Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
1296~0~
-- 1 --
l-Hvdroxyalkylxanthines, Processes for their Preparation and
medicaments containing them
1 This invention relates to a new class of xanthines,
namely l-hydroxyalkyl xanthines, to processes for their
preparation and to a medicament containing them.
EP 39780 relates to 1,3- or 1,3,8-substituted xanthines
showing sedative and anxiolytic properties. These com-
pounds were developed with a view to enhancing or making
more specific certain therapeutically useful physiological
effects of natural xanthines, such as caffeine or theo-
phylline. Despite their undeniable neuroleptic and
anxiolytic character, the compounds according to the above-
mentioned patent show side effects, particularly on the
cardiovascular function. By contrast, the cardiostimulant
activity of these compounds, taken on its own, opened up
a new channel of investigation in that direction.
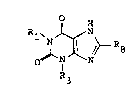
lS The present invention relates to a new class of
xanthines, namely 1-hydroxyalkyl xanthines, which show in
particular cardiovascular activity. The compoùnds according
to the invention are xanthines corresponding to the
following general formula
H
R~ XN~ (I)
o
~25 R3
in which
Rl is a C2-C5 ~-hydroxy-n-aIkyl group or a C3-C5 (~-1)-
:
, : ~ .
`-`` 1`~96i~
-- 2
1 hydroxy-n-alkyl group, R3 is a Cl-C4 alkyl group, R8 is H,
methyl or ethyl and the sum of the carbon atoms in Rl and
R3 is between 4 and 9,
or physiologically acceptable salts thereof.
S More precisely, the following compounds are more
specifically claimed:
1-(2-Hydroxyethyl)-3-propyl xanthine,
1-(2-Hydroxyethyl)-3-isobutyl xanthine,
1-(2-Hydroxyethyl)-3-isobutyl-8-methyl xanthine,
1-(2-Hydroxypropyl)-3-propyl xanthine,
1-(2-Hydroxypropyl)-3-propyl-8-methyl xanthine,
1-(2-Hydroxypropyl)-3-butyl xanthine,
1-(3-Hydroxypropyl)-3-propyl xanthine,
1-(3-Hydroxypropyl)-3-propyl-8-methyl xanthine,
1-(3-Hydroxypropyl)-3-propyl-8-ethyl xanthine,
1-(3-Hydroxypropyl)-3-butyl xanthine,
1-(3-Hydroxypropyl)-3-isobutyl xanthine,
1-(3-Hydroxypropyl)-3-isobutyl-8-methyl xanthine,
1-(3-Hydroxybutyl)-3-methyl xanthine,
1-(3-Hydroxybutyl)-3-ethyl xanthine,
: 25 1-(3-Hydroxybutyl)-3-ethyl-8-methyl xanthine,
1-(3-Hydroxybutyl)-3-propyl xanthine,
1-(3-Hydroxybutyl)-3-isobutyl xanthine,
,
~: 1-(4-Hydroxybutyl)-3-ethyl xanthine,
1-(4-Hydroxybutyl)-3-propyl xanthine,
1-(4-Hydroxybutyl)-3-propyl-8-methyl xanthine,
1-(4-Hydroxybutyl)-3-butyl xanthine,
1-(4-Hydroxybutyl)-3-isobutyl-8-methyl xanthine,
~ 35 1-(4-Hydroxypentyl)-3-methyl xanthine
: ~ -
1~96~1
1 1-(4-Hydroxypentyl)-3-propyl xanthine,
1-(5-Hydroxypentyl)-3-methyl xanthine,
1-(5-Hydroxypentyl-3-propyl xanthine et
1-(5-Hydroxypentyl)-3-propyl-8-methyl xanthine.
Physiologically acceptable salts of the compounds
corresponding to general formula (I) are understood to be
the salts which these compounds form with pharmaceutically
acceptable bases. The salts in question are salts of
which the cations are harmless with respect to animal
organisms and do not produce any side effects in thera-
peutic doses. Salts such as these include the salts of
alkali metals, such as sodium, potassium, pharmaceutically
acceptable salts of ammonium and amines known to the expert.
These salts are prepared by heating the compound corres-
ponding to general formula (I) in the presence of the
appropriate base and in the presence or ~sence of a solvent,
preferably followed by recrystallization.
The compounds according to the invention are prepared
by one of the following processes:
A) A uracil corresponding to the following formula
H~N ~ NH- Il_R8
NH2 (II)
: R3
: 30
is reacted with an alkylating agent of the formula Rl-X,
where Rl, R3 and R8 are as defined in general formula (I),
except that R1 is not ~-hydroxybutyl, and where X is a
halogen atom, preferably bromine, or monosulfate or disulfate
~ 35 or p-toluenesulfonate,
'~
~'
- . ~ - .
