Note: Descriptions are shown in the official language in which they were submitted.
~32~
HEAT_ENGINE, REFRIGERATION AND HEAT P~P ~LE~
APPROXIMATING T~ ARNOT CYCLE AND APP_RA~US T~EFOR
_ACKGRoUND ~_INTROD~TION
Thls invention r~lates to processes and
apparatus, including novel compressors and ~xpanders, by
means of which improved high efficiency vapour cycles
such as Carnot heat engine, refrigeration and heat pump
cycles can be approximated in actual practice.
In essence, the Carnot heat engine cycle is
composed of four ideal processe6: a) isothermal (zero
temparature differsnce) working fluid heat additlon at
the desired high temperature, b) i~entropic working fluid
expansion (work production~, c) isothermal (zero
temperature difference) heat re~ection at the desired low
temperature and d) isentropic working fluid compression
(work absorption).
Carnot refrigeration and heat pump cycle
approximations are also possible, a~ outllned later. For
clarity, most of the background disaussion which follows
is based on the Carnot heat engine cycle.
Until now, the most energy-efficient heat
engine cycle, the above-described Carnot cycle, has been
considered merely a theoretical basis upon which to
evaluate other practical heat engine cycles and real
; 25 machinery. This is poignantly outlined in the following
quotation rom the "Mechanical Engineer's Reference
Book", Butterworth Publishers, Boston, 11th ~dition,
1986:
... .
"The aycle for the ideal hsat engine i8 known
~ 30 as the Carnot cycle, but has little use in real
< plants as it is not composed of the steam or
gas prOCeBSe6 which are found ~uitable for
~' ~
i;`
,
,` ~, ' ' ' ' ' :~ .
~L3239~
,~
-- 2
practical machinery."
"The thermal efficiency of the Carnot cycle is
of use to the engineer as it gives him the
maximum value that he could attain between
given temperature limits".
Partly becau6e the Carnot aycle, until now,
could not itself bs actualized or alosely approximated,
other heat engine conver6ion cyales have been developed.
These heat engine cycle~ have been primarily based upon
the actual machinery and working fluidæ that were
available. For example, the Otto cycle is approximated
in practice by the spark ignition engine and the Die~el
cycle by the compression-ig ni t i o n engine. The
theoretical heat conversion cycle that is most similar to
the Carnot cycle is the Rankine cycle; it is approximated
in such applications as ~team power plants. Con~ider the
following passage from a college thermodynamics text
book, " Thermodynamics", G. J. Van Wyler, Editor, J. Wiley
& Sons, Publishers, 1962:
"... It is readily evident that the Rankine
cycle has a lower efficiency than the Carnot
cyale with the same maximum and minimum
temperatures as a Rankine cycle, because the
average temperature of heat addition is below
~5 the temperature of evaporation. The ~ue~tion
might well be aæked, why choose the Rankine
cycle as the ideal cycle? Nhy not rather
6elect the Carnot cycle? At least two reasons
can be given. The first involves the pumping
process. Great difficultie~ are encountsred in
building a pump that will handle a mixture of
liquid and vapour (aoming from the low
temperature isotherm--the condenser) and
deliver only saturated heated liquid (to the
high temperature isotherm--the boiler). It is
.
.
: . , -. :
',' ~1 ,. ': , ' '
'i ~ : . ' ~ '
~' ~3239~
- 3 -
;
much easier to completely condense the vapour
and handle only liquid in the pump, and the
Rankine cycle is based upon this fact. The
second reason involves superheating the vapour.
In the ~ankine cycle, the vapour is superheated
at constant pre~sure. In the Carnot cycle, all
the heat transfer iB at aonstant temperature,
and thexefore the vapoux is superheated
(aæsuming zingle-phase working fluid).
However, during this process, the pressure must
drop, which means that the heat must be
transferred to the vapour as it undergoes an
-, expansion process in which work is done. This
is also very difficult to achieve in practice.
Thus, the Rankine cycle is the ideal cycle that
can be approximate~ in practice".
The above conclu6ion, that for practical
reasons ons must resort to the lower efficiency RanXine
heat engine cycle rather than the Carnot cycle, has been
a persua~ive one and the classical approach to the Carnot
cycla has discouraged most people from even attempting to
closely approximate thls ideal cycle. Similar
considerations have applied in respect of refrigeration
and heat pump cycles.
BRIEF SUMMARY OF INVENTION
The present invention, which, as will be seen
,! hereafter, involves the "marriage" of innovations in
controlling and accommodating the physical phase
composition of the working fluid with new and innovative
high efficiency machines (expanders and compressors),
'~ makes possible a reaæonable approximation to the Carnot
.~ cycle in respect of hsat engine, refrigeration and heat
~ pump applications.
i,~ Accordingly, one aspect of the present
, 35 inventlon provides prooes~ and apparatus by mean~ of
J
!
i
,.':' -
:' ' ~,~
~323~
which the Carnot oycle can be approximated in practice.
The invention involves the application of novel energy-
efficient, mixed phase, high volume/ratio fluid-handling
expanders and compressors to a single-component working
fluid that exiæts as a mixture of fine droplets of
saturated liquid in saturated vapour. This combinatio n
of fluid-handling expanders and compre6~0rs with the
saturated mixed-phase working fluid enables the
approximation of isentropic saturated liquid/vapour
expansion and compression. These process approximations,
in addition to lsothermal heat addition and rejection,
enable Carnot heat engine, re~rigera~ion and heat pump
cycles to be approximated.
