Note: Descriptions are shown in the official language in which they were submitted.
~3122~
MEDICATICN XNHALER WITH WARNING
Related Patents and Applications
The present invention is related to that disclosed and clai~d in Nowacki and
Brisson in United States Pa~ent 4,470,412 and forms an improvement thereon.
Background of the Invention
A person suffering from asthma and experiencing an asthmatic attack may have
rather considerable difficulty in breathing, due to swelling in the bronchi and
due to secretion of mucus. There are various antiasthmatic pills that are
effective, but which generally are somewhat slow-acting. There are also medi-
cations available for intravenous treatment which work quite rapidly, but which
require administration by skilled medical personnel. For most asthmatic
sufferers, the promptest, immediately available relief is by way of an
inhalant. Epinephrine or other suitable asthmatic medication is packaged with
a suitable diluent in a small pressurized canister or cartridge which interfits
with a mouthpiece. The patient places the mouthpiece in his mouth, and
depresses the cartridge, thereby releasing a measured amount oE medication
which is inhaled through the mouthpiece.
Som~ patients do not inhale properly, and the mouthpiece may not be completely
effective in oooperation with the cartridge to convert the medication into a
mist which is deposited in the prcper bronchial area to relieve the asthmatic
attack. Often there are small droplets of medication, rather than a mist, and
this may be compounded by improper inhalation which results in ~uch of the
medication simply going into the throat and stomach where it is ineffective
against the asthmatic attack~
~3~224~
1 ccordance with U.S. Patent 4,470,412 mentioned above, a cylindrical chamber
is provided which has at one end a plastic or an elastomeric adapter which
receives the mouthpiece of the cartridge device. The opposite end of the
cylindrical chamker is provided with a mouthpiece or equivalent, and a one-way
valve in the chamber adjacent the downstream or mouthpiece end precludes back-
flow upon exhalation by the person suffering the asthmatic attack. Passage of
the medication through the chamber completes formation of the medication
released into the desired mist.
Some sufferers tend to inhale too strongly~ This can cause leakage of air into
the chamber, and resulting dilution of the medication~ This is undesirable.
It it also possible that medication would be drawn through the bronchi too
rapidly, thu~ not having an opportunity to deposit and take effect in the
proper areas. Inhalation should ~e at a controlled rate so that substantially
all of the medication will be effective in the bronchi, and not carried into
the lungs where it does not do any good. Although inhalation by a person
suffering from an asthmatic attack may be difficult, panic may nevertheless be
such as to cause inhalation that is too rapid.
Objects and Summary of the Invention
In accordance with the present invention, an asthmatic or the like medication
inhaler is provided having a warning device to alert the asthmatic sufferer if
he inhales too vigorously.
Thus, the principal object of the present invention is to provide an asthmatic
medication inhaler which signals a warning to the asthmatic sufferer if he
inhales too vigorously.
--2--
.~.3~.2.~
is a further object of the present invention to provide sueh a medication
inhaler having an auditory warning, speciEically an auditory warning ~hieh has
a pleasant tone, but which is readily discernible to the asthmatic sufferer.
In carrying out the foregoing and other objects and advantages of the present
invention, an air operated auditory warning device is incorporated in the plas-
tic or elastomeric adapter at the entering end of the chamber. The auditory
warning device comprises a plastic molding having an air passageway partially
closed at the downstream end by an integral tongue or reed. Any upstream move-
ment of air through the device would have no efEect on the reed. However, if
the asthmatic sufferer inhales too vigorously, then a sufficient lowering of
pressure in the medication inhaler chamber will occur to cause drawing of air
through the auditory device, t~ereby causing the reed to vibrate, and generat-
ing a musical tone, very similar to the air induced vibration of a harmonica
reed.
The Drawinqs:
The invention will best be understood with re~erence to the
following text when taken in connection wi~h the accompanying
drawings wherein:
Fig. 1 is a perspective view of a medication inhaler with warning as con-
structed in accordance with t~e present invention, shGwing the use thereof;
Fig. 2 is a side view, partially in section, of the inhaler;
Fig. 3 is an enlarged, fragmentary side view of the medication inhaler of the
present invention, partially in section, showing the movement of air and medi-
cation therethrough;
Fig. 4 is an end view taken from the left end of Fig. 3;
--3--
~ ~2~
~ S is a cross-sectional view taken subs~antially along the line 5-5 in Fig.
3;
~ig. 6 is a cross-sectional view taken substantially along the line 6-6 in Fig~
3;
Fig~ 7 is a perspective view showing the diaphram or valve of the medication
inhaler; and
Fig. 8 is a perspective view showing the auditory warning device in juxta-
position to the rubber or elastomeric ~dapter in which it is inserted.
