Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
~'7~
-- 1 --
BACKGROUND OF TIIE INVENTION
Steam-cracking is one of the most widely used
basic petrochemical processes. It is used by industries
to produce light olefins such as ethylene, propylene,
butenes and butadiene and it is also relied upon for the
production of aromatics such as benzene, toluene and
xylenes.
Basically, steam-cracking comprises a step in
which the hydrocarbon mixture to be transformed is mixed
with steam and submitted to elevated temperatures in a
tubular reactor. The different resulting products,
gaseous and liquid hydrocarbons are then collected and
separated. Thus, product distribution depends on the
nature of the initial hydrocarbon mixture as well as
experimental conditions.
Among the products obtained, C2-C4 light
olefins, as well as benzene, toluene, ethylbenzene and
xylenes have the highest commercial values and since
enormous quantities are processes throughout the world,
even small yield improvements lead to substantial profit
increases.
In recent years, ZSM-5 zeolite catalysts have
drawn considerable attention because of their abil.ity
to increase selectivity in a variety of industrial
processes such as xylene isomerization, toluene dis-
proportionation, aromatic alkylation and methanol
conversion.
, It has been shown that the zeolite's selec-
tivity properties are the result of its tridimensional
channel network and of the different pore sizes of its
structure.
One of the most interesting areas where ZSM-5
zeolites have shown substantial catalytic activity is in
the process in which methanol is converted into hydro-
carbons. Thus, by using appropriate reaction conditions,
very high yields in C5-C11 gasoline hydrocarbons can be
obtained. However, this reaction presents the drawback
of producing small quantities of durene, an undesirable
reaction product.
Furthermore, modiEications of the catalyst can
also lead to highly efficient production of light ole-
:Eins resulting :Erom methanol conversion.
Thus, it can be seen that modified zeolite
catalysts have the possibilities to present very inter-
esting properties for enhancing yields in petrochemical
reactions.
Therefore, since steam-cracking is one of the
most widespread petrochemical processes, it would be
highly desirable to provide means for increasing pro~
duction of the most valuable reaction products.
SUMMARY OF T~E INVENTION
. The present invention relates to a process for
~`7~
up-grading products resulting from the steam-cracking of
hydrocarbons which comprises bringing the steam-cracking
reaction products in contact with a multifunctional
Zn-ZSM-s zeolite/cr2o3/Al2o3 catalyst comprising of a
mixture of from 2.5 to 7.5% wt of Cr2O3, 5 to 17.5% wt
of Al2O3 and 75 to 85% wt of a Zn-ZSM-5 zeolite or a
Zn-ZSM-5 zeolite/asbestos. Such a process allows for
significant yield increases in C2-C~ olefins. Further-
more, the commonly obtained pyrolysis oil is up-graded
to a high grade gasoline, rich in mono-aromatics and
free from undesirable durenes and long aliphatic chains.
DETAILED DESCRIPTION OF THE INVENTION
The main feature of the present invention
resides in the presence of a catalytic reactor at the
outlet of -the steam-crac]~ing reactor. This catalytic
reactor contains a multifunctional catalyst which com-
prises a zeolite cf the ~SM-5 type coupled with metallic
oxides.
These oxides can either be coupled to the
zeolite by being directly deposited on the zeolite or
mechanically mixed with the zeolite.
The metallic oxides can be selected from
oxides such as Cr2O3, Al2O3, or from any metallic oxide
having a hydrogenating/dehydrogenating function.
In the case of the Cr2O3/Al2O3 proportions of
Cr2O3 ranging between 2.5 and 7.5% wt, proportions of
2~0
A12O3 ranging between 5 and 17.5% wt and proportions of
the zeolite catalyst ranging between 75 and 85% wt can
be used.
Although the catalytic reactor used in the
present invention was a fixed-bed reactor, it will be
understood that any suitable design commonly used for
catalytic reactions could have been chosen.
