Note: Descriptions are shown in the official language in which they were submitted.
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
FIBER FORMATION BY ELECTRICAL-MECHANICAL SPINNING
FIELD OF THE INVENTION
[0001] The present invention relates to fiber formation, particularly to
fibers of nano dimensions.
BACKGROUND OF THE INVENTION
[0002] Fibers of nano dimensions can be produced by streaming an
electrostatically charged liquid such as a polymeric solution through a jet or
needle with a very small orifice. Scaling up this process by using multiple
needles suffers from the difficulty of electrically isolating these needles
from
each other. Consequently, needles typically must be at least one centimeter
away from the nearest neighbor. In addition, the need to draw a Tailor cone
from a single droplet on the end of each needle limits the maximum flow rate
per
needle and increases the number of needles that are needed to achieve large
scale production.
[0003] Therefore, there is a need for a process to manufacture fibers of
nano dimensions with high throughput without the need for multiple
applicators.
The present invention provides such a process.
SUMMARY OF THE INVENTION
[0004] The present invention provides a method of fiber production
starting from a liquid material such as a polymer solution or a polymer melt.
The
liquid material is fed to an annular rotating member such as a disk or cup
rotating
around an axis concentric therewith. The rotating member has a relatively
smooth continuous surface extending from the central area to a periphery. The
liquid material is directed by centrifugal force radially from the central
area to the
periphery and is expelled from the periphery towards a target. Liquid material
is
electrically charged either by the rotating member or immediately after being
expelled from the periphery of the rotating member by passing through an
electric field. The target to which the fibers are directed is electrically
grounded.
1
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
The difference in electrical potential between the charged fibers and the
target,
the viscosity of the liquid material and the size and speed of the annular
member, the liquid delivery rate and the optional use of shaping air are
adjusted
relative to one another so that the liquid material is expelled in fibrous
form. Also
adjusting these variables affects the quality and quantity of the fibers.
DETAILED DESCRIPTION
[0005] Preferably, the continuous surface of the annular rotating member
is the interior surface of a substantially cylindrical member such as a cup.
The
sides of the cup may be divergent such that the cup is in the form of a
truncated
cone. The annular spinning member rotates around an axis concentric
therewith. The rotating member may be electrically charged to impart an
electrical charge to the liquid material being fed to the rotating member.
Alternatively, an electrical charge can be imposed on the liquid material as
it is
expelled from the rotating member in fibrous form by passing the fibers
through
an electric field. As the rotating member spins, the liquid material is
centrifugally
directed along the interior surface towards the periphery of the rotating
member.
Preferably, spinning points are located along the periphery of the rotating
member. Examples of such spinning points are V-shaped serrations extending
around the periphery, preferably extending outwardly and substantially
parallel to
the axis of rotation of the rotating member. The liquid material passes over
the
spin points and is expelled from the rotating member towards the grounded
target. The rotating member may vary in size and geometry. The rotating
member may be as a disk or rotating bell. The diameter of the rotating member
may vary from 20 mm to 350 mm, such as 20 to 160, such as 30 to 80 mm. For
fiber formation, the difference in electrical potential between the charged
fibers
and the target is preferably at least 5000 volts, such as within the range of
20,000 to 100,000 volts and 50,000 to 90,000 volts. If the electrical
potential is
insufficient, droplets and not fibers may be formed.
[0006] As the liquid material is expelled from the rotating member in
fibrous form, the fibers are directed towards a grounded target where the
fibers
2
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
are collected. Alternately, the grounded target can be positioned behind a
moving belt or conveyor where the fibers can be collected and removed from the
target area. The distance to target can vary from 2 to 50 (5 to 130 cm), such
as
2 to 30 inches (5 to 76 cm) such as 10 to 20 inches (25 -- 51 cm). Preferably,
with a rotating bell an air stream is propelled normally and concurrently
against
the expelled fibers so as to shape the fibers into a flow pattern concentric
with
the axis of rotation and towards the target. Typically air exits the rotary
applicator via ports that surround the rotating member outside diameter. Air
pressure measured at the entrance of the rotating member can typically be set
at
such as 1 - 80 PSIG (6.9 x 103 - 5.5 x 105 Pascals), such as 1 - 60 PSIG (6.9
x
103 - 4.1 x 105 Pascals) such as from 5 to 40 PSIG (3.4 x 104 - 2.8 x 105
Pascals). With a rotating disk, shaping air is usually not used.
