Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
REDUCED FAT WHEY PROTEIN CONCENTRATE AND METHOD OF
MANUFACTURE
TECHNICAL FIELD
This invention relates to reduced fat whey protein concentrate
(WPC) and a method of producing such a product, preferably, although not
exclusively, from a sweet whey.
BACKGROUND
Whey Protein Concentrates (WPC) and Whey Protein Isolates (WPI)
are two of the major whey protein product categories. WPC typically
contains > 35% protein and > 4% fat, while WPI contains > 80% protein
and < 2% fat. The high protein and low fat content of WPI imparts
greater functional attributes and a preferred composition as compared to
WPC. As a result, WPI commands a higher market value. However, the
processes used to manufacture WPI are very expensive and the yield is low
(generally 50-60% for ion exchange WPI, 50-75% for microfiltration WPI).
WPC have many different uses. Those uses may broadly be divided
into two categories, one being nutritional- based uses and the second being
functional-based uses. The nutritional-based uses would include, for
example, infant formulae, enteral formulae, and sports drinks. The
functional-based uses include, for example, for gelling, whipping,
emulsification and other valuable properties for baking products.
Functionality has been defined by Pour-EI, A. ( 1981, "Protein
Functionality: classification, definition, and methodology", Protein
Functionality in Foods, USA) as 'any property of a food or food ingredient
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
2
except its nutritional ones that affect its utilisation'. Functional
properties
of a protein ingredient are influenced by the composition of the ingredient,
the composition of the food system it is to be used in, and by the
processing conditions required to manufacture this food system. The
majority of functional characteristics of ingredients can be categorised into
hydration related (dispersibility, solubility, swelling, viscosity, gelation)
and
surface related (emulsification, foaming, adsorption at air-water and oil-
water interfaces) properties.
Traditionally, WPC is derived from whey which is the by-product of
either acid (mineral acid or lactic whey) or sweet (cheese or rennet whey)
coagulation of milk protein from milk in the manufacture of cheese or
casein. The standard methods for producing WPC from acid or sweet
whey are well known and are discussed, for example in United States
patent specification US 4,200,662.
Acidification of milk to a pH of about 4.6 causes casein to become
insolubilised and to coagulate as in, for example, casein (mineral acid casein
or lactic casein) or certain cheese types (cottage cheese). The by-product
removal of the casein curd, cheese curd, is known as acid whey. The
alternative method of producing whey is by the addition of rennin and/or
pepsin, proteolytic enzymes, to cause coagulation of casein. The resulting
whey, after removal of the coagulated casein, is known as sweet or cheese
whey.
To produce a WPC the acid or sweet whey is concentrated to
produce a product having nominally 35% whey protein solids. The process
of concentration may involve delactosing, electrodialysis, reverse osmosis,
gel filtration or ultrafiltration.
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
3
In several applications, WPC has a lower functional performance
than WPI. However, in some applications, acid WPC has a superior
functionality as compared to WPI. Typically, cheese WPC has an inferior
functional performance as compared to acid WPC. This is thought to be
due to the difference in fat content, where cheese WPC typically has a
higher fat content as compared to acid WPC. Cheese WPC has a lower
market value than acid WPC and WPI. An improvement in functionality is
expected to increase the market returns for cheese WPC. In addition,
improved functional characteristics may also be achieved for acid WPC if
the fat content could be reduced to a level similar to that for WPI.
Thus, it is an object of the present invention to provide a WPC
and/or method of its manufacture which reduces or overcomes at least
some of the abovementioned problems, or which at least provides the
public with a useful alternative.
Other objects of the invention may become apparent from the
following description which is given by way of example only.
STATEMENT OF THE INVENTION
According to one aspect of the present invention there is provided a
method for producing a WPC having a fat content of no greater than 4%,
from whey, the method including the steps of:
- concentrating the solids content of whey to produce an
intermediate WPC,
- diluting the intermediate WPC with water to reduce the ionic
strength,
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
4
- adjusting the pH of the diluted intermediate WPC to within
the range substantially 3.8 to 5.0,
- holding the pH at that level for a period to optimise formation
of a floc, and
- removing the floc to leave a reduced fat WPC supernatant.
One preferred form of the process of the invention may further
include controlling the temperature of the diluted intermediate WPC prior to
pH adjustment.
Preferably, the temperature may be increased to substantially
50°C.
Alternatively, it may be reduced to substantially 10°C.
In one preferred form of the process of the invention the pH may be
adjusted to substantially 4.2 to 4.4.
Preferably, the pH may be adjusted by the addition of a mineral or
organic acid, preferably citric acid.
Preferably, the temperature may be increased to substantially 50°C
and the removal of floc may be by a clarifier.