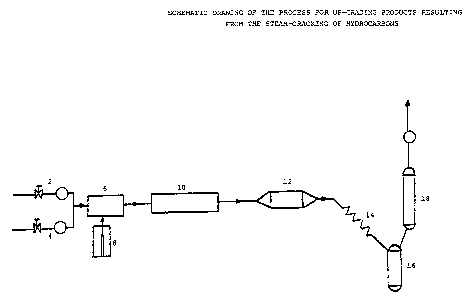
In the drawings: Figure 1 represents a sche-
matic drawing of the bench scale setting for the cata-
lytic up-grading of products resulting from the steam-
cracking of hydrocarbons.
Figure 2 represents a comparison between the
amounts of C2-C~ olefins obtained by steam-cracking
alone and by steam-crackincJ along with various zeolite
catalysts.
Figure 3 represents a comparison between the
amounts of ethylene obtained by steam-cracking alone and
by steam-cracking along with various zeolite catalysts.
Referring now to Figure 1, the starting hydro-
carbon material 2 is first mixed with a stripping gas 4.It is to be noted, however, that the use of a stripping
gas is optional. In the context of the actual experi-
ments, a stripping gas was used only for convenience.
The resulting mixture is then forwarded to a
vaporizer-mixer 6, in which steam is injected by means
of an infusion pump 8. The gaseous mixture thus
obtained enters a steam-cracking tubular reactor 10
heated at a temperature ranging between 760 and 860C.
In a further step, products coming out of the steam-
cracking tubular reactor 10 are sent into a catalytic
reactor 12 heated at a temperature ranging between 450
and 550C. The resulting products are then cooled by a
series of condensers 14 (wa-ter~cooling condensers and
ice bath). Immediately following the cooling step, the
liquid and gaseous phases are separated. The liquids
are first collected in a liquid-collector cylinder 16
while the gases flow through the liquid-collector cylin-
der to be collected for on line analysis in a dynamic
sampler cylinder 18 located at a higher position than
the liquid collector cylinder.
The present invention will be more readily
understood by reEerring to the Eollowing examples which
are given to illustrate rather than limit the scope of
the invention.
E~AMPLE 1
Propane is the starting hydrocarbon material
on which the steam-cracking process was performed. It
was introduced into the system at a flow rate of 45
ml/min. or 4.95 g/hour. It was first mixed with helium
acting as a stripping gas. After having been flown
through the vaporizer-mixer, in which steam was injected
at a rate of 1.7 g/hour, the gaseous mixture was then
sent into the steam-cracking reactor whose internal
temperature had been set to 780C at atmospheric pres-
sure. The residence time of the starting material in
the steam-cracking reactor was approximately 1 second.
The resulting product was then separated into
its liquid and the gaseous phases. The liquid fraction
was analyzed by GC using a capillary column (length:
50 m, PONA~ type, fused silica coated with a cross-
linked polymer). The gases were analyzed on line by gas
chromatography. A column having a length of 3.5 m
packed with Chromosorb9 P coated with 20% by weight of
Squalane~ was used for the analysis. The GC used was a
dual FID Hewlett-Packard Model 5790 equipped with a
3392A Model integrator. Results are shown in Table 1.
EX~MPLE 2
The same procedure as in Exc~mple 1 was re-
peated the only modification being the internal temper-
ature of the steam-cracking reactor which was set at
800C. Results are shown in Table 1.
EXAMPI.E 3
The same procedure as in Example 1 was re-
peated the only modification being the internal temper-
ature of the steam-cracking reactor which was set at
835C. Results are shown in Table 5.
EXAMPLE 4
As in Example 1 propane was chosen as the
t;~
-- 7 --
starting hydrocarbon material. It was mixed with helium
and flown through the vaporizer-mixer. The gaseous
mixture was then forwarded through the steam-cracking
reactor whose internal temperature had been set to
780C. The resulting products were then sent to the
catalytic reactor which had been previously embedded
with 4 g of a Zn-Mn-2SM-5 zeolite which was prepared
according to the procedure described in Can. Pat. Appl.
