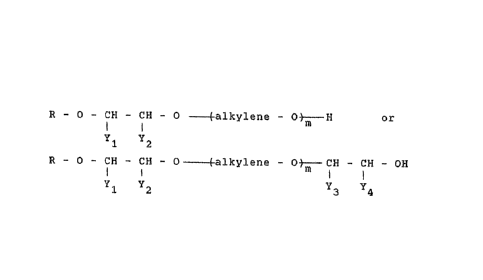Note: Descriptions are shown in the official language in which they were submitted.
2~
- 1 -
1- 17626/+
Styrene oxide products
The present invention relates to novel styrene oxide products and the use thereof as textile
processing aids, in particular as emulsifiers, dyeing assistants and especially as wetting
agents or padding assistants.
The styrene oxide products according to the present invention conform to the formula
(1) R~CH--CH--O ~aL~cylene--OtH or the formula
Yl Y2
(2) R-O- I H--I H-O~ allylene-O~ I H-l H-OH
and are also prepared in the form of their acid esters and salts thereof.
In the formulae (1~ and (2), R is an aliphatic radical having 1 to 24 carbon atoms.
"alkylene" is an alkylene radical of 2 or 3 carbon atoms such as ethylene or propylene or a
combination thereof. m is a number from 1 to 100, pre~erably from 2 to 50.
Of the substituent pairs (Yl and Y2) and (Y3 and Y4), one Y is phenyl and the other Y is
hydrogen.
The substituent R is advantageously the hydrocarbon radical of a saturated or unsaturated
aliphatic rnonoalcohol having 1 to 24 carbon atoms. The hydrocarbon radicals can be
straight-chain or branched. Preferably, R is an alkyl or aLIcenyl radical having 4 to 22
carbon atoms.
~;uitable aliphatic saturated monoalcohols are for example methanol, ethanol, propanol,
isopropanol, butanols, pen~anols, hexanols, heptanols, octanols, nonanols, decanols, lauryl
alcohol, myristyl alcohol, cetyl alcohol, stearyl alcohol, arachidyl alcohol or behenyl
alcohol, and also oxo process alcohols such as, in particular, 2-ethylbutanol,
2-methylpentanol, 5-rnethylheptan-3-ol, 2-ethylhexanol, 1,1,3,3-~etramethylbutanol,
2-octanol, isononyl alcohol, trimethylhexanol, trimethylnonyl alcohol, C9-Clloxoalcohol,
tridecyl alcohol, isotridecanol or linear primary alcohols (Alfols) having 8 to 18 carbon
atoms. Some Alfol representa~ives arc Alfol (8-10), Alfol (9-11), Alfol (10-14), Alfol
(12-13) or Alfol (16-18). ("Alfol" is a registered trade mark).
Unsaturated aliphatic monoalcohols are for example vinyl alcohol, allyl alcohol,dodecenyl alcohol, hexadecenyl alcohol and oleyl alcohol.
The alcohol radicals can be present singly or in the form of mixtures of two or more
components, such as mixtures of alkyl ancUor alkenyl groups derived from soyabean fatty
acids, palm kernel oil fatty acids or tallow oils.
(Alkylene-O)m- chains are preferably of the ethylene glycol, propylene-ethylene glycol or
ethylene-propylene glycol type; the ~Irst is particularly prefe~ed.
m is preferably 2 to 25.
The styrene oxide products of the forrnula (1) are prepared by first etherifying the aliphatic
monoalcohol with 1 mole of the styrene oxide and then by the addition of 1 to 100 moles
of an alkylene oxide (ethylene oxide and/or propylene oxide) to the resulting R-O-
substituted phenylethanol.
The styrene oxide products of the forrnula (2) are prepared by etherifying the products of
the ~ormula ~1) terminally with a further mole of styrene oxide.
The etherification of monoalcohol with styrene oxide and also the terminal addition of
styrene oxide to prepare the products of the ~ormula (2) is advantageously carried out in a
virtually anhydrous or substantially anhydrous medium at a temperature of 40 to 90C
under atmospheric or super atmospheric pressure and in the presence of an acidic catalyst,
e.g BF3 etherate, p-toluenesulfonic acid, or in particular concentrated sulfuric acid
(90-98 %). The acid is then removed by neutralizing and filtering.
The addition of the aLlcylene oxide to the substituted phenylethanol is carried out in a
2 ~ 2 L/~L
conventional manner using e,hylene oxide or propylene oxide or alternately ethylene
oxide and propylene oxide or a mixture of ethylene oxide and propylene oxide.