[0007] The rotating member is connected to a drive means such as a
rotating drive shaft connected to a member such as an electrical motor or air
motor capable of spinning the rotary member at speeds of at least 500 rpm,
such
as 1000 to 100,000, and 3000 to 50,000 rpms typically with speeds of 10,000 to
100,000 rpms. If the speed of the rotating member is insufficient, fibers may
not
form and the liquid may be expelled from the rotary member as sheets or globs,
If the speed of the rotating member is too high, droplets may form or fibers
may
break off.
[0008] Typically, the liquid material is passed through the interior of the
drive shaft and fed to the rotating member. When the rotating member is cup-
shaped, such as a rotating bell, the liquid material is fed through the closed
end
of the cup and in the central or base area of the cup. Typically, the liquid
enters
the closed end of the cup through a supply nozzle that may range in size from
0.5 to 1.5 mm, The liquid can then travel through the inside of the cup and
exits
on the surface of the cup through a center orifice or series of orifices onto
the
cup face.
[0009] The flow rate of liquid material to the rotating member is typically 1
ml/hour to 500 ml/minute, such as from 20 ml/hour to 50 ml/minute such as from
50 to 1000 ml/hour.
3
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
[0010] The liquid material that is spun into fibers in accordance with the
invention is typically a polymer solution or melt. The polymers can be organic
polymers such as polyesters, polyamides, polymers of n-vinyl pyrrolidone
polyacrylonitrile and acrylic polymers such as are described in published
application U.S. 2008/0145655A1. Alternately, the liquid can be an inorganic
polymer. Examples of inorganic polymers are polymeric metal oxides that
contain alkoxide groups and optionally hydroxyl groups. Preferably, the
alkoxide
groups contains from 1 to 4 carbon atoms such as methoxide and ethoxide.
Examples of such polymeric metal oxides are polyalkylsilicates such as those
of
the following structure:
OR OR
HO OH
OR OR
n
where R is alkyl containing from I to 4, preferably from 1 to 2 carbon atoms,
and
nis3to10.
[0011] Also, hybrid organic/inorganic polymers such as acrylic polymers
and polymeric metal oxides can be employed. Examples of such
organic/inorganic hybrid polymers are described in published application U.S.
2008/0207798A1. Also, inorganic materials such as inorganic oxides or
inorganic nitrides or carbon or ceramic precursors, such as silica, aluminia,
Titania, or mixed metal oxides can be used
[0012] The electrical conductivity of the liquid material can vary and
should be sufficiently electrically conductive such that it can accept a
charge
build up but not to the point that electrical shorting occurs. With indirect
charging, the electrical conductivity can be high since shorting is not a
problem.
The electrical conductivity can be adjusted by using appropriate amounts of
salts
such as ammonium salts and electrically conductive solvents such as alcohol-
water mixtures.
4
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
[0013] The surface tension of the liquid material can vary. If the surface
tension is too high, atomization and droplets rather than fibers may be
formed.
[0014] The liquid preferably thickens as polymer concentration increases
or polymer crosslinking occurs. In the case of a polymer solution, the
viscosity of
the solution can be controlled by controlling the molecular weight of the
polymer,
the concentration of the polymer in the solution, the presence of crosslinking
of
the polymer in solution, or by adding a thickening agent to the polymer
solution
such as polyvinyl pyrrolidone, polyvinyl alcohol, polyvinyl acetate,
polyamides
and a cellulosic thickener. If the viscosity of the solution is too high,
i.e., at its gel
point or above, it behaves more like a solid material and may not form a fiber
and may build up as solid polymer on the surface of the rotating member. If
the
viscosity of the liquid is too low, atomization and not fiber formation may
result.
[0015] The fibers that are formed in accordance with the invention
typically have diameters of up to 5,000 nanometers, such as 5 to 5,000
nanometers or within the range of 50 to 1200 nanometers such as 50 to 700
nanometers. Fibers can also have ribbon or flat face configuration and in this
case the diameter is intended to mean the largest dimension of the fiber.
Typically, the width of ribbon-shaped fibers is up to 5,000, such as 500 to
5,000
nanometers, and the thickness is up to 200, such as 5 to 200 nanometers.
[0016] In certain instances the nanofibers can be twisted around each
other in a yarn-like structure.