Further, according to another aspect of the
invention, improvements over the novel high efficiency,
high voluma ratio compressors and expanders of the
constrained vane variety illu~trated, e.g. in U.S.
Patents 4,299,097 and 4,410,305 include the provision of
unique compressor/expander chamber shapes, as the case
may be, enabling relatively high efficiencies and high
volume ratios to be achieved.
BRIEF DESCXIPTION OF DR~WIN~
Fig. 1 is a temperature-entropy diagram (T-s)
of the Carnot cycle;
Fig. 2 is a pressure-enthalpy diagram (p-h) of
the Carnot cycle;
Fig. 3 is a pressure-enthalpy diagram (p-h) of
the Rankine cycle;
Fig. 4 ~hows a Carnot cycle superimposed on a
portion of a temperature-entbalpy tT-h) diagram for
refrigerant CFC-114;
Fig. 5 is a layout of the Carnot cycle heat
engine apprcximation of the present invention;
Figs. 6 and 7 are views of high efficiency
compressors and expanders in accordance with ths present
. :. . i : , , , ~ ,
. ~ ~ . . .
.
: -: , ,
.
,
.~ ~323~
-- 5
invention, Fig. 6 being a 6implifisd and annotated
section view taken along line 6-6 of Fig. 7;
Figs. 8A and 8B are schematic (conventional)
refrigeration/heat pump systems and cycle diagrams
respectively;
Fi gs. 9 A a n d 9 B a r e s ch e ma ti c
xefrigeration/heat pump s y8 tems and cycle diagrams
respectively, illustrating a further a~pect of the
invention.
DETAI LED DESCRI PTI ON OF _PREFERR13D EMBODI ~EN~
E GARNO~ AND RANKI NE CYC~LES
To review, the Carnot cycle is defined as
consisting of four special thermodynamic proces~es: Two
isothermal heat transfer processes and two isentropic
work processes. In a temperature-entropy (T-s) diagram,
the Carnot cycle appears as a rectangle as shown in
Figure 1, with the "dome" representing the saturated
liquid-vapour phase diagram of a typical organic
compound. The two horizontal lines respectively
represent isothermal heat addition and rejection. The
right vertical line represents isentropic expansion (work
output) and the left vertical line represents isentropic
oompression (work input). On a pressure-enthalpy (p-h)
diagram, the Carnot cycle appears somewhat like a
rhomboid as depicted in Figure 2.
It is instructive to consider the Rankine
cycle, also depicted on a pressure-enthalpy diagram,
because the similarities and d~fferences between the two
cycles become readily apparent. Figure 3 shows the
Rankine cycle on a p-h diagram.
It i~ immediately apparent that both the Carnot
and Rankine aycles have i~othermal heat addition and heat
re;ection proaesses as ehown by the two sets of parallel
horizontal lines. However, oonsiderably more heat is
added in the Rankine cycle (process 4-1) than in the
,:
, ~ , . . :
;:
: . :
~ ~3239~ ~
Carnot cycle. k'urther, and consequentially, more heat i~
rejected by the Rankine cycle than the Carnot cycle
(process 2-3). Significant differences between the two
cycles occur during the work processes (1-2) (expansion)
and S3-4) (compression and/or pumping). For example,
using an organic fluid with "dome" lines as shown here,
the Rankine cyale begins (generally) slightly 6uperheated
at state point (1) and expands isentropically to state
point (2) where further superheat of the working fluid is
reached for some working fluids. On the other hand, the
Carnot cycle as dèscribed here begins its expansion
inside the "dome" at state poi.nt (l) (i.e. a mixture of
liquid and vapour) and expands at constant entropy (as
prescribed here) to a aturated vapour pha~e at state
point (2).
In the Rankine cycle, all the working fluid is
condensed to a liquid state (3) and is then pumped from
the lower pressure in the condenser to the higher
pressure in the boiler (state point 4). The Carnot
cycle, however, only partially condenses the working
fluid during the proces~ from state point (2) to state
point (3). This requires that a mixture of li~uid and
vapour phase working fluid at a state point ~3) must be
compressed as a mixture and pumped into the boiler at
state point (4). ~his compression/pumping process
accommodates the "incomplete" condensation occurring in
the condenser. The compressor/pump collapses the vapour
portion of the two-phase mixture substantially to hot
liquid. In the process of mixed-phase compression as
39 provided by the present invention, the saturated vapour
trans~ers the heat of compre~sion to finely dispexsed
liquid phase droplets entering the compressor/pump
~which finely dispersed droplets are provided by means to
be described hereafter). In a direct sense, the
condensation process i~ completed through the application
.
.
, .
. ' , ,
, ~ , ~, :
. . . : . ..
3239~
of work in the compre6sor/pump rather than by heat
transfer occurring in the condenser. ~he following
section discusses specific means to effect a real Carnot
engine. Subsequent sections discuss a detailed
embodiment ~nd presents specifics of the expander and
compressorJpump fluid-handling machinery.
Figure 4 shows a Carnot cycle superimposed on a
temperature-entnalpy diagram for refrigerant CFC-114.
The calculations for cycle efficiency ~et out below shows
how the expander and compreæ~or/pump efficiencies ~ exp
and ~ comp respectively, influence the overall cycle
efficiency.
