Note: Descriptions are shown in the official language in which they were submitted.
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-1-
UTILISATION OF DESALINATION WASTE
This international patent application claims priority from Australian
provisional
patent application 2008906021 filed on 21 November 2008, the contents of
which are to be taken as incorporated herein by this reference.
FIELD OF THE INVENTION
The present invention relates generally to processes for the production of
value
added materials utilising saline waste streams, such as those from
desalination
processes. More specifically, the present invention relates to processes for
the
production of soda ash (sodium carbonate, Na2CO3) utilising saline waste
streams and, optionally, carbon dioxide (C02) waste gases from combustion
sources.
BACKGROUND OF THE INVENTION
The provision of adequate supplies of fresh water is a continuing and mounting
problem worldwide. In many countries, the process of desalination of saline
water, such as sea water, brackish water or saline industrial waste water, is
used to supply fresh water. In Australia alone, it is predicted that
approximately
460 gigalitres per annum of drinking water will be produced from desalination
plants by 2013.
Typically, reverse osmosis (RO) membranes are used in desalination plants to
desalinate sea water, brackish ground water and/or saline industrial waste
water. The main objective of the desalination process is to reduce the salt
(sodium chloride, NaCl) concentration of saline water to less than 0.5 grams
per
litre (g/L) suitable for drinking water. The salt concentration of sea water
is
typically 35-45 g/L.
Unfortunately, the use of desalination processes to produce fresh water from
saline water presents a number of environmental issues. For example, one of
+= CA 02744264 2011-05-18 PCT/AU2009/001512
Received 30 April 2010
-2-
the by-products of the desalination process is brine waste which typically has
a
salt concentration of about 70 g/L. The highly saline liquid brine waste
stream
has to be disposed of and the typical methods used to do that include
discharging it back to the ocean, disposing of it in the sewer, injecting it
into
deep wells, applying it to land, or transferring it to evaporation ponds. In
each
case, disposal of the waste imposes significant economic and/or environmental
costs.
Ideally, brine waste from desalination processes would be used to form other,
value added materials.
Reference to any prior art in this specification is not, and should not be
taken
as, an acknowledgment or any form of suggestion that this prior art forms part
of the common general knowledge in any country.
SUMMARY OF THE INVENTION
The present invention arises from the discovery that brine waste and,
optionally,
carbon dioxide waste gases from combustion sources, can be used as starting
materials in a process for the production of soda ash.
The present invention provides a process for producing soda ash from brine
waste, the process including reacting brine waste with carbon dioxide and
ammonia to produce sodium bicarbonate and ammonium chloride in a reaction
mixture, the process including:
i. separating the sodium bicarbonate from the reaction mixture to produce
sodium bicarbonate and a first mother liquor containing ammonium chloride;
ii. converting the sodium bicarbonate to soda ash;
iii. treating the first mother liquor to produce purified water and a
concentrated ammonium chloride; and
iv. treating the concentrated ammonium chloride with a base anion
exchange resin to regenerate ammonia suitable for use in the reaction;
Amended Sheet
IPEA/AU
CA 02744264 2011-05-18 PCT/AU2009/001512
Received 30 April 2010
- 2A -
wherein at least a portion of the ammonia is regenerated from ammonium
chloride produced during the reaction..
Typically, the process will include collecting sodium bicarbonate produced
during the reaction and heating the collected sodium bicarbonate to produce
the
soda ash. Additionally, the ammonium chloride produced during the reaction
will preferably be treated with a base anion exchange resin (such as a weak
base anion exchange resin) to regenerate the ammonia. This regeneration will
preferably occur after further treatment (such as concentrating) of the
ammonium chloride produced during the reaction.
Amended Sheet
IPEA/AU
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-3-
As used herein, the term `brine waste' refers to a saline solution having a
sodium chloride concentration of between about 50 g/L and about 250 g/L,
inclusive. Advantageously, the brine waste may be a waste stream from a
desalination plant, such as a reverse osmosis desalination plant.
The process may also include treating the brine waste to remove at least some
of any undesired inorganic cations (such as calcium, magnesium and strontium
ions) present in the brine waste prior to the reaction with carbon dioxide and
ammonia. The undesired inorganic cations present in the brine waste may be
removed either partially or entirely by selectively binding those ions with a
binding agent, such as a cation exchange resin.