The acid esters can be present, depellding on the acid radical, in the form of monoesters,
diesters or half esters and as free acids or preferably as salts, for example aL~ali metal salts
or alkaline earth metal salts or ammonium salts. Suitable aL~ali metal salts are in particular
the sodium, potassium and lithium salts, suitable alkaline earth metal salts are magnesium
and calcium salts, and suitable ammonium salts are the ammonium, dimethylammonium,
trimethylammonium, monoethanolammoniunl, diethanolamtnonium and
triethanolammonium salts. Preferably, the acid esters are prepared as ammonium salts.
Mono- or diethanolammonium salts may additionally be etherified with 1 to 4 oxyethylene
units.
The acid esters are prepared by reacting the nonionic sty~ene oxide product of the formula
(1) or (2) according to the present inYention with an at least dibasic oxygen acid and if
desired converting the resulting acid ester into the abovementioned salts.
Suitable polybasic oxygen acids for fonning the acid esters are sulÇonated or sulfonyl-free,
organic, preferably aliphatic, dicarboxyl;c acids of 3 to 6 carbon atoms, e.g. maleic acid,
malonic acid, succinic acid or sulfosuccinic acid, and polybasic inorganic oxygen acids,
e.g. sulfuric acid or orthophosphoric acid. In place of the acids it is also possible to use
their functional derivatives such as anhydrides, halides or amides. Examples of these
functional derivatives are maleic anhydride, phosphorus pentoxide, chlorosulfonic acid
and sulfamic acid. The phosphoric esters are advantageously formed as mixtures of a
monoester and diester.
The esterification is in general carried out by simply mixing the reactants and heating,
advantageously to a temperature between 50 and 100C. The free acids which are
initially forrned can subsequently be converted into the corresponding aLkali metal or
ammonium salts. The conversion into the salts is carried out in a conventional manner by
adding bases, for example ammonia, monoethanolamine, triethanolamine or an alkali
metal hydroxide, for example sodium hydroxide or potassium hydroxide. In a particularly
preferred embodiment, sulfuric acid esters are directly prepared in the form of their
ammonium salts by heating the nonionic styrene oxide products with sulfamic acid in the
presence or absence of urea.
42~L
Industrially important styrene oxide products conforrn to the formula
(3) RI--O~CH~CH--O lcH2cH2ot H
Yl Y2
where Rl is aL~cyl or alkenyl each having 1 to 22 carbon atoms, preferably 4 to 22 carbon
atoms, one of Yl and Y2 is phenyl and the olher is hyc'lrogen, and ml is from 2 to 40.
Preferred acid esters prepared with an inorganic or organic acid conform to the formula
(4) Rl--O--CH~ CH--0~ CH2CH2(~ X
m
Yl Y2
or the formula
(5) R~H~ CH2CH2~ c~ ~x
Yl Y2 Zl Z2
or the formula
(6) Rl--O--I H--~ H2 l H2(~ ( CH~ C}12--tn2
Yl Y2 Zl Z2
where R~ , Y2 and ml are each as de~ined, one of Zl and ~ is mc~hyl and the other is
hydrogen, X is the maleic acid, sulfosuccinic acid, sulfuric acid or phosphoric acid radical,
and the sum nl ~ n2 is from 2 to 30, preferably from 4 to 18.
Preferred acid esters of the fo~nulae (4), (5) and (6) contain either a maleic ester group or
a sulfuric acid ester group, which are each preferably present in the form of the alkali
metal salts or ammonium salts.
Of particular interest are the acid phosphoric esters of the addition products of the forrnula
(3) in which the phosphoric acid esters are aclvantageously present as mixtures of the
corresponding salts of a mono- and diester.
2~
If desired, the styrene oxide products of the formula (3) may be terminally etherified with
a further mole of styrene oxide and if necessary then converted with an at least dibasic
oxygen acid into the acid ester and into the corresponding salt. Both the terminal styrene
oxide addition and the subsequent esterification can be carried out under the above-
mentioned conditions.
Styrene oxide products according to the present invention are suitable for a wide range of
uses in t~xtile application, such as pretreatment, dyeing or finishing. Nonionic unesterified
products are useful in particulal as assistants in the dyeing of polyamide-containing fibre
materials with anionic dyes or dye mixtures. The corresponding acid esters, in particular
dicarboxylic monoesters or acid esters of sulfuric acid and primarily phosphoric esters, by
contrast, are used in particular as wetting agents, deaerating agen~s and foam suppressants
in aqueous systems, in particular in the dyeing o~ natural or synthetic fibre material,
primarily in the dyeing of cellulose textile materials, polyester fibres or natural or
synthetic polyamide fibre materials. Both as nonionic products and as acid esters, the
styrene oxide products according to the present invention improve, if fonnulated as
described, the affinity of the dyes and thereby speed up the diffusion of the dyes into the
fibres.
~he present invention accordingly also provides a method for processing (finishing)
natural or synthetic flbre material in the presence or absence of appropriate dyes, which
comprises processing in the presence of the nonionic and/or anionic styrene oxide
products according to the present invention.
