Note: Descriptions are shown in the official language in which they were submitted.
2~76~
PATENT 211PUSQ3827
IMPROVED PIPELINE TRANSPORTATION OF HEAVY HYDROCARBONS
TECHNICAL FIELD
The present ~nventton relates to the p~pel~ne transportatton of heavy
crude otl or bttumen, and more parttcularly to a process for reducing
diluent requtrements for such pipel~ne transportatton.
:
BACKGROUND OF THE INVENTION
The product~on of heavy crude oil and bitumen will play an important
role in future energy supplles ~n many parts of the world. Large depostts
; of these heavy hydrocarbon products extst, for example, in the UnttedStates, Canada, Venezuela, and other countr~es. As the productlon of
lighter crude oil decl~nes, part~cularly ~n the Un~ted States and Canada,
the productton and transportatton of these heavy hydrocarbon products wtll
be of growing long-term tmportance. - --
These heavy hydrocarbon products are h~ghly v~scous or even soltd at
ambtent temperatures. Heavy crude olls, character~zed by gravtttes between
about 12 and 17API, may be fluid at amb~ent temperatures but usually ~ -
15 possess viscostttes whtch are too h~gh for econom~cal p~peline transporta-
tlon, especially in the winter season ~n colder reg~ons of the world. ~ -
B~tumen, character~zed by grav~t~es between about 8 and 12API, ts soltd at
ambient temperatures. In order to transport these heavy hydrocarbon
products by ptpeltne, it ts common practtce to blend them with ltghter
20 hydrocarbon dtluents so that the resulting m~xture having a ktnemattc
vtscostty of about 250 centtstokes or less at ambtent temperature can be
transported by ptpeltne to ref~ner~es. Other methods to allow ptpeltne
, transportatton of these heavy hydrocarbon products have been used or are
betng developed, such as the use of heated p~pel~nes, suspension of these
2S heavy hydrocarbons as emulsions in water, or converting the heavy
hydrocarbons to lighter products at the o~l f~eld product~on s~te. The most
wtdely used and commercially lmportant method at present, however, is the
use of light hydrocarbon dtluents to reduce the viscostties o~ these heavy
hydrocarbons. Thts method is expected to be ~mportant in future years as
30 well.
~$ ~
2~9~6~
. . .
-- 2 --
The most commonly used d11uent for this purpose is 11ght hydrocarbon
condensate from natural gas wells; a typical condensate has a grav1ty of
about 60API. Th~s condensate d~luent ~s effect1ve, reasonable 1n cost, and
has been in adequate supply. However, as the product10n of heavy crude and
b~tumen ~ncreases in the future, ava~lable suppl~es of condensate w111 be
~nsufflc~ent to meet pro~ected requ1rements. Alternate d~luents such as
llght crude oil or l~ght hydrocarbon fract~ons obta~ned 1n the ref1n1ng of
crude o~ls can be used ~n place of condensate if suff~c~ent suppl1es of
these alternates can be obtained at reasonable cost. Another solut10n to
l~ d11uent shortfall ~s to recyc1e d~luent from ref1ner~es by p1pel1ne back to
the o~l product~on f~eld for reuse. The development of economlcal alter-
nat1ves to the use of hydrocarbon d~luents, espec~ally condensate d11uent,
and the development of methods to reduce d~luent requ~rements for p1pel1ne
transportation of heavy crude o11 and b1tumen, wlll be 1mportant to the
}5 future supply of these heavy hydrocarbons as econom~cally v1able energy
sources.
In a paper ent~tled In S~tu Development at Marguerite Lake presented
at the Alberta 0~1 Sands Technology and Research Author~ty s Advances ~n
Petroleum Technolog~ Con~erence, Calgary, Alberta, May 30-31, 1983,
R. R. Capel1ng descr~bes the recovery of heavy o11 and b~tumen from the Cold
Lake reg~on of Alberta. A process flowsheet ~s presented descr1b1ng the
lntroductlon of d~luent to reduce the v~scos~ty of these heavy hydrocarbons
pr~or to p~pel1ne transportat10n.
R. D. Urquhart, ~n a paper ent~tled Heavy 0~1 Transportat~on--Present
and Future presented at the F~rst Annual Techn~cal Meet1ng of the South
Saskatchewan Sect10n, the Petroleum Soc~ety of CIM, Reg1na, September 15-17,
1985, rev1ews the p~pel~ne transportat~on of heavy o11 and b1tumen produced
~n the Alberta and Saskatchewan provinces of Canada. The problem of gas
well condensate d~luent shortages ~s d~scussed and alternatives to the use
0 of condensate d~luents are presented.
In an art~cle ent~tled Development of Heavy 0~1 and Natural B1tumen in
Western Canada in the Journal of Canad~an Petroleum Technology, January-
February 1987, Montreal, pp. 44-47, W. N. Turner reviews the development of
heavy oil and natural bitumen ~n western Canada, and part1cularly at p. 47
,,.j
6 ~
states the requ~rements for viscos~ty reductlon of these heavy hydrocarbons
for pipeline transportation and describes a number of hydrocarbon diluents
for use in viscosity reduction.
