Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
CA 02598876 2007-08-24
-1-
Title: A Method and Apparatus for Identifying and Characterizing Objects
based on Fluorescence
FIELD OF THE INVENTION
This invention relates the general field of identification or sorting
technologies, and particularly relates to identification or sorting
technologies which involve exposing the material to be identified or sorted
to an incident beam of electromagnetic radiation. Most particularly this
invention relates to identification or sorting based on detecting a material's
fluorescent reaction to the incident energy beam.
BACKGROUND OF THE INVENTION
Electromagnetic radiation, and in particular various wavelengths of
light, may be used to identify or sort various materials. One problem in
material sorting relates to identifying gems. It can be difficult to identify
different gems from the ore in which they are originally found or from
various types of look alike material, such as glass or man made materials
that may be used to imitate a gem, for example. Not all look-alike
materials can be identified by any one test, and it is common in gemology
to put a suspect material through a series of identification tests to properly
identify and certify that the gem in question is genuine.
Many different techniques and devices exist for the identification of
gems by fluorescence. The most prominent testing techniques involve
using UV light or X-rays, although other excitation methods are also
known. For example, US Patent 883,653 teaches using a blue incident
light of a higher refrangibility to produce fluorescence of a lower
refrangibility and to obviate the masking effect of the reflected light by
using a second ray filter of the yellow colour to transmit red, orange
yellow, yellow green and green fluorescence. However, in blue light, red
orange yellow, green and green fluorescence are not necessarily masked
by the blue light, and due to the large wavelength difference between the
CA 02598876 2007-08-24
-2-
incident and fluorescing wavelengths, a simple or crude filter will work well
to separate the incident light from the fluorescence.
US Patent 4,394,580 uses absorption, internal reflection and
internal excitation to try to characterize gems. However, this invention
teaches a wide source i.e. white light, and the use of red and green
optical filters before a detector. However, the red and green gems could
also be visible in the white spectrum light to the naked eye. Further, of
course white light covers the visible light spectrum including red
fluorescent wavelengths so any fluorescing will be masked by white light,
even if a filter is used.
US Patent 5,118,181 relates to a method of identifying individual
gemstones by using a series of specific incident light wavelengths in the
ultraviolet or infra red spectrum and measuring the response of the gem
stone to each separate wavelength. While useful to characterize a
particular stone, this cannot be used in a mine environment to separate
gem stones from ore for example.
Another previously known technique for identifying gems has been
to detect the presence of fluorescing material such as found in certain
gems containing the element Chromium. Such gems can red fluoresce
when exposed to particular light frequencies, and examples of gems
which can fluoresce red include emeralds, rubies and the like. In the past
the fluorescence was detected by using ultraviolet light, or even an
incident blue light on the gem in question. What has been previously
known is that such incident light can elicit the red light fluorescence from
specific gems, which could then be characterized based on the presence
of such red light fluorescence. The blue incident light can be obtained by
shining a white light through a blue liquid onto the object in question,
although more recently it is more common to use a blue light source such
as an LED. Longwave UV LEDs exist also. The technique uses incident
light having wavelengths much shorter than the red light fluorescence,
making the red light fluorescence easier to detect. It has also been
CA 02598876 2007-08-24
-3-
understood up until now that while UV and blue light elicit a relatively
strong and easily detectable red light fluorescence, wavelengths closer to
the fluorescing wavelengths tend to exhibit a weaker and more muted
excitation response. The use of a blue filter would of course eliminate red
light wavelengths from the incident light. When the object is viewed
through a red light filter, which blocks out the blue or UV incident light,
the
red fluorescence is then visible. In many cases strong fluorescence can
be detected which is easily identified by eye, or even through the use of a
crude filter which broadly transmits red light.
While blue or ultraviolet incident light works well in some
circumstances, the use of these incident lights to elicit a red light
luminescence is problematic in certain situations. For example,
in
underground mining of precious gem materials, it is not desirable for the
mine workers to be able to easily identify the individual gemstones.
These tend to be both small and very valuable so the use of any detecting
method that makes the gems more apparent to the visible eye, or easily
identifiable through the use of a crude filter is not desirable as valuable
gemstones can go missing. So, blue, ultraviolet or even wide spectrum
white lights all of which can make gem material visible to the naked eye,
are not the most desirable form of incident light for sorting the gems from
the background ore in a mine environment.
What is desired is a simple and easy way to use the red
fluorescence of certain materials to characterize the material, without
necessarily revealing the property of even the body colour of the material
to the naked eye or making it easily detectable with a crude filter.