The amounts in which the styrene o~cide products according to the present invention are
added to the processing liquors, such as dyeing liquors or pretreatrnent or aftertreatment
liquors, vary with the substrate, advantageously between 0.5 and 20 g, prefe~ably be~ween
1 and 10 g, per litre o~ liquor.
Advantageous forrnulations of the styrene oxide products according to the present
invention contain them as nonionic andlor anionic compounds with or without water in
combination with non;onic or anionic dispersants, fatty alcohols, fatty acid amides,
alkylene bis fatty acid amides, alkyl alkenyldicarboxylates, metal stearates, silicone oils,
e.g. -dialkylpolysiloxanes, or even mineral oils or alkanolamines in stable, wetting and,or
defoaming preparations. 3uch preparations are also capable of virtually completely
deaerating aqueous systems. It is thus possible to avoid air inclusions not only in the
application baths but also in the substrates. Such deaeration makes it possible to avoid
spotting in dyeing and finishing.
Preference is given to aqueous or water-free prep~ations which, based on the preparation
as a whole, comprise
(A) 2 to 50 % by weight of an acid ester of the styrene oxide product of the formula (1) or
(2),
(B) 5 to 50 % by weight of a nonionic sur~actant, preferably an aliphatic monoalcohol
having 6 to 22 carbon atoms or the product of the addition s~f 2 to 80 moles of ethylene
oxide to 1 mole of an aliphatic monoalcohol having 6 to 22 carbon atoms or a
polyoxyethylene derivative of a sorbitan fatty acid ester or of a slyrene oxide product of
the formula (1) or (2) or mixtures thereof and at least one of ~he following components:
(C) 1 to 30 % by weight of a silicone oil, for example a diaLkylpolysiloxane such as
dimethylpolysiloxane,
~ ,,
(D) 10 to 60 % by weight of a rnineral oil, for example a paraffin oil such as Shell oil
L 6189 or mineral oils Esso 301 to 312,
(E) 20 to 45 % by weight of a dialkyl ester of an ethylenically unsaturated aliphatic
dicarboxylic acid, preferably containing 4 to 12 carbon atoms per each alkyl moiety, e.g.
bis-2-ethylhexyl maleate or bis-2-ethylhexyl citraconate,
(F~ 10 to 70 % by weight of a diffusion accelerant, in particular an aliphatic or aromatic
carboxylic ester such as C4-Cl2alkyl lactate, C4-Cl2alkyl benzoate, phenyl benzoate or
benzyl benzoate, or else an alkylbenzene, such as trimethylbenzene or ethylbenzene,
(G) 0.5 to 5 % by weight of a salt of a C1O-C~4fatty acid and a polyvalent metal, e.g.
magnesium distearate~ calcium dibehenate or aluminium ~ristearate, and
(H) 0.5 to 3 % by weight of a Cl-C4aL1sylenediamide of a fatty acid having 10 to 24 carbon
atoms, e.g. methylenebisstearamide, ethylenebisstearamide or ethylenebisbeherlamide.
Further preferred aqueous preparations which are in particular used as assistants for
2~~424
- 7 -
dyeing natural or synthetic polyamide fibres, especially wool, with anionic clyestuffs,
comprise based on the preparation as a whole,
(a) 15 to 50 % by weight of a nonionic styrene oxide product of the fonnula (1) or (2),in particular a styrene oxide product of the formula (3),
(b) 1 to 5 % by weight of an acid ester or salt thereof, for example an alkali metal or
arnmonium salt~ of a compound of the fo2mula
~' ;~."
(CH-CH-03~H
(7) R'-N
(cl~H-~H-o3~H
Z' Z"
or of a quaterni~ed product of the acid ester or salt thereof, where R' is alkyl or aLkenyl
having 12 to 22 carbon atoms, one of Z' and Z" is hyclrogen, methyl or phenyl and the
other is hydrogen and sl and s2 are each integers such that their sum, s1 + s2, is from 2 to
100,
(c) 2 to 10 % by weight of a quaternary ammonium compound of the formula
z' z"
~(1HC~O3~H ~
(8) R''-NI\ An
. _ Z Z
where R" is an aL~cyl or aL1cenyl radif~al having 12 to 22 carbon atoms, Y' is a substituted or
unsubstituted alkyl, such as methyl, ethyl, benzyl or -CH2CONH2, one of Z' and Z" is
hyclrogen, methyl or phenyl and the other is hyclrogen, An~ is an anion of inorganic or
organic acid, and P1 and P2 are each integers such that the sum of Pl and P2 is from 2 to
100, with or without one or more of the following components:
(d) 0.5 to 2S % by weight of a polyalkylene glycol ether of the formula
(9) R --(~alkylene-O~ H
where R"' is an alkyl or alkenyl raclical having 4 tv 22 carbon atoms, "alkylene" is
8 21~89-7902
ethylene or propylene, and q is from 2 to 35, and
(e) 0.5 to 5% by weight of a nit.rogen-containing nonionic
compound of the ~ormula
(10) ~,~Il'fll-N-(cll2cll2-c)J;~
y' Y" c~ l2
~fll-CII-N C1121112
N-~c1l2cll2
whereln R" is an alkyl or alkenyl radica]. having 12 to 22 carbon
atoms, one of Y' and Y" is phenyl and the other ls hydrogen, and x
and y are each integers such that the sum of x and y ls from 80 to
140.
