Note: Descriptions are shown in the official language in which they were submitted.
)7~7
CATA~YTIC TWO-STAG~ CO-PROCESSING OF
COAL/OIL FE~DSTOCKS
BACKGRO~JD OF INVENTION
This invention pertains to co-processing coal/oil feed-
stock3 in a two-stage catalytic hydroconversion process. It
pertains more particulatly to such coal/oil co-processing to
produce higher percentage conversion and increased yields of low-
boiling hydrocarbon distillate liquid products, while mlnimizing
hydrocarbon gases and heavy resid materials.
Coal/oil co-processing using a single stage catalytic
ebullated bed reactor, has been shown to be an effective technique
for simultaneous conversion o coal and residual oils to produce
predominately hydrocarbon liquid products, as disclosed by
U.S. Patent 4,0~5,504 to Chervenak, et al. At high percentage
conversio. levels, the single stage hydrogenation procecs produces
undesirably high yields of byproduct hydrocarbon gas (Cl-C3) and
product quality decreases, i.e., the N2 and S contents of the
distillate llquids increase. SeVeral other processes for simui-
taneous processing of coal and petroleum feeds using two reactionstages have been proposed, such as disclosed by U.S. Patent
Nos. 3,870,621 to Arnold; 4,306,960 to Gleim, and 4,330,390 to
Rosenthal, et al. However, these processes all have shortcomings
and do not achieve the flexibilityiand high yields o low-boiling
hydrocarbon distillate liquids de~ired. Significantly ijmprove~
result~ have now ~een achieved by the present two-stage catalytic
coal/oll co-processing proces~. ~
'' ';
. i';~
. 1.
7~7
~uMM~ y ()I INVI:NI'I~)N
The present invention provides an improvecl hydrogcnntlon pro-
cess in which particulate coal and liquid hydrocarbon feedstocks
are co-peocessed in a catalytic two-stage ebullate-bed reactor sys-
tem, to produce increased yields of low-boiling hydrocarbon distil-
late liquids and minimal yield~ of hydrocarbon gas and high-boiling
resid fractions. The first stage reactor is operated at mild hy-
drogenation conditions of 650-800F temperature and 1000-4000 psig
hydrogen partiaL pressure and at 10-100 lb coal and petroleum/hr ft'
reactor volume to increase the hydrogen content of the dissolved
.. _ _ _ .., , , _ ... .. . . ... ..
coal and oil feed, and recycle oil (if ~se~) molecules,
while obtaining moderate conversion of the coal without
producing re3ressive (coke forming) reactions.
The catalyst should be selected from the metals
group con6isting of oxides or other metal compounds or
components of cobalt, iron, molybdenum, nickel, tin,
tung~ten and mixture~ thereof and other hydrocarbon
hydrogenation cataly~t metal oxide~ known in the
art, deposited on a base material selected from the group consis~-
ing of alumina, magnesia, silica, titania, and similar materials.
Useful catalyst particle sizes can range from about 0.030 to 0.125
inch effective diameter
The first stage reactor effluent material is passed to ~
direct-coupled second stage catalytic reactor, which is operated
at somewhat more severe hydroconversion conditions of 750-900F
temperature and 1000-4000 psig hydrogen partial pressure to con~
vert the remaining unconverted coal and residual oil and to pro-
duce high yields of high quality distillate liquid products, with
minimal yields of hydrocarbon gases and high-boiling resid fract-
ions. The catalyst used is similar or can be the same as that used
in the first stage reactor. From the second stage reactor the
I z~
effluent is phnse separule(l and distilled to provlde the com-
bined hydrocarbon liquid distillate products.
This process improvement permits co-processing operations on
blended coal and petroleum feedstocks at high conversion to pro-
vide distillate liquid products, without encountering compatibility
problems between the coal-derived and oil-derived products. The
addition o a :first low severity hydrogenation reaction stage to increase
the hydrogen content of thè fresh coal and oil feed materials (and
recycle oil if present) reduces sulfur and nitrogen compounds in
the liquid product and improves the solvent quality of the liquids
needed to dissolve the coal, and also significantly improves the
overall process performance and allows its successful applicat~on
to a wider range o fcedstocks. Coal conversion in catalytic two-
stage co-processing with solvent quality sensitive coals (such as
Alberta sub-bituminous coal) are equivalent to that obtained with
coal-only process derived solvent. High selectivity to hydrocar-
bon liquids with minimum by-product gas yield have been achieved.