- . .
:, ~
``` 129~
1 in a solvent suitable for all the reactants, such as for
example dimethylformamide (DMF), dimethylsulfoxide (DMSO) or
hexamethylphosphorotriamide (HMPT), at a temperature in the
range from 20 to 40C and in the presence of an alkali metal
hydroxide, for example sodium hydroxide in solid form. The
reaction is preferably carried out in DMF at 20C in
accordance with the following scheme:
H ~ ~ NH-C-R8 Rl ~ NH-C-R8
NH2 ~
R3 R3
15- The product corresponding to general formula (III) is then
cyclized at 20 to 100C in an alkali metal hydroxide solution
in accordance with the following reaction scheme:
1 ~N ~ NH-C-R8 Rl ~ ~ N
~ o~ N ~ N ~ (I)
: R3 R3
Although it is possible to isolate the derivative of
general formula (III), it is preferred to carry out cycli~-
ation directly without isolating or purifying the derivative
in question. To this end, the reaction medium is neutral-
- ized and the solvent evaporated, after which the residue is
dissolved in an alkali hydroxide solution and the resulting
~ solùtion heated to reflux temperature.
:
B) In a variant of this process, the uracil(II) is
alkylated with agent containing a function which may be
~: 35 converted into a hydroxyl group, for example a -COO-R4 function,
:
~. . - .
1~96~` 0~
1 in accordance with the following reaction scheme:
O O
H~ ~ NH-C-R8
o~N NH2 2 y 4
O O
> R4~00C~(cH2)y~ N ~ NH-C-R8
~ N ~ (IV)
I NH2
R3
in which R3, R8 and X are as defined above, y is a number of
from 1 to 4 and R4 is an alkyl group. This alkylation
reaction is carried out in the same way as for A) in a
solvent suitable for all the reactants, such as DMF, DMS0
or HNPT, at a temperature of from 20 to 40C and in the
presence of an alkali metal hydroxide in solid form. The
alkylation step is followed by cyclization and simultaneous
hydrolysis of the ester (IV) at 20 to 100C in an alkali
metal hydroxide solution in accordance with the following
reaction scheme:
O
R4-ooC-(cH2)y N ~ NH C0~ R
~N
: ~ 1 2
HO0C-(cH2)y~N ~ N ~ ~ (V)
R3
, ~:
Although the acid (V) may be directly reduced to alcohol,
`` lZ96f~
-- 6 --
l it is preferred to re-esterify the acid (V) with an alcohol,
such as methanol, ethanol or propanol, under reflux to
obtain the corresponding ester which is then reduced in
known manner, for example in the presence of LiAlH4 in
THF, to obtain the followinq product according to the
invention:
~O-C~2-(CN2)~ R8 (I)
Accordingly, it can be seen that ~-hydroxyalkyl
xanthines are always obtained in this second embodiment of
the process according to the invention.
C) In a final embodiment of the process according to the
invention, (~-l)-hydroxyalkyl xanthines are prepared by
alkylation of the uracil (II)
H ~ ~ NH-~-R8 (II)
O ~ N NH2
R3
with an alkylating agent of the formula R5X, where R3, R8`
and X are as defined above and R5 is a C3-C5 (~ alkenyl
group. The alkylation reaction is carried out under the
same conditions as for A) and B), namely in a solvent
suitable for all the reactants, such as DMF, DMSO or HMPT,
` at a temperature in the range from 20 to 40C and in the
presence of~an alkali metal hydroxide in solid form. The
intermediate product obtained is then cyclized at 20 to
100C in an alkali metal hydroxide solution in accordance
with the following reaction scheme:
Rs~ ~ NH C R8
o ~ Nl NH2 ~ N N
R3 3
The final step comprises hydration of the double bond of
R5 by an addition reaction of the Markownikoff type. This
addition may be carried out in two ways:
Method A: using dilute sulfuric acid at a temperature of
approximately 100C over a period of several days.
Method B: by the reductive oxymercuration-demercuration
method in the presence of mercury acetate and then NaBH4
(Larock, R.C.: Solvomercuration/Demercuration Reactions in
Organic Synthesis, Springer Verlag, Berlin, 1986, Chap. 2).
The present invention also relates to a pharmaceutical
composition containing a compound of general formula (I)
in combination with an inert, pharmaceutically acceptable
support.
