Note: Descriptions are shown in the official language in which they were submitted.
~2~8~%
-- 1 --
The ~resent invention relates to a process of
removing polluants from exhaust gases from burning processes
carried out in the processing of stones and earths, particu-
larly in the production of cement~ In order to comply with
the ecological regulations which are in force, plants for
processing stones and earths are increasingly equippe~ with
means for a removal of gaseous polluants.
In the known wet purifying processes, the exhaust
gases are subjected to dust collec-tion and are then treated
with a sorbent, which is circulated. In that step the
exhaust gases are cooled to such an extent that they must
be reheated to a temperature which is about 20 C above the
dew point temperature in order to avoid damage by corrosion.
As a rule, the reaction products become available at such
a high rate that they cannot be entirely recycled in the
production process. For this reason, wet purifying processes
usually involve high plant costs and operating costs and said
costs often oppose a quick adoption of a purification of
exhaust gases.
In the production of cement it has also been
attempted to subject the exhaust gases to a dry purification
at 300 to 850 C. In that practice it has been found,
however, that
~.
-- 2
at temperatures above 500C the exhaust gases often contain
less then lO0 mg/sm3Of SO2, which is the polluant that is
primarily of interest here, because a major part of the sulfur
that is introduced into the cement-producing process with the
fuel is absorbed by the ground mixture of raw materials in that
temperature range and is incorporated in the clinker.
Such favorable conditions are not obtained in other
burning processes, which are carried out in the processing of
stones and earths and which require a removal not only of the
sulfur contained in the fuel but also of large amouts of other
polluants, such as chlorides or fluorides, from the exhaust gases
whereas the product cannot absorb the polluants in an appreciable
extent.
In the cement-producing process, sulfur is intro-
duced into the burning process also by the ground mixture of raw
materials. Part of said sulfur comes from organic or sulfidic
sulfur compounds, which are decomposed even at low temperatures
so that SO2 is released during the preheating of the ground
mixture of raw materials and S02 contents upn to 6000 mg/sm
may be obtained. At the exhaust gas temperatures which are
usually obtained in the cement process, virtually no S02 is
absorbed by the raw material so that cement-producing plants must
also be provided with an exhaust gas desulfurizer even though the
exhaust gas contains only a small part of the sulfur which is
introduced with the fuel.
Independently of the sorbent employed and of the
ration of the quantity of sorbent to the quantity of polluant, a
dry purification at temperatures from 200 to 400C will not be
particularly effective and usually permits only less then 60% of
the polluants to be removed from the exhaust gas unless catalytic
purifying processes are employed, which are expensive and are not
highly reliable in operation because dust is usually raised at a
high rate.
~2~3~8~2
- 3 -
For this reason it is an object to propose for -the
removal of polluants from the exhaust gases from the burning
processes carried out in the processirig of stones and earths
a process which is generally applicable and which is more
econom:ical than the known processes.
According to the present invention, there is
provided a process of removing polluants from exhaust gases
carried out in the processing of stones and earths,
particularly in the production of cement, wherein the
exhaust gases are contacted at temperatures from 50 to 100C
with a solid particulate sorbent in a solids-circulating
system consisting of a sorbent-containing fluidized bed, a
solids separator and a recycling line and the resulting
product is entirely supplied to a cement-producing process,
wherein said sorbent consists of limestone containing more
than 90~ CaCO3, quicklime, slaked lime or a partly calcined
ground mixture of raw materials which are used in the
production of cement.
In the process in accordance with the invention
the contact time at relatively low temperatures, which is
usually too short in other dry purifying processes, may be
increased as desired, and the reaction product otained can
be subjected to further processing or can be disposed of
whithout a need for special precautions, by which the
economy of other processes is often adversely afEected to a
high degree.
The exhaust gases are suitably at a temperature
between 50 to 100C before they enter the solids-
circulating system. If a tempexature in that range cannot
be obtained, the exhaust gases may be cooled in the
fluidized bed to a temperature from 50 to 100C by means of
sprayed water. Because plants for burning stones and earths
usually comprise a final dust collector, the latter may be
used as a solids separator in the solids- circulating
'1~
-- 4
system.
The main advantage of the process in accordance
with the invention resides in that the sorbent may consist
of materials which are used in the production of cement,
such as a ground mixture of raw materials, limestone
containing more than 90~ CaCO3, quicklime, slaked lime or a
partly calcined ground mix-ture of raw materials and said
materials can subsequently be recycled to a cement-producing
process.
The temperature in -the fluidized bed may be
controlled to an optimum value by a spraying of waterand the
rate at which the sorbent is supplied may be controlled in
dependence on the polluant content of the pure gas.