Detailed Disclosure of the Illustrative Embcdiment
Reference should first be made to Fig. 1 and 2 wherein there will be seen a
prior art pressurized canister or cartridge 10 , sometimes referred to as a
nebulizer which is charged with epinephrine or other suitable antiasthmatic
medication and a suitable diluent, and under pres6ure. m e cartridge fits into
the receiving end of a right angle mouthpiece 12, the opposite end of which is
designed to be placed in the asthmatic sufferer's mouth. The cartridge is
pressed down, being sgueezed between the index finger and the thumb underlying
the mouthpiece. This causes a valve stem in the cartridge to press against a
reaction base in the mou~hpiece to discharge a measured quantity of medication
into the mouthpiece. The discharge is supposed~o be in the ~orm of a mist, but
in fact often contains small droplets~ The patient inhales, and the mist
passes into the m~uth, and hopefully into the bronchial tubes to provide asth-
matic relief. The patient is then supposed to hold his breath for a short
time, and subsequently to exhale slowly through nearly closed lips. However,
as heretofore noted, some of the medication may si~ply be in the form of drop-
~4--
~3~2~
;, not mist, and the droplets generally do not reach the bronchial tubes
to effect their intended p~rpose.
In accordance with U.S. Patent 4,470,412, mentioned above, the drops can bebroken up into a mist, and the patient can be more or less forced to inhale
properly with the use of the medication inhaler shown in such patent, and
detailed hereinafter
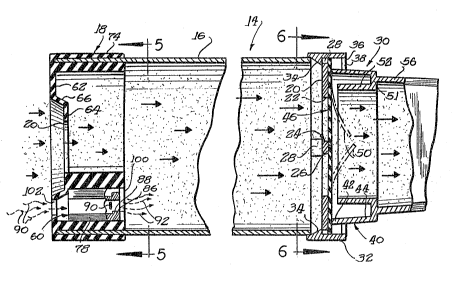
The medication inhaler is shown generally in Figs. 2 and 3, and attention
should no-~i be directed to these two figures. The medica~ion inhaler is indi-
cated generally by thenume~al 14 and includes a cylindrical body 16 molded of
semi-rigid plastic resin. The chamber is open at both ends. A resinous plas-
tic or elastomeric adapter or fitting 18 is received on the entering or up-
stream end of the chamber and includes a central opening 20 adapted resiliently
to grip the horizontal portion of the mouthpiece 12 which is intended to be
placed in a person's mouth. Further details of the adapter or receiver 18 will
be sent forth in greater detail hereina~ter.
At the opposite end of the cylindrical chamber 16 there is a right angle
inwardly extending flange 20 integral with the side wall of the cylinder and
defining a central circular aperture 22 having a spider 24 (Fig. 6) there-
across and comprising equally arcuately spaced legs 26. m e flange 20 and
adjacent sidewall of the cylindrical chamber 16 are provided with four equally
ately spaced recesses or notches 28u An end cap 30 is provided at the
downstream end (the E~ht end in Figs~ 1-3) and is m~lded of the same plastic
resin material as the cylindrical chamber. The end cap 30 includes at the left
end thereof a cylindrical flange of proper diameter to embrace the open end of
the cylindrical cartridge 14. The flange 32 is provided with inwardly directed
cam teeth 34 for snapping into the notches 28 to secure the end cap 30 on the
_5_
~3~22~
c ndrical chamber 16. A radial flange 36 extends inwardly from the right
edge of the cylinder 32 and is molded integrally therewith. apertures 38 are
formed in this flange to provide enhanced flexibility in the area o~ the teeth
34, and also to provide for clearance for various portions of the m~ld. A
generally cylindrical, but slightly tapered wall 40 is formed lntegrally with
the f~ange 36, and extends to the left thereof at 42, and also through the
right thereof at 44.
A flat plastic or elastomeric diaphram 46 (see also Figs . 6 and 7) having a
circular periphery fits within the cylinder 32 of the end cap 30 and is held
against the flange 20 by the leftwise projection ~2 of the wall 40. This
leaves the oe nter portion of the diaphram entirely free, except that it is
backed by the spider 24. The diaphram is provided with a pair of right angle,
intersecting slits 48 that are backed by tw~ of the spider legs 26. When one
uses the inhaler as will be describ~d in greater detail later, the diaphram
deflects to the right as shown in broken lines at 50 in Fig. 3, to open the
slits, and to allow air and medication to pass freely past the diaphram. How-
ever, if one attempts to exhale through the inhaler, the diaphram presses
tightly against the spider 24 and air cannot pass to the left of the diaphram.
A flange 50 extends inwa~dly from the right end of the tapered cylindrical wall
40 and has integral therewith a cylindrical wall 52 extending to the left.
This wall terminates short of the diaphram 46. A pair of diametrically spacèd
apertures 54 (Fig. l) are provided in the wall 51 so that if one attempts to
exhale thrcugh the inhaler, the exhaled air passes around the left end of the
cylindrical wall 52 and back and out through the openings 54O
The end cap 30 is completed by a ~apered extension 56 whi~h is adapted to be
received in a person's mouth, and which therefore is wider than it is high.