The medicament according to the invention may be made
up in various pharmaceutical forms containing the usual
excipients or vehicles, such as tablets, capsules, supposi-
tories, solutions, suspensions, and may be administeredorally, sublingually, rectally, subcutaneously, intra-
muscularly, intraveneously or by inhalation.
Before actual examples of the synthesis of the
compounds according to the invention are given, Table I
below shows the method of preparation and the alkylating
agent to be used for 27 compounds according to the
; invention. Table II shows the melting point and recrystal-
Iization solvent for these same 27 compounds and Table III the
3C NMR values.
.
12~6~ nl
-- 8 --
W ~ I- O U~ ~ w ~ ~- o o
~g
&)
~ r ~ ~ ~ ~ w w w w w w w w w w w ~v ~ ~ ~ ~ î~
g ~ g g ~ g ~ - ~
_ _ _ ~a
- - - - - l l l l l l l l l l - - - - - - - - - l l l
l l l l l wwwww wwwww l l l l l l l l l w~w ~
wwwww l l l l l l l l l l wwwwww www l l l
~Ç ~ X~ ~
X ~ X I X I ~ ~
X~D
w ~ g
g ~ ~
~ n W W ~ :q W ~ ~ ~ ~ ~W w w w w wD~ ~ ~ ~ ~
w ~ w w w w w ~ ~ w ~ w w w
o ~ ~ ~ ~ o ~ o ~ o ~D
~t ~ t ~ ~t ~ ~ ~ ~ ~J tD ~ ~ tD (D ~ ~ ~ r~l ~ ~ ~ 1_
D D ~J tD n ~ 3 ~ ID S~ 1 ID iD D ::1
O ~ ~ o o ~ 30
~ lD lD ~) iD
~ ~29~'`(31
Table II
Xanthine l~o. Mp. (C) Recrystallization solvent
1 178-179 Acetone
2 200-202 Acetone
3 227-229 Chloroform
4 150-152 Chloroform
196-197 Methanol
6 157-158 Water
7 145-146 Methanol
8 221-222 Methanol.
9 196-198 Water
111-112 Water
11 159-160 Acetone
12 248-250 Ethanol
13 200-201 Acetone
14 217-218 Water
212-213 Water
16 163-164 Acetone
17 141-142 Acetone
18 207-208 Water
19 173-174 Acetone
192-193 Water
: 21 122-123 Acetone
22 211-212 Methanol
23 180-181 Acetone
24 153-154 Acetone
: 25 190-191 Water
26 151-152 Water
~: : 27 184-185 Dioxane
::
~: - :
~: :
.,
1296COl
-- 10 --
W ~ ~-- O ~ Ul ~ W ~> 1-- 0 a~ ul ~ W N 1-- i~
~ tD
tD H
~ H
OOO OO OOOOO OOOOO oOOOOO~ n
W CO ~ ~ o o o o O ~ E~ ~z
o ~ ~9 ~ ~ ~D ~ O~ W CO O ~ O~ 3
ooo oo ooooo ooooo oooooo ~ ~ .,
~
~ ,n ,p o a~ P ~ W W ~ ~n o w a~ Ul ~n
w ~ r` ~ ~ w ~ w ~ ~ ~ $~
~I ~ ~ ~ r~ o~ w co w o ~ w ~ w w ~ i--w ~ 0 CJ~ P .P ~
o o o o o o o ~P O O O O ul w ~ Ul ~.n ,P ~`~
~n ~ W ~ .P O ~n ~ Vl O ~ ~ ~ o ~ ul ~ ~n Ul ,~ ,p ~r~ W W ~ ~.
o
,~ ,
o o o o o o o o o o ~ co co co c~
n Vl ~ W ~ W ~ ~ ,~ W W ~ i~ W V
W W ~ W W . . . . W W W ~~ ¢
N W W t~ W W W h~ ~ o
~ ~ ~ a~ ~ ~o o o o u~ ~ ~ ~ ~ ~ OD 00 Ot~ ;~ X a:~ O O O S~ I
... .. ---- --...... .... --. ~ ~.
~IV ~OOO~1 WJ~WWW O~ ~
w ~ w a~ ~ ~ ~ tv ~ ~.
N 1~ n Ul O O O O O W W W W W
' o o ~ 3
J~ ~ W ~ ~ W W ~ ~ ~ .P ~ ~ .P ,p ,p~ ,
w ~ cr~ P ~ ~ 0~ w ~ ~ ao ~ o o ~ u~ w ~ ~ 0~ ~p ,r CD ~ , W C
O O O C~ ~D O O ~) a~ o w w ~ o~ ~ o ~ o o a~ a~ o .-
O O O ~ ~D O O ~D O ~ D O O O c> o o ~ ~ o .
