Note: Descriptions are shown in the official language in which they were submitted.
1 335708
TONOM~-.KIC CA~n~ COMBINATION
Back~round and 8ummary of the Invention
This invention relates to medical diagnostic equipment
and methods and is particularly concerned with hollow viscus
tonometry and remote electronic and optical sensing.
The prior art (see U.S. Patent No. 4,643,192) has
recognized that intestinal ischemia, and to a lesser degree,
stress ulceration, are two problems that plague physicians
involved in th~ management of patients in intensive care units.
Intestinal ischemia, in particular, has an insidious onset and
may not be detected until days after the intestine has become
completely and irreversibly compromised. A delay in the
diagnosis of intestinal ischemia may have devastating
consequences for a patient. The availability of means for early
diagnosis and management of patients with these problems would
have immediate applicability in all intensive care units,
especially where the procedure can be conveniently conducted with
reasonable safety and reliability.
It has been established ~hat a fall in the intramucosal
pH may precede the development of intestinal ischemia and stress
ulceration. As I reported in my prior U.S. Patent No. 4,643,192,
entitled "Hollow
Viscus Tonometry" a fall in intramucosal pH also occurs within
minutes of inducing intestinal ischemia in dogs. The fall in pH
in intestinal mucosa, and hence the likelihood of ischemia or
stress ulceration, can be reliably calculated from a PC02
(partial pressure of CO2), or other indicia of pH, in luminal
fluid and the bicarbonate concentration in arterial blood. The
i
1 335708
method of calculating the pH in intestinal mucosal tissue,
pursuant to principles of my prior patent, has been validated by
directed measurements under a variety of conditions ~imulating
clinical problems. A correlation coefficient in the order of
0.92 to 0.95 has been obtained in each of 16 dogs. The validity
of the procedure is inherently extensible to humans, and indeed
may also be useful in assessing the vitality of other hollow
organs and tissue. See R.G. Fiddian-Green et al. "Splanchnic
Ischemia and Multiple Organ Failure".
To measure the PC02 in the lumen of the gut it has
heretofore been necessary to obtain and remove a ~ample of fluid
that has been in contact with the wall of the gut for a certain
time period, usually at least half an hour. It has now been
observed that it i6 ~omewhat difficult to manually aspirate the
~ampling fluid or medium from a tonometric catheter located in
the gut or other internal focus with any consistency. It is much
easier to obtain such ~amples from the ~tomach, but samples
obtained from the ~tomach frequently contain foreign material
that can damage a gas analyzer.
As taught in my prior patent, the desired 6ample or
~amples can be obtained from the gut using a catheter tube
(called a tonometric catheter) having a walled sampling chamber
on the tube with the ~ampling chamber being in ~ample-specific
communication with the hollow interior of the tube. The wall of
the sampling chamber comprises a material which iB ~ubstantially
impermeable to liquid yet is highly permeable to gas. One
suitable material i5 polydimethylsiloxane elastomer.
In use the catheter is introduced into a patient to
place the ~ampling chamber at a desired ~ite within the gut. An
-- 2 --
-
~ ~ J
1 335708
aspirating liquid or medium i8 employed to fill the interior of
the gampling chamber. The sampling chamber i8 left in place at
the desired sampling site long enough to allow the gases present
to diffuse through the wall of the 6ampling chamber into the
aspirating liguid. The time should be long enough for the gases
t~ eguilibrate. The liguid impermeable nature of the sample
chamber wall material prevents both the aspirating liquid from
leaking out of the chamber and also the intrusion of any liquids
into the aspirating liquid. After the appropriate or desired
amount of placement time has elapsed the aspirating liquid is
aspirated along with the gases which have diffused into it. The
~ample thus obtained is analyzed for gas content, in particular
for pC02. In this way the PC02 within the lumen of the gut can
be reliably measured with the fluid being free from lumenal
debris.
In carrying out the diagnostic method taught in my
prior patent the PC02 measurement i8 utilized in conjunction with
a measurement of the bicarbonate ion concentration (HC03 ) in an
arterial blood ~ample of the patient for determining the pH of
the tract wall. .'
