Note: Descriptions are shown in the official language in which they were submitted.
35~ 'r
FIELD OF_INVENTION
This invention relates to the processing of grain
millfeed. In particular this invention relates to a process Por
preparing light coloured purified stable protein and dietary fibre
concentrates from grain mill~eed such as wheat millfeed. The
protein and dietary fibre produced have many possible applications
as ingredients in food systems.
DESCRIPTION OF PRIOR ART
During the milling of grain and in particular wheat,
purified flour and germ are separated from other components of the
grain kernel. These other components are referred to as millfeed, a
term which includes wheat bran and shorts. Wheat millfeed i5
derived from the outer layers of the wheat kernel. The outermost
layer is the pericarp which is comprised of the thick cell walled
epidermis and hypodermis. Immediate]y beneath this layer, is the
inner pericarp which is composed of cross cells and tube cells. To
the interior of these two layers is the seed coat (testa) and the
nucellar epidermis. Immediately below the nucellar tissue is
situated the aleurone layer which is a layer mainly one cell thick
(about 25-75 ~m in thickness). When wheat is milled for flour the
break takes place within the endosperm which is situated below the
aleurone layer, and close to the junction of the two layers.
Therefore the millfeed may contain in addition to all the layers
mentioned above, some endosperm material which is high in protein.
:: ~
'; .: ~ ' ~ ' ' :
.'
. . .
. '` ~ ., . ,:
". .
Because oE the presence of these layers millfeed has superior
protein, vitamin, mineral and lipid content when compared to the
primary product, flour. Millfeed as a by-product constitutes an
important component of the wheat milling industry. Approximately
25~ of the weight of wheat milled across Canada is sold as
millfeed. Literature information on the chemical composition of
bran (the outer layers of the wheat kernel down to and including the
aleurone cell layer) gives proximate analysis values of: moisture
3.7 - 17.7% w/w, protein 11.9 - 2~.9~ w/w, fat 3.0 - 6.8% w/w and
ash 3.8 - 9.6% w/w. The carbohydrate component constitutes about
70% of the total dry weight of the bran. The carbohydrate portion
is composed of hemicelluloses including pentosans (about 45% w/w),
cellulose (about 35~ w/w), starch (about 12~ w/w) and free sugars
(about 8% w/w). The sugars are mainly present in the aleurone
layer. Seventy-five percent of these sugars are sucrose, raffinose
and neokestose with stachyose, fructosylraffinose, glycerol, xylose,
arabinose, glucose, fructose and low molecular weight fructosans,
present at lower levels. In addition to the carbohydrates, lignin
is another component of the bran which can be present in quantities
up to 10~ w/w of the bran. Other components include vitamins and
minerals.
.
The digestible components of millfeed ~protein, vitamins
and lipids) have significant nutritive value in addition to the food
value of flour, the primary product of wheat milling. In recent
years it has been demonstrated that the indigestible components of
~illfeed are also of great value in the human diet. Four groups of
compounds present in millfeed are important components of dietary
-~ '
- ~ 3 --
.
~ ~r~
fibre in terms of their function in human metabolism. These
compounds are cellulose, hemicelluloses, pectins, and lignin. Each
of these components plays an intricate role in human metabolic
function. For example lignin has been shown to have beneficial
metabolic effect through its ability to combine with bile salts
which are secreted into the duodenum. Subsequently this action
prevents the resorption of these salts in the large intestine. This
in turn is thought to result in a lowering of blood cholesterol
levels. In spite of the superior quality components of millfeed, it
is generally ignored as a human food source and sold as animal
feed. This is probably due to the poor palatability of millfeed.
It is known to be possible to further process millfeed into
potentially higher value food products by alkali extraction of the
protein from the millfeed. The practice of using alkali for
dispersing plant proteins is known and has been used to produce soy
protein concentrates and isolates. Alkali extraction of oats is
also documented.
,
The first study (Fellers, D.A. et al. 1966, Solubilization
of Recovery of Protein from wheat Millfeeds. Cereal Chemistry 43:1)
of the applications of wheat bran and millfeed included
solubilization of the protein at alkaline pH, centrifuging to remove
the non soluble matter and acid precipitating the dissolved
protein. Only about 65~ w/w of the total protein was dissolved and
a yield of approximately only 45% w/w was obtained. The protein
-- 4 --
. .