BRIEF DESCRIPTION OF THE DRAWINGS
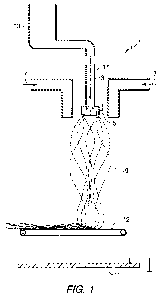
[0017] FIG. 1 is a schematic vertical cross-section through a centrifugal
spinning apparatus in which the process of the invention may be practiced.
[0018] FIG. 2 is a bottom elevation of a spinning member in accordance
with the process of the invention,
[0019] FIG. 3 is a section along line 111-lll of FIG. 2.
[0020] FIG. 4 shows photomicrographs at various magnifications of
nanofibers prepared in accordance with Example 1.
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
[0021] FIG. 4a (comparative) shows photomicrographs at various
magnifications of droplets prepared in accordance with Example 1a
(comparative).
[0022] FIG. 5 is a chart showing how the variables of rotating member
speed, shaping air and liquid flow effect fiber formation for the polymer
solutions
of Examples I and la (comparative).
[0023] FIG. 6 shows photomicrographs at various magnifications of
nanofibers prepared in accordance with Example 2.
[0024] FIG. 6a (comparative) shows photomicrographs at various
magnifications of droplets prepared in accordance with Example 2a
(comparative).
[0025] FIG. 7 is a chart showing how the variables of rotating member
speed, shaping air and liquid flow effect fiber formation for the polymer
solutions
of Examples 2 and 2a (comparative).
[0026] FIG. 8 shows photomicrographs at various magnifications of
nanofibers prepared in accordance with Example 3.
[0027] FIG. 8a (comparative) shows photomicrographs at various
magnifications of droplets prepared in accordance with Example 3a
(comparative).
[0028] FIG. 9 shows photomicrographs at various magnifications of
nanofibers in the form of a twisted yarn prepared in accordance with Example
4.
[0029] FIG. 10 shows photomicrographs at various magnifications of
nanofibers prepared in accordance with Example 5.
[0030] With reference to FIG. 1, the apparatus I contains a cup-shaped
rotating member 5 and an air plenum arrangement 7 through which air is
directed to shape the fibrous stream 9 as it is directed towards the target
11.
Positioned before the target is a conveyor 12 for removing the fibrous product
from the apparatus 1. A container 13 for the liquid material 15 includes a
suitable feed mechanism (not shown) for feeding the liquid material to the
rotating cup 5 via a feed supply line 17 mounted concentrically with the axis
3.
The supply line 17 has an exit in the rotating cup 5 adjacent to closed end.
6
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
Preferably, the feed supply line is located within a rotating drive shaft for
rotating
the cup-shaped rotary member 5. As shown in FIG. 1, a voltage is imposed on
the rotating cup to impart a charge on the liquid material and the fibers that
are
expelled from the rotating cup.
[0031] With references to FIGS. 2 and 3, the rotating member 5 is cup-
shaped having a planar base or closed end 21 and divergent walls 23 extending
from the base 21. The base 21 has a central aperture 25 through which the feed
supply line extends and fixing elements 27 by which the rotating cup 5 is
mounted on the drive means for rotation around the axis 3. The interior
surface
29 of the wall 23 is relatively smooth over the region extending from the base
21
to the edge 31 of the cup 5. The edge of the cup 5 is serrated such that there
are spinning points 33 defined by V-shaped serrations 35 on the external
periphery of the cup 5. V-shaped serrations 35 lie in a plane parallel to the
base
of the cup 5. In using apparatus 1, the cup 5 is spun at the desired rate and
the
liquid is fed to the rotating cup in the central area of the base of the cup
and is
directed to the periphery of the base 21 and across the interior surface 29 by
centrifugal force. The liquid that is electrically charged flows across the
interior
surface 29 of the rotating cup through the spinning points 33 from which the
liquid is expelled in fibrous form towards the grounded target 11.
[0032] The following examples are presented to demonstrate the general
principles of the invention. However, the invention should not be considered
as
limited to the specific examples presented. All parts are by weight unless
otherwise indicated.
EXAMPLES
EXAMPLE A
[0033] An acrylic-silane polymer was prepared as follows.
[0034] With reference to Table 1 below, a reaction flask was equipped
with a stirrer, thermocouple, nitrogen inlet and a condenser. Charge A was
then
added and stirred with heat to reflux temperature (75 C-80 C) under nitrogen
atmosphere. To the refluxing ethanol, Charge B and Charge C were
7
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
simultaneously added over three hours. The reaction mixture was held at reflux
condition for two hours. Charge D was then added over a period of 30 minutes.
