Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
CA 02278965 1999-09-22
-2-
TECHNICAL FIELD
The present invention relates to mineral recovery processes and particularly
but not
only flotation of valuable minerals which use oxygen as a conditioning and/or
flotation
gas.
BACKGROUND TO THE INVENTION
The use of flotation processes to recover valuable minerals is well known in
the
art. The control and optimisation of these processes, however, can sometimes
be a "hit
and miss" affair.
A mineral recovery process such as froth flotation which may work extremely
well
in one geographic location and with one particular type of ore but may be
entirely
unsuitable in another location due to the different reactivities of the ores.
Further, even at one location and one ore body the reactivity of the ore may
change
on an hourly, daily or weekly basis. There is significant variability in the
characteristics
of the ore processed by flotation at any particular mine. Changes are
unpredictable and
are caused by: ore bodies that are not homogenous, mining practices,
stockpiling,
crushing, and milling conditions. Even slight changes in the ore's
characteristics can
upset the delicate balance in the flotation cells and have a substantial
negative impact on
the recovery of the valuable sulphide mineral.
Researchers have established that most sulphide minerals require some oxygen
for
complete flotation. There are three collector reaction mechanisms generally
accepted for
xanthate type collectors (the most common collector type used) that lead to
making
valuable minerals floatable:
Electrostatic collector attraction
MeS + H20 + 202 = Me(H20)2+ + S042-
The ionically charged collector attracts to the mineral surface
Chemisorption
1) MeS + 202 = MeSO4
2) MeSO4 + C032- = MeCO3 + SO42-
3) MeCO3 + 2X- = MeX2 + C03 2-
Metal xanthate layer builds up on mineral surface.
Electrochemical oxidation
CA 02278965 1999-09-22
-3-
1) 2X- + 1/2 OZ + H20 = X2 + 2OH-
2) MeS + X2 = (MeS)x2ads
The collector is initially oxidised and then attaches to the mineral surface.
Oxygen is required for each of the collector mechanisms recognised. Oxygen is
the principal electron acceptor. Oxidising conditions favour reaction with
xanthate type
collectors including dithiophosphates. Thiocarbamate and thiourea collector
actions also
require oxygen for complete flotation.
Many flotation pulps are oxygen deficient. Milling produces a reducing
environment. Grinding media and minerals corrode. Oxygen in the pulp is
consumed.
1o Some minerals present in the ore can be significant oxygen consumers eg
pyrrhotite,
marcasite, pyrite.
Valuable sulphide minerals are also prone to over oxidation that can reduce
flotability. Previously, where an oxygen deficiency has been recognised air
was used as
the oxidation gas. Due to its low dissolved oxygen saturation point (5-8 ppm),
the
danger of over oxidation was minimal. However, the applicant has discovered
that the
intensity of oxidative conditioning in the full scale application can usually
only be
achieved by using an oxygen-rich gas which has a higher saturation point. This
in turn
may lead to over oxidation over the valuable mineral, if the addition of the
oxidation gas
is not adequately controlled.
In the flotation of sulphide ores it has been found that the oxidising
environment in
the pulp, as measured by oxidation - redox potential (or Eh) has a strong
influence on the
flotation result. Eh has frequently been measured but not often used as a
control
parameter. The reasons for this include unreliable electrodes, changes in Eh
being more
attributable to changes in pH, and the difficulty in controlling Eh by some
reagent
addition.
Dissolved oxygen (DO) concentration in the pulp has also been tried as an
indicator of the status of the flotation process. Air has been occasionally
used as an
aeration gas to raise the dissolved oxygen concentration of the slurry.
Measurement of
dissolved oxygen concentration though does not give sufficient information on
whether
ore oxygen gas flow requirements have been optimised.
CA 02278965 1999-09-22
-4-
The applicant has found that the oxidative conditioning step in the ACTIFLOAT
process, which is subject of Australian patent no 670,163 and application no
37917/95
and several overseas patents and co-pending applications, provides substantial
improvements over conventional flotation methods. There is no doubt that with
many
ores an oxidative conditioning step conducted prior to or simultaneously with
the
flotation step increases the recovery of the valuable mineral over
conventional treatment.
Current and potential users of the process, however, have indicated that it
would be
useful to have in place methods and equipment to rapidly and accurately
determine the
oxygen gas flow requirements of the slurry being processed, fluctuations in
the make up
of the ore will change the oxygen gas flow requirements. Optimising oxygen
addition is
an important component of the overall efficiency of the ACTIFLOAT process.