S.N. 471,463. The temperature of the catalytic reactor
had been previously set at 500C, with a pressure of 1
atmosphere and a W.~I.S.V. (weight hourly space velocity)
of 1 h 1, The final products were analyzed using the
techniques discussed in Example 1. Results are shown in
Table 2.
EX~MPLE 5
The same procedure as in Example 4 was re-
peated, the only modification being the internal temper-
ature of the steam-cracking reactor which was set at
800C. Results are shown in Table 2.
EXAMPLE 6
The same procedure as in Example 4 was re-
peated, except for the following modifications the
catalytic reactor was embedded with 4 g of a Zn-Mn-ZSM-5
zeolite/asbestos catalyst prepared according t~ the
procedure described in Can. Pat. Appl. S.N. 471,463.
Results are shown in Table 3.
,
- ~7~
EXAMPLE 7
The same procedure as in Example 6 was re-
peated, the only modification being the internal temper-
ature of the steam-cracking reactor which was set at
800C. Results are shown in Table 3.
E~AMPLE 8
The same procedure as in Example 4 was re-
peated, except for the following modification: the
catalytic reactor was embedded with a Zn-ZSM-5 zeolite/
asbestos/Cr2O3/A12O3 catalyst. The Zn-ZS~-5 zeolite/
asbestos catalyst was prepared according to the method
described in Can. Pat. Appl. S.N. 471,463. Then, 4.5 g
of the Zn-ZSM-5 zeolite/asbestos catalyst obtained were
wet with a solution prepared from 0.3 g of Cr2O3 and
0~4 g of sodium aluminate dissolved in 5 ml of distilled
water. The resulting multifunctional catalyst was dried
at 120C for 12 hours and actuated at 500C for another
12 hour period. Finally, the catalyst was reduced in
hydrogen at 350C for at least 1 hour. Results are
shown in Table 4.
EXAMPLE 9
The same procedure as in Example 8 was re-
peatedt the only modification being the internal temper-
ature of the steam-cracking reactor which was set at
~00C. Results were shown in Table 4.
When studying the results obtained from the
- 9 -
various examples, it is to be noted that in the steam-
cracking process alone (Table 1) significant increases
in highly valuable compounds such as ethylene, benzene
and toluene are observed when the internal temperature
of the reactor is increased from 780 to 800C. The
amount of less valuable products such as methane is
higher at 800C but this increase is compensated by a
decrease in C2-C4 paraffins.
As for the aromatic content, there is a
dramatic decrease in less valuable C5-Cll aliphatics,
resulting in the obtention of more interesting products
such as benzene, xylenes and toluene. In examples 4 to
7, æn-Mn-ZSM-5 zeolite and Zn-Mn-ZSM-5 zeolite/asbestos,
two known catalysts were used to form the catalytic bed.
As it can be seen in Tables 2 and 3, and in Figures 2
and 3, inferior results were obtained when compared to
steam-cracking alone as far as the olefin content is
concerned, regardless of the temperature at which the
reactions were performed.
As for the aromatic content, better results
were obtained, but these results are at the best suf-
ficient and no more, to compensate the quality loss on
the side of the olefin production, especially, as far as
ethylene is concerned, since ethylene is the most valu-
able steam-cracking product.
Thus, in the light of these results, one could
~;~7~
~ 10 ~
tend to be led away from using zeolite catalysts as
means to improve steam~cracking processes.
In Examples 8 and 9, the results obtained by
using a multifunctional catalyst point out better
results in both olefin and aromatic productions. Thus,
it has been discovered as it can be seen in Figures 2
and 3, that the use of metal oxides co~catalyst coupled
with a zeolite type catalyst unexpectedly increases the
amounts of valuable steam~cracking products. In fact,
the total amount of C2-C4 olefins and especially ethyl-
ene obtalned by using the multifunctional catalyst after
a steam-cracking reaction of 780 (55.8~ wt) is even
superior to the amount obtained when performing the
steam-cracking reaction alone at 800 (47.1 wt).