X W , X X X W W X X ~
:: ~ :
C~
rt
.~
: O
n
~ :
~ .
. ~ .. . ...
1296~1
1 The invention is illustrated by the following
Examples in which the quantities are by weight, unless
otherwise indicated.
EXAMPLE 1
1-(5-hYdroxyPentYl)-3-Propyl-8-methyl xanthine
(Method A: xanthine no. 27)
22.6 g (0.1 mole) of 1-propyl-5-acetylamino-6-amino-
uracil (II) ~R3 = methyl, R8 = methyl) are suspended under
nitrogen in 200 ml of anhydrous dimethylformamide (DMF).
21.7 g (0.13 mole) of 5 bromo-1-pentanol are then added,
after which 6 g (0.15 mole) of powder-form solid NaOH are
added with stirring in portions of 1 g at a time at intervals
of 1 hour. After the third addition, the suspension is
completely dissolved. The solution is left standing over-
night to complete the reaction. The solvent is evaporated
in a rotary evaporator at 40C/0.1 mm Hg. The oily
residue, which is the uracil (III) (Rl = 5-hydroxvpentyl,
R3 = propyl, R8 = methyl), which is not purified, is dissolved
in 100 ml of 10% NaOH and the solution heated under reflux
for 2 hours. The solution is then cooled in a ice bath
and neutralized to pH 5 with acetic acid. The product
precipitated is filtered, washed with cold water and then
dried in a vacuum oven. The dried product is dissolved in
500 ml of ethanol and the solution is decoloured overnight
with active carbon. The product is precipitated by
addition of 1 litre of water, filtered and the treatment
with active carbon repeated. The dried product is
recrystallized twice with 120 ml of dioxane. Yield: 23.5 g
(80%); colourless crystals; Mp. 184 - 185C.
EXAMPLE 2
1-(4-hydroxYbutYl)-3-butyl xanthine
(Method B, xanthine no. 21)
11.3 g (0.05 mole) of 1-butyl-5-formylamino-6-amino-
. .~ .
,, .~ ,.. . . . .
12~6~
- 12 -
1 uracil (II) (Rl = butyl, R8 = H) are dissolved under
ni1:rogen in 200 ml of DMF. 10.6 ml (0.075 mole) of
ethyl 4-bromobutyrate are added, after which 2 g (0.05 mole)
of solid powder-form NaOH is added with thorough stirring
in portions of 0.5 g at intervals of 1 hour. On completion
of the addition, the mixture is left to react overnight.
The solvent is then evaporated and the oily residue of
~IV) (substituent in the l-position - (CH2)3COOEt) is
dissolved in 100 ml of 10~ NaOH. This solution is then
heated under reflux for 0.5 hour and then cooled, neutralized
to pH 5 with acetic acid and filtered. The precipitate
formed is dried. Yield: 9.2 g (60~) of crude xanthine (V~
(substituent in the l-position = (CH2)3COOH, R3 = butyl,
R8 = H).
Without purification, the product is esterified under
reflux for 5 hours with 200 ml of methanol containing 1 ml
of concentrated sulfuric acid. The solutlon is concentrated
to a small volume and 50 ml of water are added. The ester
precipitate formed (substituent in the l-position = (CH2)3-
COOMe) is filtered, washed and dried. Yield: 9 g (93~).
Without purification, this product is dissolved in
200 ml of anhydrous tetrahydrofuran (THF), cooled to between
0 and -I0C and added dropwise to a solution containing
2.13 g (0.056 mole) of LiAlH4 in 150 ml of anhydrous THF.
On completion of the addition, the mixture is left to react
for another 2 hours at 0C, after which the excess LiAlH4`
is destroyed by addition of 50 ml of water. The mixture
`~ is acidified to pH 2 with concentrated HCl, after which the
THF is evaporated. The residual aqueous solution is
continuously extracted overnight with dichloromethane.
The organic extract is dried and the solvent evaporated.
The slightly coloured solid residue (6.1 g) is recrystallized
twice from acetone. Yield: 4.6 g (33% from II); colour-
less crystals; Mp. 122 - 123C.
.,~ .. , : .