Equally, some ores may not be susceptible to oxidative conditioning or
alternativeiy such conditioning may in fact be detrimental to mineral
recovery. In such
instances it would be beneficial to determine whether the oxygen gas flow
requirements
of such ores are negligible and thereby characterise the flotability of the
slurry.
The present invention seeks to provide a method for controlling a mineral
recovery
process which overcomes at least some of the disadvantages of the prior art or
provides a
commercial alternative thereto.
STATEMENT OF INVENTION
In a broad aspect, the present invention provides a method of optimising a
mineral
flotation recovery process comprising extracting a representative sample of a
slurry
containing the mineral to be recovered, treating the sample with an oxidising
gas,
measuring one or more parameters before and/or after said oxidative treatment
wherein
the change in said parameter(s) is indicative of the flotability of the
minerals contained
in the slurry, characterising the slurry as a function of said measured
parameter(s), and
controlling the mineral recovery process in accordance with said slurry
characteristic.
The present applicant has found that the inventive method is particularly
suitable
for optimising the ACTIFLOAT process ie a process which has oxidative
conditioning
of the slurry. It will be recognised, however, that the inventive method is
also suitable
for other flotation processes such as MAXIFLOAT and CLEANFLOAT both of which
use non-oxidising gases to condition or float the desired minerals.
CA 02278965 1999-09-22
-5-
The present invention provides a mechanism for optimising a mineral flotation
recovery process in several ways, namely:
a) characterising a slurry by providing a measure of the flotability of the
minerals contained thereilrafter an oxidative gas treatment,
b) determining the effect of various control regimes on the slurry and indeed
the
entire mineral recovery process eg what effect do different dissolved oxygen,
pH, electrochemical potential levels, different mixing times, different
intensities of mixing etc have on the flotation recovery of the valuable
minerals, and
c) providing an historic record of the correlation between the effect of
different
parameter alterations, ore types etc and the flotability of the minerals
contained within the slurry thereby allowing an operator to predict what
control parameters are required to optimise mineral recovery..
In another broad aspect, the present invention provides an apparatus for
optimising
a mineral flotation recovery process comprising means for extracting a
representative
sample of the slurry, means for treating the sample with the oxidising gas,
means for
measuring one or more parameters before and/or after said oxidative treatment
wherein
the change in said parameter(s) is indicative of the flotability of minerals
contained in
the slurry, and means to determine a slurry flotability characteristic as a
function of said
measured parameter(s), said apparatus being operatively linked with said
mineral
flotation recovery process to thereby control said mineral flotation recovery
process in
accordance with said slurry characteristic.
The parameters to be measured may be selected from any one of dissolved oxygen
concentration, electrochemical potential, pH, temperature, chemical species in
solution,
mineral content, mineral surface composition and mineral surface properties.
It will be
appreciated by persons skilled in the art that the change in these parameters
is indicative
of the flotability of the minerals contained within the slurry.
In accordance with the invention, as many parameters of the slurry as are
required
are measured to provide a reliable characterisation of the flotability of the
contained
minerals in the slurry. By the phrase "indicative of the flotability of
minerals contained
in the slurry" we mean indicative of the flotability of minerals in the slurry
per se, but
CA 02278965 1999-09-22
-6-
also indicative of the flotability of the minerals in slurry when subjected to
the respective
mineral recovery process. A mineral slurry may be perfectly flotable in one
process yet
difficult to float in another process where the changes in process parameters
are quite
minor.
The list of parameters mentioned above is not meant to be exhaustive. One of
the
advantages of the present application is that the type and number of
parameters to be
measured may be tailored by an individual operator to suit the particular
mineral
recovery process. For instance, if consistent water supply is not reliable and
water must
be obtained from different sources, it may also provide helpful to measure
various
1 o parameters of the slurry water to determine its effect on the flotability
of the slurry.
Another advantage of the present invention is its ability to apply a series of
experiments to the representative sample under conditions which do not
replicate the
mineral recovery process but which serve to optimise the mineral recovery
process. To
explain, in an embodiment where the mineral recovery process includes an
oxidative
conditioning step, it may be determined that the oxidative gas treatment
applied to the
representative sample should replicate the oxidative conditioning step in the
mineral
recovery process. Alternatively, a series of experiments may be applied to the
representative sample under conditions which do not replicate the oxidative
conditioning
step. These experiments may include different oxidising gas types, different
mixing
times, different intensities of mixing and different temperatures.
In some mineral flotation recovery processes, little or no oxidative
conditioning is
required or applied. The inventive method and apparatus, however, is still
useful in
characterising the slurry. The tests applied to the representative sample may
be used to
confirm whether or not the mineral recovery process may be optimised by
including an
oxidative conditioning step.