Depending upon the particular condition of a given
patient, the catheter may be left in place and 6amples may be
taken at periodic intervals 60 that pH values may be periodically
calculated. The procedure has a high reliability in accurately
determining the adequacy of organ tissue oxygenation, and
diagnosing intestinal ischemia in its incipient ~tages. Such
determination or detection can be useful in treating the patient
~o that the potentially devastating consequences resulting from
less timely detection may often be avoided.
1 335708
While the sampling techniques taught in my prior patent
have provided highly accurate and reliable results, it has now
been observed that there are instances (in the care of the
critically ill in intensive care units, for example) in which
remote sensing of the organ or organ-wall condition and automatic
calculation of the organ or organ-wall pH would be advantageous
and easier to effectuate. This method would thus partially or
totally eliminate the need for the Qomewhat cumbersome aspiration
of the sampling fluid or medium which fills the sampling chamber;
it may also eliminate the need for the ~ampling chamber to be in
Qampling-medium communication with any other part of the device.
There is also a need to extend the benefits of tonometric
sampling and eensing to other internal hollow viscous organs. To
thig end, there is a need for new and different tonometric
devices 6pecifically adapted to allow my 6ensing and sampling
techniques to be performed with ease in a clinical environment,
and in combination with other procedures.
The importance and significance of determining the pH
of the wall of a given hollow visc~us organ has been recently
7~ dramatically magnified as a result of the recent r~cogn~tion that
the pH of the wall of a given organ can be employed to accurately
evaluate the vitality and/or ~tability of that organ as well as
others; this is in contrast to merely determining whether such an
organ is experiencing an ischemic event. Further, certain organs
can be ~elected for monitoring, either alone or in combination,
and evaluation of this organ or these organs can aid in
predicting the overall condition of the patient, or the onset of
a multitude of pathologies, including predicting or identifying
~uch events a6 multiple organ failure. Such a methodology can be
- 4 -
1 335708
employed to greatly enhance and ~upplement the monitoring of the
critically ill, for example.
In one aspect, the pre~ent invention provides a new
apparatus and method for remotely sensing organ condition and
conveying an electromagnetic signal, e.g. an electrical current
or optical 6ignal, to an electronic or optical apparatus located
outside the organ under investigation. In one embodiment, a
chemically sensitive electronic transducer (or plurality of
transducers), 6uch a6 a field effect-tran6istor, i~ attached to a
tonometric catheter for introduction into the organ along with
the tonometric catheter. The first electronic sensor, preferably
non-temperature, generates and conveys an electromagnetic signal
indicative of 60me desired aspect of organ condition, e.g.,
indicative of the pC02, pH and/or P02 level of the organ or
organ-wall. For example, in one preferred embodiment, mean
ambient pC02, pH and/or P02 of lumenal fluid or the like is
~easured or monitored via wire or other suitable electromagnetic
energy conveying ~eans to an electronic circuit which interprets
the electromagnetic signal and produces a report of the organ
condition.' The electronic circuit may includé an ~nput for
receiving a ~eparately determined signal indicative of the blood
pH of the patient. Using this pC02, pH and/or P02 measurement
along with blood (preferably arterial) pH data, the electronic
circuit determines the pH of the organ wall under test and
thereby provides information for determining the organ'~ current
condition or perhaps predicting the organ's future condition.
The electronic circuit may be 6uitably constructed from analog
components, digital components or both.
1 335708
In another embodiment, a pH, PC02 or P02 6ensitive
colorimetric 6ubstance is injected into an area adjacent to the
organ, e.g., into the sampling chamber of the tonometric
catheter, and an optical 6ensor is employed to detect color
change in order to determine the pH of the wall of that organ.
The optical sensor can either be disposed in or on the tonometric
catheter for introduction into the area adjacent the organ or it
may be disposed outside the organ with fiber optic cable
optically coupling the 6ensor to the tonometric catheter 6ite at
which the pH sensitive substance has been injected.
In another aspect the present invention provides a
variety of new and different tonometric catheter devices for
sensing and/or sampling a fluid or gas property (such as pH, Po2~
pC02, and the like) which is indicative of the condition of an
internal organ, in conjunction or combination with a walled
catheter tube adapted for delivery or draining fluids, such as
nasogastric tubes, urinary catheters, ureteric catheters,
intestinal feeding tubes, wound or abdominal drains (suction or
regular) and biliary tubes, catheters and stents, with or without
remote sensing means for pH, PC02 and/or Po2-
In still another aspect or embodiment, the deviceemploys two separate walled catheter tubes, one tonometric
catheter tube for the measurement of a fluid or gas property,
that is in communication with the sampling chamber; and a second
walled catheter tube adapted for delivering or draining fluids.