12Y 6~
- 13 -
1 EXAMPLE 3
1-(3-hYdroxYbutYl)-3-isobutYl xanthine
(~ethod C/Variant A, xanthine no. 17)
11.3 g (0.05 mole) of 1-isobutyl-5-formylamino-6-amino-
uracil (II) (R3 = isobutyl, R8 = H) are dissolved in 220 mlof DMF. 8.8 g (0.065 mole) of 4-bromo-1-butene are added,
after which 3 g (0.075 mole) of powder-form NaOH are added
with stirring in portions of 0.5 g at intervals of 1 hour.
The mixture is left standing overnight, after which the
solvent is evaporated in vacuo. The crude residue (R5 =
3-butenyl) is taken up in 100 ml of 10% NaOH and heated
under reflux for 0.5 hour. The solution is then cooled
and neutralized to pH 5 with acetic acid, after which the
precipitate formed is filtered and dried. 8.5 g of crude
solid product (VI) (R5 = 3-butenyl, R3 = isobutyl, R8 = H)
are obtained. This product i purified by passage through
a column of silica (85 g) using chloroform as eluent:
6.5 g of colourless product.
A mixture containing 6 g of the above compound in
100 ml of 20% sulfuric acid is heated at 100C for 4 days,
cooled and neutralized to pH 5 with 50~ KOH. The solution
is evaporated to dryness, the residue is taken up in 100 ml
of boiling absolute ethanol and the insoluble fraction is
filtered. The filtrate is freed from the solvent, leaving
a coloured solid residue (6.4 g) which is passed through
a column of silica (650 g) eluted with a gradient of
chloroform/methanol (0-5~ methanol). The xanthine is re-
crystallized twice from acetone. Yield: 4.5 g (33~ based
on II); Mp. = 141 - 142C.
; EXAMPLE 4
(2-hvdroxYPropvl)-3-butyl xanthine
(Method C:Variant B, xanthine no. 6)
16 g (0.050 mole) of mercury (II) acetate are dissolved
3S in 250 ml water. 12.4 g (0.05 mole) of 1-allyl-3-butyl
.,j,~.~, ....... . .
lZ~6~ 0~
- 14 -
1 x,anthine (prepared as described above from 1-butyl-5-formyl-
amino-6-aminouracil and allyl bromide) dissolved in 250 ml
of THF are added dropwise with stirring over a period of
10 minutes. After a few minutes, a precipitate is formed.
Stirrin~ is continued for 30 minutes, after which the
mixture is cooled in an ice bath. 68 ml of 3 N NaOH are
added, followed by the dropwise addition of 60 ml of a
freshly prepared 0.5 M solution of NaBH4 in 3 N NaOH. On
completion of the addition, stirring is continued for
another 15 minutes. ~he solution is neutralized to pH 4-5
with 6 N HCl (approximately 60 ml), saturated with NaCl and
the xanthine extracted with dichloromethane (3 x 200 ml).
The organic extract is dried and the solvent is evaporated:
12.9 g of crude product containing 20% of starting product.
The products are separated by passing the mixture through a
column of silica using chloroform as eluent for the starting
product and chloroform/methanol (g5/5) for the hydroxylated
xanthine (9.5 g). The xanthine is recrystallized twice
from water: 8.5 g (64~); Mp. 157 - 158C.
EXAMPLE 5
.
The compounds (I) according to the invention have been
found to show low toxicity and have little or no effect on
the central nervous system. By contrast, they were found
to be active in the in vitro tests for inotropic (cardiotonic)
activity. Table IV shows the results of these tests.
1296~'J1
- 15 -
Table (IV)
Xanthine No. Cardiotonic activity1) (minimum effec-
tive concentration in ~g/ml producing
a sigr.ificant response)
2 2.5
3 2
4 3.3
0.6
6 ; 4.2
7 1.3
8 i.2
9 2
0.4
11 0.7
12 0.5
13 50
14 0.8
0.6
16 0.1
17 1.3
18 3.3
19 0.4
0 7
21 2
: 22 25
: :23 . 33.3
24 0.7
; 25 6.7
26 0.2
~ 27 0-3
,~,...~:..,,. ,..,,, :,
,
12~6G (~1
- 16 -
References
Theophylline 15
Amrinone2) 100
Milrinone ) 0.5
IBMX3) 0 5
1) Measurement of the force of contraction of the isolated
and electrically stimulated left auricle of a guinea
plg in accordance with Erjavek, F. and Adamic, S., Arch.
Int. Pharmacodyn. 155: 251, 1965. A response is consi-
dered to be significant when the basic contraction force
is increased by more than 40%.
2) Prior art cardio stimulant medicament.
3) 1-methyl-3-isobutyixanthine.
~,
.