SUMMARY OF THE INVENTION
The present invention is directed to a method of sorting out
material based on its red light fluorescent properties. However, rather
than using blue, ultraviolet or white incident light, the present invention is
directed to using red incident light to excite the material being identified
to
CA 02598876 2007-08-24
-4-
cause it to fluoresce. In particular, the red light source will cause
fluorescence to arise in a wavelength band only slightly above the red
light wavelength band of the preferred incident light source, and thus can
be used to characterize and identify the presence of a certain fluorescing
material, based solely on the fluorescing property. However, the use of a
red incident light means that the unfiltered red fluorescence will be
completely swamped by the red reflected light and the red fluorescence of
the gemstones will remain invisible to the naked eye. Further, the red
fluorescing light is separated from the red incident light by a relatively
small difference in wavelength, and is much harder to detect, requiring a
much more specific filter. Thus, separating gemstones from the ore,
without the use of the specific filter capable of blocking incident red light,
but passing fluorescence having only slightly longer wavelengths is much
more difficult. In this way the valuable gem materials are only visible to
the appropriate people through the use of an appropriate filter or detection
means.
For example, in a mine environment, the use of a narrow band red
light source, such as an LED will not permit a red fluorescence light
response to be visible the naked eye but will be easily detectable by the
presence of the higher wavelength fluorescence by means of a
wavelength detector or even by visual examination with an appropriate
narrow band filter.
Thus, the present invention provides a method which is particularly
useful in being able to sort material, for example, when trying to select the
gem materials from a background such as common ore. Although the use
of red light causes the gem to fluorescence in the longer wavelength red
light spectrum, this fluorescing is masked by the incident light to the
naked eye. However, by means of a detector or through the use of a
specific red light filter, the fluorescence can be detected and this can be
used as the basis for sorting the gem material from the ore.
Therefore there is provided, according to one aspect of the present
CA 02598876 2014-09-03
-5-
invention, a method of characterizing objects, said method comprising the
steps of:
illuminating said object with an incident red light having
wavelengths shorter than a maximum red light wavelength, said object
being characterized as exhibiting red light fluorescence of a longer
wavelength than the wavelength of the incident light; and
detecting the presence of said longer wavelength red light
fluorescence emanating from said object.
According to a further aspect of the present invention, there is
provided an apparatus for sorting materials comprising:
a source of red incident light including wavelengths of at least
between 600 and 650 nms,
a station to apply the red incident light to an object;
a detector to detect red light fluorescence in a wavelength between
650 as 700nms; and
a means to physically separate said object exhibiting said red light
fluorescence from other objects being tested.
There is also provided, according to another aspect of the present
invention, a method of using red light fluorescence, said method
comprising the steps of:
directing incident red light having wavelengths shorter than a
maximum red light wavelength at a material to detect the presence of
chromium, and
detecting the presence of red light fluorescence of a longer
wavelength than the wavelength of the incident light emanating from said
material to prove the presence of chromium is said material.
BRIEF DESCRIPTION OF THE DRAWINGS
Reference will now be made, by way of example only, to drawings
which depict various embodiments of the present invention and in which:
Figure 1 is a view of the present invention according to a first
CA 02598876 2014-09-03
-6-
embodiment;
Figure 2 is a view of the present invention according to a second
embodiment;
Figure 3 is a spectrometer graph of the wavelengths and intensities
of the wavelengths produced by a red LED suitable for use in the present
invention;
Figure 4 is a view of the wavelength intensity graphs or the light
emanating from various natural and synthetic emeralds;
Figure 5 is a view of the wavelength and intensity readings for the
light emanating from natural and synthetic ruby based on an incident light
from a red LED;
Figure 6 is a view of the wavelengths and intensities produced by
natural and synthetic alexandrite when illuminated by an incident light
from a red LED;
Figure 7 is a view of the present invention as applied to a mining
environment;
Figure 8 is a plot of a red laser incident light source, applied to a
flame fusion synthetic ruby; and
Figure 9 is a plot of a red LED light source overlayed on a red laser
light source.
DETAILED DESCRIPTION OF THE PREFERRED EMBODIMENTS
The present invention is directed to a method of identifying or
detecting the presence of certain materials, based on the fluorescence of
such materials under the exposure of such materials to incident red light.
The term red light includes light generally in the visible light spectrum in
the wavelength range of about 620 to 750 nms. Most preferably, to be
easily visible, the incident light wavelength is less than about 700 nms. In
this specification the term fluorescing means that electromagnetic energy
is released from an object which is subject to an incident light, at a
frequency that is different from the incident light. Thus fluorescing is an
CA 02598876 2014-09-03
-7-
optical phenomenon in cold bodies in which the molecular absorption of a
photon triggers the emission of another photon with a longer wavelength.
