Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
-- 1 --
MAKING COMPOSITE METAL DEPOSIT BY SPRAY CASTING
This invention is concerned with a method of
making a composite metal deposit by spray casting.
The technique of spray casting is well known and
comprises the steps of atomising a stream of molten
metal to form a spray of hot metal particles by
subjecting the stream to a relatively cold gas directed
at the stream, and depositing the spray on a substrate.
The provision of rapid and controlled cooling permits
the production of deposits having unusual
microstructures, which can be rolled or formed into
shaped articles. But with only one source of molten
metal, there is a limit to the range of compositions
and microstructures that can be obtained.
GB 8606733 describes a spray casting method which
includes the step of applying to the stream or spray
fine, solid particles of a material of different
composition from the metal. The particles are
incorporated in the deposit. The use of refractory
particles e.g. of alumina or silicon carbide can result
in metal matrix composites having enhanced
properties.
US Patent 4522784 describes a casting method in
which two streams of molten metal are mixed just prior
to casting, the smaller stream having a higher
temperature than the larger stream and being chosen so
that a fine intermetallic precipitate is formed during
and prior to casting. With the DC casting methods
described, it is difficult to remove heat fast enough
from the system to prevent to re-solution of the
intermetallic precipitates.
GB 1359486 describes a spray casting technique for
casting two immiscible metals of different density.
A single flow of molten metal consisting of concentric
streams of the two metals is atomised and the drops
collected on a substrate. The range of alloy
-- 2 --
compositions that can be cast in this way is quite
restricted.
This invention provides a convenient way of making
composite metal deposits by spray casting, which is
characterized over the above prior art by the fact that
separate streams of molten metal are atomised
separately. As a result, there is much less
restriction on the compositions of the two metals.
GB 1083003 describes a method of making bearing
materials by spraying Al and Pb simultaneo~sly onto a
backing strip. This results in a microstructure
comprising alternating regions, of size corresponding
(substantially) to the molten spray droplets, of Al and
Pb. U~ 3826301 contain a similar disclosure.
The invention provides a method of making a
composite metal deposit which method comprises
providing a first metal stream, a second metal stream
and a substrate, atomising the two streams to form
first and second sprays of hot metal particles by
subjecting the streams to relatively cold gas directed
at the streams, and depositing the sprays
consecutively on the substrate to form thereon a
laminated deposit.
The first and second metal streams may be provided
by gravity flow from holding vessels containing
supplies of the molten metals. The invention
contemplates the use of two, three or more molten metal
streams, each being atomised separately, and references
to first and second metal streams should be construed
accordingly.
Atomisation conditions may be chosen, as is known
in the art, to control the size, velocity, direction
and temperature of the sprays of hot metal particles.
On being atomised, the particles of molten metal spread
out in a conical spray pattern, which may be of
~ 8~
circular cross-section or may be modified, as known in
the art, to form a different cross-section or a more
even spr~ad of metal particles.
The substrate may be a metal surface, which may
for example be flat or tubular with the metal spray to
be deposited on the inner or the outer surface. It
is generally preferred that the metal particles be
still at least partially liquid on impact, otherwise
the deposit may be too porous. By suitable control
of the atomising conditions, the metal spray can be
arranged to be partial ly or ful Iy ~ iquid but super-
cooled on impact, so that solidification takes place
immediately on impact and there is no need to extract
large amounts of heat through the substrate.
It is possible to provide fibres, whiskers or
particles of refractory material, e.g. carbon or
silicon carbide, on the substrate in such a way that
they become embedded in the coherent composite metal
deposit and provide reinforcement for it. Also if
desired, particles of refractory material can be
incorporated in the first and/or second spray by the
technique described in GB 8606733 noted above.
Three ~or more) sprays may be used. For example
the spray patterns of two may be superimposed and
operated simultaneously. This can result in a
laminated structure in which alternate layers have a
microstructure resulting from these two sprays. Or
two superimposed sprays may be chosen to interact as
described in the aforesaid US patent 4522784.
There is no critical range of ratios of the two
metals constituting the first and second sprays.