Further details and pre~erred comblnations and use
forms of components (b), (c), (d) and ~e) may he found in
DE-A-1,568,258, publication date April 9th, 1970, DE-A-1,619,385,
publication date June 9th, 1971, EP-A-8900~, publicatlon date
September 21st, 1983 and EP-A-312493, publicatlon date Aprll l9th,
1989.
In the preparatlon and use examples below, percentages
and parts are by weight, unless otherwise stated. The dye
quantities are based on commerclal, i.e. extended material, and
the auxiliary quantitles are based on pure substance.
PreParati~
ExamPle l,a) 540 g of 3,5,5-trimethylhexanol are heated to 80C
to~ether with 15 g of 96~ sulfurlc acid. 300 g of styrene oxide
are added dropwi~e over 70 minutes, during which the temperature
ls maintalned at between 80 and 90C. The reaction mixture is
8a 21489-7902
then stirred at 86C for 5 hoursl and thereafter s~irred until
cold, neutralized with sodium bicarbonate solution and filtered,
and the aqueous phase is separated off. After the excess 3,5,5-
trimethylhexanol has been distilled off, the residue is subjected
to a fractional distillation under a high vacuum.
The result obtained is 304.6 g of a colourless product
of formula
A
;24
ICH3
(ll) CH3--f--CH2--I H--CH2--CH2-- FH CH2 OH
~H3 CH3
b.p.: 105-106C/10-2 OH number: 213
b~ 263 g of 3,5,5-trimethylhexyloxyphenylethanol of the formula ~11) are reacted in the
presence of 1 % sodium methoxide with 88 g of ethylene oxide at 140C and 8 bar
pressure.
The result obtained is a product of the forrnula
(12) (CH3)3C--CH2--fH--CH2CH2--~ CH--CH2--O--( CH2CH20 2tH
CH3
~3
OH number: 156
c) 70.2 g of the product of the formula (12) are heated to 60C and stirred with 20.8 g of
urea for 15 minutes. 20.8 g of sulfamic acid are then added, and the mixture is heated to
70C, stirred at 70C for 1 hour, heated to 95C and stirred at 95C for a further 2 hours.
Addition of 111.8 g of water gives a viscous, gellike solution of the product of the formula
(13) (CH3)3C--CH2--fH--CH2CH2--0~ CH--CH2--O--( CH2CH2~ SO3NH4
CH3
~1
Example 2: a) 333 g of n-butanol are heated to 65C together with 13.4 g of 96 % sulfuric
Z~ 4;~
- 10-
acid. 345.6 g of styrene oxide are added dropwise over 30 minutes during which the
temperature rises to 108C. The reaction mixture is subsequently stirred at 75C for 2
hours, then stirred until cold and neutralized with sodium bicarbonate solution, and
filtered. The excess butanol is distilled off, and the product is subjected to a fractional
distillation under a high vacuum. The result obtained is 201 g of n-butyloxyphenylethanol
of the formula
4) n--C4Hg--O--CH--CH20H
b.p.: 96-100C/10-2 OH number: 286
b) 192 g of n-butyloxyphenylethanol are reacted in the presence of 1 % of sodiummethoxide with 81.8 g of ethylene oxide at 140C and 6 bar pressure.
The result obtained is a product of the fonnula
(15) ~--Ca,Hg--O--CH--CH20--( CH2CH20~ H
OH number: 199
c) 56.3 g of the adduct of the forrnula (15) are reacted with 95 g of phosphorus pentoxide
at room temperature with rapid stirring. In the course of the reaction, the temperature rises
to 65C. The mixture is subsequently stirred at room temperature for 4 hours, producing a
mixture of monophosphoric and diphosphoric esters of the formulae
'2~ L24
(16a) /OH
n--C4Hg--O--CH--CH2O ( CH2CH2 )2 1 1 OH
( 1 6b) n ~ C4Hg ~ O ~ CH--CH2--t C~2CH2)2\
~ \
~ /P--OH
n--C4Hg--- O~ CH--CH2--Ot CH2CH20)2
The reaction product is clear and viscous.