This invention relates to the useful application of this phenomenon to
characterize or separate articles based on specific forms of such
fluorescence as described below.
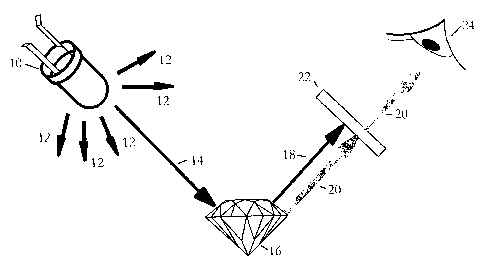
Figure 1 shows an example of the method of the present invention
being implemented. A red light source, for example an LED, is shown at
with red wavelength light emanating as indicated by arrows 12. In this
specification the term red is used to describe the wavelengths of the
10 incident light, and it will be generally understood to include the
wavelengths within the visible red spectrum (620 ¨ 750 nms). In a
preferred form of the invention a red LED is used which produces a
relatively narrow range of wavelengths of light, in the 610 to 650
nanometre range, primarily within the 620 to 640 range. It will be
understood by those skilled in the art that the present invention is not
limited to LED's which produce photons within this specific band, provided
the wavelengths of the incident light are generally within the visible red
light band as explained in more detail below. For example, a red light
laser having wavelengths of a narrow band peaking at 655 will also
produce adequate results as explained below. Whether the light source is
an LED or laser light, the light intensity will need to be sufficient to
excite
fluorescence which may vary from gem to gem, as will be understood by
those skilled in the art. A laser light, with it is tightly focussed beam will
provide good intensity at greater distances.
Turning back to Figure 1, arrow 14 represents an incident light ray
directed onto a gem 16 and produces two resultant light rays 18 and 20.
The light ray 18 is simply a reflected or transmitted red light ray having the
same wavelength as the incident light ray. The light ray 20 however is a
fluorescent light ray which is of a different, and longer, wavelength from
the reflected or transmitted light ray 18. A filter 22 is shown which blocks
or absorbs the reflected or transmitted red light ray 18 but permits the
CA 02598876 2014-09-03
-8-
fluorescent red light ray 20 to pass. The passed fluorescing light ray 20
can then be readily observed, as indicated by eye 24. Although longer
than the incident light, the fluorescing light wavelength is still in the
visible
light spectrum.
Figure 2 discloses a further embodiment of the present invention.
In Figure 2, the red wavelength light source is noted as 30 having a
plurality of red light rays 32 emanating there-from. Again, the light rays 32
include incident red light ray 34 which impinges upon a gem 36. Two
different light rays extend from the gem 36, shown as 38 and 40. The
light ray 38 is merely a reflected or transmitted red light ray, whereas the
light ray 40 is a fluorescent light ray having a longer wavelength than the
reflected or transmitted red light ray. Both of these light rays impinge
upon a detector identified by 42. The detector includes a means for
detecting the presence of the longer red fluorescent wavelengths of the
light ray 40, which can be used as a basis to identify the presence of red
light fluorescing materials in the object or gem itself. For example, the
detector can measure the wavelengths of the light emanating from the
object, determine if there is a sufficient amount of longer wavelength red
fluorescent light present and provide an electronic output to signify that
there is. The electronic output can then be used as a trigger signal, for
example for an actuator, such as an arm or a blast of compressed air, to
separate the detected object from a stream of objects moving for example
past the detector on a conveyor belt or other form of mover. In this way
the detector can signal and separate gem bearing ore from non-gem
bearing ore for example. An advantage of using a detector as described
is that it can also detect red light fluorescence that may be above the
easily visible red light spectrum.
A preferred light source according to the present invention is a light
emitting diode or LED. An LED is a semiconductor device which often
looks like a tiny light bulb without any filament. LEDs have the advantage
that they do not heat up, are relatively energy efficient and have a
CA 02598876 2014-09-03
-9-
potential life span of 100,000 hours or about 20 times longer than
conventional light bulbs. While some LEDs can produce very bright white
light or light of specific colours, in the present invention, it is preferred
to
use a somewhat monochromatic red light laser LED. A red light LED
also uses less energy than an LED which produces a shorter wavelength
light, such as a UV or blue light LED, leading to longer battery life for hand
held red LED devices. Laser light sources are also comprehended, as
providing both an intense and narrow red incident light source.
