of figures 2-7 which however should in
no way be considered as limiting the scope of the invention
itself.
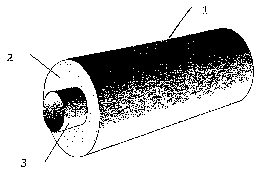
The tube or tubes contained in the reactor are repre-
rented in figures 2, 3, 4, 5, 6 and 7; more particularly,
the tube is illustrated in figure 2 in a perspective view,
in figure 3 in a partially sectional perspective view, in
figure 4 in a longitudinal section, in figure 5 in a trans-
vernal section with the core filled with inert material and
with the catalyst uniformly distributed, in figure 6 in a
transversal section with the core hollow and with the cata-
lyst uniformly distributed and in figure 7 in a longitudi-
nal section with the core hollow and with the catalyst laid
- 11 -
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
along both the internal concave wall with a higher radius
(2) and along the internal convex wall with a lower radius
(3 ) .
The numbers of the figures have the following meaning:
1 outer wall of the tube
2 internal concave wall with a higher radius
3 internal convex wall with a lower radius
4 tube core
Some examples are provided for a better illustration
of the present invention.
For each example the operating conditions are the follow-
ing:
Feed: vacuum gas oil
H~0/feed ratio: 0.8 w/w
Catalyst: 12Ca0~7A1a03 (mayenite)
Diameter of reactor: 1 inch
Length of the iradiated bed: 750 mm
EXAMPLE 1 (Comparative)
Reactor filled with a catalytic bed consisting of spherical
particles having a diameter of 5 mm.
Results:
Wall T = 859°C
Delta P = 1.8 kg/cma
~ = 0.095
- 12 -
CA 02510878 2005-06-17
WO 2004/056943 PCT/EP2003/014780
Reactor with an inert cylindrical element having a diameter
of 5 mm.
Catalyst in spherical particles of 5 mm arranged in the
circular crown.
Results:
Wall T = 855°C
Delta P = 2.6 kg/cm2
i = 0.090 (5o reduction with respect to the basic case)
EXAMPLE 3
Reactor with an inert cylindrical element having a diameter
of 10 mm.
Catalyst in spherical particles of 5 mm arranged in the
circular crown.
Results:
Wall T = 793°C
Delta P = 2.3 kg/cm2
i = 0.079 (17% reduction with respect to the basic case)
It can. be deduced that, with the same thermal exchange
surface and respecting the constraints and optimal operat
ing conditions for the reaction, it is possible to signifi
cantly reduce the residence time.
- 13 -