Example 3: 5 I g of the adduct of the formula
(17) CH3--O--fH--CH2--O~CH2CH2Ot H
OH number:219
prepared as described in Example lb), are heated to 60C and stirred with 20.8 g of urea.
20.8 g of sulfamic acid are then added, and the mixture is heated to 70C, stilTed at 70C
for 1 hour, heated to 95C and stirred at 95C for a further 2 hours. Addition of 92.6 g of
water gives a water-miscible, slightly viscose solution of the product of the formula
z~
- 12-
( 1 8) CH3--O--CH--CH2--O~ CH2CH2~ SO3NH4
Example 4: 63.75 g of the adduct of the formula (17) are slowly heated to 70C together
with 24.5 g of maleic anhydride. The mixture is then stirred at 70C for 1 hour, heated to
90C and stirred at 90C for a further 3 hours. The result obtained is a highly viscous
product of the formula
(l9) CH3--O--fH--CH2--O~CH2CH2OtC--CH=CH--fi--OH
O O
The acid number is 161.
Example 5: 11.35 g of phosphorus pentoxide are added with stirring at room temperature
to 61.2 g of the adduct of the formula (17). In the course of addition, the temperature rises
to 80C. The mixture is subsequently stirred at room temperature for 4 hours, turning into
a viscous mixture of phosphoric esters of the forrnulae
OH
(20a) CH3 O--CH~ CH2--(~ ( CH2CH2O )--P OH
~3
(20b) r CH3--O I~CHz--~CH2CUZO~\
4~L
The procedure of Examples 1 to 5 is repeated using appropriate starting materials to obtain
the following nonionic and anionic addition products:
(21) CH3--O--fH--CH2 O~CH2CH20~ H
,~,,
OH number:92
~22) CH3--O--CH--CH2--O~ CH2CH20t SO3NH4
~3
(23) CH3--O--CH--CH2--(~CH2CH20~ CO--C~F C~ COOH
Acid number: 78.8
Mixture of a phosphoric monoester and diester of the formulae (24a) and (24b)
OH
(24a) CH3--O--C~--CH2-- ~ CH2CH2~t P--OH
0
~&~24~
- 14-
(24b) [ C~13 O~CHr O~C1~2C~2 -- \p_
(25) C2H5--O--j H--CH20~ CH2CH20~ H
OH number: 223
(26) C2H5--O--CH--CH2ot CH2CH20~ S03NH4
(27) C6 H13 0 f~l C~120~ CH2CH20t--H
OH number: 185
(28) C6 H13----fH--CH20--( CH?cH2ot S03NH4
4;~:~
- 15-
(29) C4H9 --FH--CH2Ot CH2CH20t CO--C~F CH--COOH
Acid number: 148.5
(30) C6H13 --ICH CH2t CH2cH2ot CO--CH= CH--COOH
~13
Acid number: 145
(31) C8H17--o--ICH--CH20 tcH2cH2otH
OH number: 156
(32) C8H17--O--CH--CH20 ( CH2CH20t SO3NH4
(33) C8H17 O CH--CH2t CH2CH20~ CO--CH= CH--COOH
~,3
24
- 16-
Acid number: 127
34) ( CH3 ) 3 C--CH2fH--CH2CH20--CH--CH2--0~ CH2CH20t--S03N~14
CH3
(35) ( CH3 ) 3 C--CH2fH--CH2CH2(~ CH--CH2--t CH2CH20t COC~F CH--COOH
c~3
Acid number: 98
(36) ( CH3 ) 3 C--CH2fH--CH2CH2(~ CH--CH2--0~ CH2CH20t H
CH3
OHnumber: 116
Mixture of a phosphoric monoester or diester of the formulae (37al and (37b)
OH
(37a) ( CH3 ) 3 C--CH2CH~ CH2CH20--CH--CH2--0~ CH2cH2ot ~ OH
10 11
CH3 O
~3
(37b) 1 (C113)3C--CII~CIl--CII~C1121~ :~ Cll~--OtCll~Cll~C~¦ \
(38) C1~3--(CH2)11--O--Cll--CH20~ CH2CH20~ H
g~
OH number: 154
(39) CH3 (CH2)11 fl-l cll20tC~I2CH20~ co c~ Cl~ COOH
~3
Acid number: 124
Mi~cture of a phosphoric monoester and diester of the forrnulae (40a) and (40b)
o}~
(40~) C~ 3--O--CH--CH2--O~C112CH20~11 OH
~3
- 18
- C6H13--O--CH--CH2 O~ CH2CH2~
(40b) ~3 ~Il OH
(41 ) CH3--( CH2 )11--O--C~ CH2-O~ CH2CH20~ SO3NH4
Mixture of a phosphoric monoester and diester of the formulae
OH
(42) CH3--( CH2 )11--O--fH--CH2-~ CH2CH2~ ~ l OH
(43) ~ CH3--(c~l2)lr~cll2-o~cH2cH2o~ P--OU
(44) CH3~CH2~--fH--CH2--o--CH--CH20 t CH2CH20~ H
C2H5
~3 '
O~ number: 177
& ~ 2 L~L
- 19-
(45) CH3(CH2)3 fH CH2--O--CH--CH20~ CH2CH20~ S03NH4
C~H5
~3
(46) CH3(CH2)3 ICH CH2--O--CH--CH20t CH2CH20~ C--CH= C~ COOH
C2H5 ¦ o
~\
Acid number: 140
Mixture of a phosphoric monoester and diester of the formulae
OH
(47a) CH3~CH2~3--fH--CH2--O--CH--CH20~CH2CH20~ P OH
C2HS ¦ O
(~7b) CH3 ( CH2 )3--CH--CH2--O--CH--CH2(}t CH2CH20~
C~H5 ~ P--ON
CH3 ( CH2 )3 CH--CH2--O--CH--CH20~ CH2CN2
C~H5 [~
- 20 -
(4~) Cg Cll alkyle--O--CH--CH2--OtcH2cH2o~H
~3
OH number: 160
(49) Cg Cll alkyle O--CH--CH2--OtCH2CH20~ SO3NH4
Example 6: 120 g of the addition product of the formula (36) (OH number: 116) are
admixed with 1.8 g of concentrated sulfuric acid (96 %), and the mixture is heated to 65.
30 g of styrene oxide are then added dropwise over 30 minutes, during which the
temperature rises to 82C. The reaction product is then stirred at 75C for 5 hours,
n~utralized with sodium bicarbonate solution and filtered. The filtrate is concentrated, and
the residue is dried. The lwsult obtained is a clear, yellow product of the formula
(so) (CH3) 3 C--CH2--CN--CH2CH2--O--CH--CH20t CH2C~12~ CH--~ CH2 OH
OH nurnber: 95
~:
Use examples : ~ :
::
Example 7: 100 g of wool serge are wetted out in a circulatlon machine at 40C in the :
course of lS minutes with a:~liquor comprising 1.1 Iltres of water and~the followmg
additives~
: ~
. .
24
21 -
3.2 g of 80 % acetic acid
5.~) g of calcined sodium sulfate
1 g of the styrene oxide product of the formula (38).
5 g of the dye ~cid Black 172 C.I. 15711 are then added. ~fter 10 minutes, the dyeing
li~uor is heated to 85C in the course of 45 minutes, and the textile material is maintained
at that temperature for 60 minutes with constant circulation. Tlle dyebath is then
cornpletely exhausted (dye on the ~lbre). The textile is then rinsed and dried. ~ deep and
fast black dyeing is obtained on the wool without the conventional heating to the boil. The
fastness properties correspond to a dyeing at the boil (98C).
Similar results are obtained Oll replacing the styrene oxide yroduct of the formula (38) by
the same amount of the bisstyrene oxide product of the formula (50).
Example 8: A circulation dyeing machine is entered with 100 kg of wool packages on a
spindle support. In the make-up vessel, 12001 of water are heated to 60C. A solution is
prepared therein of 1200 g of an aqueous formulation which, based on the formulation,
contains
12 % of silicone oil, for example dimethylpolysiloxane,
15 % of 2-ethylhexanol
15 %ofparaffinoil
2 % of a water-soluble, surface-active siloxaneoxyaL~cylene copolymer
8 % of thç anionic styrene oxide product of the formula (39) and
2 % of the nonionic styrene oxide product of ~he formula (38)
and which has been adjusted tv pH 8 with monoethanolamine. The liquor is then pumped
from the make-up vessel through the material into the dyeing machine and subsequently
circulated in alternating direction~ Owing to the addition of the product, the dyeing system
is spontaneously deaerated and consequently the textile material is ~horoughly pene~ated.
Thereafter 2 kg of the dye of the forrnula
- 22 -
NH--CH3
S03H
~N=N~
( 101) CH2 = C ~ CO--NH~
Br H0 ~
S03H
are added to the liquor, followed by 1 kg of a 1:1 mixture of the
chloroacetamide-quaternized product of the addition of 7 moles of ethylene oxide to 1
mole of a Cl6-Cl8fatty amine mixture and the ammonium salt of the sulfuric acid ester of
the product of the addition of 7 moles of ethylene oxide to 1 mole of a Cl6-Cl8fatty amine
mixture (50 % aqueous preparation), the dyeing liquor is heated to the boil in the course of
30 minutes, and the wool is dyed at the boil for 60 minutes. Virtually no foam develops
~uring dyeing. The result obtained is a strong and uniform dyeing of the package.