Suitable proportions of the two (or more) metals are
chosen for particular applications. The spray
patterns of the first and second sprays may, but need
not, overlap; that is to say, the two sprays may be
-- 4
arranged to impact on the same or different areas of
the substrate. The substrate may be translated, or
reciprocated, or rotated in order to pick up the two
metal sprays. These features can be used to
exercise further control over the structure of the
deposit. For example, if the spray patterns of the
two sprays do not overlap and the substrate is
reciprocated between them, the deposit may consist of
alternating layers of first and second metal.
1~If the spray patterns of the two sprays do
overlap, then it is necessary to operate the two sprays
- alternately in order to achieve the desired laminated
structure. In order to operate both sprays
continuously, it is therefoe preferred that the two
spray patterns can be arranged not to overlap, and to
reciprocate or rotate the substrate so that alternating
layers of the two metals are deposited thereon.
Other variations in structure can be achieved by
supplying one of the metal streams continuously and the
other only intermittently. Or a first metal stream
may be supplied initially, followed by the second, so
that the deposit consists of the first metal with a
surface coating of the second. The supply of molten
metal in two or more streams gives the operator a great
deal of latitude in determining the structure of a
deposit.
Preferably, the laminated deposit comprises at
least two layers of each metal in alternating
superimposed relationship. The thickness of the
alternating layers has a significant effect on the
properties of the laminate. In the as-sprayed deposit,
each layer preferably has a thickness in the range 0.01
- 100 mm, particularly 1 - 10mm. The as-sprayed
deposit may be subjected to rolling. For many purposes
- 5 -
it is preferred that each layer in the rolled product
have a thickness of from 10 to 500 microns, particularly
! 30 to 200 microns.
Since the two metals do not contact one another
S prior to deposition on the substrate, which may be
followed by immediate solidification, there is very
little restriction on the nature of 'he metals that may
be used. It would be disadvantageous if the
temperature of one metal on deposition were so high
that substantial mè~ting of the other took place on ~he
deposit. It is often convenient to use two different
alloys of the same base metal. The method is of
particular interest for spray casting aluminium alloys,
for which purpose an inert gas such as argon or
nitrogen is generally desirable though not essential.
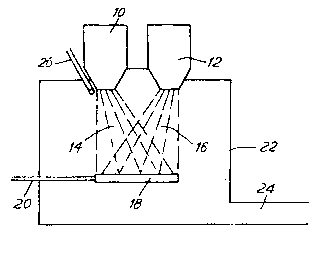
Reference is directed to Figures 1 to 3, each of
which is a schematic diagram of a system for making a
composite metal deposit according to the invention.
Referring to Figure 1, the system comprises first and
second furnaces 10 and 12 for supplying first and
second streams of molten metal, which are atomised (by
means not shown) to form first and second sprays, 14
and 16, of hot metal particles. The spray patterns
overlap, and each spray is operated in turn while the
other is shut off. The sprays are deposited in turn on
a substrate 18, whose position and orientation are
controlled by means 20. The metal sprays and the
substrate are contained within a spray chamber 22,
which is closed except for an exit vent for gas and any
overspray powder.
A refractory material contained in a stream of
carrier gas is supplied via a pipe 26 to the region
where the first metal is atomised, and becomes
incorporated in the first metal spray 14.
' ' :.
- 6 -
In the system shown in Figure 1, the substrate 18
might be maintained stationary, so as to build up a
composite metal body thereon; or it might be
translated, rotated or reciprocated in order to build
up a uniform composite metal layer. The substrate 18
might have taken the shape of a mould, with the
intention of working the deposit while on the substrate
to form a shaped article. In the system shown,
provided that the metal drops are still liquid on
impact, the deposit will be substantially non-porous
and will comprise alternating layers of the first and
second metals.
Figure 2 corresponds to Figure 1, except that the
spray patterns of the two metal sprays 14 and 16 are
shown as being non-overlapping. In this case, the
sprays are operated continuously and the substrate 18
is reciprocated in order to obtain a deposit consisting
of alternating layers of first and second metal.
In Figure 3, the spray patterns of the two metal
sprays 14 and 16 are shown as partly overlapping when
they impact on cylindrical substrate 18 which is caused
to rotate round a horizontal axis 28. A fibre 30 is
supplied from a spool 32 and becomes incorporated in
the deposit.
There follow examples of combinations of metals
and alloys that can be used to make composite metal
deposits according to this invention. In each case,
the first metal or alloy mentioned will generally be
present in the deposit in a volume concentration as
great or greater than the second metal or alloy.