The carbon dioxide used in the process of the present invention will
preferably
be obtained from a waste gas stream derived directly or indirectly from
combustion sources, such as coal or gas or diesel fired power stations, steel
works or petroleum refineries. In this form, the carbon dioxide may be treated
to remove some or all of any gaseous contaminants that may be present in the
stream.
The reaction of sodium chloride and carbon dioxide to produce soda ash is
catalysed by ammonia, and in the present invention at least a portion of the
ammonia that is used as the catalyst is regenerated from ammonium chloride
produced during the reaction. As mentioned above, the ammonium chloride
produced during the reaction will ideally be filtered, after collection of the
sodium bicarbonate, by a high pressure filter such as a reverse osmosis
membrane to provide purified water that can be reused, and at the same time to
concentrate the remaining ammonium chloride solution. Furthermore, the
concentrated ammonium chloride solution will then be the solution treated with
a base to regenerate ammonia suitable for reuse in the reaction, and in one
form the base will be an anion exchange resin.
As mentioned above, at least a portion of the ammonia used in the reaction of
the brine waste and the carbon dioxide is the regenerated ammonia. In this
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-4-
respect, the regenerated ammonia may make up at least 10%, and preferably
somewhere between 20% and 80%, of the ammonia required for the reaction.
Ideally, all of the ammonia regenerated will be returned to the reactor, and
an
amount of fresh ammonia will be added to the reactor to make up the ammonia
solution strength as required for the reaction. It will thus be appreciated
that
the amount of fresh ammonia used in the reactor will be dependent upon the
operating parameters of each individual operation that might utilise the
process
of the present invention.
The basic chemical reactions involved in the process of the present invention
are similar to the Solvay process which is currently used industrially to
produce
soda ash. The traditional Solvay process produces soda ash from concentrated
brine (as a source of sodium chloride (NaCl)) and from limestone (as a source
of calcium carbonate (CaCO3)).
Unfortunately though, the traditional Solvay process is an energy intensive
process. Firstly, the reactant carbon dioxide gas is obtained by heating
limestone in a kiln to drive off carbon dioxide gas. This is an energy
intensive
step, not only consuming a large amount of thermal energy (typically, it
requires
2.2-2.8 GJ/ton of soda ash), but also producing surplus greenhouse gas carbon
dioxide (typically, 200-400 kg/ton soda ash) which is usually released to the
atmosphere.
As mentioned, the brine used as a starting material in the traditional Solvay
process is a concentrated brine solution, typically having a salt
concentration of
about 300 g/L. However, we have discovered that brine having a significantly
lower salt concentration can successfully be used in a modified Solvay process
to produce soda ash. This leads to the environmentally positive outcome of
being able to use brine waste as a starting material in soda ash production.
Advantageously, in the process of the present invention the brine waste may be
a waste stream from a desalination plant. Desalination plants are used to
reduce the salt concentration of sea water, brackish water, saline industrial
CA 02744264 2011-05-18 PCT/AU2009/001512
Received 30 April 2010
-5-
waste water, and the like. The desalination plant may be a reverse osmosis
desalination plant, in which case, the brine waste may have a sodium chloride
concentration of greater than about 50 g/L and less than about 250 g/L. In
some
forms of the present invention, the sodium chloride concentration of the brine
waste is greater than about 50g/L and less than about 250 g/L. In other forms,
that lower limit of the range may be 60 g/L, 70 g/L, 80 g/L, 90 g/L, or 100
g/L.
The present invention therefore also provides a process for producing soda
ash,
the process including the steps of:
a) providing brine waste having a sodium chloride concentration of
between about 50 g/L and about 250 g/L;
b) treating the brine waste to remove at least some of any undesired
inorganic cations present to provide pre-treated brine waste;
c) reacting the pre-treated brine waste with carbon dioxide in the presence
of ammonia, at least a portion of which is regenerated ammonia from
step (g);
d) separating sodium bicarbonate produced during the reaction of the
brine waste with carbon dioxide from the reaction mixture to provide
collected sodium bicarbonate and a first mother liquor containing
ammonium chloride;
e) heating the collected sodium bicarbonate to produce soda ash;
f) filtering the first mother liquor to produce purified water and a second
mother liquor containing concentrated ammonium chloride; and
g) treating the second mother liquor with a base anion exchange resin to
regenerate ammonia suitable for use in the reaction of step (c).
Of course, the 'present invention provides soda ash produced by the above
described processes. Soda ash is an important industrial chemical. Soda ash is
used to regulate pH in many chemical process streams. For example, it is the
most widely used fixed alkali for the manufacture of other alkali products,
sodium
salts, glass, soap, sodium silicates, detergent, bicarbonates, bichromates,
cellulose and rayon, iron and steel, aluminium, cleaning
Amended Sheet
IPEA/AU
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-6-
compounds, textiles and dyestuffs, drugs and many other materials. It is also
used as an alkali for household purposes and as washing powder by laundries.
It is used in the manufacture of glass, chemicals, such as sodium silicates
and
sodium phosphates, the pulp and paper industries, the manufacture of
detergents and for the treatment of water. The superior buffering capacity of
soda ash versus caustic soda offers advantages in adjusting plant wastewater
pH ranges.
The present invention provides a more sustainable approach to soda ash
production. Firstly, the process of the present invention utilises brine waste
as
a starting material, thereby ameliorating the need to dispose of brine waste
from
desalination plants in the typical manner. Secondly, the present invention is
also able to release at least a small portion of the water contained in a
brine
waste as purified water for re-use. Thirdly, the present invention can use
carbon dioxide that is obtained from emissions from a combustion source, such
as coal or gas or diesel fired power station, steel works or petroleum
refinery, as
a starting material for the reaction. Finally, and in contrast with the
traditional
Solvay process, limestone and subsequent lime milk need not be used in the
process of the present invention.
BRIEF DESCRIPTION OF THE FIGURES
The present invention will now be described in relation to a preferred
embodiment, of which some aspects are illustrated in the accompanying
figures. In the figures:
Figure 1 is a general flow diagram of a process for producing soda ash
according to the present invention;
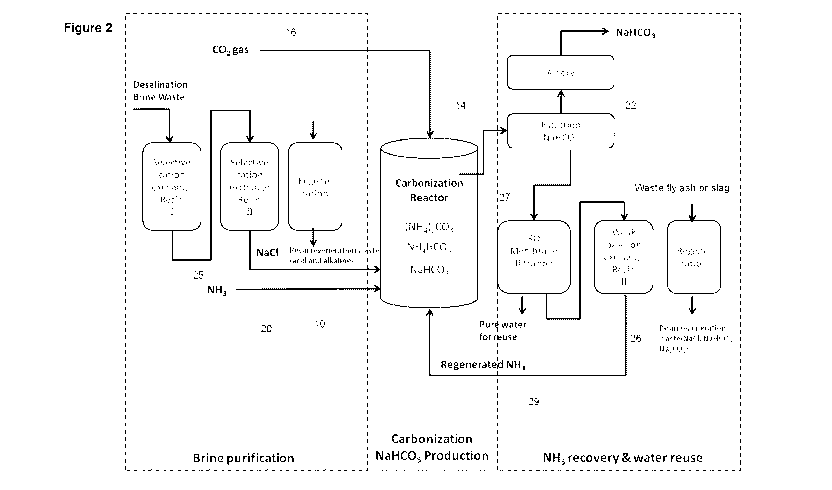
Figure 2 is a detailed flow diagram of a process for producing soda ash
according to the present invention;
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-7-
Figure 3 is a graph showing a comparison of different concentrations of
sodium carbonate for use as a regenerant solution for ion
exchange resin; and
Figure 4 is a plot of sodium bicarbonate (NaHCO3) conversion rates with
varying initial brine concentrations.
DESCRIPTION OF A PREFERRED EMBODIMENT
The present invention provides a process for producing soda ash from brine
waste. The process includes treating brine waste with carbon dioxide and
ammonia under conditions to produce soda ash. In this respect, the term soda
ash means a composition that contains predominantly sodium carbonate
(Na2CO3). Soda ash may not be pure sodium carbonate and may contain other
compounds including, for example, sodium bicarbonate (NaHCO3).
Referring to Figure 1, brine waste 10 from a desalination plant 12 is fed into
a
reaction vessel 14. The brine waste 10 may be subjected to the process of the
present invention on-site at a desalination plant, or it may be stored and/or
packaged for transportation to an off-site processing facility. Of course, to
reduce the costs and environmental impact of transport, the brine waste is
ideally treated on-site at a desalination plant.
Indeed, it should also be appreciated that the brine waste could be obtained
from waste streams from other desalination plants, including plants that use
thermal desalination processes such as Multistage Flash Distillation (MFD),
Multiple Effect Distillation (MED) and Mechanical Vapour Compression (MVC).
These processes all generate concentrated brine streams that can be utilised
for soda ash production as described herein.