Moreover as described in Example 3,a run
without catalyst was performed at 835C. This tempera~
ture was fairly close to temperatures used in industrial
steam-cracking facilities usin~ propane as a starting
hydrocarbon material. When the product distribution oE
such a run is compared to the run performed in presence
of the Zn~ZSM~5 zeolite/asbestos/Cr2O3/~12O3 catalyst
and with the steam~cracking reactor temperature set at
800C, as described in Example 9, it can be seen, as it
is shown in Table 5, that in the presence of the multi-
functional catalyst and with a much lower steam~cracking
temperature, higher yields in ethylene and propylene
were obtained. The propylene yield was nearly doubled
(due mainly to a lower steam-cracking temperature) and
the ethylene yield was increased by S wt percentage
points while methane formation was significantly lower.
Furthermore, the liquid yield was much lower
for the run performed at a lower steam-cracking tempera-
ture in the presence of the multifunctional catalyst.
However, the BTX aromatics (benzene, toluene, ethyl-
benzene and xylenes) content in the licluid hydrocarbon
ln products was much higher and there was no formation of
undesirable hydrocarbons.
Thus, by performing the steam-cracking of
propane at a lower temperature and by using a multi-
functional catalyst, the total "ethylene -~ propylene"
yield can be increased by 10 wt percentage points and
the ethylene/propylene wt ratio can be decreased to a
very large extent (see Table 5).
~ rom an industrial viewpoint, this would
represent a real advantage since the present market
trends are Eor a lower demand in ethylene and an in-
creasing demand in propylene.
It will be appreciated that even though yields
increase in valuable products ranging from 5 to 10% wt
do not seem to be of significant importance, because of
the enormous amounts of hydrocarbon material refined
every day throughout the world, even a 0.5~ wt yield
3~
- 12 -
increase represents millions of dollars of profits for
petrochemical industries. Therefore, it is submitted
that every invention increasing production yields in the
petrochemical conversion processes has tremendous com-
mercial values for these industries.
It will also be understood that although the
process of the present invention has been developed for
up-grading steam produets, it can also be applied to
every situation where starting materials such as pyroly-
sis oil, pyrolysis gasoline, mixtures of light olefins,
light paraffins or mixture thereof are flown directly
into the catalytic reactor without requiring any passage
through a steam-cracking reactor.
In such cases, the multifunctional properties
of the catalyst are expressed through several actions
such as acid-catalyzed reactions (cracking, oligomer-
ization, isomerization, transmutation) and redox re-
actions on intermediates leading to the final products
or on the products themselves.
Therefore, in the framework of the present
invention, the reactor containing the multifunetional
catalyst can be located either after the steam-cracking
reactor or after the liquid/gases separation operation
~thus intercepting the liquid or gaseous products) and
still obtain similar end results.
-- 13 --
E~ ~ ~ o O O O O _ .