--6--
13~22~
P h the side walls and the top and bottom walls are arched, being convex out-
wardly. The entire end cap is a single, one-piece r~lding. A cover cap 57 may
be provided for sanitary purposes to cover the portion of the extension 56 that
fits in a person's m~uth, and to this end the wall is notched at 58 for best
positioning of the cover cap.
Attention now should be directed to Figs. 2 - 5 and 8 wherein the adapter or
receiver 18 will be seen in greater detail, along with the sonic signalling
device 60 forming the subject matter of the present invention. The adapter 18
comprises a front wall 62 adapted to abut the inlet end of the chamber 16~ The
front wall 62 is provided with a central thinner portion 64 which is offset at
66 inwardly (in a downstream direction) and i~ prcvided with the central appur-
ture 20 which is slightly smaller than the portion of the mouthpiece 12
designed to be received in the mouth. This shape includes vertical side walls
70 and outwardly arcuate top and bottom walls 72. The adapter 18 further
includes a peripheral flange which extends axially of the chamber 16 and
e~braces the outer end thereof. This flange is circular, and an inner-circular
flange 76 engages the inner wall of the inlet end of the chamher t6.
The sonic or audible signalling device 60 comprises a one-piece molding of
polycarbonate plastic resin material. m e signaling device is shown as m~unted
in t;le lower part of the adapter 18, but it could be in the upper part. It is
described as positioned in the lower part for reference purposes. The si~na~l-
ling device comprises a bottom wall 78 which is ~ylindrical in shape and con-
forms to the inner wall of the flange 76. An outer wall 80 is also of cylin~
drlcal ~hape, but of sllghtly less r~d~us than the bottom wall 72 of the appur-
ture 20. The inner or upper wall 80 is narrower than the wall 78, and lateral
flat wall extensions 82 extend outwardly to vertical side walls 84~ Longitu-
dinal ribs 87 and 88 are respectively formed on the outer and inner walls 78
and 80 centrally thereof for reinforcement, and to direct airflow.
~122l~
The signalling device is ~pproximately 7/16 inch long, and at the exit end
(the right end in Fig. 3, and the end viewed in Fig. 8) there is a vertical
wall 86 which is substantially 3/32 inch thick, and which is provided with a
rectangular oriface 88. At the inner face of the wall 86, there is provided an
integral reed 90 which is joined at one end to one of the side walls 84, and
which is spaced at the opposite en~ and along the sides from the margins of the
oriface 90. The reed is approximately 1/2 inch long, and slightly less than
1/16 inch wide. If air passes from the inlet end of the sonic device 60 and
through to the outlet end as indicated by the arrows 90 and 92, respectively,
the reed will vibrate. When the reed vibrates it generates a musical tone
similar to that generated by a harm~nica, approximately at a note of C one
octave above middle C. This provides a pleasant tone which is not annoying to
those who might overhear it, yet one which is of somewhat higher pitch than
most ambient sounds, thereby being readily heard. If air is forced through the
sonic devioe in the opposite direction, no sound is generated.
The inner face of the flange 76 and extending rearwardly from the wall 64 are
provided with side walls 94, and inner arcuate wall 96, and lateral extension
walls 98 which form a cavity 100 w~lich snugly receives the sonic device 60.
The wall 62 is provided with a retangular aperture 102 leading into the inlet
end of the sonic device 60.
When the shaped end 62 of the outlet fitting 30 is placed in the patient,'s
nouth, the patient inhales, and medication is drawn from the mouthpiece 12 upon
depression of the cylinder or canister 10 which emits a measured amDunt of
medication. This medication is thoroughly misted within the chamber 16 before
entering the patient's m~uth, and ultimately his bronchi and lungs. An adult
patient should inhale the mist at a rate of less than 30 liters per minute. If
he inhales at greater than 30 llters per minute, the medication is not fully
~8--
~3~22~l~
~ctive, as much of it is drawn past the bronchi and into the lungs where it
has no effect on an asthmatic attack. If a patient does inhale at greater than
30 liters per minute, then the pressure differential between the inside of the
device 14 and the outside becomes sufficiently great that air is dr~wn through
the sonic device 60 to cause the reed 90 to vibrate and generate a sound which
is readily discerned by the patient or b~ an overseeing medical person. This
alerts ~he patient and any overseeing medical person to have the patient slow
down his inhalationO
The reed does not generate sound with reverse flow, as previously noted, but no
reverse flow i5 to be e~pected, since if the patient exhales through the device
14 the diaphram 46 will be pressed against the arms of the spider 24 to close
the slits 48 thereof, the exhaled air then being diverted past the forward end
of the wall 52 and exiting through the openings 38 and 54.
The specific example of the invention as herein shown and described is for
illustrative purposes. Various changes in structure may occur to those skilled
in the art, and will be understood as forming a part of the present invention
insofar as they fall within the sphere and scope of the appended claims.