Carnot = T1-T2 = 180 - 40 = 21.7%
~1 180+460
cycle = f88.07-75.81~ exp -(52.687-48,1)x-~ comp
88.07-52.687
= ( 12.26~_exp - 4.587~x ~ aQmp
35.383
= 21.7% (~or isentropic expansion and compression,
numerical values being taken from ASHRAE, 1981
Fundamentals Handbook pp. 17, 23.
With a perfect expander and compressor, the cycle
efficiency equals the Carnot efficiancy. However, it is
apparent from calculations that with an inefficient
expander and compressor pump, the actual Carnot cycle
engine efficiency can fall well below the Rankine
effioiency. ~he reason that an actual Carnot engine is
more sensitive to machine efficiencies than the Rankine
cycle is because tha compressor/pump "back-work" term is
considerably larger than the liquid pump term of the
Rankine cycle. Typically, the Carnot engine's
compressor/pump energy requirement is on the order of 1/4
-1/3 of the expander wor~ output. In the Rankine cycle
this term is often less than 2% of expander work output.
: . .
~. ~
- . `, ' ` `
132399~
THB CARN~;)T ENGI NE
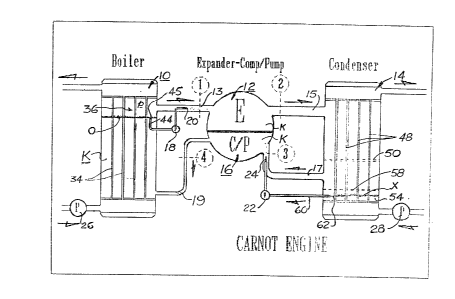
Figure 5 presents a detailed s ahematic layout
of the Carnot engine approximation accordlng to the
present invention. The engine as shown comprises four
primary components: The boiler 10, the expander 12, the
condenser 14, and the compressor/pump 16. Boiler 10 is
connected to the inlet of expander 12 by a boiler outlet
line 13 while the expander outlet for "spent" gas is
connested to the condenser inlet via condenser inlet line
15. Compressor/pump inlet line 17 leads from the
condenser outlet to the aompressor/pump inlet. The
compre~sed hot li~uid from the ~ompressor/pump enters the
boiler 10 through the boiler inlet line 19. Secondary
components include an expander inlet injection pump
18,the outlet of which is connected to expander inlet
liquid spray nozzle 20 located in boiler outlet line 13.
A compressor/pump inlet injection pump 22 has its outlet
connected to a compressor/pump inlet liquid spray nozzle
24 disposed in inlet line 17 leading to the
~0 compressor/pump inlet. Also noted in Figure 5 is a
boiler hot watsr circulating pump 26 and a condenser cold
water circulating pump 28. The working fluid, which
displaces the inside volume of the engine loop, is
denoted K.
In the pre~ent layout, it is convenient to
begin with considering a flow of high temperature water
from a heat source (not shown) into the boiler as a
result of the action of boiler hot water airculating pump
26. As the hot water flows upwards in the boiler heat
exchanger tubes 34, heat is transferred to the
surrounding organic working fluid K. This heat input to
the boiler 10 cau6es the working fluid K to vaporize and
emerge at the top region 36 of the boiler. ~he interface
between the liquid and vapour in the boiler is indicated
3 5 as 0. The saturated vapour, denvted p, thPn leaves the
... .
:
~,
.~
. .
132~9~1
boiler via outlet line 13. In the meantime, liquid
injection pump 18 draws liquid from the boiler via an
open draw line 44 haviny an up-turned inlet end 45. The
vertical position of the upturned inlet end 45 of this
liquid draw line 44 determines the liquid level in the
boiler if the pumping capacity of the liquid injection
pump 18 is sufficiently high. This (su~ficient pumping
capacity) is a desirable condition, of course, because
the liquid flow rate will be caused to stabilize at the
required value at design operation and working fluid
charge level. It also ensures that the maximum boiler
heat transfer tube area i~ in contact with liquid phase,
thus maximizing the perfoxmance of the boiler 10.
The action of the injection pump 18 in
combination with the spray nozzle 20 and the inlet
saturated vapour p yields a finely dispersed high
pressure mixture of very small liquid droplets suspended
in the vapour. This homogeneous dual-phase working fluid
then enters the expander 12 at state point (l). Next,
the working fluid at state point (1) expands in the
expander 12 to state point (2). For analytical and
practical purposes, the amount of li~uid spray injected
into the vapour at state point (1) should be such that
the low pressure expanded or "spent" gas reaches state
point (~) with a quality of 100% (i.e. saturated vapour).
This can be seen in Figure 2 in the lower right-hand
corner.
During the expansion process, the lowering of
the pressure of the vapour surrounding the suspended
liquid droplets causes the droplets to evaporate. This
evaporation process is tantamount to adding heat to the
gas during expansion. Such action, of courss, increases
the work done as the expansion process proceeds, and
therefore the net expander power output.
As the "spent" vapour enters the condenser 14
. . . .
:.. ";'~
. . . .. .