The formulation used in this Example is prepared as follows:
12 parts of silicone oil are dissolved in 15 parts of 2-ethylhexanol. 15 parts of paraffin oil,
2 parts of water-soluble, surface-active siloxaneoxyalkylene copolymer, 8 parts of the
maleic monoesters of the formula (39), 2 parts of the styrene oxide product of the formula
(38), 47.6 parts of water and 0.4 parts of monoethanolamine are then added with
continuous stirring, and the mixture is subsequently stirred for a further 30 minutes. ~he
result obtained is a stable formulation having a wetting, deaerating and in particular
foam-suppressing effect.
Example 9: In a winch beck, 100 kg of cotton knitwear are wetted out in 30001 of water
containing 3 kg of an aqueous preparation containing, based on the product,
7 % of a silicone oil
11 % of 2-ethylhexanol and
11 % of the anionic styrene oxide product of the forrnula (41). The cotton is completely
wetted and deaerated within 30 seconds. 2 kg of a dye of the formula
0~4;Z 4L
N~N
SO3H OH NH~ NH2
(102) ~N=N ~
HO3S SO3H
are ~hen added to the liquor together with customary chemicals such as electrolytes and
alkalis, and the knitwear is then dyed at the boil for two hours. There are no problems with
fo~ming, The dyed material has a uniform and non-spotty shade.
The preparation used in the Example is prepared as follows:
First, 70 parts of silicone oil are dissolved in 110 parts of 2-ethylhexanol at room
temperature. 110 parts of the styrene oxide product of the formula (41) are then added,
followed 30 minutes later by 710 parts of water, and thereafter the mixture is stirred for a
further 30 minutes. The result obtained is a stable formulation having excellent wetting
and deaerating properties.
Example 10: In a short liquor jet, 100 kg of cotton tricot are wetted out in 600 litres of
water at 40C. The liquor is then turned into a dyebath ~by adding 36 kg of sodium
chloride, 5 kg of the dye of the formula (102) and 0.5 kg of a preparation containing
186.75 g of rnineral oil (for example Shell Oil L 6189),
185 g of bis-2-ethylhexyl maleate,
g of magnesium distearate,
8.25 g of N,N-ethylenebisstearàmide, ~ ~ ~
g of a polyoxyethylene derivative of sorbitan tristearate with 20 oxyethylene
units, e.g. Tween 65, and
g of the anionic styrene oxide product of the forrnula (13).
The textile material is dyed at 40C for 45 minutes. 0.6 kg of calcined sodium~carbonate is
then added, followed after a funher 5 minutes by;1.2 kg of ~aqueous 36 %~ sadiumhydroxide solution. The~ tricot is then dyed `for a~ further 40 rninutes, and then~ rinsed and
washed. The result obtained ls a fast,~level~red dyeïng on the tncot. During the dyeing
;, . ::
- : :
2~
- 24 -
process, there is no foam and there are no problems with the circulation of the textile
material.
The preparation used in this Example is prepared as follows:
186.75 g of mineral oil, 185 g of bis-2-ethylhexyl maleate, 10 g of magnesium distearate
and 8.25 g of N,N-ethylenebisstearamide are heated with continuous stirring at 110C
until a clear solution is s)btained. The solution is cooled down to 45C in the course of 5
minutes, and S5 g of a polyoxyethylene derivative of sorbitan tristearate with 20
oxyethylene units, e.g. Tween 65, and SS g of the anionic styrene oxide product of the
fo~nula tl3) are dispersed therein. Thc result is a stable formulation which is very
effective in particular as an antifoam in aL~aline liquors and at high shearing rates.
Example 11: 100 parts of polyester fabric are introduced into a hot dyeing liquor at 60C
containing 1300 parts of water, 2 g/l of ammonium sulfate, 2.5 parts of a dye of the
formula
Cl N~
( 10~) N2 ~ N = N ~ NH--C~12CN3
N02 NH--CH2CH20H
and 2 parts of an assistant fonnulation consisting of
16 parts of the anionic styrene oxide product of the formula (39),
24 parts of the product of the addition of 18 moles of ethylene oxide to 1 mole of a
Cl2-Cl8fatty alcohol mixture and
60 parts of benzyl benzoate
and has been adjusted to pH 5 with formic acid. The liquor temperature is raised to 130C
in the course of 30 minutes and left at that temperature for 60 minutes.
The liquor is then cooled down to 70C, and dropped, and the polyester fabric is rinsed.
Even without the customary reduction clearing a level and rub-fast red dyeing is obtained
4~4
- 25 -
with a high colour yield.
The assistant forrnulation used in this Example is prepared as follows:
In a heatable stirred vessel, 60 parts of benzyl benzoate are heated to 60C with
continuous stirring, and 24 parts of the product of the addition of 18 moles of ethylene
oxide to 1 mole of a Cl2-Clgfatty alcohol mixture and 16 parts of the styrene oxide
product of the formula (39) are then stirred in. Cooling down to room temperature leaves a
stable formulation which is suitable in particular for dyeing polyester fibres.