The reaction mixture was held at reflux condition for two hours and
subsequently
cooled to 30 C.
TABLE I
Example A
Char e A (weight in grams)
Ethanol SDA 40B1 477.5
Char e B (weight in grams)
Methyl Methacrylate 0.2
Acrylic acid 11.5
Silquest A-1742 134.4
2-hydroxylethylmethacrylate 45.8
n-Butyl acrylate 0.2
Acrylamide 7.2
Ethanol SDA 40B 206.5
Charge C (weight in grams)
Vazo 673 8.1
Ethanol SDA 40B 101.7
Charge D (weight in aranns)
Vazo 67 2.0
Ethanol SDA 40B 12.0
% Solids 21.3
Acid value (solution) 10.5
Denatured ethyl alcohol, 200 proof, available from Archer Daniel Midland Co.
2 gamma-methacryloxypropyltrimethoxysilane, available from GE silicones.
3 2,2'-azo bis(2-methyl butyronitrile), available from E.I. duPont de Nemours
& Co., Inc.
[0035] A hybrid organic-inorganic polymer was prepared as follows:
8
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
[0036] The ethanol solution of acrylic-silane polymer solution, prepared as
described above, 200 grams, was poured into a jar, and deionized water (30
grams) was added. An ethanol solution of ethyl polylsilicate (Silbond 40, Akzo
Chemical, Inc) was added to the polymer solution along with
polyvinylpyrrolidone
(4 grams, Aldrich, Catalog 437190, CAS [9003-39-8], and MW 1,300,000).
While warming the jar with hot tap water, the mixture was hand shaken, and
hand stirred with a spatula until a homogeneous solution was obtained. After
this
solution was allowed to stand at room temperature for about 3.5 hours, its
viscosity of was determined to be C+ by the method of ASTM-D1545.
EXAMPLE B
[0038] An acrylic-silane polymer was prepared as follows.
[0039] With reference to Table 2 below, a reaction flask was equipped
with a stirrer, thermocouple, nitrogen inlet and a condenser. Charge A was
then
added and stirred with heat to reflux temperature (75 C-800C) under nitrogen
atmosphere. To the refluxing ethanol, Charge B and Charge C were
simultaneously added over three hours. The reaction mixture was held at reflux
condition for two hours. Charge D was then added over a period of 30 minutes.
The reaction mixture was held at reflux condition for two hours and
subsequently
cooled to 30 C.
9
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
TABLE 2
Example A
Charge A (weight in grams)
Ethanol SDA 4081 288.0
Charge B (weight in prams)
Methyl Methacrylate 16.0
Acrylic acid 6.9
Silquest A-1742 81.1
2-hydroxylethylmethacrylate 0.1
n-Butyl acrylate 0.1
Glycidyl Methacrylate 11.6
Ethanol SDA 40B 124.5
Charge C (weight in grams
Vazo 673 49.0
Ethanol SDA 40B 61.1
Charge D (weight in grams)
Vazo 67 1.2
Ethanol SDA 40B 7.2
% Solids 18.5
Acid value (solution) 8.9
1 Denatured ethyl alcohol, 200 proof, available from Archer Daniel Midland Co.
2 gamma-methacryloxypropyltrimethoxysilane, available from GE silicones.
3 2,2'-azo bis(2-methyl butyronitrile), available from E.I. duPont de Nemours
& Co., Inc.
[0040] Deionized water (30 grams) was pored into a jar, and
polyvinylpyrrolidone (4 grams, Aldrich, Catalog 437190, CAS [9003-39-8], and
MW 1,300,000) was added. The mixture was warmed on a hotplate to promote
dissolution, and the resulting solution was allowed to stand at room
temperature.
The acrylic-silane polymer solution, 170 grams, was added to this aqueous
polyvinylpyrrolidone solution. While heating the contents of the jar with warm
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
water on a hot plate, the mixture was hand shaken until a homogeneous solution
was obtained. This organic polymer solution was allowed to stand at room
temperature to cool before use.
EXAMPLE C
[0041] An inorganic sol gel polymer was prepared as follows.
[0042] Deionized water (36 grams) was placed in a jar, and polyvinyl
alcohol (4 grams, Aldrich, Catalog 36311, CAS [9002-89-5], 96% hydrolyzed,
and MW 85,000-100,000) was added to the water while stirring magnetically.