In yet another aspect or embodiment, the device employs
a walled sampling chamber in communication with a sensing means,
and a 6econd walled catheter tube adapted for delivering or
draining fluids.
1 335708
Optionally, when a non-temperature sensing-means is
employed, a second sensing-means may be employed as well.
For a more complete understanding of the invention, its
objects and advantages, reference may be had to the following
specification and to the accompanying drawings. Also, see
applicant's co-pending Canadian applications filed of even date
herewith entitled "Remote Sensing Tonometric Catheter Apparatus
and Method~ and "Hollow Viscus and Solid Organ Tonometry",
bearing respective serial numbers 609,067 and 609,066.
Brief Doscription of the Dr~winqs
Figure 1 is a plan view of a first embodiment of the
tonometric catheter;
Figure 2A is a partial cross-~ectional view of the
tonometric catheter illustrating a first means for attachment of
an electronic field effect transistor sensor;
Figure 2B is a partial cross-sectional view of the
tonometric catheter illustrating a ~econd means of attachment of
the field effect transistor sensor;
Figure 3 illustrates the method of use of the
tonometric catheter in measurement of the pH of the colon and
also of the ~tomach, the specific embodiment illustrated for
colonic measurement being that of Figure 5 and the specific
tonometric catheter for ~astric measurement being that of
Figure 4;
Figure 4 i6 another embodiment of the tonometric
catheter with nasogastric tube;
1 335708
Figure 4A i8 a cros6-~ectional view of the tonometric
catheter of Figure 4 taken 6ubstantially along the line 4A-4A of
Figure 4;
Figure 4B i6 a cros6-~ectional view of the tonometric
catheter of Figure 4 taken substantially along the line 4B-4B of
Figure 4;
Figure 5 i6 yet another embodiment of the tonometric
catheter having multiple sensing/sampling portions;
Figure 5A is a cross-sectional view of the tonometric
catheter of Fig~re 5, taken sub6tantially along the line 5A-5A of
Figure 5;
Figure 6 is a detailed view illu6trating the tonometric
catheter of Figure 4 in u6e within the stomach;
Figure 7 i8 a detailed view illu~trating the tonometric
catheter of Figure 5 in use within the colon:
Figure 8 is a similar view illustrating the tonometric
catheter of Figure 1 in use within the colon;
Figure 9 i6 an electrical schematic diagram
illu6trating one embodiment of electronic circuit in accordance
with the invention;
Figure 10 is an électrical schematic diagram
illustrating another embodiment of the optical mea6urement of pH
in accordance with the invention;
Figure 11 i6 another embodiment of a tonometric
catheter with a urinary catheter;
Figure llA i6 a cro66-sectional view of the tonometric
catheter/urinary catheter of Figure 11, taken substantially along
the line llA-llA of Figure 11.
1 335708
n~cr~ption of the Proferred ~bodi~ents
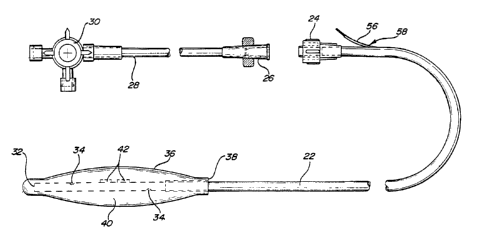
~ igure 1 illustrates a fir6t embodiment of tonometric
catheter 20. The tonometric catheter comprises a length of
cuitable tubing 22, one end 32 of which i6 closed, and the
opposite end of which has a connector ~uch as a luer-lock 24.
~uer-lock 24 is adapted to receive a complementary fitting 26,
which in turn couples through a second length of tubing 28 to a
three-way stopcock 30. Three-way 6topcock 30 may be used to
6electively connect tubing 28 to various ~ources of irrigation or
a6piration.
Adjacent the closed end 32, tubing 22 i~ perforated as
at 34. A balloon-like tonometric catheter membrane 36 is fitted
over the closed end 60 that the perforations 34 are enclosed, as
illu~trated. The tonometric catheter ~embrane 36 has an internal
61eeve diameter at 38 which forms a tight fit with tubing 22.