E~c~ O _ _
W ~0 3~ ~^ c~oo oo ,~, o
~ ~ ~P~ _ o o o o o ~ o
~ _ 't .__._ 3,3
H _ ~ _ .~
h ~ ~ ~ ~ U O ~ _
a ~ P. a m X ~ ~ U ~ z
_ = ' _ = = _ _~
U~ ~ ~ b
~ ~1 o ~ Il~ ~ ~) .E~ 8 ~ ,~ ~o .~. ~ o N u~ t~
H C: o o _ _ __ _ u
D ~ O . O~ ~ ~ ~ I` . _~ ~ O
m ~ ~ ~ ~ O~ ~ ~
~ 11 O t`') r-l H _I r l _ O
,~ .~ J- 3
~ . ~ > ~3) Z
. __ .._ . ~r;
~J~ !~ Z; +Z ~Z ~ wz ~ ~3~ o
O s ~ o ~
2 ~o ~ o ~w ~ y o ~ ~ ~ ~
. _ .E~ ~ DD ~ ~ tr
-- 14 --
-- ~ I` ~ 111 N ~ I`
O (d ~ D O O O ~i 0 ~ ~9
O N C) _
h 1~~ ~1 ~ _
0 3 rl O ~1 O~ ~r ~1 0 ~ Ul 1~ N
a~ --o " 3 a ~ ~ o o o o o i
w ~ ~ _
U~ ~ O '3~ , ~1 ~ r0, ~1 O. ~1
JJ t` dP ~ a C~
~ t~ U~ ~ ~ _ ,1::
Z ~ o3~0~ N O O O O O ~I O ~
Z W O O ::~
~C E-l _ _ . . . __
W ~ ~ .C ~ 3 ~H') 1:~
E-l m C~ P 4 . ~ ~3 ^ S~
1:~ ~ ... __ W ..... _._ ;~; C`P O
U~ H ~ t/- ~-- ~0
~0 w ~ o Cl ~zW W ~ zW z ~ ~7 ~ E ~
N ~ H 3 1'-~ CI~ E Z O
_ _ . .____ ~:
~ ~ .~17'~ CO ~ ~ CO
u~ ~ ~ o la ~! ~ ~ o ~ r i ~ ~
z ~ ~a ON _ -.-. .___ _ ~1
EHI H o 3 ~i O ~1 11~ 1~ . ~ al C~
~, ~ ~ ~ o 'a ~ 'a ~ o~7 ,1 o, N ~1 ~i ~:
H ~ . . . _. __ _ _
a ~ ~
O C) o ~ a~ ~D I~ a
H ~ N V N N 00 " ~ ~r .~
JJ O _ .4
j~0 3,a 0 ~ ~ w ~ ~ ~1O
a~ dP ~ ~ ~ J i O U~ I` O ~` ~
N .... _.. _ . _ _ .~:
m ~ ~ ~ ~ o o
~ O ~1 p . ._ ._ _ Z dP H dP h
3 ~1 7 7w ~7 ~, w z ~ w 3 _
O ~:1 ~ ~1 7 ~
U. o tr~ o E~ c~ Y ~ O E~ O g W
a ~ ~ ~ ~m C,,N~ P~ W ~ 0~
~ 15 --
Y Y ~ , _
o ~ 02 ~ ~ a~
/U 1~ 3 T ~ ~ ~D O O O ~ ~ N 111
N 111 1 _ _
~1 3~ ~1 o ,~ ~J
~3 ~ o~, ~ O O O O O _~ _
E~ S u~
U~ ~r~ V
~ .Y O ~0
~ _ ~ J . ~
t~ ~ ~ O ~ ~D
.Q C ~ Dl u~ O O o o _~ ~ u~
O O N N al _ . S
W U~ O ~- Ia O O O O O N O 0
I` dP N O O O O O t~ O S
~1_1 _ .. U~
~ ~ ~ ~ ~
E~ ~ ~ ~ S ~ 0 ~ O
o ~ ~, a w ~ O~ ~ ~ a
D ~ ul = i-
~ U~ __ l U~
~0 ~ 0 N al O U~ ~
~ H o ~ .~ a ~N a) ,~ ul ~ N , ~) . I` O N