1323~
-- 10 --
through the condenser inlet line 15, it comes in contact
with heat exchanger tubes 48. These tubes are cooled
through the action of cold water flowing through them
that is pumped by the condenser water pump 28. Since in
a real machine some losses will occur, the temperature of
the working fluid at 6tate point (2) will be slightly
above the ideal saturated value that should enter the
compressor/pump 16. ~herefore, baffle 50 ensures that
the upper tube~ 48 chill the vapour to the saturation
temperature.
Next, the chilled vapour leaves the condenser
14 on its way to the compressor/pump 16 through pump
compressor/inlet line 17. In the meantime, the condensed
liquid collects in the bottom region 54 of the condenser.
The interface between the vapour and liguid phase in the
condenser is denoted X. ~affle 5~ ensures that liquid
"splashing" does not occur so that no liquid will enter
compressor/pump inlet line 17. The collected condensed
liquid W then enters the liquid injection line 60 at the
line's end, 62. Again ,the use of an "over capacity"
liquid pump 2~ ensures that all of the condensed liquid
enters the compressor/pump and that the condenser remains
essentially "dry". This is important because the maximum
amount of condenser tube area should be in contact with
vapour.
Through the combined action of the liquid
injection pump 22 and spray nozzle 24, the condensed
liquid is "atomized" at 24 as very small liquid droplets
and mixes with the vapour passing through compressor/pump
inlet line 17. This mixed-phase working fluid, K, then
exists at state point (3) just prior to entering the
compressor/pump 16.
As the finely mixed saturated liquid droplets
and vapour are captured by the aompressor/pump 16, the
vapour phase is compressed. This input work causes an
~3~3~9~
increase in the vapour temperature and pressure. As the
vapour temperature increases, the tiny liquid droplets
absorb the heat, 80 that the temperature of the dual-
phase mixture stays lower than it would without the
liquid droplets. Since the pressure is al60 increasing
as a result of the compression, but the temperature is
being simultaneously lowered by heat flowing to the
existing liquid droplets, the vapour phase portion of the
mix converts to liquid. This (essentially) fully-
condensed hot li~uid then enters the boiler throughboiler inlet line 19 where it re-evaporate6 in order to
continue and repeat the cycle.
It is important to understand that thi~
invention is not limited to the liquid atomization means
(pump and spray nozzle) as outlined herein. For example,
common Venturi embodiments can be used that are similar
to the action of internal combu~tion engine carburetors
that "atomize" the liquid gasoline. It is also important
to realize that the level of ~pproximation to isentropic
compression and expansion processes is a function of
droplet size. This is because there is (~ssentially) no
limit to the area that can be made available for the
intra working fluid heat transfer processes. Said
differently, by greatly decreasing the size of the
individual liquid particles (and, therefore, greatly
increasing their number), extremely large heat transfer
areas are available. Large intra-fluid heat transfer
area permits very close temperature "tracking" between
the two phases of the working fluid.
By injecting the "misted" liquid working fluid
component into the vapour component of the working fluid,
a "homogeneous" mixed-phase working fluid is created.
This mixed-phase working fluid thus accrues special
properties. The property arises as a reæult of the
continuous thermodynamic property changes that the
.
'
,, .
,
, . :
,
. ! - ,
" ' , . ' ~' ~ .,, ~
~.~
- 12 -
~239~
mixed-phase working fluid undergoes as heat is
transferrsd across the liquid-to-vapour or vapour-to-
liquid boundaries created by the fine mixture of liquid
and gas.
Consider the organic working fluid CFC-114.
When undergoing expansion, for example, this single-
oomponent mixed-phase working fluid naturally experiences
ever-lowering pressure and temperature. The
thermophysical properties of CFC-114 cause the liquid
10 droplets to evaporate into the existing vapour. This
process, if carried out adiabatically on the macro~copic
scale, but isothermally on a "microscopic" scale (heat
transfer between the droplets and the Rurrounding
vapour), can approximate an isentropic expansion process.
15 That is, as entropy is gainad by the vapour component
(heat being tran ferred to the vapour), entropy is lost
by the liquid component (heat being transferred from the
liquid) in equal amount, thereby approximating an actual
two~phase isentropic expansion process. Of course, the
20 mixed-phase compression process is directly similar to
expansion, except that heat sntropy is gained by the
liquid and lost by the vapour.
In the limit (infinitely small liquid droplets
and infinite heat transfer area), the mixed-phase working
25 fluid volume-changing processes would actually be
i~entropic, assuming no maahine irreversibilities or heat
transfer. Because in practice it requires only small
amounts of energy to "atomize" liquids into small
droplets, the net area for heat exchange between the
30 liquid and the vapour pha~es can become very large at
low snergy expense. It is believed to be these facts, in
~ combination with high e~ficiency high volume ratio
- machines, that make the approximation of the Carnot cycle
po sible.
.
i,.
: ~i
. .
.
- .. , -
, .
~' :
",. . . : :,
- 13 -
~323~
~Y~
Due to the extxame changes in volumetric
requirementæ resulting from actualizing the Carnot cycle
with dual-pha~e working fluid~, new fluid-handling
machines were, a6 a part of thiR invention, requixed to
manage the~e large change6 in volumP. Of cour~e, ~ecause
the most dra6tic changes i~ displa~ed volume take place
in the compressor/pump, thi6 maohine pre~ented the
highest design challenge. In a ~pecific example, using
n-Butane (R-600) as the working fluid across 180F and
40F, the volume ratio for the expander i~ approxlmately
8. 8 to 1. Whila thi~ i~ a relatively large value which
cannot be accommodated by prior art imachine~, the
compre~sor/pump volume ra~io requirement under these fiame
conditions is i n the order of 70 ~o 1 as will be seen
from the example whiah follows.