The preferred form of tonometric catheter ~embrane is
polydimethylsiloxane elastomer. The membrane may be 6ealed to
the tubing 22 with appropriate adhesive ~o that the tonometric
catheter membrane i6 6ealed in a closed relationship to the outer
wall of tubing 22, thereby forming ~ campling chamber 40 adjacent
closed end 32. The tonometric catheter ~embrane has a certain
elasticity to allow the membrane to expand when filled with an
aspirating liquid in order to contact the wall of the organ under
examination, as will be explained below.
The membrane 36 i8 preferably constructed such that at
least a portion of it is selectively permeable to the gas or
fluid property of interest. In a preferred embodiment, it is
selectively permeable to hydrogen, oxygen, or H~, ~o that pH,
PC02 and/or P02 can be measured. It i6 also preferably
1 335708
impermeable to other materials that would interfere with the
desired measurements, such as other gases, proteins, and the
like. In a highly preferred embodiment, an ion-~elective
membrane is employed.
Bonded to either the inner wall or the outer wall of
tubing 22 are one or more sensors 42 for detecting a property
indicative of pH and/or temperature. Two such 6ensors are
illustrated in Figure 1, bonded to the outside wall of tubing 22
with suitable adhesive. Figures 2A and 2B illu~trate two
alternate means of sensor attachment, Figure 2A illustrating the
sensor attached to the inner wall of tubinq 22 and ~igure 2B
illustratin~ the sen~or attached to the outer wall of tubing 22.
In a preferred embodiment, at least a portion of the
tubing, but not all of it, i~ made of a C02 impermeable material,
such a8 polyester elastomers derived from the reaction of
dimethylterephtalate 1,4-butanediol ~nd -hydro- ~ -hydroxypoly
(oxytetramethylene). In a highly preferred embodiment, this is a
material such as Hytril, sold by DuPont.
For purposes of 6en~ing temperature, thermistor devices
are presently preferred. For ~ens~ng properties indicative of pH
chemically responsive field effect transistors or ~Chemfets" may
be employed. In this regard, Chemfet 6ensors 44 have been
~llustrated in Figures 2A and 2B. Chemfet sensor 44 comprises a
field effect semiconductor device 46, which is encapsulated in a
solution impervious material 48, such a8 a polymerized epoxy
resin. The encapsulation material 48 in turn may be encapsulated
in a housing 50 (~igure 2A). Semiconductor device 46 is
electrically coupled by bonding wires 52 to a terminal 54.
Suitable electrical conductors such as conductor 56 are attached
* Trade-mark
1 3357~8
to terminal 54 for electrically communicating between the Chemfet
device 44 and the electronic circuitry described below in
connection with Figure 9. Conductor 56 is preferably routed
through tubing 22 and exits through ~ ~ealed aperture at or near
the luer-lock end of tubing 22, as at 58. A more detailed
description of a 6uitable electronic sensor may be found in U.S.
Patent ~o. 4,020,830 to JohnRon, entitled "Selective Chemical
Sensitive FET Transducers ", In
order to allow a solution to contact the chemically 6ensitive
6urface of sem~conductor device 46, tubing 22 may be provided
with ~n ~perture 60 when implementing the embodiment of
Figure 2A. Such an aperture i8 not needed in the embodiment of
Figure 2B, since the semiconductor device 46 i6 exposed to
~ampling chamber 40 by virtue of the external mounting
configuration.
The ~ampling ch~mher 40 can be filled with an
~spiration or sampling medium that i~ used to absorb or otherwise
provide a means for incorporating and delivering or measuring the
the fluids or gases of interest. Such a ~edium is selected
depending upon many factors, incLuding the properties of the
fluids or gases of interest, the type of sensor 42 employed, and
the type of calibration that is necess~ry. Such mediums include
bicarbonate ~olutions and saline solution. It might be noted
that gases often behave as fluids and are therefore frequently
considered to be fluids.
As noted above, when the sensor employed does not
require frequent recalibration, the need for the sampling
chamber 40 to be in communication with the proximate end of the
tonometric catheter (that remains outside the patient) may be
~- i
1 335708
eliminated ~ince no aspiration is needed. However, in many
instances ~uch communication may still be desirable as aspiration
may be required to calibrate the 6ensor or ~ensors, to replace
the aspirating or ~ampling medium with a fresh ~edium, and to
incorporate the gas or gases of interest.