Further details will be explained with reference
to an example in which exhaust gases from a cement-producing
process are desulfurized and with reference to Figure 1,
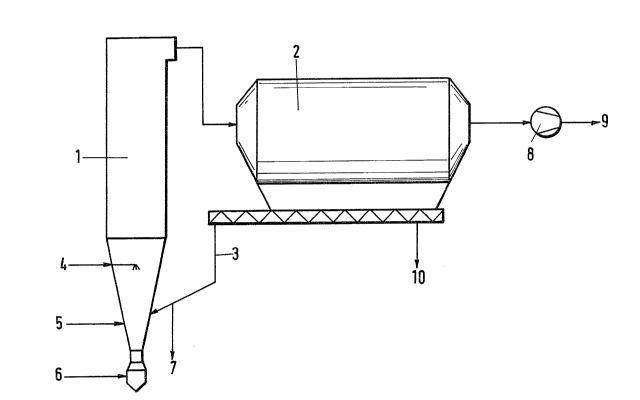
which is a simplified flow scheme of the process.
The solids-circulating system, consisting of a
fluidized bed reactor 1, a solids separator 2 and a
recycling line 3~is supplied through line 4 with water to be
sprayed, through line 5 with sorbent and through line 7 with
the exhaust gas that is to be desulfurized. Exhaust gas
which has been substantially desulfuriæed leaves the
fluidized bed reactor 1 at its top and is subjected to dust
collection in th~ solids separator 2 and is then delivered
via the fan 8 and the line 9 to the chimney. A conveyor
provided below the solids separator 2 serves to convey the
separated dust to the recycling line 3, in which the dust is
finally returned to the fluidized bed reactor 1. Part of
the circulating solid particles is removed through line 7
from the solids- circulating system and is suppl:ied to the
cement-producing process at a suitable location.
Individual components contained in the residual
material, such as heavy metals, may be selectively separated and
discharged through line 10 separately of the remaining substances.
In dependence on the provision of heat-recovering
means (waste heat boilers, water heaters, mills), and on the
nature thereof, the exhaust gases become available at a tempera-
ture from lO0 to 450C. They may contain dust at a rate of as
much as lO0 g/m3 unless they are passed through a mill in that
case their dust content may amount to as much as
lO00 g/sm3 and it may be desirable to effect a preliminary dust
collection by means of an electrostatic precipitator or a mecha-
nical separator so that the circulation of solids in the solids-
circulating system is limited to the optim~m rate for the desul-
furization. In such cases an addition of a separate sorbent may
be omitted entirely or partly. In the solids separator the dust
content of the exhaust gases is decreased to a value below 50
mg/sm3.
In dependence on the composition of the raw mate-
rial the exhaust gas may contain as much as 6000 mg/sm3 SO2.
That So2 content may be decreased by means of the process in
accordance with the invention to values below 50 mg/sm SO2.
That SO2 content may be decreased by means of the process in
accordance with the invention to values below 50 mg/sm3. The
S2 contents which are legally permissible at the present time
are 400 mg/sm3 in Germany and 500 mg/sm in Switzerland.
Another advantage afforded by the process resides
in that the economy can be improved because the f~els used in the
burning processes may contain heavy metals, inclusive of class I
polluants, such a cadmium, thallium, mercury and the like. Sub~
stantially the entire content of such polluants is bonded in the
fluidized bed adsorber to the particularly fine-grained
solid particles and in a solids classifier, e.g. in a multifield
eletrostatic precipitator, can be almost entirely removed sepa-
rately from the other residual materisls and can be removed from
the process independently of those reaction products which are
recycled to a cement-producing process.
The solid particles which are thus removed from the
process contain polluants in a high concentration and constitute
only a small part of the entire quantity of reaction products
which become available. Because said solid particles consist of
stable, water-insoluble compounds, they can be recylcled to the
prodcution process at a suitable location unless such recycling
would adversely affect the product. In the production of cement,
said solid particles may be admixed to the clinker or cement as
it is groud. Alternatively the solid particles which have been
removed fromt he process may be processed for a revovery of pol-
luants in a metallic form or another usable form. 90% of said
polluants can be removed from the exhaust gas by means of the
process in accordance whith the invention and can then be dis-
posed of in an ecologically satisfactory manner.
The process in accordance with the invention hasexperimentally be carried out in conjunction with cement-produc-
ing plant. The raw materials contained 0.3 to 0.4% sulfide, based
on the weight of the raw material, and the SO2 contents in the
partial stream of exhaust gases to be examined amounted to as
much as 3600 mg/sm3 if the process in accordance with the
invention was not used. By the use of the process, the SO2
content has been decreased to values which are dlstinctly beow
400 mg/sm3. In that case the sorbent employed consisted of a
ground mixture of raw materials whith an admixture of slaked lime
in such an amount that the skaled lime provided about 5O% of the
calcium which was stoichiometrically required.