In this way, an operator can experiment with the current slurry passing
through the
mineral recovery process without interrupting or upsetting the mineral
recovery process
itself. It will be appreciated, that this is a very useful mechanism for
applying alternative
process parameters to optimise the mineral recovery process.
The method and apparatus has been discussed above as being used to optimise a
mineral recovery process. It will be appreciated, however, that the method and
apparatus
CA 02278965 1999-09-22
-7-
may equally be used simply to characterise a slurry by determining the
flotability of its
contained minerals. The characterisation of the slurry can be used for a
number of
purposes other than optimisation of the mineral recovery process. For example,
it is
useful to determine which ore is more-easily flotable or more compatible with
current
process equipment and limitations attached thereto. An example may be where it
is
intended to change the ore body from which the slurry originates. Clearly
there would
be significant advantages in being able to test several alternative ore bodies
to determine
their compatibility with current process equipment.
In a third aspect, the present invention provides a method of characterising
the
l0 flotability of a slurry in a mineral flotation recovery process comprising
extracting a
representative sample of the slurry to be floated, treating the sample with an
oxidising
gas, measuring one or more parameters before and/or after said oxidative
treatment
wherein the change in said parameter(s) is indicative of a flotability of the
minerals
contained in the slurry, and characterising the slurry as a function of said
measured
parameter(s).
In a fourth aspect, the present invention provides an apparatus for
characterising
the flotability of a slurry in a mineral recovery process comprising a slurry
feed to
extract and provide a representative sample of the slurry to be floated, an
oxidising gas
supply to contact and treat the sample with an oxidising gas, an analysis
means to
measure one or more parameters both before and/or after said oxidative
treatment
wherein the change in said parameter(s) is indicative of the flotability of
the minerals
contained in the slurry, and a calculation means to determine the flotability
characteristic
of the slurry as a function of said measured parameter(s).
Unless the context clearly requires otherwise, throughout the description and
the
claims, the words `comprise', `comprising', and the like are to be construed
in an
inclusive as opposed to an exclusive or exhaustive sense; that is to say, in
the sense of
"including, but not limited to".
BRIEF DESCRIPTION OF THE DRAWINGS
The present invention will now be described by way of example only with
reference to the accompanying drawings in which:
CA 02278965 1999-09-22
-g-
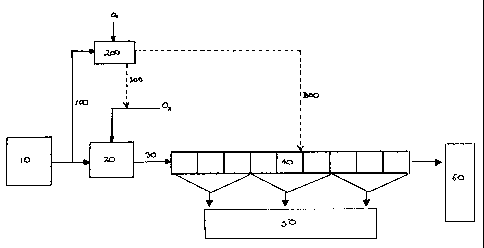
Figure 1 is a flow diagram of a mineral recovery process employing an
optimisation method in accordance with a first embodiment of the present
invention.
Figure 2 is a diagram of an optimisation apparatus in accordance with a second
embodiment of the present invention, and
Figure 3 is a graph of the output of the analysis equipment of"figure 2.
BEST MODE(S) FOR CARRYING OUT THE INVENTION
The present invention will now be described with reference to figure 1 and
with
particular reference to the ACTIFLOAT mineral recovery process. It should be
understood, however, that the present invention is suitable for other mineral
recovery
processes.
Referring to figure 1, in the ACTIFLOAT process a slurry or pulp is prepared
by
milling the ore in a liquid eg water until it reaches the desired particle
size. This feed
slurry 10 is then passed to a conditioning step 20 where the slurry is mixed
with an
oxidising gas eg oxygen or ozone and optionally other reagents, collectors,
frothers etc.
The conditioned slurry 30 is then fed to the flotation circuit 40 and the
valuable minerals
recovered as a concentrate 50. The remainder of the slurry is rejected as
tails 60.
As will be clear to persons skilled in the art, the oxidative conditioning
step 20 acts
to "prime" the slurry before the flotation circuit. In the flotation of
sulphide ores, it has
been found that the oxidising environment in the slurry, as measured by redox
potential
and/or dissolved oxygen concentration, has a strong influence on the flotation
result.
The presence of the oxidising gas is suspected to activate the surface of the
sulphide
mineral in the slurry to thereby make it more susceptible to bonding with a
collector. It
is important, however, that the duration of the oxidative conditioning is
controlled to
provide sufficient oxidation while still avoiding over-oxidation which may
create
difficulties in terms of less efficient collector usage in at least two ways,
namely that the
collector itself may be destroyed or the mineral surface may be rendered less
susceptible
to bonding with the collector than would ordinarily be the case.