Another embodiment of the tonometric catheter ls
illustrated in ~igures 4, 4A and 4B. As illustrated, the
tonometric catheter i8 appropriately configured to also ~erve as
a nasogastric 6ump, either with or without gastric suction. With
reference to F~gure 4, the tonometric catheter 20a comprises a
multipa~sage tubing 62 which defines three individual
noncommunicating (between each other) pas6ageway6 or lumens, an
air lumen 64, ~n optional suction lumen 66 and a tonometric
catheter lumen 68. A tonometric catheter membrane, similar to
that previously described, i6 attached at an intermediate
location on tubing 62, allowing a portion of the tubing to extend
beyond the end of membrane 36 to define the nasogastric sump 70.
Tubing 62 i6 provided with a plurality of perforations 72 which
communicate between tonometric catheter lumen 68 and the 6ampling
chamber 40 defined by membrane 36, If desired~, one or more
~ensors 42 can be included in ~ccordance with the above
teachings, in which case a ~uitable conductor 56 may be routed
through tonometric catheter lumen 68 to exit at ~ealed
aperture 58.
The nasogastric ~ump portion 70 is suitably provided
with a plurality of openings 74 through which the ~tomach may be
aspirated.
At the opposite end of tubing 62 the tubing ~plits to
form three separate connections. Air lumen 64 communicates with
- 12 -
1 335708
air lumen pas~ageway 76, 6uction lumen connects with 6uction
lumen pa6sageway 78 and tonometric catheter lumen 68 communicates
with tonometric catheter lumen pa6sageway 80. The tonometric
catheter lumen pa66ageway is fitted with three-way stopcock 30,
6imilar in function and purpose to the three-way ~topcock 30
described in connection with Figure 1. If desired, a quick
connect fitting 82 may be used to couple the suction lumen
passageway 78 with an aspiration source. As illustrated, the
quick connect fitting preferably has angularly cut ends and a
61ightly enlarged midsection, making it easy to insert into the
end of passageway 78 and al60 into the ~6piration hose coupling
(not 6hown). The enlarged midsection helps form a ceal with the
adjoining pa~sageways. Preferably the quick connect fitting is
fabricated of dispo6able plastic.
Yet another embodiment of the tonometric catheter i8
illustrated in Figures 5 and 5A. This embodiment is a multiple
tonometric catheter embodiment employing ~ tubing 84 having a
plurality of pa66ageways or lumen a6 shown ~n the cros6-Fectional
view of Figure 5A. Specifically, tubing 84 includes an air
lumen 86a which communicates ~ith the endmost tonometric
i
catheter 36a and three additional tonometric catheter lumens 86b,
86c and 86d, which communicate respectively with tonometric
catheters 36b, 36c and 36d. As with the other embodiments, each
tonometric catheter may be provided with one or more 6ensors such
as ~ensor6 42. A radiopaque L~l,y~Len plug 88 i8 positioned
within each of the three tonometric catheter lumen 86b, 86c and
86d adjacent the distal end of each tonometric catheter, ~erving
to block the remainder of the tonometric catheter lumen
passageway and thereby en~uring that fluid pres6ure introduced
- 13 -
~ 3357~8
into each tonometric catheter lumen will cause the ~ssociated
tonometric catheter to balloon outwardly a~ required during use.
Similarly, a radiopaque tungsten rod 90 is fitted a8 a plug in
the end of ~ir lumen 86a, serving to terminate the end of the air
lumen passageway. Being radiopaque, the tungsten plugs and
tungsten rod aid in properly positioning the tonometric catheters
by being vi~ible under fluoroscope or x-ray. In addition, if
desired, tubing 84 can be prov~ded with a radiopaque fitripe along
all or part of its length.
At the proximal end of tubing 84 the lumen 86a-86d
diverge to define four ~eparate tubes 92a-92d. Each tube i6
fitted with a three-way ~topcock ~imilAr to tho6e de~cribed
~bove. Each sampling connector may optionally be coded
numerically by color, etc. While four approximately equally
spaced tonometric catheter~ have been illustrated in ~igure 5, it
will be under6tood that the invention can be modified to include
a greater or fewer number of tonometric catheter6 at different
6pacing as reguired for a particular application. It will also
be understood that ~ome or all of the tonometric catheters can
include one or ~ore 6ensors coupled to conductors 56, each
preferably routed through the co~-e_~onding lumen pas6ageway.