W O X ~ ,~ _i r 1 00 0 CO 1~.--i ~1
N O N N 10 U
~ ~ ~J ~ O . ~1
O ~ O ~ In O~ U~ . , . r~7 O~ ~
~, ~ dP X ~ ,a r-- o rr''~ N ~ r-i C:
-- N t~ _ _ S
Z W h ~n
O O b` ~ u~ ~ ~ ~ .~
E~ W ~ a) ~ ~ o o
D ~_) .1 _ ~ ~ I` ~ CO ~ N 1~ G)
m w ~ ~ ~ 3 ~ ~ ~ D ~ ~ ~
U~ ~1; ~ .q ~ 111 N~( ~1 ,~~_1 a~ ~ .Q
~ W .~ _ _ . _ ~1
~ ~ a~ ~ ~I
O ~ o 3-~ O~ . . ~ _~ O
r~ ~ ~ I~
O S ~1 ~ ~I H ~ oo .;r
. _ wO
o ~ ~ ~ zO; O)
t~ C la IU ~1 ~ H
~ ~ _ ~3 j~_ ~dP 0
~ ~_ O ~P I ~ ~ ~
~ C~ D. ~ ~ ,,~ O D N ~ ~ ~ 3 ~ a
C~ m W ~ m m m u ~ ~ ~
_ _ -- -- _ _ _
4~3
-- 16 --
~ ,~ `9,~
o ~ o o ~ , J ~ ~ ,~
J- ~0 CP ~ ~ O .q N ~ ~ O O O O O r'l
) IO ~ N 111 U ¢ U
E E~ .. _ _ _
1:~ o 3 ~ ~ _ ~ N u~ I`
~ U l~ ~`I O O O O ~iC`l
~0_ _ _ .
O ~ ~u~oq ~
Y ~ ~ X ~
o 3 u~ I _ o o o o o ~_ U
~ ~ ~ ~ ~- _
Z 1'~ o 3 . ~ _ O O O O O N O
H O I~ d~ ~ O O O O O N O ~
b Q u _ _ ._ __ . _ c
O ~, ~ ~ ~ oq c~ O
El 8 ~ _ _ ~ ~
c ~ u a ~~ N ~ U ~U ~ O O U
P~ ~ U E~ U ~! E-~ ~1
Z ~ .. ____ _ = ._ ____ =: = O
O ~ CJ
~ 3 1 o ~ 0N ~ rd N ~ N a~ o o a:
~ 0 ~ N 1~I U ,e U ..
O 1~_ _ . _ . __ - _ . . _ _ N
z o ~ 30~ ~ ~ ~ u~
O ~C ^ O ~.) ~ O N CO _~ C
~ ~ 'xoU -- --- - ----- - _ C
~ ~ _~l~o ~J L~
U o ~ ~ ~ ~ ~ ~ ~ . . c~ u~ N ~ CO ~O a)
O 13 O N N ,a U 1'1 U _I _I N O ~ ~ ~--i .~ Cz Z ~a ._ ... _ . _ . .. _ _. _ c
0 1~ ~ I` I` ') ~
~o 3~ C ~ ~ ~_~ o
r~ d~ o o l~ ~
l ---- ~c ----- - ->~ z ~
~! oo ~ ~ ^ udP z Q~
1~ ~ 1 D D N ~ ~ O
-- 17 --
_ -_ _
E~ O
~0 F ~
21 _ O 3 .,~ ~ .~ ~ ~ ~ ~1 N O
O O O C~ ~1 0 ~ ~ ~ ~ O
~13 O ~ N ~
~3 ~ ~ u~3~0~ CO er ~ O
H ~ V n t~ IJ a V ~ CO CO d'
O~N _ ~
v ~ , a ~ -
= : __
0 ~1 n,
~H ~! ~0 0~3-~1EOO~' N ~ N ~ O Cn O a~ ~
n~. ul ~ _
O ~ ~ In ~ rl 0 ~ ~ In N 1~ . I U~ a~ 1~ N
~U~ C0 0~ ~ ,~ ~:r ~ I` N
~ 0 1/ ~ .U _
..~ (d S - > 3 HO Z Z
u~ ~1 u~ ~
. .____ ~ _ ~ dP P:;
V ~ ~ E ~ ~ ;q 3 ~ V