Turning to Figure 3, a wavelength and intensity graph for a
preferred form of red LED according to the present invention is shown.
As can be seen, the red light intensity peaks at approximately 635
nanometres, rising from a base line of approximately 600 nanometres and
ending at a comparable base line of approximately 650 nanometres. The
preferred wavelength range of this incident light source is therefore
between 600 and 650 nanometres, although an even narrower or different
range can also be used as will be understood from the detailed
description below.
Figure 4 is a view of the wavelength and light intensity of the red
light fluorescence produced by two types of emeralds when exposed to
the red light LED of Figure 3. The line 50 depicts a natural Columbian
emerald and illustrates a red fluorescence light intensity sub-peak just
>640 nms. This sub-peak is a fluorescence phenomenon at a longer
wavelength than the incident red light, and is clearly visible when the
incident red light wavelengths are filtered out. As can be seen from the
graph, line 52 represents the incident red light wavelength produced by
the preferred LED light source according to the present invention (and
matches the plot of Figure 3). Line 53 represents the red fluorescing light
emanating from a synthetic flux emerald and includes a smaller but still
visible intensity peak just beyond 650 nanometers in wavelength. In this
sense synthetic means a lab grown counterpart to the natural material
which is chemically, optically, chrystallographically and otherwise
CA 02598876 2014-09-03
-10-
substantially identical to the natural gemstone. Interestingly, the synthetic
emerald also includes a second intensity peak at around a wavelength of
about 710 nanometers some of which is also possibly visible when the
wavelengths of the incident red light are filtered out. Thus an aspect of
the present invention is identifying a specific fluorescing pattern
associated with a specific gem material, and separating one gem material
from another based on such red light fluorescing patterns. Above 710 can
be actually somewhat hard to see, but does form part of the visible red
light spectrum, and can be detected by a detection device as previously
described. Interestingly, in respect of natural emeralds, the intensity of
the red light fluorescence produced by an incident red light source is often
observed to be significantly greater than the intensity of red light
fluorescence produced by a UV incident light.
Figure 5 is a plot or graph of the wavelength versus intensity for a
number of natural and synthetic ruby elements exposed to a red light
incident light source. Again, the red LED incident light source peaking at
635 nanometers is shown at line 60. A natural Be treated ruby is shown
at line 62, a natural treated ruby from Burma is shown at line 64 and a
synthetic flame fusion ruby is shown at line 65. Be treated rubies usually
start out as a poor coloured sapphire, often iron quenched, thus
explaining the poor red fluorescence shown here. The purpose of the Be
treatment is to improve the colour, but it does not improve the
fluorescence. As will be understood by those skilled in the art, both rubies
and sapphires are classed as corundum. Rubies are red corundum and
all other colours of corundum are sapphires. As can be noted, the natural
ruby from Burma exhibits a high degree of fluorescence, the synthetic
flame treated ruby also exhibits fluorescence, and the natural Be treated
ruby exhibits a very minor fluorescence all centered around 690
nanometers. As can be seen from Figure 5 each of these gem materials
has responded by fluorescing at a red light wavelength longer than the
incident light wavelengths.
CA 02598876 2014-09-03
-11-
Figure 6 shows the wavelength and intensity graphs for natural and
synthetic alexandrite exposed to incident red light. As can be seen, the
red LED source peaking at 635 nanometers is shown at line 70, the
natural alexandrite from India is shown at line 72 and the synthetic
hydrothermal alexandrite is shown at line 74. Each of these alexandrite
gems has a fluorescing peak at about 680 nanometers. The synthetic
hydrothermal alexandrite has a very intense fluorescence up to 4,000
counts, whereas the natural alexandrite has a much weaker fluorescence
of just under 1,000 counts. However, in each case, the longer wavelength
fluorescence is clearly visible with an appropriate filter or detector capable
of detecting light at the red light fluorescing wavelength peak around 680
nanometers.
Figure 7 depicts the method of the present invention being applied
to separate valuable gem bearing ore from worthless background ore
being produced in a mine for example. A narrow band red incident light
source 100, such as a red light LED peaking at 635 nanometers is shown
with rays 102 and having ray 103 impinging on ore samples 104 and ray
110 on ore sample 106. Sample 104 has no gem content, and as a result
merely reflects the impinging light directly as shown at 105. A red light
filter 107 which is configured to block red light across the range of light
produced by the incident light source, passes no light to the observer 109.
In other words, when observed through the filter 107 the ore sample 104
remains dark. However, when observed without the filter the whole ore
sample 104 appears red due to the reflected light.