Also, it has been determined that the Watson characterization ~actors
in relation to the mean average boiling point for the hydrocarbon
liquid products produced by the present catalytic two-stage
coal/oil co-processing process are intermediate those produced
by a catalytictwo-stage coal liquefaction process and by catalytic
petroleum hydroconversion processes.
; In the present invention, if the petroleum oil feed exceeds
needed for slurrying the particulate coal ~eed to provide a
pumpable fluid mi~ture, the recycle of hydroconverted hydrocarbon
liquids or such slurrying may not be required. Otherwise, such
recycle o heavy distilled hydrocarbon ractions is usuaLly done
to provide increased conversion and yields o' low-boiling hydro-
~lcarbon liquid products.
il -3-
i~
B
~ Z 8~)7 13~ -
-3a-
The present invention, therefore, resides in a two-
stage continuous process for catalytic hydroconversion
of a fluid blend of a solid carbonaceous material and
heavy hydrocarbon liquid, comprising;
(a) mlxing a solld carbonaceous particulate
material with sufflcient heavy hydrocarbon liquid
having at least about 90 V% normally boiling above
650-F to provide a flowable B lurry mixture, the
total hydrocarbon liquid/coal feed weight ratio
being between about 1.0/1 and 3/1, with the solid
aarbonaaeous material being between about 25 and 50
W % o~ the total feed material;
(b) feeding the flowable slurry mixture with
hydrogen into a first stage back-mixed catalytic
reaction zone containing an ebullated catalyst bed
of particulate hydrogenation oatalyst, said
reaction zone having an internal liquid recycle;
said catalyst containing an active metal component
wherein the metal in said component i5 selected
from the metals group consisting of cobalt, iron,
molybdenum, nickel, tin, tungsten, and mixtures
thereof on a support material, said catalyst bed
being maintained at 650-800-F temperature, 1000-
4000 psig hydrogen partial pressure and feed rate
of 10-100 lb carbonaceous materi.al plus heavy
hydrocarbon liquid feed per hour per ft3 reaotion
zone volume for hydrogenation reaction to partially
hydrogenate and hydrooonvert the feed materials to
hydrocarbon material contalning less. than 6 W% C1--
C3 hydrocarbon gases, 15-25 W% 650-F-light li~uld
~raction and 60-70 W% 650-F-~ hydrocarbon material
fraction;
(c) passing the total effluent material from
~aid first stage reaction zone wlth addltional
hydrogen directly to a close-coupled second stage
back-mixed catalytic reaction zone containing an
~ 30~7~7
-3b-
ebullated catalyst bed &O as to avoid forming
retrograde materials in the effluent, said catalyst
containing an active metal component wherein the
metal ln said component la selected from the ~atalq
group consisting of cobalt, lron, molybdenum,
nickel, tin, tungsten and mixtures thereof on a
support material, said second stage reaction zone
being maintalned at a higher temperature than the
~irst s-tage reaction zone, and at 750-900'F
temperature and 1000-~000 psig hydrogen partial
pres~ure to convert the remaining unconverted
carbonaceous materlal to hydrocarbon gases,
hydrocarbon liquld fraction normally boiling
between 400-650-F and including a high boiling
residuum fraction;
(d) passing the resulting effluent material
from said second stage reaction zone to successive
phase separation and distillation steps to separate
the gas material fraction; and
(e) removlng unconverted coal and ash solids
material and a heavy hydrocarbon bottoms liquid
material, and thereby producing low-boiling
hydrocarbon liquid products normally boiling
between 150 F and 975 F.