Carbon dioxide 16 that is obtained from emissions from a combustion source 18
is introduced into the reaction vessel 14 and passed through the brine waste
10
in the vessel. The carbon dioxide 16 is ideally obtained from emissions from
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-8-
coal or gas or diesel fired power stations, steel works, petroleum refineries,
and
the like. Ammonia 20 is also introduced into the reaction vessel 14 and passed
through the brine waste 10. Typically, ammonia 20 is introduced into the
reaction vessel 14 first followed by introduction of the carbon dioxide 16.
The
overall reaction process is:
NaCl + C02 + NH3 + H2O -* NaHCO3 + NH4CI
The reaction of the brine waste 10 with carbon dioxide 16 and ammonia 20 can
also be carried out in two stages. In the first stage, ammonia 20 would be
bubbled through the brine waste 10 so that it is absorbed by the brine waste
to
produce ammoniated brine. In the second stage, carbon dioxide 16 would be
bubbled through the ammoniated brine.
During or after the reaction, sodium bicarbonate (NaHCO3) precipitates out of
solution. The sodium bicarbonate precipitates because in a basic solution,
sodium bicarbonate is less water-soluble than sodium chloride. The ammonia
(NH3) buffers the solution at a basic pH; without the ammonia, a hydrochloric
acid by-product would render the solution acidic, and arrest the
precipitation.
The sodium bicarbonate that precipitates out in reaction is filtered using a
filter
24 and then dried to form a dried sodium bicarbonate precipitate 22. This
dried
sodium bicarbonate precipitate 22 is subsequently converted to soda ash by
calcination (160 - 230 C), producing water and carbon dioxide as by-products,
as per the following process:
2 NaHCO3 -* Na2CO3 + H2O + C02
The water and carbon dioxide produced during this final calcination step (not
shown in Figure 1) may be captured and re-used as a starting material in the
process or they may be fed directly into the process as required.
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-9-
The solution remaining from the reaction vessel 14 contains ammonium chloride
(NH4CI). In the traditional Solvay process, the mother liquor obtained after
filtration is reacted with quicklime (calcium oxide (CaO)) left over from the
calcination step (CaCO3 -> C02 + CaO) to regenerate ammonia which is
recycled back to the initial brine solution. Unfortunately, and as mentioned
above, this step generates large amounts of calcium chloride solids that are
typically disposed of into a waterway. In the general process of the present
invention illustrated in Figure 1, ammonia is regenerated from the ammonium
chloride by passing the solution containing ammonium chloride through a
regeneration step such as an anion exchange resin 26. No solid wastes are
generated in this regeneration step and, therefore, the environmental impact
of
the process is further reduced.
Referring now to the more detailed flow diagram of Figure 2, it will be seen
that
in some embodiments the process can also include treating the brine waste 10
to remove at least some of any undesired inorganic cations present in the
brine
waste prior to its treatment in the reactor 14 with carbon dioxide and
ammonia.
Examples of undesired inorganic cations are ions that may interfere with the
soda ash production process include, but are not limited to, calcium,
magnesium or strontium ions.
At least some of any undesired inorganic cations present in the brine waste 10
may be removed by selectively binding those ions with a binding agent. Thus,
the flow diagram of Figure 2 shows the brine waste moving through dual cation
exchange treatment processes, where the brine waste is treated with a binding
agent that selectively binds calcium ions, a binding agent that selectively
binds
magnesium ions, and/or a binding agent that selectively binds strontium ions.
In this embodiment, the binding agent is a cation exchange resin. For example,
the cation exchange resin may be AMBERLITE IRC748, which is an
iminodiacetic acid chelating cation exchange resin with high selectivity for
calcium, magnesium and strontium.
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-10-
Additionally, in some reverse osmosis desalination plants it is a standard
practice to dose the feed sea water with an antiscalant, such as polyacrylic
acid,
to prevent membrane fouling. The antiscalants are rejected by the filtration
membrane and remain in the brine waste. An anion exchange resin can also be
used to remove these antiscalants and other organic matters. This step may
alternatively be an activated carbon absorption step, an advanced oxidation
step, or a combination of all of these described steps.
The carbon dioxide gas 16 used in the reaction may be pumped directly from its
source to the reaction vessel 14 containing the ammoniated brine solution.
Alternatively, the carbon dioxide gas may be captured off site and stored in
an
appropriate vessel (such as a cylinder) for transport to the site where the
reaction is carried out.