This mixture was warmed to 80 C in a hot water bath to affect dissolution.
More
deionized water (40 grams) was added to this warm aqueous polyvinyl alcohol
solution while continuing to stir. To this warm, diluted aqueous polyvinyl
alcohol
solution was added colloidal silica dispersion (120 grams, MT-ST Silica,
Nissan
Chemical Industries, LTD., about 30% silica in methanol) while continuing to
stir.
Viscosity of this polyvinyl alcohol, silica solution was determined to be A'
by the
method of ASTM-D1 545.
EXAMPLE D
[0043] A solution of polyacrylonitrile was prepared by dissolving 12 weight
percent of polyacrylonitrile resin (Aldrich, Catalog 181315, CAS [25014-41-9],
MW 150,000) in dimethylformaldehyde solvent while warming on a hot plate.
EXAMPLE I
[0044] The polyacrylonitrile resin solution of Example D was loaded into a
300 ml positive pressure fluid delivery system. A rate of 300 milliliters per
hour
was fed through a 3/8 inch (9.5 mm) outside diameter teflon tube system to a
rotary spray applicator via a 1.1 mm diameter fluid nozzle. The outlet of the
nozzle was connected to a rotary bell cup 55 mm in diameter. The fluid nozzle
inserts to the back of the bell cup where approximately 80-100% of the fluid
exits
through a circular slit of approximately 40mm diameter. The fluid then forms a
thin sheet across the bell cup and spins off the edge of the rotary bell cup
to form
11
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
fibers. This rotary bell was set to spin at a rate of 12,000 rpms. The bell
cup
edge geometry is configured with straight serrations. The perpendicular
distance
from the circular slit to the edge of the bell cup is approximately 7.85 cm.
The
bell cup referred to in this experiment is a Durr Behr Eco bell cup model
N16010037 type. The bell shaping air was set at 25 psig (1.72 x 105 Pascals)
at
the back of the bell via a 1/2 inch (12.7 mm) outside diameter nylon tube. The
rotary applicator was connected to a high voltage source with a 75,000 Volt
indirect charge applied potential. The entire delivery tube, rotary applicator
and
collector were in a booth that allowed the environmental condition to maintain
a
relative humidity of approximately 56% to 60% at a room temperature of 70 F to
72 F (21-C - 22 C). Nanofibers were collected on the grounded target onto
aluminum panels set at a target I collection distance of 15 inches (38 cm)
from
the rotary bell and were characterized by optical microscopy and scanning
electron microscopy. The nanofibers were essentially cylindrical and had
diameters of 600 to 1800 nanometers (nm). Some large diameter fibers were
observed that appear to be assemblies of the smaller diameter fibers. The
scanning electron micrograph is shown in Figure 4 and shows many fibers with
little or no drops.
[0045] A Design Analysis was completed for the solution of Example 1 to
determine application factors with respect to this solution. The application
factors studied for this work were bell speed from 12K rpms to 28K rpms,
target
distance from 10 inches to 20 inches (25.4 - 50.8 cm), voltage from 60KV to
90KV, fluid delivery rate from 100ml/ hour to 300 ml/ hour and bell shaping
air
from 15psig to 35psig (1.03 x 105 - 2.41 x 105 Pascals). The results reported
in
Figure 5 showed that fluid delivery rate, shaping air, and bell speed were the
most influential application factors followed by target distance and KV.
[0046] In Figure 5 "BS" refers to Bell Speed". "SA" refers to Shaping Air.
"FF" refers to Fluid Delivery Rate.
[0047] The values of the vertical axis are the product of the thickness of
the nanofiber mat that is formed multiplied by the ratio of nanofiber to
drops.
12
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
The thickness of the mat is given a subjective value of 1 to 10 and the ratio
of
nanofibers to drops is given a subjective value of 1 to 6.
[0048] The higher the number of the value on the vertical axis, more
volume of good fibers is generated.
EXAMPLE 1A (Comparative)
[0049] In this example, the procedure of Example 1 was repeated with the
following differences:
Bell Speed 28,000 rpms
Target collector distance 10 inches (25.4 cm)
Fluid delivery rate 200 ml/hour
[0050] Nanofibers were attempted to be collected on the grounded
aluminum target onto aluminum panels set at a part 1 collection and were
characterized by scanning electron microscopy as shown in Figure 4a. The
electron microscopy shows very little fiber formation and many wet drops.