In gen~ral, the prs~e~t lnventton lncorporate~
Yane-type rotary compres~ors and expander~ of the type
disolosed in U. S. Patents 4, 299, 097 issued Nov~m}3er 10,
1981 and 4,410,305 issued OctabQr 18, 1~83. Figures 6
and 7 shc~w a vane type compres~;or ~;imilar to the
compre~sor described in the above two patent~ but
differing therefrom in several important respect~;
insofar as the geometry of the chamber or stator
interior is concerned. (This same discussion can be
applied to expanders). All of them enjoy the advantages
conferred by vanes riding on rol:Lers located in grooves
or cam contours of predetermined shap~ so that vane tip
friction is es~entially eliminated; inlet and outlet
port configuration is optimized and numerous other
mechanical advantages are conferred thereby to provide
for extremely high operating efficiency.
~ urning a~ain to the drawings there is ill-
uætrated in Figs. 6 and 7 a compressor 70 comprising a
!A
. .
. .
. - ~ . . -
. . . -
: . . .
-- . ,
3 2 ~
- 14 -
stator housing 72 defining a chamber having opposed
parallel end walls 74, 76 and a curved interior wall 78
extending about a chamber axis 80.
Forming the end walls 74, 76 of the chamber are
end plates 82, 84 which are respectively mounted upon end
pieces 86, 88 whiah are clamped together by bolts 90.
The end pieces carry anti-friction bearings 94, 96 and an
associated seal 97 centered about a rotor axis 98.
.~ The bearings 94, 96 serve to journal a rotor
100 of cylindrical shape supported upon a shaft having a
driving end 102, and a remote end 104. The rotor,
dimensioned to fit between the end walls, has a plurality
of spaced radially extending slots. Occupying the slots
for sliding movement in the radial direction is a set of
vanes 106-110 of rectangular shape and profiled to fit
~` the stator chamber to define enclosed compartments
between them.
Each vane has a pair of axially extending,
aligned stub shafts having rollers mounted thereon. Each
set of rollers, indicated at 114-118, i6 guided in a cam
contour 120 having parallel side walls 122,124. The
outer side walls 122 form tracks for the vane rollers,
the tracks being so profiled that when the vanes are
urged outwardly the outer edges of the vanss follow in
; 25 closely spaced proximity to the inner wall 78 of the
, stator chamber.
There is provided, on the stator chamber, an
! inlet port 126 for aspiration of gas into each
compartment between ad~acent vanes. There is also
provided an outlet port 128 for discharging gas from each
compartment in the compressed state. The curved
, .
`; interiox wall 78 is rece~ed to provide peripheral
pockets 130, 132, re~pectively, which extend the ports to
minimize inlet and outlet fluid dynamic losse6. A "tuck
in" seal region 133 of the stator interior wall located
i.
,
"
:
... . .
.~,~ . ~ " . .
.;j, ~:
. . ~. .
:~ . :
i, ~: . ,,
~3~3~
- 15 -
';
; between pockets 130, 132 i~ in cl~se sealing engagement
~ith the smooth out~r periphery of th~ rotor thereby to
prevent lea~age of fluid from the high pressure outlet
to the low pressur~ inlet side.
An expander according to the invention is also
as described above and illustrated in FigsO 6 and 7
except that the direction of the rotor i6 reversed and
the positions of inle~ and ou$1st ports 126, 128 and
~; :their associat~d pock~t~ 130, 132 ~re interchanged.
It has been found that high volume ratio
machines of the constrained rotary vane type as described
can be created by three primary individual geometrical
components and a single ~x-o~fsat" between the rotor 100
and the stator chamber inner wall 78. From Figure 6, the
stator cham~er inner wall pr~file can be seen as
including: (1) a quarter circle section 134; (2) a three-
quarter elliptical section 136; ~3) a short straight-line
segment 138 betw~en ths quaI~er ~ircle section 134 and
~ (4) a rotor ~x-of~s0t" 140 from the center axis of the
` 20 stator cham~er profil~ on the x-~xis. It will be noted
that the le~t-bo~tom quadrant of the s~ator chamber in
:I Fig. 6 arbitrarily co~tains the ~uarter circle section
~1 134, the top two and lower right quadrants together
con~in the 3/4 ellip~e secti~n and the short straight
line segment 138 lies across the bottom of the lower
, righ*~hand q~a~ra~t from the bottom end-point of the
;~ quarter circle section to the buttom left end-point of
the 3/4 ellips~ section. From point D to point E the
sta*or ellipse is descri~ed as ~eing "imaginary" since
the ~ctual stator interior wall in this area is occupied
j by ~he peripheral pockets 130, 132 and the seal region
.! 133, the latter region ~ct~ally defining a sylindrical
-sur~3ce centered with ~he axis of rotation of the rotor
100. From poi~t E *o poi~t F ~the remaining portion of
the 3~4 ellipse) t~ ~tator i~ner w~ll 78 conforms to the
, j
~ .
.
.
,, , . ~ , .
:, ~ ! ' ' .: ' ; :
: ,, . :
,: , :
~ ` '' ' ' , :
.. : ', : :
-. ~ , .; .:
'~: ' ' ' . ', , ','
.' ' , .