- BRIEF DESCRIPTION OF DRAWINGS
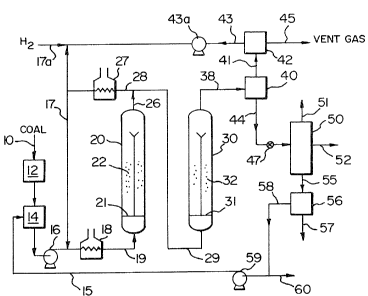
Fig. 1 is a schematic flow diagram of a two-stage catalytic
process for hydroconversion of coal/oil feedstocks according to
the present invention.
;'
.. . .
,
DESCRIPTION OF INVENTION
In the present invention, improved hydrogenation and hydro-
conversion of blended coal and oil feedstocks is provided in a
two-stage catalytic process using ebullated catalyst bed reactors.
As is ~hown in the Fig. 1 process flow diagram for catalytic two-
stage coal/oil co-processing, a coal such as bituminous, sub-bitum-
inous or lignite, is provided at 10 and is passed through a coal
preparation unit 12, where the coal is ground to a desired particle
~size such as 50-375 mesh (U.S. Sieve Series) and dried to a desired
moisture content such as containing 2-10 W % moisture. The par-
ticulate coal is then blended with fresh hydrocarbon liquid feed
such as petroleum resid, heavy crude oil, tar sand bitumen, or
shale oil provided at 11, and are mixed together at slurry tank 14
~, ; . .
'. I, . ' i
;l '' \
~`! ' '
I _4-
I . '
;~æ
to provide a pumpable coal-oil slurry feed material. A total
oil/coal weight ratio between about 1.4/1 and 3/1 can be used. If
desired, a recycled process-derived slurrying oil at 15 can be ad-
ditionally mixed with the coal and oil feedstocks. The resulting
cosl-oil slurry is pumped at 16 to reactor pressure, preheated at
18, mixed with hydrogen gas at 17 and is fed into the lower end of
the first stage reactor 20.
The first stage reactor 20 is preferably a catalytic ebullated
;bed reactor containing catalyst bed 22 and operating at moderate
jconditions of 650-800F temperature and hydrogen partial pressure
; of 1000-4000 psig for hydrogenation and hydrocon~ersion of the
! blended feed materials. In the reactor the blended upflowing coal-
oil feed material is effectively contacted with hydrogen in the
presence of a particulate hydrogenation catalyst, as generally
described by U.S. Patent No. Re 25,770. Conventional hydrogenation
catalysts, including nickel molybdate, cobalt molybdate or nickel-
tungsten on an alumina or silica support such as employed in the
H-Coal~ or H-Oil~ Processes are utilized in the well-mixed ebul-
lated bed reactor. Useful total feed rates are in the range of
lO to 100 lb/hr/ft' reactor volume for each stage, with feed rates
of 15-75 lb/hr/ft' usually being preferred depending on the par-
ticular proportions of coal and oil in the feed and the products
desired.
1 From the first stagr-. reactor 20,the total effluent stream 26
i! iY mixed with additional preheated hydrogen at 28 as needed and
!1 is fed directly into the lower end of second-stage reactor 30 The
dded hydrogen is preheated to increase the ternperature of the
!1 first-stage reactor effluent to the desired second stage reactor
temperature conditions. The second stage reactor 30 is preferably
;la catalytic ebullated bed reactor contain~g catalyst bed 32 and 2
operating at essentially the same pressure conditions as the first
5-
1,,~,,"'1,,,, , ~
~} ~ 7(:~7
st~ge res~to~ (slightly low~r to allow ~or pressur~ drop nnd for-
ward flow of ms~erials) ~nd a~ hi~her temperatures of 750-900F
utili~ed for further hydroconversion reactions. The second stage
reactor uses catalysts which are similar to those for the first
stage reactor. The first and second stage reactors may hsve equal
volumes or they may be substantially different in volume depending
' ,on the product yield and product quality objectives.