In some cases, when the carbon dioxide gas is derived from a waste stream, it
may be desirable to scrub the waste stream to remove some or all of any
gasesous contaminants that may be present in the stream. Examples of
gaseous contaminants that may need to be removed include nitrogen oxides
(NOX) and sulphur dioxide (SO2). In some cases, the impurities NO2 and SO2
can be converted into nitrate ion and sulphate ions. The sodium salts of these
ions are more soluble than sodium bicarbonate so the salts remain in solution
and can be finally removed by, for example, membrane filtration before the
reuse of water.
At least a portion of the ammonia 20 that is used to treat the brine waste may
be obtained from any suitable source. However, it is possible to regenerate
and
then reuse ammonia from ammonium chloride produced during the reaction in
the reaction vessel 14, and thus it is advantageous to re-use the ammonia
regenerated in this manner, such that at least a portion of the ammonia used
in
the reaction is regenerated ammonia.
In the flow diagram of Figure 2, the ammonium chloride produced during the
reaction is filtered (after filtration 26 to collect the sodium bicarbonate
formed
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-11-
from the brine waste, the carbon dioxide and the ammonia in vessel 14) in a
membrane filter 27 to concentrate the ammonium chloride solution. This
concentrate may then be treated with a base in an ion exchange process 26 to
generate ammonia suitable for reuse in the reaction. Any suitable base may be
used in this ion exchange process. Advantageously, the base is a weak base
anion exchange resin.
For example, the concentrated ammonium chloride may be passed through a
column containing an anion exchange resin, such as AMBERLYST A23 or
AMBERLITE IR 45, which are highly porous granular weak base anion
exchange resins, both showing sufficient alkali capacity to be used for the
purpose of ammonia regeneration, or such as AMBERLITE IRA 400, a strong
base anion exchange resin. The chloride ions in the concentrate will be
absorbed, and the hydroxyl ions will be exchanged to the concentrate from the
resin. The effluent of the anion exchange resin column contains ammonia with
high alkalinity, and can be returned (via stream 29) back to the carbonisation
reactor.
Sodium carbonate (soda ash) may also be used to itself regenerate spent anion
exchange resin, as represented schematically in Figure 2 by the stage 31. The
weak base anion exchange resins can be efficiently regenerated by much
weaker sodium carbonate solution, such as 1% of sodium carbonate (noting
that the recommended regeneration concentration is 5% sodium carbonate).
Experiments have shown that after an initial difference, the effluent from
both
resins quickly stabilises to the same pH. This implies that the resin has the
same usable capacity under both regeneration conditions. The advantage of the
lower concentration is that much less material is wasted (a five fold
reduction in
chemical consumption). In this respect, it will also be appreciated that the
alkaline waste produced from the regeneration of the weak base cation resin
(stage 25) can be used to regenerate weak base anion exchange resin (stage
26).
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-12-
Advantageously, and as mentioned above, the present invention avoids the
need to use energy intensive lime milk and steam distillation for ammonia
regeneration, as in the traditional Solvay process. The weak base anion
exchange resin can be regenerated by weak waste alkali which is available
from several industries, such as alkaline fly ash from brown coal combustion
or
slag from steel mill.
With particular regard to the use of the base anion exchange resins, testwork
has been conducted on the weak base anion exchange resins to confirm that
they exhibit the most potential to be regenerated by weak waste alkali.
With that in mind we have tested AMBERLITE IR 45 at both the recommended
regeneration concentration of 5% sodium carbonate and also at a much weaker
solution of 1 % sodium carbonate. Both the quantity of regenerant required and
the degree of regeneration of the resin was tested. This was done by passing
regenerant solution through the resin until the effluent was as close in pH to
the
regenerant as practical and then rinsing with deionised water until the
effluent
was neutral. For both solutions this was 8 bed volumes of regenerant solution.
Then, 5% sodium chloride solution was introduced and the pH of the effluent
was monitored.
Plots of these experiments (see Figure 3) show that, after an initial
difference,
the effluent from both resins quickly stabilises to the same pH. This implies
that
the resin has the same usable capacity under both regeneration conditions.
The advantage of the lower concentration is that much less material is wasted
(a five fold reduction in chemical consumption). This is probably because the
pH of a 5% solution of sodium carbonate is 11.5 whereas the 1% solution is
11.1. This small difference is due to the equilibrium dissociation of
carbonate
and water to give hydroxide anion. Because the active material in this
solution
is not sodium (Na+), but hydroxide ion (OH-), there is little difference in
actual
concentration and both are in excess of what is required.
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-13-
DESCRIPTION OF AN EXAMPLE
In order to illustrate an embodiment of the present invention, the following
experimental work was conducted, the results of which are reported in Table 1
below, the work being conducted primarily in order to investigate the effects
of
CO2 concentrations and brine concentrations.
The experimental procedure was as follows:
1) Making reaction solution - NaCl prepared in different concentration levels
was weighed and put into a flask, in concentrations of 216g/I, 147g/I, and
100g/I. 125mL of 28% ammonium water and 275mL of miliQ water was added
to the flask. The flask was capped and the NaCl dissolved with the use of a
magnetic stirrer. The solution was transferred into a gas washing bottle and
covered, and for 15 minutes the washing bottle was placed in a bath set up at
temperature 20 C.
2) Gas mixture - gas cylinders supplying C02, N2, SO2 and NO2 were set up
with mass flow controllers (based on the "multibus" concept) for certain flow
rates and for desired mixing ratios. The percentage of CO2 was set up at 5%,
7.5%, 10% and 15% to simulate expected industrial conditions, and accordingly,
the percentages of N2 were set up at 95%, 92.5%, 90% and 85% respectively),
following which the gas tube was connected to the gas washing bottle.
3) Chemical reaction - the chemical reaction was allowed to proceed to
equilibrium (for about 15 hours), following which the gas was turned off and
the
gas tube disconnected. The reacted solution was allowed to sit overnight in
the
gas washing bottle for crystallization.
4) Filtering - the reacted solution was mixed well in the gas washing bottle,
and
the reacted solution was discharged into a membrane filtering device. The
material collected by the filter was removed after 30 minutes and placed into
an
evaporating dish.
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-14-
5) Drying - the evaporating dish was placed in a fume cupboard at 298K for
drying, and the product was weighed after 2 days of drying.
The experimental work showed the following relationship between C02
concentrations and NaCl concentrations on the process reaction rate, showing
an ideal reaction rate at C02 concentrations above about 7.5% and NaCl
concentrations above about 147g/L.
15% Legend
10% Slow reaction
Medium reaction
75 I 0
Fast reaction
5 Io ........................................................
100g/L 147g/L 216g/L
Additionally, the experimental work showed the following comparative
recoveries from the relationship between C02 concentrations and NaCl
concentrations, which confirms the production of NaHCO3 at different NaCl and
C02 concentrations.
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-15-
Data presented in the order of C02 concentrations
Concentration Percentage Total equilibrium time Unwashed product
of NaCI(g/L) of CO2
216-15 216 15 10hrs 13.36
216-15 216 15 20hrs 39.68
100-15 100 15 14hrs 12.06
147-10 147 10 14hrs 9.84
100-10 100 10 15hrs 5.67
147-7.5 147 7.5 15hrs 7.47
100-7.5 100 7.5 more than 20hrs 4.48
216-5 216 5 more than 20hrs 7.84
147-5 147 5 More than 20hrs 6.13
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Data presented in the order of NaCl concentrations
Concentration Percentage Total equilibrium time Unwashed product
of NaCI(g/L) of C02
216-15 216 15 10hrs 13.36
216-15 216 15 20 hrs 39.68
216-5 216 5 more than 20hrs 7.84
147-10 147 10 14hrs 9.84
147-7.5 147 7.5 15hrs 7.47
147-5 147 5 More than 20hrs 6.13
100-15 100 15 14hrs 12.06
100-10 100 10 15hrs 5.67
100-7.5 100 7.5 more than 20hrs 4.48
CA 02744264 2011-05-18
WO 2010/057261 PCT/AU2009/001512
-16-
It will be appreciated that Figure 4 represents the conversion rates to sodium
bicarbonate based on to its initial Na+ concentrations. Figure 4 shows that
the
conversion rates, represented as (Co-Ce)/Co, increased with increasing Na+
concentration in a logarithmic shaped trend.
Finally, it will be appreciated that various modifications and variations of
the
methods and compositions of the invention described herein will be apparent to
those skilled in the art without departing from the scope and spirit of the
invention. Although the invention has been described in connection with
specific
preferred embodiments, it should be understood that the invention as claimed
should not be unduly limited to such specific embodiments. Indeed, various
modifications of the described modes for carrying out the invention that are
apparent to those skilled in the art are intended to be within the scope of
the
present invention.