~ ~15~7~
derived was dark in colour and it is not known how acid
precipitation altered its functional properties. Several other
studies have been made since but none has greatly improved upon this
study. Attempts have also been made to reduce the colour of protein
concentrates obtained by acid precipitation. Fellers et al. 1966
suggested the use of solvents namely ether, acetone or ethanol to
decrease the colour. This resulted in a light gray product.
Johnson and Anderson in U.S. Patent 3,127,388 used hydrogen
peroxide with heat on alkaline slurries, prior to alkaline
extraction and acid precipitation of protein. This only lightened
the colour to straw yellow, at the expense of decreasing the
protein yield. Increased protein yields and purity have been
reported for some vegetable proteins namely soybean (Okubo, K. et
al. Preparation of Low Phytate Soybean Protein Isolate and
; Concentrate by Ultrafiltration. 1975 Cereal Chemistry 52~263) and
sunflower (Maubois et al U.S. Patent 3,993,636) when ultrafiltration
was used as the method of protein concentration in place of acid
precipitation.
~'
The utilization, as a source of dietary fibre~ of the
fibrous residue remaining after the extraction of protein from
millfeed has not been investigated. Indeed the main efforts so far
have only been concerned with the extraction of the protein fraction
Erom millfeed with no interest in the use of the remaining fibrous
residue in human diets. Although it had been suggested as early as
1975 (Saunders et al. Cereal Chemistry 52:93) that the fibrous
residue could be used as a ruminant feed, only recently (Roberts et
-- 5 --
~ ` `
,: ,
~, ' . .
~ ., .
:
al. J. Sci, Food Agric. 1985 36:5) has it been suggested that the
protein depleted bran would be useul in the formulation of high
fibre based products. Great interest has recently been generated in
the use of dietary fibre in foods~ Numerous investigations have
shown that dietary fibre plays a very important role in human
nutrition and metabolism.
SUMMARY OF INVENTION
It is an object of the present invention to provide an
economical and efficient process for preparing light coloured
purified stable protein concentrate from grain millfeed.
It is a further object of the present invention to provide
an economical and efficient process for preparing light coloured
dietary fibre concentrate from spent grain millfeed residue after
extraction of the protein.
It is yet a further object of the present invention to
provide an economical and efficient process for preparing light
coloured dietary fibre concentrate directly from grain millfeed.
It is still a further object of the present invention to
provide an economical and efficient integrated process for preparing
light coloured purified stable protein and dietary fibre
concentrates from grain millfeed.
- 6 -
;'
These and other objeets o~ this invention have been
aeeomplished by the processes hereinafter summarized and deseribed
in detail.
The integrated process comprises the treatment of millfeed
with alkali solution and the separation into two proeess streams
with the inal produet from one stream being bleached protein
eoneentrate and bleached dietary fibre eoneentrate from the other.
Protein extraetion is aeeomplished by utilizing millfeed
having redueed partiele size, an appropriate extraetion pH range
reeovery of the solubilized protein by ultrafiltration and
decolourization of the protein concentrate with hydrogen peroxide.
These conditions result in a marked inerease in solubilization of
the protein (in exeess of 75% w/w) and recovery (in excess of 55%
w/w of the protein in the millfeed) in the form of light coloured,
pwlatable ood grade protein eoncentrate containing approximately
70% w/w protein.
,
As a separate stream the fibrous residue is treated to
produce a light coloured product containing in excess of 85% w/w
dietary fibre that ean be used as an ingredient in food products.
In particular the present invention provides a process for
preparing bleached purified stable protein coneentrate from grain
millfeed, comprising the steps of: eombining grain millfeed of
redueed partiele size with an alkali solution to produee a slurry
'
_ ~ '^j 7 ~
,
' . .,' ' ' :
':
3S~'7~
with solvent/millfeed ratio in the range of 7:1 to 10:1 v/w and
having pH in the range of pH 9-11; separating the alkali liquid from
the millfeed residue to isolate alkali extract; removing starch and
fat from the alkali extract; ultrafiltration of the alkali extract
to form a retantate using a semi-permeable membrane; bleaching the
retantate by adding hydrogen peroxide and heating; cooling and
drying the retantate to provide a light coloured protein concentrate.