Example 12: 10() parts of textured polyester yarns in package form are introduced into an
HT dyeing machine which contains 8û0 par~s of hot water, 40C, 2 parts of ammonium
sulfate, 4 parts of a dye of the forrnula
-
(104) ~--CH2CH2U~N--N ~ N = N ~3OH
OCH3
and 2 parts Gf the assistant formulation described in Example 11 and whose liquor hasbeen adjusted to pH 5.5 with forrnic acid. The temperature of the liquor is then raised to
128C in the course of 40 minutes, and the textile material is dyed at that temperature for
60 minutes; During the heating-up phase no increase in the differential pressure between
the inside and the outside of the yarn package is detectable. Thereafter the liquor is cooled
down to 70C, and the substrate is as usual reduction cleared, rinsed and dried. The result
obtained is a strong and level orange dyeing which is notable for good penetration and
good fastness properties.
Example 13: A nylon 6.6 loop pile carpet is impregnated on a padder at 25C with a
preparation which contains per litre 1.2 g of a dye of the formula
Z~ 4
- 26 -
(105) [~N N~N=N~30CH3
OCH3
S03Na
0.8 g of a dye of the formula
NH2
I~N = N ~3
C~}N--S2 HC~
CH3
S03H
0.8 g of a dye of the formula
NH2
07) ~ ~ ~ ~ 503H
O NH~ CH3
S02NHC~H2CH20H
S g of the seyrene oxide product of the formula (13) prepared as described in ~3xample 1
2 g of a thickener and
1 g of sodium acetate
and has been adjusted with acetic acid to pH ~-5.5. The liquor pick-up is 320 %. The
impregnated carpet is steamed with saturated steam in a dog house steamer at 100C for 5
minutes. The carpet is then rinsed with cold water and ~ied. The r~sult obtained is a deep
brown dyeing, without frosting, which has the expected fastness properties.
2~
- 27 -
Similar results are obtained on replacing the styrene oxide product of the formula (13) by
the same amount of the anionic s~yrene oxide products of the formulae (32) or (37a)
combined with ~37b).
Example 14: 100 g of wool serge are wetted out in a circulation machine, for example an
A Ahiba-turbomat, at 40C in the course of 15 minutes with a liquor comprising 1.1 Iitres of
water containing the following additions:
3.2 g of 80 % acetic acid
5 g of calcined sodium sulfate
2 g of an aqueous preparation containing, based on this preparation, 30 % by weight
of the styrene oxide product of the formula (38), 10 % by weight of the assistant
mixture Al described in ~P-A-0089004 and
20 % by weight of triethylene glycol monobutyl ether.
4 g of the dye Acid Black 172 C.I. 15711 are then added. After 10 minutes, the dyeing
liquor is heated to 90C in the course of 50 minutes, and the textile material is kept at that
temperature with contimlous circulation for 60 minutes. The textile material is then rinsed
and dried.
The result obtained is a deep, level and i~ast black dyeing on the wool. The dye bath is
95 % exhausted, although the dyeing was carried out at 90C. The fastness properties
correspond to those of a conventional dyeing at the boil (98C).
Example 15: The representatives of the styrene oxide products prepared as described in
Examples 1 to 5 are tested in wetting power and foam characteristics compared with
known products.
A. Wetting power
By test method AL-10-1 using the sink technique
Substrate: Ladies wool cloth
Amount used: 1 g of active substance per litre
rk
2~
- 28 -
B. Foam characteristics:
Foam test by foam plunger method ,
; Apparatus: Ahiba-Texomat *
Test solution: 200 ml of a 0.1 % aqueous solution of the
styrene oxide product
Temperature: 25C
Number of plunging movements: 60
Assessment of volume of foam: 5 minutes after foam production
C. Surface tension
0.1 g of the styrene oxide product is dissolved in 99.9 g of distilled water. The
surface tension (dynfcm) of the solution is determined with the aid of a tensiometer.
The results in the table below show that the styrene oxide products have better wetting
power and foam characteristics than the comparative products.
Table
. .~ . . . _
Styrene oxide product Wetting Foam height dyn/cm
of (sink time) (ml) (mN/m)
. .__
Formulae (40a) and (40b) 4" 25 34.3
Forrnula (27) 2" 0 35.0
Formula (31) 7" 0 33.8
Formula (33) 7....... 0 35.4
Formula (35) ~ ~ 20 30.4
Formula (36) 6" 10 30.4
Formula (12) 8" 0 30.9
Ammonium salt of the sulfuric
ester of the addition product 8" 225 30.1
of 2 mol of ethylene oxide to
I mol of p-nonylphenol
k