EXAMPLE 2
[0051] The hybrid organic - inorganic polymer solution of Example A was
spun into nanofibers in accordance with the procedure of Example 1, but using
a
Dur Behr Eco bell cup model N16010033. The nanofibers were characterized by
optical microscopy and scanning electron microscopy. The nanofibers were
somewhat flat-faced with cross-sectional dimensions that ranged from 700
nanometers (nm) to 5000 nm. The scanning electron micrograph is shown in
Figure 6 and shows many fibers with little or no wet drops.
EXAMPLE 2A (Comparative
[0052] In this example, the procedure of Example 2 was generally
followed with the following differences:
Bell Speed 28,000 rpms
Target/collector distance 10 inches (25.4 cm)
Fluid delivery rate 200 ml/hour
13
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
Nanofibers were attempted to be collected on the grounded aluminum panel
target and were characterized by scanning electron microscopy as shown in
Figure 6A. This electron microscopy shows very little fiber formation and many
wet drops.
[0053] A Design Analysis as described in Example 1 was completed for
the solution of Example 2. The application factors studied for this work were
bell
speed from 12K rpms to 28K rpms, target distance from 10 inches to 20 inches
(25.4 - 38.1 cm), voltage from 60KV to 90KV, fluid delivery rate from 100
ml/hour to 300 ml/hour and bell shaping air from 15 psig to 35 psig (1.03 x
105 --
2.41 x 105 Pascals). The results reported in Figure 7 showed that fluid
delivery
rate, shaping air, bell speed and target distance were the most influential
followed by KV. Figure 7 uses the same terminology as used in Figure 5.
EXAMPLE 3
[0054] The inorganic sol gel polymer solution of Example C was spun into
nanofibers in accordance with the procedure of Example 2 using a fluid
delivery
rate of 100 milliliters per hour, a spin rate of 28,000 rpms, a voltage of
90,000
volts and a target collector distance of 20 inches (50.8 cm). The bell shaping
air
was set at 15 psig (1.03 x 105 Pascals) at the back of the bell. Nanofibers
were
collected on the grounded aluminum panel target and were characterized by
optical microscopy and scanning electron microscopy.
[0055] The nanofibers were essentially cylindrical and had diameters of
100 to 700 nm. Some of the fibers appeared to have small beads along the
linear axis that had not drawn into a fiber. The scanning electron micrograph
is
shown in Figure 8 and shows many small fibers with little drop formation.
EXAMPLE 3A (Comparative)
[0056] In this example, the procedure of Example 3 was repeated with the
following differences:
Bell Speed 12,000 rpms
Fluid flow rate of 300 ml/hour
14
CA 02703958 2010-04-23
WO 2009/055413 PCT/US2008/080693
Target collector distance 10 inches (25.4 cm)
Shaping air 35 psig (2.4 x 105 Pascals)
[0057] Nanofibers were attempted to be collected on the grounded
aluminum target and were characterized by scanning electron microscopy as
shown in Figure 8A. The electron microscopy shows little fibers with wet
drops.
EXAMPLE 4
[0058] The polyacrylonitrile resin solution of Example D was spun into
fiber in accordance with the procedure of Example 1 using a voltage 86,000.
Fibers were collected on the grounded aluminum panel target and were
characterized by optical microscopy and scanning electron microscopy. Large
fibers collected on the panel. One large fiber was removed from the panel and
was evaluated microscopically as shown in Figure 9. A low resolution optical
image (left-most image) indicated that the large fiber might be an assembly of
smaller fibers. Scanning electron microscopy (center image) revealed that
these
large fibers are a twisted yarn 100 microns in diameter comprised of several
much smaller fibers. The yarn is formed as the smaller fibers rotate from the
spinning bell cup. Higher magnification (right-most image) revealed that these
smaller fibers are nano-scale in diameter within the yarn.
EXAMPLE 5
[0059] The organic polymer solution of Example B was spun into fibers in
accordance with the procedure of Example 1 with the following differences:
Fluid flow rate 200 ml/hour
Shaping air 35 psig (2.41 x 105 Pascals)
Target collector distance 20 inches (50.8 cm)
The nanofibers were somewhat flat-faced with cross-sectional dimensions and
had diameters of 300 to 700 nm. The scanning electromicrograph is shown in
Figure 10. The micrograph shows many small fibers with little drop formation.
[0060] The invention is now set forth in the following claims.