In a further preferred form the whey may be concentrated to a solids
concentration in the range substantially 12 to 30% total solids, more
preferably > 18% and in one preferred form substantially 23%.
Preferably, the solids concentration may involve ultrafiltration.
Preferably, the intermediate WPC may be diluted with water at a
ratio in the range substantially 1 :3 to 1:20, retentate:water.
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
Preferably, the intermediate WPC having a solids content of > 18%
total solids may be diluted in the ratio substantially one part retentate to
nine parts water. Preferably, that water is demineralised water.
Preferably, the starting whey for use in the process of the invention
5 is sweet whey.
In a further preferred form the process of the invention may include
further processing of the reduced fat WPC supernatant to concentrate the
solids content and thereafter prepare a dry powder product by conventional
means.
According to a further aspect of the present invention there is
provided a WPC derived from a sweet whey, having functional
characteristics at least equivalent to those of a WPC derived from acid
whey by conventional means.
According to a further aspect of the present invention there is
provided a WPC derived from acid or sweet whey having functional
characteristics substantially equivalent to those of a conventional WPI.
Other aspects of the present invention may become apparent from
the following description which is given by way of example only and with
reference to the accompanying drawings and/or Examples.
BRIEF DESCRIPTION OF THE DRAWINGS
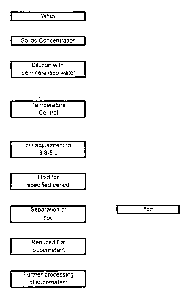
Ficture 1: is a flow diagram of the general features of the
process of the invention, in one preferred form;
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
6
Figure 2: is a flow diagram of one specific embodiment of the
invention.
DETAILED DESCRIPTION OF THE INVENTION
The process of the invention is broadly identified in Figure 1, and
involves the processing of whey (preferably sweet whey) to produce a
reduced fat WPC. As indicated in Figure 1 the process, in broad terms,
involves concentrating the solids component of the whey, preferably by an
ultrafiltration process, to produce an intermediate WPC with a solids
content in the range 12% to 30% total solids. The solids content is
preferably > 18%, and 30% is a maximum practical content. The
intermediate WPC is then diluted with water to produce a WPC retentate
with a lower ionic strength. The dilution ratio will vary depending on the
solids content of the intermediate WPC, but will be in the range 1 :3 to
1 :20, retentate:water. With a preferred solids content > 18% a ratio of
about 1 :9 is appropriate. The pH of the diluted retentate is then adjusted
to a selected level in the range 3.8 to 5.0, the diluted retentate is held
under those conditions for a specified period during which a floc forms and
the floc, which contains a high proportion of fat, is then separated to leave
a low fat supernatant which can subsequently be processed to produce, for
example, a dried, powder, reduced fat WPC.
The end product has a reduced lactose content, as well as a reduced
fat content, and this combination results in an increase in protein content.
Starting with a sweet whey the fat content may be reduced to 1 % or
lower. Conventional sweet WPC has a fat content of 6 to 7% and
conventional acid WPC has a fat content of generally above 4%. Thus,
reductions in fat content of a sweet WPC produced from the process of the
invention to 4% or less provides at least functional equivalence to a
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
7
conventional acid WPC product. Reductions below 4% offer functional
advantages which, as the fat content is reduced to 1 % or less, approach
the functional performance of WPI. It will be appreciated, therefore, that
the functional characteristics of an acid WPC may also be improved by the
process of the invention.
Specific details of the process of the invention, and the product
resulting therefrom, in one preferred embodiment, are now provided by way
of example.
Example
Figure 2 summarises the process employed in a pilot plant trial.
Sweet whey was dosed with citric acid to reduce the pH to 6.00.
The whey was then thermalised by heating indirectly to 73°C and
holding
for 15 seconds, before cooling regeneratively to 55°C. The thermalised
sweet whey was stored prior to ultrafiltration (UF). OF was used to
increase the total solids ITS) (brix) of the whey from about 6% to 23%
using 13 loops of 1 OkD spiral wound membranes. Diafiltration (DF - the
addition of demineralised water to allow a high concentration ratio to be
used) was used after loop 8. After UF, the retentate was chilled to about
4°C, for microbial control, prior to storage.
100 L of this sweet whey retentate (at 23% brix) was then diluted
using 900 L of demineralised water at about 50°C; an approximate ratio
of
10% retentate to 90% water. This mixture, having a preferred ionic
strength for the process, was agitated to ensure a homogenous solution.
Once mixed, the dilute retentate was heated to 50°C using indirect
heating.