The invention further provides a process for preparing
bleached dietary fibre concentrate from spent grain millfeed residue
from which protein has been extracted comprising the steps of:
adding water and 35-70% w/v hydrogen peroxide to said millfeed
residue to produce a slurry; heating said slurry to a temperature of
50-100C; decanting the liquid and drying the residue to produce a
light coloured dietary fibre concentrate.
The invention still further provides an integrated process
for preparing bleached puriEied stable protein and dietary fibre
concentrates from grain millfeed comprising the steps of: mixing
grain millfeed of reduced particle size with an alkali solution to
produce a slurry with solvent/millfeed ratio in range of 7:1 to 10:1
v/w and having pH in the range of pH 9-11; separating the alkali
liquid from the millféed residue to isolate alkali extract; removing
starch and fat from the alkali extract; ultrafiltration of the
alkali extract to form a retantate using a semi-permeable membrane;
bleaching the retantate by adding hydrogen peroxide and heating;
- 8 -
: :
:~' ' ' ~ ,. , '
' ;~'
~ ' .
3S17~
cooling and drying the retantate to provide a protein concentrate;
adding water and 35-70% w/v hydrogen peroxide to said millfeed
residue to produce a slurry; heating said slurry to a temperature of
50-100C; decanting the liquid and drying the residue to produce a
light coloured dietary fibre concentrate.
The invention still further provides a process for
preparing bleached dietary fibre concentrate from grain millfeed
comprising the steps of: mixing grain millfeed of reduced particle
size with an alkali solution to produce a slurry with
solvent/millfeed ratio in the range of 7:1 to 10:1 v/w having pH in
the range of pH 9-11; separating the alkali liquid from the millfeed
residue to isolate alkali extract; adding water and 35-70% w/v
hydrogen peroxide to said millfeed residue to produce a slurry;
heating said slurry to a temperature of 50-100C; decanting the
liquid and drying the residue to produce a dietary fibre concentrate.
The invention still further provides for light coloured
purified stable protein and dietary fibre concentrates produced by
the aforesaid processes.
DESCRIPTION OF INVENTION
_
The following is a description by way of example of certain
embodiments of the present invention, reference being had to the
accompanying diagram in which:
-- g _
'' ' ~ - ~
:
s~
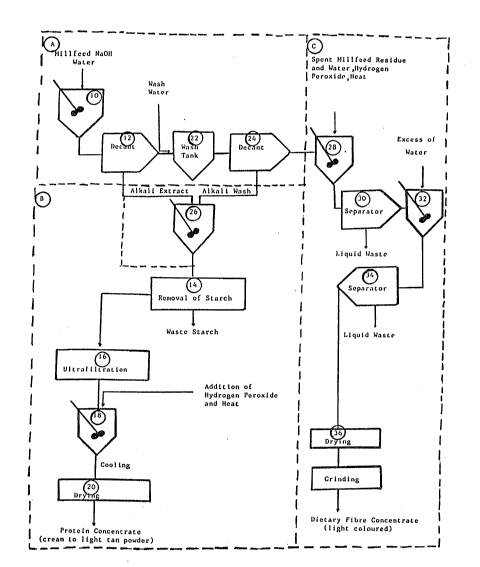
Figure 1 is a Elow diagram oE the total integrated process.
Referring to Figure 1 the total integrated process is
represented by the processes set out in boxes A, B and C.
A combination of shorts together with finely ground fine
and coarse wheat bran in proportions produced under normal milling
conditions is used. Millfeed from grain other than wheat may also
be used as raw materials for the proc:ess. The raw materials are
passed through a grinding roll and screened so that the particles
used for the extraction are less than 1680 um (micrometers) in
size, The reduced particle size provides for higher extractions.