Referring now to Figure 9, a 6uitable electronic
monitoring circuit will now be described. In Figure 9 CHEMFET
semiconductor device 46 has been shown ~chematically by the
equivalent circuit model enclosed in dotted lines. The device 46
thus comprises drain electrode 150, source electrode 152 and
reference electrode 154. The chemically selective system, ~uch
as a ~embrane system is depicted diagrammatically at 156. The
substrate i~ ~ou..ded as at 158.
1 33~708
Source electrode 154 is coupled to an input lead of
operational amplifier 160 which includes feedback network
diagrammatically depicted at 162. Operational amplifier 160
6enses the drain ~ource current flowing through device 46 and
converts this 6ignal into a voltage signal which i8 output on
lead 164. Tbe drain ~ource current changes in accordance with
changes in the chemical system under test. More specifically, as
the PC02 level changes in the fluid exposed to device 46, the
drain ~ource current changes accordingly. Hence the output
voltage signal on lead 164 is likewi6e an indication of the PC02
level of the organ under test. Thi6 voltage signal on lead 164
i8 coupled to an input of comparator 166 which also receives a
reference voltage Vref, which ~ay be 6upplied using a voltage
divider network (not shown) or which may ~lternatively be
provided by a digitally controlled voltage source 168. The
output of comparator 166 is fed to reference electrode 154 to
provide a ~table reference bias voltage. If a digitally
controlled voltage source i6 used, thi~ reference voltage can be
ad~usted ~nd calibrated by a computer circuit yet to be
~i~c~sed. The voltage ~ignal o~lead 164 is also fed to an
analog to digital convertor 170, which i6 in turn coupled to a
microprocessor-based microcomputer 172.
In order to automatically determine the pH of the wall
of the hollow viscous organ under test, a separate gas analyzer
~ensor 174 i6 used to determine the bicarbonate concentration in
the arterial blood of the patient. The ou~ of sensor 174 is
coupled through analog to digital convertor 176 to
microcomputer 172. Microcomputer 172 is preprogrammed to
calculate the pH of the organ wall u~ing the values provided by
- 15 -
!
1 335708
analog to digital convertors 170 and 176. Conversion of PC02
measurements can be converted into pH measurements automatically
by microcomputer 172 using ~arious equations and references
well-known in the art.
Although many different types of output devices may be
employed, 6trip chart recorder 178 and CRT monitor 180 have been
illustrated. Strip chart recorder 178 and monitor 180 are
coupled as output devices to microcomputer 172. Strip chart
recorder 178 offers the advantage of developing an easily
readable, permanent record of the fluctuations in organ wall pH.
Monitor 180 offer6 the advantage of providing digital readout of
the pH value as well as displaying the upper and lower excur6ions
of pH fluctuation. If desired, microcomputer 172 can be
preprogrammed using keyboard 182 to compare the instantaneous pH
value with doctor-selected upper and lower alarm limits. If the
measured instantaneous pH fluctuates outside those limit6,
microcomputer 172 can 60und an alarm to alert hospital staff.
While a 6ingle 6emiconductor device 46 has been
illustrated in con~unction with the electronic circuit of
Figure 9, the circuit may be readily adapted for use with a
plurality of 6emiconductor devices in order to measure the pH at
different locations substantially ~imultaneously. In 6uch an
embodiment, the data coming from each 6ensor can be fed to a
6eparate I/0 port of microcomputer 172. In the alternative, a
single I/O port can be used with the individual ~nput ~ignals
being time multiplexed.
As an alternative to electronic pH sensors, the
invention may also be practiced using optical sensor technology.