In contrast, the ore 106 contains some chromium bearing gem
material. This is illustrated as 108. Upon the incident red light 110
impinging on the gem material 108, both red light 112 and red light
fluorescence 114 result. The reflected red incident light 112 is blocked by
the filter 118 but the red light fluorescence ray 120 passes and is
observed at 122. In other words, when the illuminated ore is observed
through the appropriate red light filter, the fluorescing gem is very visible
CA 02598876 2014-09-03
-12-
and easily distinguished. On the other hand, when observed by the
naked eye, all that can be seen is the whole ore sample as red, due to the
incident red light being reflected off the ore sample. A broad spectrum or
crude red light filter will not work as it must be precise enough to pass
visible red light fluorescence at wavelengths above the incident light while
blocking the red incident light.
Figure 8 is a plot of a red light laser incident light source shown as
130 impinging on a synthetic flame fusion ruby which gives of a red light
fluorescence as shown by trace 132 centered on wavelength just below
700 nms.
Figure 9 is a plot of two different suitable incident red light sources
according to the present invention. The trace line 134 is for a LED light
source and the line 136 is a laser light source. Red light LEDs are widely
available from a number of manufacturers, as are pocket laser pointers.
While each one will likely have different wavelength distributions, even
with the same brand, a suitable one will be characterized as producing
red light of sufficient intensity to elicit a fluorescing response from
fluorescing materials, wherein the incident red light has a sufficiently short
red light wavelength so as to permit the longer red light fluorescence to be
within the visible spectrum.
The present invention can be used to characterize a number of
materials providing they red fluoresce and this has been observed in
certain natural materials such as emeralds, rubies, pink sapphires,
alexandrites, spinnel, garnets, and tourmaline. The present invention can
also be used to characterize a number of manmade materials, providing
they red fluoresce, and this has been observed in materials such as
certain coated manmade diamonds or other synthetic gem stones such as
man made rubies and emeralds and other materials which exhibit the
property of red light fluorescence. The other materials include certain
forms of material or fabric as well. As will be appreciated by those skilled
in the art, not every gem has the same composition. Thus, certain gems
CA 02598876 2014-09-03
-13-
are known to be inactive or non-fluorescing, even if they are from a class
of gems that would be typically considered active. This is due to the
peculiarities of the mineralization that occurs in nature in forming the
gems in the first place including iron quenching of fluorescence. Thus,
the present invention is of course limited to being used on those gems or
other materials which exhibit the red light fluorescence property.
It can now be appreciated that the red light fluorescence that can
be elicited from a red light incident source has certain advantages. The
red light fluorescence is masked in a red incident light environment. The
advantage is that this makes it harder to detect with the naked eye. Also
the red light luminescence produced by the object, such as a gem as
noted in the examples, tends to be in a relatively narrow band located
close to the wavelength of the incident light source and so requires a
specific filter. However, these very properties are an advantage in a
detection technique which is on the one hand reliable and yet on the other
not visible to the naked eye, even with the help of crude filters. Thus, in a
separation facility for ore from a mine, the whole facility can be lit with
red
light, which is bright enough to allow the workers to move around and
accomplish the required physical tasks, but does not provide to the
workers enough visual information to permit the workers to identify the
valuable gem material.
Thus the present invention is directed to the realization that a red
light incident source which includes red light wavelengths between 610
and 650 nms can be used on a object to detect fluorescence generally in
a light band slightly above the incident light band, generally below 750
nms in wavelength and at least part of which is typically below 700 nms in
wavelength. Such red light fluorescence is detectable with a specific
wavelength filter which blocks light wavelengths generally below 650 nms,
and this can be an effective method for sorting objects such as gem
materials in, among other things, a mine or ore processing environment.
In the event that more than one chromium bearing gem material is found
CA 02598876 2014-09-03
-14-
in a particular site, all of them can be located with the red incident light.
So, where emeralds and alexandrite are found together, both can be
characterized by this method. Of course the present invention can also
be used to characterize already cut and polished gems from other visually
similar gem materials and from materials which do not fluoresce red by
this method such as glass or the like.
While reference has been made to certain preferred embodiments
of the present invention, this is by way of example only, and it will be
appreciated by those skilled in the art that the true scope of the invention
is broadly defined by the attached claims. For example, while certain red
light LED and laser light sources have been identified, other red light
sources are also comprehended. What is required is that the red light
source have a sufficient intensity to provoke fluorescence, and be of a
wavelength to permit a longer red fluorescent light wavelength to be
produced, most preferably in the easily visible red light range.