From the second stage reactor~effluent 38 vapor and liquid
ractions are separated at the existillg high pressure in separator
40, and the vapor fraction ~1 passed to hydrogen purification unit
~2 to provide a hydrogen recycle stream 43. The liquid fraction
~4 is pressure-red~ced at 47 to recover distillate liquid products
in an atmospheric pressure fractionator 50 to produce desired
distillate liquid prod~cts 51 and 52. The bottoms liquid stream 55
is passed to a liquid-solids separation step 56, from which fine
solids material of unconverted coal and ash are removed ~t 57.
If desired, a portion of the atmospheric bottoms
liquid from the liquid-solids separation step 56 can be
advantageously recyaled to the iirst stage reaotor as
~lurrying oil 15. If sufficient liquid hydrocarbon
feedstock is used to slurry the coal eed, use o~
recycled process-derived slurrying oil can be
eliminated, to provide a once-through type operation ~or
the feedstocks. Process-derived hydrocarbon streams
which may be used for the coal slurrylng oil include
distillate liquid product, and product oils which are
recovered from the liquid-solids separation step, which
may utilize hydroclones, filters, centriEuges, or
solvent deashing technigues. The remainder of the
a-tmospheri.c bottoms material rom separation step 56 is
vacuum distilled to recover a vacuum gas oil stream and
a pumpable vacuum bottom~ slurry material.
This invention will be further described by
reEerence to the Eollowing Examples of operations, which
should not be construed as limiting the invention.
-6-
~ 7~7
I .
/ . ' '.
EXAMPLE 1
Feed materials consisting of Alberta sub-bituminous coal
alone, and also the coal mixed with equal portions of Cold Lake
atmospheric rcsiduum, wcrc processed in a bench scale two-stage
catalytic co-processing unit in accordance with this invention.
Inspection analysis of the Alberta sub-bituminous coal is provided
in Table 1, and analysis for the Cold Lake residuum material is
provided in Table 2. The first and second stage catalytic reactors
were operated at 750~F and 825F temperature, respect~ly, and
2400 psig hydrogen partial pressure and at feed ratios for oil/
coal/recycle liquids as indicated in Table 3. Comparative results
of these operations are shown in Table 3.
TAaLE 1
ANALYSIS OF ABBERTA SUB~BITUMINOUS COAL
Moisture, W Z 8-9
Ultimate Analysis, W /. Dry Basis
Carbon 67.7
Hydrogen . 4.3
Nitrogen 1,.5
Sulfur 0 7
Ash 8.0
Oxygen (by difference) 17 8
tlydrogen/Carbon Atomic Rrelo 0~76
_7_
. ~'~
t .
ANALYSIS Ol COID LAKE RrSlDUUM
Gravlty, ~API 5 2
Uydrogen, W % 10 2
Nitrogen, W % 0.50
Oxygen,W %
Ni~kel, ppm
Vanadium, ppm 240
Weight percent 975F~ ~aterial7~.2
'~ydrogen/Carbon Atomic Ratio1.46
.
y ~
. , .,. --_
~ ~ 8(:~707
I
I
TABEE 3
SUMMARY OF OPERATING CONDITIO~S AND YIELDS
A B
, Condition Coal OnlyCo-Processing
W % Coal Feed 100 50
W % Oil Feed 0 50
Recycle Oil, W % on c081 170 70
First Stage Reactor Feed
W % Co~l 37 37
Oil/coal/recycle0/1/1.71/1/0.7
Feed Space Velocity
- lbs coal/hr/ft' reactor 20 20
- lbs coal+oil/hr/ft' reactor 20 40
First Stage Temperature, F 750 750
Second Stag~ Temperature, F 825 825
Reactor Pressure, psig 2500 2500
Yields, A % Dry Feed
; C4-390F Liquid 260 3 15 7
390-650DF Liquid37.0 25.8
¦ 650-975F Liquid 7.6 27.5
975F~ Resid 2.3 11.8
Unconverted Coal8.1 4.2
Ash 8.0 4.0
H20, CO~ C02 16.0 8.0
NH3 1.5 0.7
H2S 4 2.7
Total (lOO+H2 reacted) 107.3 104.2
C4-975F liquid 64.9 69.0
j Performance Parameters
Coal Conversion, ~ % MAF Coal91.3 91.2
975F~ Conversion, W % MAF Feed88.7 80.4
C4-975F Liquid, ~ % MAF Feed70.5 71.9
Barrels of C~-975F/Metric
lll Ton MAF Feed 5.1 5.2
j'l _9_
~' P
____
~, ,.