~.323~
shape of the actual ellipse to be described hereafter.
The geometrical relationships are fairly simple
and, if the radius of the quarter circular portion 13~ of
the stator chamber wall contour is called "R", then the
3/4 ellipse portion 136 of the stator wall contour has a
major axis equal to twice the sum of R and the x-offset
between the ellipse center and the circle aenter, both of
which lie on the x-axis. Also, it has been found that a
very convenient value for the semi-minor axis of the
elliptical portion of the stator chamber contour is
simply the radius R of the ciraular portion 134 of the
stator profile. (The radius of ~he rotor is only slightly
; less than radius R as shown in Fig.6). Since the
eccent.ricity of an ellipse is defined here as the arc
cosine of the ratio of the minor to major axes of the
ellipse, the eccentricity of the elliptical portion of
the stator chamber can be easily computed. The X and Y
coordinates of all points along the elliptical wall can
~ also be easily calculated using standard mathematlcal
. 20 techniques.
In Figure 6, it can be seen that the center of
the rotor 100 iB coincident with the center of the
quarter circle section 134 of the stator chamber profile
-- again, on the x-axis. This choice, with four rotating
`~j 25 vanes 106-llO, precisely causes the rate of inlet flow
' (as an expander) or the rate of outlet flow (as a
~` compressor) to be a constant function of rotor Epeed.
1 Furthermore, by choosing R as the value of the semi-minor
;~ axis of the stator chamber ellipse, it coincides nicely
~; 30 with an x-offset equal to about 1/5 of the rotor radius.
~' This fraction, however, can change considerably with the
i
choice of volume ratio. Nonethele~s, these geometric
values result in a configuration that is not only easy to
understand and calculate, but its manu~acture and
dimensional inspection will be easier than with the
;
,
~ ., .
j~,
: . ,
~., ' : - '
,,
~ 323~ ~
- 17 -
earlier doubly-offset machine shown in U.S. Patent
4, 410, 305.
It is noted that the high volume ratio
machines described above have two specific
characteristics related to gas dynamics: 1) the high
; pressure side, whether considering the machine a
compresor or expander, has constant volume flow rate at
constant rotor ~peed, and 2) the low pres6ure side,
; whether considering the machine a compressor or expander,
has varying volume flow rate at constant rotor speed.
However, the low pressure side is designed as described
above in such a way that the rate of volume change dwells
. at zero or nearly zero during a large angular change of
rotor position. This is impoxtant because this
characteristic ensures that a) when behavin~ as a
compressor (such as in the Carnot compressor/pump
embodiment), this zero-volume change secures an
opportunity for the vane cavity to fill completely ~i.e.
there are no "wire-drawing" fluid pressure losses), and
,~ 20 b) when behaving as an expander (such as in the Carnot
expander embodiment) no vane cavity pressure build-up
occurs during the exhaust process.
The inventio~ will be better understood from
the following non-limiting example.
EXAMPLE
The various values of the sta~e points of the
Carnot engine cycle are computed below. The fundamental
assumption is that the single-component mixed-phase
working fluid exchanges heat rapidly enough to comprise a
quasi-static thermal equilibrium. Further, the analysis
assumes that the processes are, by initial definition,
isentropic.
To start the analysis, state point (~) (post
; ~xpansion) and state point (4) (post compressor/pump) are
selected. For example, assume (specify) that state point
~.'
,.. - : : :
,
.
.."
.. . .
```~` 1~23~9~
- 18 -
(2) is 6aturated vapour at 40F, and that point (4) is
saturated liquid at 180F. The problem is to find the
properties of state points (1) (pre-expansion) and (3)
(pxe-compressor/pump). Since the state points in
question t1 & 3) fall within the P-s dome, the quality of
the mixture i~ non-zero and it exists, of course, at
saturated conditions. The quality of the mixture is
defined as the ratio of the mass of the mixture in vapour
~ form to the mass of the whole mixture.
- 10 In the following analysis:
h = enthalpy BTU/lb
s = entropy BTU/lb.F
x = quality
f = liquid
g = vapour
THERMAL OPERATING CO~DITION~
Normal butane R-600 is the working fluid
High Side: T high=180F, psat2=154.7 psia
Low Side: T low=40F, psatl=17.62 psia
Pressure Ratio: psat2
pratio:= pratio=8.779796
. psatl
sf2 =1.0547 sg1-1.2473 hg1= -564.1
vg1=0.5976 vf1= 1 hf1= -687.5
, 31.17
2 =1-2369 (This is imposed upon the cycle)
The quality at state point (1) is calculated from:
82 - sfl
X1= x1 = 0.946002 (quality)
Sgl - Sfl
Specific enthalpy:
1 = xl hy1 + [1 - x1] hf1; h1 = -570.763344
~ Specific volume:
`, 35 V1 = X1 vg1 + [1 - x1] vf1; v1 = 0.567063
.~
:~
,.,
, .
: .
, ~ ,, .
,. : .
.,~ .. ..
.,.
.~: ~ , .