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
8
The heated dilute retentate was then acidified. Citric acid was
dosed in-line, to reduce the pH from approximately 6.00 to approximately
4.30. The solution was then held for about 15 minutes prior to clarification
using a pilot clarifier (GEA-Westfalia model KNA31. This was run at 750
L/hr, 4-4.5 bar back pressure, with continuous discharge, and two 0.5mm
nozzles to separate the floc from the supernatant.
It will be appreciated that other acids or acid combinations may be
employed in the acidification step. Furthermore, whilst the preferred pH
may be about 4.30, floc formation may be achieved, although perhaps less
effectively, with a pH in the range 3.8 to 5Ø Adjustment of the time the
solution is held may be appropriate to optimise floc formation for removal.
The supernatant was then heated to 50°C prior to OF through 5kD
spiral wound membranes. This second OF step concentrated the product
to about 20% TS (brix). The volume concentration fraction (VCF) used
was approximately 10. No diafiltration water was added.
The retentate was then pH adjusted to about 6.8 (to give a pH in
the final powder of 6.8 - 7.0) using a mix of KOH and NaOH prior to spray
drying.
It will be appreciated by those skilled in the art that different
processes may be involved in the initial solids concentration of sweet
whey, resulting in higher or lower proportions of solids than described in
this example. To produce the required ionic strength of the WPC retentate
will then require a greater or lesser rate of dilution.
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
9
It will also be appreciate by those skilled in the art that alternative
methods may be employed for separating the floc from the supernatant, for
example gravity settling, filtration or hydrocyclones.
Whilst the process described in the example includes the step of
heating the diluted WPC retentate to about 50°C, the optimum
temperature
for the process will depend on a number of variables, including the holding
time and the method of separation used. The process may, for example,
involve chilling of the diluted WPC retentate to about 10°C prior to
acidification, rather than heating, where the separation method is other
than by clarification.
Product Composition
Table 1 provides compositional data for product manufactured using
the process of the invention in a pilot plant.
Table 1
Dried WPC lend-product) Dried Floc (by-product)
Fat 1.00 12.9
Protein 87.30 74.0
Moisture 3.60 3.00
Lactose 1.02 1 .20
Ash 4.40 1 .50
The table shows that the by-product floc has a fat content of about
13%. The dried WPC end-product has a protein content of about 87% and
a fat content of only 1 %.
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
Functional Testing
End-product from the pilot plant trial was assessed for functional
characteristics (solubility, viscosity, gelation (aqueous and brine), foaming
5 (with sugar and without sugary and performance in an acid beverage rapid
test) and in model food systems (meringues, infant formula and yoghurtl.
The results were as follows.
1. Functional Test Systems
10 a) Solubility.
It is desirable for products to have good solubility over a wide pH
range to avoid sediment forming in final product applications, particularly
beverages. Table 2 illustrates the solubility of each sample over a range of
pH's. The pilot plant sample had a much greater degree of solubility than
that of an acid WPC. This is shown by the narrow pH range over which
sediment formed, and also by the small amount of sediment which formed
in comparison to the Acid WPC.
Table 2. Solubility results - amount of sediment formed at varying pH
values for Pilot Plant Sample and Acid WPC.
Sediment volume
Icm3)
pH Acid WPC Pilot Plant
Sample
Natural (6.911 < 0.1 (7.23) < 0.1
pH
2.0 0.15 < 0.1
3.0 2.5 < 0.1
3.5 2.4 < 0.1
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
11
4.0 2.9 < 0.1
4.5 2.5 0.6
5.0 1.6 0.6
5.5 0.1 < 0.1
6.0 < 0.1 < 0.1
6.5 < 0.1 < 0.1
7.0 < 0.1 < 0.1
8.0 <0.1 <0.1
b) Viscosity.
The relationship between viscosity and total solids for the pilot plant
trial sample as compared to Acid WPC is shown below. Viscosity is a
measure of rheological properties and is important in applications for
example, sauces, gravies and salad dressings. The viscosity characteristic
for the pilot plant sample is equivalent to acid WPC.
250
0 200
- -
v
O
O
150 010%
- Ts
~ 20%
TS
~ 30%
TS
100 - 40%
TS
- - ~
is
c~
50
. -
~oi iii viii
Acid WPC Pilot Plant Sample
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
12
c) Foaming.
Foaming is important for applications such as, for example, cakes
and meringues. Table 3 shows that the pilot plant sample has a maximum
overrun greater than that of the target product for the application (WPI) in
the foaming system without sugar. In both systems, the pilot plant sample
has greater foam stability than WPI.
Table 3. Functional results for a pilot plant sample and a WPI in
a foaming test system.
With Sugar Without
Sugar
WPI Pilot PlantWPI Pilot Plant
Sample Sample
Overrun @ 15 minutes1 280% 1 280 % 2 030% 1 900
Max. Overrun 2 280% 1 280 % 2 030% 2 15C~ ~o
Foam Stability 25.1 1 28.09 15.11 19 minutes
(time for first serumminutes minutes minutes
drop)
d) Gelation.