Accordingly, the present invention seeks to optimise the mineral recovery
process
by characterising the slurry and in accordance with the results of such
analysis
controlling the tlotation process in particular the conditioning step to
optimise the
flotability of the minerals contained in the slurry.
CA 02278965 1999-09-22
-9-
In the embodiment shown, a sample stream 100 is directed toward analysis
equipment 200. In this analysis equipment 200, the sample slurry undergoes an
oxidative treatment whereby it is mixed with an oxidising gas. One or more
parameters
eg DO levels, pH, Eh, temperature are measured before and after mixing with
oxygen
and the change in this parameter determined. From the change in these values
an
indication of the flotability of the contained minerals is provided and the
slurry may then
be characterised as a function of these measured parameters. Once this has
been
determined the control unit, via control lines 300, can optimise the process
conditions for
the mineral recovery process as a whole and particularly the oxidative
conditioning step
1o 20. The analysis equipment 200 may be linked to any of the control elements
in the
mineral recovery process or in the oxidative conditioning step 20, such as the
oxygen
modulating valve and/or slurry feed pump, via a single control loop or a pic.
This feed
forward control will reduce large lag errors between the measurement of the
controlled
variable and the effect of the control action.
Conventional bench scale testing may establish that for a particular ore, a
dissolved
oxygen level of between 10 and 40 ppm and a conditioning time of 1 to 8
minutes is
required for maximum recovery of the valuable mineral. In the full scale
plant, however,
the ore and slurry characteristics are continually changing for reasons
previously
mentioned and therefore the optimum settings for mineral recovery are also
changing.
With the inventive method a technique is provided whereby the analysis
equipment
200 can continuously or intermittently determine the optimum mineral recovery
process
including requirements for oxygen gas flow and other additives eg collectors,
frothers,
non-oxidising gases and then put into effect the most appropriate process
control
including oxidative conditioning regime to meet this target and thereby obtain
maximum
recovery of the valuable mineral prior to the slurry entering the flotation
circuit. This
optimisation of the recovery process can of course be accomplished in many
ways eg
control of oxygen flow, different intensities or times of mixing, using
different oxidising
or flotation gases, dosage with other components eg ozone instead of oxygen.
These various alterations may also be tested on the sample slurry by the
analysis
equipment 200 after it has determined for example the oxygen gas flow
requirements to
ascertain which is the most efficient for meeting these oxygen gas flow
requirements
CA 02278965 1999-09-22
-10-
prior to entry into the flotation circuit. The analysis equipment may not only
measure
the effect of different DO levels on the mineral recovery process but also the
differences
in intensity of mixing, different mixing times, different oxidising gases and
different
concentrations of oxidising gases on the oxidative treatment of the slurry and
accordingly the mineral recovery process overall.
Each of the parameters may be measured separately or in'conjunction with each
other to determine what combination of control features optimises the
oxidative
conditioning step in the mineral recovery process and recovery of the valuable
mineral.
It is also possible to include the various equipment constraints in any
experiments
conducted by the analysis equipment 200. Generally, there will be various
constraints
on the variability of the oxidative conditioning step 20 in the mineral
recovery process.
It may only be possible to increase power by say 10% or, for example, increase
the
quantity of oxidising gas to the conditioning step by 10-15%. The analysis
equipment
can include these equipment constraints so that it conducts its own oxidative
treatment
step on the sample slurry within these equipment constraints thereby
optimising the
mineral recovery process within the constraints of the process overall.
It is also possible to take samples of the slurry at different points in the
mineral
recovery process. For example, an operator may determine that the samples
should be
taken upstream of the conditioning step, during the conditioning step and/or
downstream
of the conditioning step and correlate these samples with the mineral recovery
process
itself.
Still further, the analysis equipment may include a recordal function for
storing the
results of its own oxidative treatment on the sample slurry but also the
effect of altering
various process parameters in the mineral recovery process and how its own
sample test
data correlate to the historical data of the mineral recovery process.
Figure 2 provides a simplified diagram of analysis equipment 200. In this
example, feed slurry 10 enters analysis equipment 200 via sample stream 100. A
metering pump 110 measures the quantity of slurry entering the equipment. In
this
example, the equipment comprises two tanks 210, 220 with a metering pump 230
therebetween. Metering pump 110 provides a continuous or intermittent stream
of slurry
to first tank 210. Oxygen 400 is supplied to the first tank to provide a
dissolved oxygen
CA 02278965 1999-09-22
-11-
concentration of between 12 and 20 ppm. The outlet port of the first tank
carries slurry
via metering pump 230 to the second tank 220. Both tanks are agitated via
mixers 250
to ensure uniform oxygen concentration throughout the slurry. The dissolved
oxygen
concentrations are measured via DO meter 500. Volumetric flow rates are
measured via
the metering pumps. Preferably the dissolved oxygen concentration is measured
at both
the inlet and exit port of the second tank 220.