Referring to Figure 10, the presently preferred optical ~ensor
- 16 -
1 335708
embodiment use~ a first fiber optic cable 94 which i~ optically
coupled through a series of lences 96, 6electable color
filters 98 and heat absorber 100 to an illumination source 102,
such as a 100 watt tungsten-halogen lamp. Fiber optic cable 94
is routed through the tonometric catheter lumen in a fashion
similar to the conductor 56 of the above-described embodiments,
with the end thereof protruding through the tubing and into the
6ampling chamber 40. A second fiber optic cable 104 is routed
parallel to the fir6t fiber optic cable 94, with one end
protruding through the tubing and held in place ad;acent the end
of first cable 94 with a collar 106. Collar 106 may be
adhesively bonded to the out~ide wall of the tubing. The
oppo6ite end of second fiber optic cable 104 iB positioned for
optically coupling with a phototran6i6tor 108 which i~
electrically connected to an operational amplifier circuit 110.
The operational amplifier circuit can be coupled to an analog to
digital converter, 6uch as A/D converter 170 of ~igure 7.
~r~
In use, fiber optic cabye 94 ill-~minates a region
within the ~ampling chamber 40 whi~h i6 fill~d with a ~ampling
fluid containing ~ colorimetric pH~indicator~ The illumination
from fiber optic cable 94 reflect6 from the molecules ~uspended
in the pH indicator solution, with ~ome of the reflected
illumination passing back through second fiber optic cable 104 to
the phototransistor. By 6electing the appropriate filter 98, a
monochromatic illumination or illumination of otherwise known
spectral content is employed to illuminate the colorimetric pH
indicator solution. When the color of the filtered illumination
matches that of the indicator, the illumination i6 absorbed and a
low illumination signal i6 received at the phototransi~tor. When
- 17 -
1 335708
a pH change causes a color change in the indicator away from the
color of the filtered illumination, more illumination is
reflected back to the phototransistor, with an attendant increase
in detected signal output. In this fashion, the proper selection
of indicator dye and illumination filtration can be used to
detect pH ranges. For a further description of fiber optic pH
sensor technology, refer to G. G. Vurek "A Fiber optic PC2
Sensor, n Annals of Biomedical ~nqineerinq, Vol. 11, pp. 499-510,
1983, which is available from Pergamon Press, Ltd.
While the preferred embodiments have been disclosed in
connection with monitoring of the gastrointestinal tract and the
urinary and ureteric tracts it will be appreciated that its
principles are applicable to other hollow internal organs to
monitor pH and hence perfusion of those organs. Also while
several presently preferred detailed constructions for tonometric
catheters have been disclosed, it will be appreciated that other
constructions may be developed which are equally suitable. The
disclosed constructions are presently preferred for the reason
that they are readily fabricated using existing available
materials. Other embodiments may include other, but equivalent
materials for the tonometric catheter membrane and/or connective
tubing. They may also differ in the specific fabrication
details. As an example, the sampling chamber may be eccentric
rather than symmetric about the connective tubing.
In still another embodiment, conventional gas analyzers
may be employed externally. A device such as that shown in
Figure 1 may be used in combination with a pump or aspiration
means (not shown) for continuous or regular intermittent
- 18 -
1 3~708
aspiration of a 6ample of the aspirating liquid or medium that is
used to fill the sampling chamber 40. The gample removed by pump
or aspiration means via attachment to the luer-lock 24 can be
optionally designed 80 that the sample a6pirated at each sampling
interval can be brought in contact with an exterior, ~eparate gas
analyzing means or sensor (not Fhown) to determine the pH, pO2~
PCO2 and/or the like, of the sample. Such automatic ~ampling can
be conducted employing a sy6tem as ~hown in Figure 12. In the
a6sembly a sampling ~ystem employs a personal computer to conduct
evaluations and analysi6 of the ~amples withdrawn from the
tonometric catheter 299.
Pump 203 i8 loaded with the ~ampling or aspirating
~edium such as 6aline. Next, valve 201 iB activated to withdraw
a de~ired amount of the sampling fluid. The valve 201 is
deactivated and pump 203 is u~ed to enforce the sampling chamber
of the tonometric catheter 299 using a calibrated amount or
optionally a pressure transducer 215. The sampling fluid or
~edium i~ allowed to come to equilibrium with the wall of the
organ or area of interest. Next the ~dead space, n i.e., the ~rea
of the lumlen filled with the ~4mpling fluid that is not in
equilibrium, is removed by activating valve 205, activating
pump 207, activating valve 209 and infusing pump 207; the
waste 219 is discarded. A ~ample for analy~i6 is then withdrawn
by deactivating valve 209, activating pump 207 to then deliver
the sampling to a gas analyzer (not ~hown) that provides data
from the sample to the PC 217, and the evaluation i~ conducted as
described herein.