A. The first metal is the alloy designated 7010
in the Aluminum Association Register, and the second
metal is a softer Al alloy such as 6061 or pure Al
metal. 7010 is typically used for aircraft
~ 8~
structures, and the second metal improves fracture
tou~hness by a micro-laminated structure to reduce
fatigue and blunt cracks.
B. The first metal is 7010 alloy and the second
metal is 6010 alloy to confer ductility, toughness and
fatigue resistance.
C. The first metal is 7010 alloy and the secund
metal is an Al/Zn alloy to improve stress corrosion
resistance.
D. The first metal is 7075 all~y, used for
armour plating, and the second metal is an Al/Si alloy
to increase resistance to spalling.
E. The first metal is 7075 alloy, and the second
metal is Pb to increase density, improve ballistic
properties, and provide a microstructure to break up
shock waves.
F. The first metal is any Al alloy and the
second metal is Zn, applied on the surface as a layer
amounting to 1 to 5~ of the total thickness of the
deposit, to aid diffusion bonding.
G. The first metal is 6061 alloy and the second
metal is 7475 alloy + SiC to provide a product having
improved ductility and toughness.
EXAMPLE
Laminated material was sprayed using a combination
of 6061 Al alloy from one atomizer and 7475 Al alloy +
SiC from the other atomizer.
1. Spraying conditions.
Both crucibles used were alumina with zirconia
nozzles. The atomizing gas was nitrogen. The
collector was an aluminium plate 300 mm long by 150 mm
wide. The plate was reciprocated beneath the sprays at
a frequency of 1 Hz.
~9 ~84
The following are the spraying conditions
for the 6061 alloy
Temperature of melt = 730C
s
Primary gas Pressure = 1.~ bar
Flow rate = 0.6 m3/min
Yelocity - 240 m/sec
Secondary gas Pressure = 5.5 bar
Flow rate = 8.0 m /min
Yelocity = 300 m/sec
The conditions for the 7475 alloy were the same
apart from the melt temperature which was 710C. 5iC
particulate (F600) was fed to the 7475 spray only, SiC
was fed at a rate of 1.72 kg/min to the atomizing
nozzle.
Several batches of material were made using
similar conditions.
2. Microstructure of product
Measurements of the elemental distribution across
the bands indicate that there has been a degree of inter-
diffusion between adjacent layers indicating that an
effective metallurgical bond has been made between them.
The SiC content of the 7475 layers was 10-12X by volume.
3. Mechanical properties
The as - sprayed material was consolida'ced by hot
rolling at 430C to approximately 2 mm in thickness and
then further cold rolling to 1 mm. Any as - sprayed
porosity was found to close up during this process to
form a fully consolidated product. The sheet was
solution heat treated by holding for at least 30 min at
500C and cold water quenching. The material was
artificially aged for 20 hr at 120C
- 9 -
Tensile properities were determined on several
batches of sheet containing different thicknesses of
laminate. The following table contains a summary of
the results.
i
Material 0.2X PS (MPa) TS (MPa) Ef (X)
5 layer laminate 270 369 4.4
(=200 micron layers)
17 layer laminate 273 406 9.9
(=70 ~icron layers)
7475 alloy with SiC525 5~1 1.7
6061 alloy 240 260 B.0
6061 alloy with SiC322 340 0.9
Crack initiation and propagation were also
determined with the following results.
Material Initiation Propagation
energy energy
(J/m ) (J/m2)
5 layer laminate
(=200 micron layers) 13433 40597
17 layer laminate
(=70 micron layers) 17623 55788
7475 alloy with SiC3189 8182
- lo ~
Th~se results illustrate that the combination of
the two materials in the laminate can result in
improvements in mechanical properties over the
individual constituents. In this instance the results
- 5 indicate an improvement in strength and ductility over
6061 and an improvement in ductility and crack
initiation and propagation energy (indicating improved
toughness) over the 7475 alloy with SiC. The results
also indicate a significant improvement in the
properties (notably ductility) over that of the 6061
alloy with SiC. It is also notable that the thickness
of the laminated regions is highly important in
controlling the final properties of the material -
specifically in this instance the ductility and
toughness of the laminate.