Coarser particles provide lower yield. Still finer particles would
give better yields but are difficult to obtain economically. As
shown in Figure 1 the millfeed is then combined (10) with alkali to
obtain a pH in the range pH 9 to 11. A pH outside of this range
diminishes extraction. Sodium hydroxide is the preferred alkali but
ammonium or potassium hydroxide or other alkali may also be used.
It is preferred that the alkali is dissolved in water prior to the
addition of the millfeed. The desired ratio of solvent to millfeed
is in the range of 7:1 to 10:1 v/w. Agitation of the resulting
slurry is continued for about 45 to 90 minutes with the temperature
maintained between 20 to 25C. The pH is monitored throughout the
process and additional alkali is added if the pH falls below the
range pH 9-ll. After the 45 to 90 minute contact time between the
millfeed and the alkali solution, the spent millfeed is separated
(12) from the liquid extract by known mechanical means such as a
- 10
A
:
decanter centriEuge, filter press or screwpress. The residue is
washed with water and the alkali wash of residue along with the
liquid extract is subjected to centrifugation (14) or other process
to remove the starch and fat. The starch-free or clarified protein
liquid is subjected to ultrafiltration (16) by passing it over a
semipermeable membrane haviny a nominal molecular weight cut-off in
the range of 5,000 to 25,000 and preferably between 8,000 to 15,000,
at a temperature in the ranye of 10C to 35C. The pressure of the
liquid entering the ultrafiltration module is between 40 to 60 psi
and that leaving the module between 20 and 40 psi. Ultrafiltration
is continued until the nitrogenous matter content of the retantate
is 70~ w/v (N X 6.25) or greater of the total dry matter o~ the
retantate. The resultant purified and concentrated protein liquid
is decolourized or bleached (18) by the addition of hydrogen
peroxide at a level of 1 to 4~ w/v, and by heating the mixture to
between 50 and 65C and maintaining it at that temperature for
between 120 and 180 minutes. After cooling to 2C the mixture is
held for a period of up to 24 hours. At this time, the pH of the
protein liquid is adjusted to a pEI of 6.5 to 7.0 with a food grade
organic acid. The pH adjusted protein liquid is then dried (20) by
spraying or other means to give a cream to light tan powder protein
concentrate containing 5-8% w/w moisture. This process stream is
represented by boxes A and B of Figure 1~
:
In a second process stream, represented in boxes A and C of
Figure 1, the residue remaining after separation (12) from the
liquid alkali extract is washed (22) with water and subjected to
.~ .
. ~ .
, ~
:~
: :
, :' '
centrifugation (24) to produce an alkali wash. The alkali wash is
added (26) to the a]kali extract in the process stream leading to
the protein concentrate (Box B). Returning to the second process
stream, the wet spent grain milleed residue from which protein has
been extracted is decolourized or bleached (28) by adding water and
35% to 50% w/v hydrogen peroxide to form a slurry which contains
10-15% w/v residue solids and l to 1~% w/v hydrogen peroxide. The
slurry is heated to 50-100C and held for 30 to 40 minutes. The
liquid is then decanted (30) and the bleached residue is washed (32)
with an excess of water and decanted (34) again. The collected
bleached product is then dried (36) at temperatures o~ 40-80C to
moisture levels of 8 to 12~ w/w, to produce a light coloured dietary
fibre concentrate.
It will be readily apparent to those skilled in the art
that various components of the integrated process (Boxes A, B and C)
may be independantly utilized. Three component processes of the
integrated process are represented by boxes C, A plus B and A plus C.
- 12 -
. .
'-
Example l: Laboratory Scale Production of Light Coloured Wheat and
Dietary Fibre Concentrates from Milleed.
A 2 kg sample of wheat millfeed analyzed as having 15.58%
w/w protein and 12.29% w/w moisture was slurried in 14 kg of water
in which 30 g of sodium hydroxide had been dissolved. This was
agitated for 60 minutes at 23C. The insoluble residue was
separated from the soluble protein/starch liquid. To remove any
residual protein/starch liquid from the insoluble residue, 7 kg of
water was added to the residue and it was stirred for 30 minute.s.