-- 19 --
t
1 335708
The cample gas analyzer or a ~eparate gas analyzer may
be employed to determine the bicarbonate concentration in the
arterial blood of the patient, as described above.
Another embodiment of the tonometric catheter is
illustrated in Figures 11 and llA. As illustrated, the
tonometric catheter is appropriately configured to also serve as
a urinary or ureteric catheter, either with or without ~uction,
which optionally employs sensors. With reference to Figures 11
and llA, the tonometric catheter 220 comprises a multipassage
tubing 262 whi~ch defines three individual noncommunicating
(between each other) pas6ageways or lumen~, an optional air or
irrigation lumen 264, a drainage or suction lumen 266 and a
tonometric catheter lumen 268. A tonometric catheter membrane,
6imilar to that previously described, is attached at a distal
location on tubing 262, allowing an intermediate portion of the
tubing not exten~;ng beyond the end of membrane 236 to define the
uretary or uretary catheter 270. Tubing 262 is provided with a
plurality of perforations 272 which communicate between
tonometric catheter lumen 268 ~nd the ~ampling chamber 240
defined by membrane 236. If desired, one or ~ore sensors 242 can
be included in ~ccordance with the above teachings, in which case
a suitable conductor 256 may be routed through tonometric
catheter lumen 268 to exit at ~ealed aperture 258.
The urinary catheter or ureteric catheter portion 270
is suitably provided with a plurality of openings 274 throu~h
which the bladder or ureters may be aspirated or lrrigated.
At the opposite end of tubing 262 the tubing 6plits to
form three separate connections. Air or irrigation lumen 264
optionally communicates with air lumen passageway 276, urinary
- 20 -
1 3357û8
lumen connect6 with suction or drainage lumen pa6sageway 278 and
tonometric catheter lumen 268 communicate6 with tonometric
catheter lumen pa~sageway 280. The tonometric catheter lumen
pa6sageway i~ fitted with three-way stopcock 230, similar in
function and purpo6e to the three-way ~topcock 30 de6cribed in
connection with Figure 1. If desired, a quick connect fitting 82
as seen in Figure 4 may be u~ed to couple the ~uction urinary
passageway 278 with an aspiration source. As illustrated, the
quick connect fitting preferably ~ag angularly cut ends and a
slightly enlarged midsection, making it easy to in6ert into the
end of passageway 278 and al~o into the a~piration ho6e coupling
(not shown). ~he enlarged midsection help~ form a ~eal with the
adjoining pa~ageway~. Preferably the quick connect fitting is
fabricated of di6posable plastic.
Yet ~nother embodiment of the urinary
catheter/tonometric catheter combination illustrated in
Figures 11 and llA may employ a multiple tonometric catheter
embodiment employing a tubing having a plurality of passageways
or lumen a~ 6hown in the cro6s-sectional view of Figure 5A.
In ~nother embodiment of the present invention, a
tonometric catheter may be ~dopted to deliver a
pharmaceutically-active agent, either for sy~temic, local or
topical activity, or a combination thereof. For example, an
~dditional lumen may be added such as that and for irrigation or
~piration, to deliver the active. For example, the
irrigation/aspiration lumen 264 shown in Figure 11 ~nd llA, may
be used to deliver an active agent. In another embodiment, a
portion of the device may be modified ~o as to provide 6ustained
release of the active aqent of interest.
- 21 -
1 335708
Thus, for example, the problems of nosacomial infection
associated with catheter insertion can be overcome by
incorporating an antimicrobial into At least a portion of the
polymeric material used to manufacture the tonometric catheter,
or by coating at least a portion of the device with a sustained
release composition, or by delivering the antimicrobial via the
tonometric catheter. Such modifications are well known to those
~killed in the art. See U.S. Patent No. 4,677,143,
Classes of useful agents include antimicrobial agents,
nonsteroidal anti-inflammatory agents, topical anesthetics,
topical vasodialators, ~etabolic suppressants, and other agents
that could be delivered for absorption at the sites of the
tonometric catheter.
Accordingly, while several preferred embodiments of the
invention have been disclosed, it will be appreciated that
principles of the invention, as set forth in the following
claims, are applicable to other embodiments.