7~
From these results, it is seen that for essentially
twice as much feed material being co-processed through
the two-stage-catalytic reactors, the present co-
processing proce~s provides improv2d yields of C4-975F
liqu.ids and reduced yields of C1-C3 gas. Furthermore, it
is pointed out that the yield of C4-975F material is
actually increased.
Example 2
Other similar catalytic hydroprocessing operations
were carried out separately on the Cold Lake atmospheric
residuum material and on Alberta sub-bituminous coal
blended with different ratios of the residuum Eeed and
recycled processed-derived oil. In an alternative
process arrangement, the coal/oil co-processing of Cold
Lake atmospheric residuum and Alberta sub-bituminous coal
was carried out in a once-through operati.ng mode, i.e.,
without recycle of any process-derived liquid, with
results being shown in Table 4.
TABLE 4
CATALYTIC TWO STAGE CO-PROCESSING YIELDS
Alberta Sub-Bituminous Coal/Cold Lake Atmospheric
Residuum
Oil/Coal/Recycle Weight Ratio 1.7/1/0 1/1/0.7
YIELDS, W % M.A.F. Coal Plus Oil
C1-C3 Gas 2.7 3.8
C4-975F Liquid 74.4 71.9
Coal Conversion 92 91
975F ~ Conversion 80 80
Hydrodesulfurization, % 77 87
Hydrodemetallization, %
Hydrogen Efficiency 21 16
C4-975F,
Bbl/Metric Ton Fresh Feed 5.4 5.2
These results show that comparable coal conversion,
975F -~ conversion material and liquid product yields and
hydrodesulEurization were
-- 10 --
i`-'~
.
.2~7
.
~1
/ ¦ achieved by catalytic two-stage hydroprocessing in accordance with
~ the present inv~ntion, as compared to separate catalytic hydrocon-
¦ version of these feed ma~erials. Also, as shown in Table 4, the
1 low tempernture ~irst stage renction zon~ hydrocJenat~s th~ Ee~d coaland oil suf~iclen~ly ~:ue~ h~ u~ oL l~ro~ r.lvc~l rocyc:l o
liquids can be elimin~ted. R~sults Eor the once--throu~ll
l . operatiny mod~ similar to 13xam~1e~ 1 ~oal/o.Ll co ~ro~ lny
¦ ¦ were obtained . EXAMPLE 3
l . '.
Other comparable two-stage catalytic operations were carried
out which shows chc advantage of recycling unconverted coal and ash
solids to the first stage catalytic reactor in this two-stage coal/
oil co-processing process, the results being shown in Table 5.
. .
TABLE 5
TWO-STAGE CO-PROCESSIN~ WITH LIQUID RECYCLE
Coal in Fresh F~ed, W % 50 50
~irst Staee Feed Ratio,
Oil/Coal/Recycle 1/1/0.7 1/1/0.7
~ Recycle Liquid ~sed 550FtFiltered 550F~Liquid
I Liquid Product Containing Solids
Cl-C3 Gas~W % dry coal 3.6 3.8
C4-975F Liquids, W % dry coal 70.5 69.0
Coal Conversion, W % MAF Coal 88.7 91.2 (~2.5)
C4~Liquid, W % MAF Coal Feed 82.1 84.2 (~2.1)
From the results, it is seen that for otherwise equivalent
operating conditions the recycle of unconverted coal and ash solids
,results in approximately 2. 5% increase in the coal conversion
iand 2.1% lncrease in the production of C4~ liquids, based on the
~M.A.F. coal feed.
¦ Although this invention has been described broadly and in
Illterms of certain preferred embodiment thereof, it will be under-
jistood that modification and variations of the process can be made
within the spirit and scope of the invention, which is defined by
I~the following claims. -11-
l _~
. . .