~ ~3~3~
- 19 -
~ ~ate Point (2):
.~ .
vg2= 4.998 1 hf2= ~770 7
vf2=
37.22
~` X2 = 1.000 (S~tllrated gas condition)
Speaific enthalpy:
.' h2= X2 h~2 + {1 - x2~ hf2 h2 ~ -606.9
Specific volu~e:
., 10 v2= x2 vg2 + [1 - x2] vf2 v2 = 4.998
State PQint (3):
~ sf3= 0.9085 sg3= 1.2369 hg3= -606.9
., vg3= 4.998 vf3= 1 hf3= -770.7
. 37.22
. .
~ 15 Quality at State Point 3:
i.
. S4= 1.0547 (~his is ~y~l t~ the 8 aturated 1iquid
~ntropy at state point 4.)
~`' S4 - ~f3
~ 20X3 = X3 = 0.445189
"j sg3 - 8f3
Speci fi c enthalpy:
~` h3 = x3 hg3 ~ l1 - ~3J h~3 ; h3 = -697.778076
l Specific volume:
;.~ 25 V3 - x3 vg3 ~ [1 - x3] vf3 ; v3 = 2.23996
i S~ate Point (4).,_ -
:-.
.'J
~ sf4 = 1.0547 ~g4 = 1.2473; hg4 = -564.1
`~`! vg~ = O. 5976 V~4 = 1 ; hf4 = -687.5
"t 31. 17
:~ 30 x4 = O. 000 (Saturated liquid condition)
.`.i ..
q
:,:,
~.~
~ . ~
.~,i,;
~"i .
. . . . .
~.323~
- 20 -
.
..
S~ecific enthalpy:
,
h4 = X4 hg~ + [1 - x4] hf4; h4 = -687.5
` Specific volume:
V4 = x~ vg4 ~ [1 - x4] vf4; v4 = 0.032082
CYCLE CAL~ULATIQNS:
Specific Power:
p: = [h1 - h2] - [h4 - h3] ; p =25.858581BTU/lb.
.,
Expwork: = [h1 - h2l i Expwork=36.136656BTV/lb.
Compumpwork:= [h4 - h3] Compumpwork =10.278076BTU/lb.
Ideal Thermal Conversion Efficiency:
eff = P _ 25.858 = eff = 0.221512
l - h4 116.736
EFFY = 22.151209 lapprox.same as below)
.~
CARNOT - HIGH - TLOW x 100=21.8856(The difference
i 15 EFFICIENCY THIGH + 459.69 from the above represents
a .4% error in tabulated
property data)
VOLUME RATIOS:
.~ Expander:
V2
Vre= _ = 4.998~ ~ = Vre = 8.813832
,. v1 .567063
Compressor/Pump: = 9:1 approx.
.; !
' V~
Vrcp=~ = 2.2399 - Vrcp =69.819549(about
,~ V4 .032082 70:1)
;~ Vrcp - 70:1 approx
',1,
~!
!
~:i
. ~'
.1
,.1
. ,
:'.
: " ':
`,'~ ' ' '
., .
~ . .
1323~
- 21 -
IDEAI, MASS F~L~OW_RATES.
Q = 100000 Watts, nominal engine output
Mdot = O 3.412969 = 10-x3,4129~9
25.858
Mdot = 1.319859x104 Pounds/Hr.
Mdotmin= _~Q~ ; Mdotmin = 219.976561
Pounds/Min
n-Butane Flow Per Expander Revolution at RPM speed:
,i
RPM = 1800
. 10 Mdispl = MdQ~n
RPM
. Mdispl = 0.122209 lb.
Maximum _~umetric Dlsp~ e~~ 9~ rL
;: 15
Expdispl = Mdispl [v2]
.~ Expdispl = 0.610802
Max Displacement per Segment (4-vane):
,
, Displ Exp = Expdispl
,. 20 4
Displ Exp = 0.1527 Cubic Feet per r~v per Segment
= 263.366 cubic inches per rev per vane
Segment
!
Maximum VQlumtric Displacement of C.ompre8~0r/Pump
~ Compumpdispl = Mdotmin x
i~, RPM 4
i.i
$l Compumpdispl = 0.068436 cubic feet per rev per
Segment
= 118.2573 cubic inches per rev per
~i; Segment.
.;,
s-
~: i
,i ,, , . ~ . .
, . .
~.: . . .
": - ~
1323~v ~
- 22 -
:;
'
The above ideal example therefore not only
establishes the ~alues of the state points under the
conditions given, ~ut it also enables specific power to
be calculated along with thermal e~ficiency of the cycle,
volume ratios for the aompressor/pump and expander, mass
flow rate6 and maximum volume~ric displacements for the
expander and ~om~r~30r~pump. Using the geometrical
relationships described above together with these values
the detailed enyineering design for both the
`~ compressor/pump and expander can be accomplished. By
providing expanders and compressors of the "volume
change" or positive displacement type described above as
i- opposed to tur~i~e machi~es, problems o turbine blade; 15 pitting and ero~ion ars non-~xi~tent. The dual phase
mixtuxe of droplets ~u~pended in vapour iB tolerated very
well in the ~a~e ~ype c~m~x~sors and expanders as
de~cribed. M~reover, these ~ame machines provide the
very high volume ratios needed ~or the reasons as
. 20 described above.