Gelation is a functional characteristic important in systems, for
example, yoghurts and restructured meats. Table 4 illustrates the similarity
between the pilot plant sample and Acid WPC (target product) in 0% NaCI
gels. No data was available for comparison in 0.4% NaCI systems, and it
can be seen that the pilot plant sample does not perform as well overall in
the 2% NaCI system.
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
13
Table 4. Functional results for a pilot plant sample and Acid WPC in
gelation test systems.
0 % NaCI 0.4% NaCI 2% NaCI
Added Added Added
Acid WPC Pilot Acid WPC Pilot Acid WPC Piiot
Plant Plant Plant
Sample Sample Sample
Syneresis 3.6 3.9 (no data)11.8 25.3 46
(%)
Fudoh 1234 1257 (no data)1626 783 665
Rheometer
19/cm31
TA-XT2 44.47 34.22 (no data)54.52 25.38 16.68
Stress (kPa)
TA-XT2 1.18 1.05 (no data)0.96 0.86 0.96
Strain (kPa)
TA-XT2 29.38 32.59 (no data)17.34 29.38 17.34
Rigidity
e1 Acid Beverage Rapid Stability Test
Absorbance of pilot plant sample and WPI over range of pH's after
heating at 80°C for 20 minutes is shown below. For acid beverages, pH
3.8 is the most relevant. It can be seen that the two products, where WPI
is the target product for the application, are very similar in absorbance over
the pH range.
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
14
3.5
c 2.5
0.5
0
Sediment is undesirable and was observed in the pilot plant sample
only at pH 4.0 ( 14.5 ml sediment), compared to the WPI which had 2m1
sediment at pH 4.0 and 1 ml sediment at pH 3.8.
2. Model Food Systems.
a) Set Yoghurt.
Table 5 shows the similarity between the pilot plant sample and acid
WPC in a set yoghurt system.
WPI Pilot Plant
Sample
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
Table 5. Free whey, total solids content (TS) and Texture Analyser
results for set yoghurt samples.
Sample Day Texture Free TS
Profile
Results
Peak 1 Peak Positive Whey (%)
(9) 2 Area (%)
(g) (g.s)
Acid WPC 1 21 22 361 0 9.55
Pilot Plant 1 22 25 408 0 9.63
Sample
5 Peak 1 - represents elasticity / brittleness
Area - calculated from the area between Peak 1 and Peak 2,
represents mouthfeel / body
b) Stirred Yoghurt
10 Table 6 shows the similarity between the pilot plant sample and
Acid WPC in a stirred yoghurt system.
Table 6. Viscosity, drained syneresis and total solids (TS) results for
stirred yoghurts.
Viscosity Level TS
(mPas) of
Sample Shear Rate Whey (%)
50s-' 100s-' Drainage
Acid WPC 230 130 31 9.38
Pilot 250 140 33 9.23
Plant
Sample
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
16
c) Infant Formula.
The following results show the similarity between the pilot plant
sample and Acid WPC in a model infant formula system.
Acid WPC Pilot Plant Sample
Heat Coagulation Time 7.5 minutes 7.5 minutes
Mean particle diameter 1 1 (d 0.9) 15 (d 0.9)
(Malvern Mastersizer)
Table 7. Particle Size distribution of pilot plant sample and Acid WPC
as determined using the Malvern Mastersizer.
Peak One Peak Two
0.28-0.35pm 0.35-0.43~m 6.52-8.04~m 8.04-9.91
(%) (%1 (%) ~m
(%)
Acid WPC 7.1 1 6.17 14.81 17.86
Pilot Plant5.41 5.64 8.07 9.98
Sample
Thus, it can be seen that the process of the present invention
enables the processing of a whey (preferably a sweet whey) in a manner
which produces a readily removable floc containing a substantial portion of
the fat, leaving a WPC end-product having a high protein and reduced fat
content, and with improved functional performance. The resulting product,
when derived from a sweet whey may have a functional performance at
least equivalent to that of a conventional acid WPC, and a similar
composition. Furthermore, a WPC product, whether derived from sweet or
acid whey, may be produced with functional performance substantially
equivalent to that of a WPI.
CA 02379169 2002-O1-28
WO 01/08502 PCT/NZ00/00140
17
Where in the foregoing description reference has been made to
specific components or integers of the invention having known equivalents
then such equivalents are herein incorporated as if individually set forth.
Although this invention has been described by way of example and
with reference to possible embodiments thereof it is to be understood that
modifications or improvements may be made thereto without departing
from the scope or spirit of the invention.