This analysis equipment can either intermittently or continuously provide a
reading
of the oxygen gas requirements of the slurry as follows. The time course of
dissolved
oxygen concentration within second tank 220 is dependent upon the ore/oxygen
reactiveness ie the rate of oxygen uptake by the slurry, and the dwell time of
the slurry
within tank 220. Expressed formally, the differential equation for the oxygen
concentration "C" within second tank 220 is
V. dC
= Q.(Cin - Cout) - k.C (1)
In equation (1) V represents the tank volume, Q is the slurry flow rate and k
is the
slurry oxygen consumption characteristic. A solution to this equation leads to
the
following expression for the rate constant (3 of oxygen consumption within the
slurry
k = _ (Cin - Cout) V - ~ T.Cout (2)
In equation (2), T is the residence time for the slurry within second tank
220,
which is given by V/Q. The dissolved oxygen levels C;,, and Co,,t are obtained
by using
the oxygen probes shown respectively at the input and exit of the second tank,
as
described earlier.
The rate constant (3 for oxygen consumption of slurry is obtained by using
steady
state measurements of the parameters Q, C,,, and C. This method allows precise
measurements of P. An error analysis of equation (2) yields a fractional error
in B
A,8 - AT + 1 ~OCin - ACoutI (3)
~3 T 1- Cout Cin Cout
( Cin )
A principal error source arises from the measurement of the dissolved oxygen
at
the input and exit of the second tank 220. Further error is introduced in the
measurement
of the slurry volumetric flow rate. Using equation (3) and the known precision
of
CA 02278965 1999-09-22
-12-
measurement for dissolved oxygen and slurry flow, the overall precision in the
evaluation of (3 by this method can be shown. The measurement precision for (3
is found
to be a strong function of the slurry residence time. The greatest error
occurs at high
values of P. A measurement precisiorf of less than 5% can, however, be
achieved if the
residence time is constrained to be less than two minutes.
At low values of (3, a short residence time may" cause the arithmetic
difference
between Ciõ and CoõI to approach the sum of the measurement precision of the
dissolved
oxygen probes. Under these conditions, a longer residence time can be
selected.
j3 is a value representative of the oxygen gas requirements of the ore. It is
to a
1o certain extent artificial. You cannot directly measure P. Rather, it is a
function of other
measured parameters which, while each one is important, none of which alone
give a
complete picture of the flotability of the ore.
Further, measurement of such standard parameters only gives a snapshot of a
slurry. It does not take into account the large number of other variables
which influence
the flotability of the slurry ie water quality, fluctuation in ore flow rates,
temperature
changes, changes in quality of other additives such as collectors, frothers
etc.
Accordingly, it is desirable that the analysis equipment include a recordal
function
such that values of P can be compared with historical data which correlate (3
with
mineral recovery. A typical example is shown in figure 3.
To attain figure 3, the analysis equipment was used intermittently and
continuously
over a period of approximately 10 days. As can be seen from figure 3, the
values of j3
fluctuated wildly from just above 0 to 0.8. As can be seen from figure 3, it
took some
time for the analysis equipment to reach steady state and from approximately
day 6
onward consistent values of (3 were obtained. These values of 0 characterise
the slurry
undergoing mineral recovery and can be used to control both the oxidative
conditioning
step and mineral recovery process to obtain maximum mineral recovery.
(3 in this instance is a characteristic of the slurry. Of course, by altering
the design
and operation of the analysis equipment eg such that it replicates the
oxidative
conditioning step, one may also determine the effect of different oxidative
conditioning
steps on the slurry and thereby determine the most appropriate oxidative
conditioning
CA 02278965 1999-09-22
-13-
parameters for a particular ore. In this way 0 becomes characteristic of the
slurry and the
mineral recovery process.
Accordingly, it can be seen that the present invention provides a self-tuning
control
mechanism for optimising the mineral-recovery process. It takes into
consideration
different ore reactivities and quickly adapts the oxidative conditioning step
to optirnise
the slurry entering the flotation circuit and thereby increasing the recovery
of the
valuable minerals from the flotation process.
It will be appreciated by those skilled in the art that the method described
can be
embodied in other forms without departing from the spirit or scope of the
present
invention.