The soluble portion was again separated as before. The above
process gave 3779 g of insoluble residue with a protein content of
1.90% w/w. The remainder of the initial millfeed protein was found
in the protein!starch liquid and represented 77.0% w/w of the
original milleed protein. To further purify the soluble components
of the millfeed, the liquid was centrifuged to remove the starch.
This resulted in 18,863 g of protein liquid with a protein content
of 1.25% w/v giving a recovery of 75.67% w/w o~ the original
millfeed protein. Further purification o the protein was obtained
through ultrafiltration which gave 3116 g of concentrated protein
containing 6.3% w/v protein. From the above data, the laboratory
process as stated recovered 63% w/w of the original protein in the
millfeed. The colour of the protein concentrate was lightened to a
cream colour by the addition o 97 ml of 50% w/v H22 and heat.
After a temperature of 65C was reached, the liquid was held at this
temperature for a period of 3 hours. During this holding period it
was agitated periodically. The lightened liquid was cooled rapidly
and held at 2C overnight prior to pH adjustment down to 6.8 with an
- 13 ~
'
- : :
'
- ': ' ':
- ~ , . - ~ .
.
organic acid.
The second stage of the process resulted in the production
of a residue which was high in dietary fibre (87~ w/w, dry basis)
and light in colour. To the 3779 g of wet residue, 624 ml of 50
w/v H22 and 6991 g of H2O was added. This was heated to a
temperature of 100C and held for 1 hour with continuous agitation.
When the reaction was complete, the bleached insoluble residue high
in dietary fibre was separated from the liquid portion and washed
thoroughly with an excess of water and separated again. The dietary
fibre concentrate was dried in a convection oven at a temperature of
60C to a moisture content of 8% w/w.
~ ,
~ ,
~ ~ '' ' ' ~ , ..
,
' . :
: ~ ,
~ 3~
Example 2: Batch Pilot Scale Production oE Light Coloured Wheat
Pxotein and Dietary Fibre Concentrates from Millfeed.
Wheat millfeed having a protein content of 16.32~ w/w and a
moisture content of 12.89% w/w was employed. A 150 kg quantity of
the millfeed was slurried in 1050 kg of water to which 2.25 kg of
sodium hydroxide pellets had been added and brought into solution.
The slurry was agitated for one hour at 21C. Ater the extraction
period, the slurry was centrifuged to remove the protein liquid and
the starch from the insoluble residue. The insoluble residue was
agitated with 500 kg of water and centrifuged as before. The wash
from the insoluble residue was added to the protein/starch liquid
from the first separation and the remaining insoluble material was
decolourized as described in the second stage of process. A further
centrifugation step of the protein/starch liquid resulted in a
"starch-free" protein liquid. The protein liquid had a total weight
of 1360 kg and contained 2.74% w/v total solids and 1.30~ w/v
protein. From the above data, the process example as stated
provided 72.2% w/w of the protein as an extract from the original
wheat millfeed. This was sub]ected to ultrafiltration to further
purify and concentrate the protein. The result was 230 kg of
concentrated protein liquid which was 6.0~ w/v protein giving a 56%
w/w recovery of the protein in the initial wheat millfeed. To this
protein li~uid, 17.7 kg of 35% w/v hydrogen peroxide was added and
heated to 65C with continuous agitation for 3 hours, cooled to 2C
and held overnight. The pH was lowered with an organic acid to 6.8
and spray dried to yield a protein concentrate which was cream to
light tan in colour. For the second stage of the process, to the
- 15 -
- -
. ' .
.
35~72
325 kg of insoluble wet residue at 70.98% w/w moisture level, 103.8
kg of 35% w/v H22 and 388.19 kg of H2O were added. The mixture
which contained 94.31 ky wet residue solids and 36.33 kg of H2O2
(4.45% W/V H2O2) was heated to 100C and held at this temperature
for 60 minutes. The pH of the reactions mixture was 6.3 - 6.7. The
liquid was decanted and the bleached residue was washed with an
excess of water and decanted again. The collected bleached product
was then dried in a convection oven at temperatures of 45~50C to a
moisture level of approximately 10% w/w.
.~
.
- 16 -
'
:
~'
~: