Note: Descriptions are shown in the official language in which they were submitted.
ak 02915305 2016-11-09
54106-1940
- 1 -
Description
Method for operating a heat pump arrangement, and heat pump
arrangement
The invention relates to a method for operating a heat pump
arrangement, in which a first fluid flows through a first heat
pump and a second fluid flows through a second heat pump, and
in which heat is transferred from the first fluid to the second
fluid using a heat exchanger. The invention also relates to a
heat pump arrangement, having at least one first heat pump
through which a first fluid flows, and a second heat pump
through which a second fluid flows, in which heat may be
transferred from the first fluid to the second fluid using a
heat exchanger
Such heat pump arrangements are used for example for industrial
heat provision. A'heat pump is a machine which, by using
technical work, absorbs thermal energy in the form of heat at a
lower temperature from a heat source and, together with the
drive energy of a compression machine, releases it as waste
heat at a higher temperature to a heat sink. For temporary
storage or to transfer heat, a fluid is used, which is conveyed
within the heat pump in a cycle process by means of the
compression machine. This cycle process is also known as a
thermodynamic vapor compression cycle.
In the absence of suitable fluids and suitable compression
machines for high temperature heat pumps (HTHP), the useful
heat from heat pumps which are currently commercially
obtainable is limited to temperatures of up to at most 10000.
Mk 02915305 2016-11-09
54106-1940
- 2 -
The object of the present invention is accordingly to provide a
method and a heat pump arrangement by means of which useful
heat at particularly high temperatures may be provided.
According to one aspect of the present invention, there is
provided a method for operating a heat pump arrangement, in
which a first fluid flows through a first heat pump and a
second fluid flows through a second heat pump, and in which
heat is transferred from the first fluid to the second fluid
using a heat exchanger, wherein Useful heat is extracted from
the second fluid at a fluid temperature of at least 150 C,
wherein the useful heat of the second fluid is released' at a
volumetric heating capacity of the first fluid and of the
second fluid of at least 500 kJ/m3, wherein at least one
fluoroketone flows through the first heat pump as the first
fluid.
According to another aspect of the present invention, there is
provided a heat pump arrangement having at least one first heat
pump through which a first fluid flows, and a second heat pump
through which a second fluid flows, in which heat may be
transferred from the first fluid to the second fluid using a
heat exchanger, wherein useful heat may be transferred using
the second fluid at a fluid temperature of at least 150 C,
wherein the first fluid and the second fluid have a volumetric
heating capacity of at least 500 kJ/m3, wherein at least one
fluoroketone may flow through the first heat pump as the first
fluid.
To provide a method of the above-stated type, by means of which
useful heat at particularly high temperatures may be provided,
CA 02915305 2016-11-09
=
54106-1940
- 2a -
provision is made according to the invention for useful heat to
be extracted from the second fluid at'a fluid temperature of at
least 120 C, wherein the useful heat of the second fluid is
released at a volumetric heating capacity of the first fluid
and of the second fluid of at least 500 kJ/m3. The volumetric
heating capacity (VHC) is crucial for the theoretically
achievable coefficient of performance (COP) of heat pumps. In
other words, the higher the VHC, the more efficiently the heat
pump thus operates. Thus, the higher is the volumetric heating
capacity above the stated 500 kJ/m3, the higher also is the
coefficient of performance (COP) of the respective heat pump.
By means of the second heat pump, particularly high fluid
temperatures may be achieved. As a consequence, useful heat at
a particularly high temperature may be extracted from the
second fluid as a. function of the heat transferred by the first
fluid.
It has proven advantageous for the useful heat to be extracted
from the second fluid at a fluid temperature of at least 150 C
and particularly preferably of at least 160 C. By means of the
second heat pump, particularly high fluid temperatures may be
achieved. Consequently, useful :heat at a particularly high
temperature may be extracted from the second fluid, whereby for
example useful heat may be provided all the more effectively
for industrial use.
=
CA 02915305 2015-12-11
PCT/EP2014/061528 - 3 -
2013P05829W0US
In an advantageous configuration of the invention, at least one
fluoroketone flows through the first heat pump as the first
. fluid. Fluoroketones are particularly safe to use in industry,
since there is no need to take special protective measures in
the event of an incident. Since the use of fluoroketones is not
governed by environmental requirements, use thereof is
particularly future-proof. Moreover, they have a particularly
low global warming potential and are non-flammable and non-
toxic. For this reason, fluoroketones are particularly suitable
for use as fluids in heat pump arrangements, in particular if
industrial process heat, in particular useful heat at a
temperature of greater than 120 C, is provided using these heat
pump arrangements.
In a further advantageous configuration of the invention, water
or at least one fluoroketone is used as the second fluid. Since
they are both environmentally friendly and unobjectionable from
the point of view of safety, both water and fluoroketones are
suitable in particular as fluids in applications in which high
fluid temperatures occur, given that they are neither flammable
nor toxic.
It is particularly advantageous for different fluids to be used
as the first and second fluids. The coefficient of performance
(COP) of the respective heat pump depends on the respective
temperature rise. The temperature rise of a heat pump is
understood to mean the temperature difference which may be
achieved between a respective condenser of the heat pump and a
respective evaporator of the heat pump. In accordance with the
achievable temperature rise of the first heat pump, waste heat
at a particularly high temperature may thus be provided and
CA 02915305 2015-12-11
PCT/EP2014/061528 - 4 -
2013P05829W0U5
= transferred by means of the heat exchanger to the second fluid
of the second heat pump. The maximum temperature of the second
fluid which can be reached using the second heat pump thus
depends directly on the quantity of heat transferred by the
first fluid. Particularly high coefficients of performance may
be achieved using particularly large temperature rises of the
respective heat pump, wherein it will be explained below with
reference to an example why it is advantageous for fluids of
different composition to be used in each case for the first and
second fluids. If fluid temperatures of at most 140 C are to be
achieved for example using the first heat pump, the use of
fluoroketone NOVEC 524 is particularly recommended.
Fluoroketone NOVEC 524 has a particularly high volumetric
heating capacity (VHC) in the range from 100 C to 140 C. Since,
however, NOVEC 524 is suitable only up to the stated maximum
fluid temperature of 140 C, in order for example to achieve a
temperature rise from 140 C to 200 C using the second heat
pump, it is recommended that water be used as the second fluid,
water also being suitable for greater fluid temperatures than
140 C.
It is particularly advantageous for the heat release from the
first to the second fluid to proceed largely isothermally.
Through isothermal heat release, the temperature of the
released quantity of heat is kept particularly constant,
whereby temperature fluctuations are particularly largely ruled
out and thus also a largely constant temperature rise may be
achieved using the second heat pump. To achieve isothermal heat
release using the heat exchanger, the first fluid has to be
operated sub-critically, i.e. the first fluid can only be used
CA 02915305 2015-12-11
PCT/E22014/061528 - 5 -
2013P05829W0US
below its critical temperature. In other words, the first fluid
thus has to be operated at a temperature at which both the
. liquid and the gaseous physical states may be present.
In a further advantageous configuration of the invention, the
useful heat is released at a volumetric heating capacity of at
least 1000 kJ/m3, preferably of at least 1200 kJ/m3 and
particularly preferably of at least 1500 kJ/m3 of the second
fluid. Although the theoretically achievable coefficient of
performance (COP) depends on the design of a compression
apparatus by means of which the respective fluid of the
respective heat pump is compressed, the fluid in the heat pump
arrangement should be operated at a point at which a volumetric
heating capacity of at least 1000 kJ/m3 is present. The higher
the volumetric heating capacity above the stated 1000 kJ/m3,
the higher too is the coefficient of performance (COP) of the
respective heat pump. If at least 1000 kJ/m3 are needed for the
volumetric heating capacity of the respective fluid, water at a
temperature of below 150 C cannot for example sensibly be used
as the fluid. If a volumetric heating capacity of at least
1500 kJ/m3 is present for a respective fluid, the coefficient
of performance (COP) of the respective heat pump is
particularly great.
In the heat pump arrangement according to the invention with at
least one first heat pump through which a first fluid flows,
and a second heat pump through which a second fluid flows, heat
may be transferred from the first fluid to the second fluid
using a heat exchanger.
CA 02915305 2015-12-11
PCT/EP2014/061528 - 6 -
2013P05829W0US
In this case, useful heat may be transferred using the second
fluid at a fluid temperature of at least 120 C, wherein the
, first fluid and the second fluid have a volumetric heating
capacity of at least 500 kJ/m3. The higher the volumetric
heating capacity, the greater too is the achievable coefficient
of performance (COP) of the respective heat pump. Useful heat
at a particularly high temperature may in this case be
extracted from the second fluid as a function of the heat
transferred by the first fluid. To provide a heat pump
arrangement, which is also known as a heat pump cascade, in
which the second heat pump may provide useful heat at a
particularly high temperature, a maximally high volumetric
heating capacity of the first fluid of the first heat pump is
favorable, wherein it is favorable if the quantity of heat
transferred from the first to the second fluid is transferred
at a particularly high temperature.
The advantages described above in relation to the method
according to the invention apply in the same way to the heat
pump arrangement according to the invention and vice versa.
In one advantageous configuration of the heat pump arrangement,
at least one temperature rise resulting from a relatively high
pressure ratio of the first fluid and/or the second fluid may
be increased by means of at least two-stage compression. If
particularly large temperature rises are to be achieved with a
fluid in a heat pump, two- or multistage compression is
recommended. In this case, intermediate cooling may be fitted
between the compression apparatuses effecting the respective
compression stage. This is sensible in particular where water
is used as the fluid. The heat of the intermediate cooling may
CA 02915M52015-12-11
PCT/EP2014/061528 - 7 -
2013P05829WOUS
be fed in a particularly energy-efficient manner to an
evaporation apparatus of the respective heat pump. To bring
, about very high temperature rises, cascades of more than two
heat pump circuits are moreover also possible.
It has been demonstrated that, in a further advantageous
configuration of the heat pump arrangement, the second fluid is
largely isothermally compressible using a liquid ring
compressor. Compression of the fluid may proceed largely
isothermally using a liquid ring compressor. The liquid ring of
the liquid ring compressor is then in direct contact with the
fluid to be compressed, whereby heat of compression may be
transferred particularly effectively from the fluid to the ring
liquid from which the liquid ring is formed. In other words,
the heat transfer resistance is thus particularly low, since
the fluid and the ring liquid are not divided from one another
by a wall.
Further advantages, features and details of the invention are
revealed by the claims, the following description of preferred
embodiments and with reference to the figures, in which:
FIG. 1 is a schematic representation of a heat pump cascade
according to the prior art, which corresponds to a
heat pump arrangement with in the present case two
heat pump circuits;
FIG. 2 is a schematic diagram of the respective curves of
the volumetric heating capacities of various fluids
of the heat pump arrangement plotted against
temperature; and
CA 02915305 213112-11
PCT/EP2014/061528 - 8 -
2013P05829WOUS
FIG. 3 is a schematic representation of a heat pump cascade
which corresponds to a heat pump arrangement with two
heat pump circuits, wherein one of the heat pump
circuits is operated with a fluoroketone as fluid.
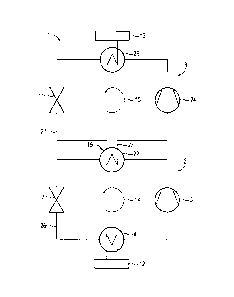
FIG. 1 is a schematic representation of a heat pump arrangement
which comprises two heat pump circuits and is known in
accordance with the prior art as a cascade heat pump 1. The
cascade heat pump 1 comprises a first heat pump 2 through which
a first fluid flows and a second heat pump 3 through which a
second fluid flows. The first and second fluids are coupled
together for heat transfer by means of a heat exchanger 19. In
this case, the heat exchanger 19 comprises a condenser 6 of the
first heat pump 2 and an evaporator 8 of the second heat pump
3. The first fluid of the first heat pump 2 is evaporated by
means of an evaporator 4, wherein the evaporator 4 is supplied
with thermal energy by means of a heat source 12. The first
fluid heated by means of the evaporator 4 is delivered in the
direction of an arrow 14 by the first heat pump 2 by means of a
compressor 5 of said first heat pump 2. Then the heated and
compressed first fluid in the condenser 6 releases heat to the
evaporator 8, wherein the second fluid of the second heat pump
3 is evaporated by means of the evaporator 8. Subsequent to
this release of heat, the first fluid is expanded by means of
an expansion valve 7 of the first heat pump 2 and thereupon
once again absorbs heat through the evaporator 4. The circuit
of the first heat pump 2 is thus closed. The second fluid of
the second heat pump 3 heated by means of the heat exchanger
19, i.e. by release of heat by means of the condenser 6 of the
first heat pump 2 to the evaporator 8 of the second heat pump
CA 02915305 2015-12-11
PCT/EP2014/061528 - 9 -
2013P05829W0US
3, is compressed by means of a compressor 9 of the second heat
pimp 3 and in a condenser 10 of the second heat pump 3 releases
,heat to a heat sink 13. Then the second fluid flows in the
direction of an arrow 15 through an expansion valve 11 of the
second heat pump 3 and is expanded there. Then the second fluid
again absorbs heat by means of the heat exchanger 19 and the
circuit of the second heat pump 3 is thus closed.
FIG. 2 is a schematic diagram of different curves of volumetric
heating capacities, wherein the y axis of the diagram plots a
volumetric heating capacity 20 and the x axis plots a fluid
temperature 21, which corresponds to the condensing temperature
of the fluid. This diagram shows that a heating capacity curve
16, which corresponds to the heating capacity curve of a
fluoroketone known as NOVEC 524, in each case has higher values
for the same fluid temperatures 21 than a heating capacity
curve 17, which corresponds to the heating capacity curve of a
fluoroketone known as NOVEC 649. As is clear from the diagram,
neither the heating capacity curve 16 nor the heating capacity
curve 17 extends over the entire length of the x axis, on which
the fluid temperature 21 is plotted. Thus, the heating capacity
curve 16 of the fluoroketone NOVEC 524 is limited by reaching a
critical point 28 at 148 C and the heating capacity curve 17 of
the fluoroketone NOVEC 649 is limited by reaching a critical
point 29 at for instance 169 C. Although, as is clear from the
diagram, a heating capacity curve 18 which corresponds to the
heating capacity curve of water has the lowest volumetric
heating capacity 20 in each case for the same fluid
temperatures 21 compared with the two fluoroketones, water may
be used over a particularly wide range of fluid temperatures
CA 02915305 213112-11
PCT/EP2014/061528 - 10 -
2013P05829W0US
= 21, without the critical point thereof being reached. As is
additionally clear from FIG. 2, although the heating capacity
.curve 18 of water at fluid temperatures below the critical
point 28 and the critical point 29 is respectively below the
heating capacity curve 16 and the heating capacity curve 17,
the heating capacity curve 18 of water rises at high fluid
temperatures 21 to greater values than can be achieved with the
heating capacity curve 16 and the heating capacity curve 17 as
a consequence of the respective critical points 28 and 29
respectively being reached. It is moreover clear that a
quantity of heat with a working temperature of at least 160 C
may be released by means of the cascade heat pump 1 to the heat
sink 13 if the fluoroketone NOVEC 649 is used as the first
fluid of the first heat pump 2. Since, on the basis of the
quantity of heat transferred by means of the heat exchanger 19
from this first fluid to the second fluid at a temperature of
up to 160 C, the second fluid of the second heat pump 3 is
heated further, a temperature of the useful heat even higher
than 160 C may be reached by means of the second heat pump 3.
Only subcritically operated fluids are options for the fluid
for the first heat pump 2 of the cascade heat pump 1, since
heat release from the first fluid to the second fluid should
proceed isothermally by means of the heat exchanger 19. To
allow isothermal heat release, the first fluid of the first
heat pump 2 is operated at a fluid temperature 21 which lies
below the critical temperature of the respective critical point
28 or 29. The higher the volumetric heating capacity 20 of a
fluid of one of the heat pumps 2, 3, the more efficiently the
respective heat pump 2, 3 operates. Thus the theoretically
CA 02915305 2015-12-11
PCT/EP2014/061528 - 11 -
2013P05829W0US
achievable coefficient of performance thereof also increases
with a respective higher volumetric heating capacity 20.
As is clear when FIG. 2 and FIG. 3 are looked at together, it
is particularly advantageous for a fluoroketone 26 such as for
example NOVEC 524 to be used as the first fluid of the first
heat pump 2. It should be noted in this respect that the fluid
temperature 21 to which the fluoroketone 26 is heated remains
below the fundamental critical temperature of the critical
point 28, in order to allow isothermal heat release by means of
the heat exchanger 19 to the second fluid of the second heat
pump 3. Water 27 is used, for example, as the second fluid of
the second heat pump 3.
The cascade heat pump 1 illustrated in a schematic
representation in FIG. 3 substantially comprises the components
already described in FIG. 1, for which reason only the
differences will be examined below.
Instead of the condenser 6 and the evaporator 8, the heat
exchanger 19 according to FIG. 3 comprises a high temperature
condenser 22 of the first heat pump 2 and a high temperature
evaporator 23 of the second heat pump 3. Moreover, as is
visible in FIG. 3, a liquid ring compressor 24 is used to
deliver the water 27 rather than the compressor 9. By means of
the liquid ring compressor 24, the water 27, which has been
previously evaporated as the result of an input of heat by
means of the high temperature evaporator, is now compressed and
supplied to a high temperature condenser 25.
Due to the particularly high volumetric heating capacity of the
fluoroketone 26 of over 3000 kJ/m3 and thus markedly over 1500
CA 02915305 2015-12-11
PCT/EP2014/061528 - 12 -
2013P05829W0US
kJ/m3, a quantity of heat is transferred at a fluid temperature
21 of 140 C of the fluoroketone 26, which does not exceed the
. critical temperature of the critical point 28, by means of the
high temperature condenser 22 of the first heat pump 2 to the
high temperature evaporator 23 of the second heat pump 3. Thus,
a quantity of heat at a particularly high temperature may be
released to the water 27 by means of the high temperature
evaporator 23, wherein as a consequence a quantity of useful
heat at a particularly high temperature may be released to the
heat sink 13 by means of the high temperature condenser 25. If
the water 27 which is conveyed as a fluid in the second heat
pump 3 is heated to a temperature of for example 200 C by the
quantity of heat at 140 C transferred by means of the heat
exchanger 19 from the fluoroketone 26 to the water 27, this
corresponds to a rise in temperature of 60 C of the water 27.
At 200 C the volumetric calorific value 20 of water 27 amounts
to over 4000 kJ/m3, i.e. a markedly higher value than 1500
kJ/m3.