.3 Those ~illed in this art will realize that
;' the ideal expa~der -and ~mpressor designs can only be
approached as a li~it. Hence, all references to
i6entropic expan6ion and comprecslon are to be
interpreted in a gener~l s~nse ~only and not in a narrow
restricted sen~e. There wlll always be some losses
.~, during expansion and compre~ion. At the same time it
will be appreci~ted that c~mpressor and expander
efficiencies of over ~0% cr thereabouts will be required
if the Carnot cycle approximation here described is to
have any appreciable adva~tage over the conventional
:' Rankine cycle. T~is ls particularly true in the ¢ase of
,. the compres~or owin~ to the fact that the pump work
;~ factor in a Car~ot cycle is a relatively large percentage
,,
,, .
.~ .
,, .
~,', ~ . .
. : . .
: . .
. ~ .
~ ~323~
- 23 -
.
; of the expander output woxk as compared with the
conventional Rankine cycle as noted previously. The low
friction roller mounted vanes and favourable fluid
dynamics associated with the compressor and expander
described above greatly assist in providing the high
efficiencies needed.
: THE CARNOT REFRI GE~TI ON AND HEAT PUMP CYÇLES
Referring now to Figs. 8A and 8B there is shown
a conventional refrigerator or heat pump and its vapor
cycls. The working fluid or refrigerant is compressed
between state points (1) and (2) by compressor 200,
ending with uperheated vapor. Cooling and condensing
takes place between state points (2) and (3) in condenser
202 with heat being transferred out of the system.
Throttling between state points (3) and (4j by way of
s throttling valve 204 then occurs with the enthalpy
remaining unchanged. (There is no heat transfer).
~` Evaporation, a constant pressure process, occurs between
(4) and (1) in boiler 206 to complete the cycle, this
being the process in which the refrigerating effect
occurs as heat is transferred to the evaporating fluid.
Referring now to Figs. gA and 9B there is shown
a Carnot refrigeration and heat pump cycle. The
equipment uses a two phase rotary expander 212 and a two-
phase rotary compresæor 208, both constructed as
~ des~ribed with reference to Figs. 6 and 7 60 the detailed
-~ mechanical de~cription need not be repeated here.
.I Furthermore, the inlet line to the compressor 208 is
~ provided with a liquid pha~e in;ection pump and spray
i:~ 30 nozzle essentially the same as pump 22 and nozzle 24
- described with reference to the Carnot engine and with
reference to Fig. 5. Similarly, the inlet line to the
two-phase expander 212 is provided with a liquid phase
injection nozzle and pump essentially the same as the
' 35 nozzle 20 and pump 18 again as described with reference
, ~
.
~ `
:,. ~ , .
~323~
- 24 -
to Fig. 5. The condenser and boiler may be of a
generally conventional nature except that means should
be provided to control the liquid levels in both units to
ensure good heat transfer efficiency, as by suitably
arranging the levels of the inlets to the liquid phase
pumps as described previously.
With reference to Fig. gs compressor process
. (1)-(2) (which is approximately isentropic) starts with
saturated liquid and ends "inside the dome" with a
compressed two phase fluid. Cooling and condensiny from
state points (2) to (3) ends at the 6aturated liquid line
with subsequent expansion (approximately isentropic) in
the two phase expander 212 from point (3) to (4)
providing a two-phase fluid which is then evaporated in
boiler 214 to produce the desired cooling effect. During
:; the expansion in expander 212, some useful work is
produced and this energy is fed back into the system,
i.e. to complement the shaft work input to the compressor
,. 208 in any suitable manner.
;: 20 The phenomena de cribed previously in
connection with the Carnot engine, i.e. the continuous
.~ thermodynamic property changes that the mixed-phase
working fluid undergoes as heat is transferred across the
~;'' liquid-to-vapour and vapour-to-liquid boundaries created
by the fine mixture of li~uid and gas, applies equally in
~, this case during the compression and expansion processes.
The coeffecient of performance (COP) of a
', refrigeration or heat pump machine can be expressed as:
r', COP = useful thermal ~ffeçt
-- 30 net power input
In case of the refrigeration apparatus in
Figure 9A and 9B the useful thermal effect is the heat
absorbed (1-4), while in a he~t pump the useful thermal
effect is the heat output (2-3). Using the values of
Figure 4 for perfect isentropic expansion and
.,
. .
:,:
~,
~ .,
:::
1323~
- 25 -
'
compres~ion we obtaln:
:; refrigeration maohine COP = ~ LDL L-=LL~
(88.07-75.81)-(52.687-48.1)
~ ~
: 5 heat pu~p COP = ____Q~07 -_ S2.687
8.07-75.81) - (52.687-48.1)
,` = ~
~ sy way of comparison, the heat pump COP when
.~ 10 using a prior art expansion valve is only 2.9 so the two-
phase cycle of the present invent~on could provide a COP
improvement approaching 60% if compressor/expander
efficiencies can be made to approa~h 100%. As
compressor/expander efficienaie~ drop off the COP
improvement will of course be reduced.
.~ The comments made previsusly noting that ideal
expander and compressor designs can only be approached as
. a limit and that all references to isentropic expansion
and compression are to be taken in a general sen~e and
not in a narrow restricted sen6e apply to the
.. refrigeration/heat pump cycle as well. High compressor
~, and expander effiaienci~s (90 + %) are required as noted
before and the low friction roller mounted vane- type
machines described herein greatly assist in providing the
~5 required efficisncies as well a~ handling the very wet
~; vapours requirsd by the cycle.
;1
.
J
: i
"
r, ~
~'~
. .