R. T. Bailey in a paper entitled The Heavy Oil Dilemma--Problems
Solut~ons and Responsib~l~ties presented at the 38th Canadian Chemical
Engineering Conference October 2-5 1988 Edmonton Alberta reviews
problems in heavy o~l production and pipeline transportation in Alberta and
in particular ~n Figure 6 presents pro~ect~ons of diluent ava~lab~ y and
the future shortfall thereof. The availability of the currently favored
diluent gas field condensate ~s projected to decline from about 100 000
bbl/day ~n lg90 to about 50 000 bbllday in 2005.
U.S. Patent No. 1 762 423 d~scloses a method for the transportatlon of
crude oil and natural gas through a pipeline in which natural gas is com-
pressed and cooled the resulting condensate and uncondensed gas are mixed
with pressurized crude oil and the resulting m~xture is transported through
a p~pellne. The method allows s~multaneous transportat~on of crude oil and
natural gas and the v~scosity of the crude o~l ~s reduced which decreases
the pu~p~ng power requ~rements.
U.S. Patent No. 3 344 583 d~scloses a method for transporting ethane in
a crude oil pipeline by d~ssolving ethane in the crude oil before flow~ng
into the pipeline. Methods for d~ssolv~ng ethane and other l~ght hydro-
carbons in crude o~l are d~sclosed as are methods for recovering ethane
from m~xtures with crude oil after pipel~ne transportatlon.
U.S. Patent No. 3 596 437 d~scloses a method for reducing the viscosity
of crude oil in a p~peline by add~ng to the crude oil a fluid containing at
least 50% by volume of carbon d~ox~de and less than 10% by volume of
ethane. The viscosity of a crude oll at 70F for example can be reduced
by at least 30~ when the crude o~l ~s mixed w~th such a fluid. Carbon
dioxide for use in this method is generated in part from combustion of
natural gas.
U.S. Patent No. 3 618 624 discloses the use of methane ethane carbon
dioxide or mixtures thereof for introduct~on into crude oil for viscosity
reduction. Carbon dioxide when mixed with crude oil is claimed to promote
the format10n of two liquid phases between 40 and 200F and this two-phase
3S .
7 ~ ~
-- 4 --
condit~on is preferred for improved pipeline flow of the mixture. The
temperature and pressure conditions are controlled in the pipeline to
prevent the formation of a gaseous phase therein.
French Patent No. 80 07955 discloses the reduction of heavy oil
v~scoslty by d~ssolv~ng carbon d~ox~de ~n the o~l to y~eld a C~/o~l molar
rat~o of 0.2 to 0.7 and preferably 0.25 to 0.5. C~ is produced by
burning a portion of the crude o~l and recover~ng the C~ from the
result~ng combust~on gases. Related~French Patent No. 80 01687 discloses
additlonal data and points out that the best results ~n oil viscosity
.
;~ 10 reduction are obtained by dissolving 20 to 120 and preferably 25 to 80
cubic meters (S.T.P.) of C~ per cub~c meter of oil.
Japanese Patent No. J60069400 discloses the reduction of heavy hydro-
carbon viscositles by dissolution of C~ therein. The viscosity of a heavy
oil of 15.1 API was reduced to approximately 100 cP at ambient temperature
by disso1ution of C~ at about 80 psig.
SUMMARY OF THE INVENTION
The present inventlon is a process for reducing the viscosity of a
heavy hydrocarbon product wh~ch at amb~ent cond~t~ons exh~b~ts a fluid
viscosity sufficiently high to render p~peline transportation infeasible.
The process comprises m~xing the heavy hydrocarbon product at a super-
amb1ent temperature w1th an a~ount of hydrocarbon diluent separating water
and noncondens~ble gas from th~s first mixture cool~ng the mixture and
adding an additional amount of hydrocarbon diluent heating the resultlng
second mixture and contacting this second mixture with a miscible fluid
which dissolves ln the m~xture. The resulting reduced-viscosity mixture of
heavy hydrocarbon product hydrocarbon d~luent and m~scible fluid is
suitable for pipeline transportation.
I Heavy hydrocarbon products amenable to viscosity reduction by the
- 30 process of the present invent~on include heavy crude oil having a gravlty
between about 12 and 17API and bitumen having a gravity between about 8
and 12API.
In the process of the present invention the second heavy
hydrocarbon-diluent mixture is heated to about 80 to 217F preferably
y"
976~
-- 5 --
about 95 to 166F, by ~nd~rect heat exchange w~th the first heavy hydro-
carbon-d~luent mixture. The heated second mixture is then mixed with a
misc~ble fluid, preferably carbon diox~de, in a gas-l~quid contacting device
such as a bubble-cap tower, packed tower, or other suitable device at a
temperature of between about 80 and 217F, preferably about 95~ to 166F,
and at a pressure between about 400 and 1200 psig. The carbon dioxide
dlssolves 1n the heavy hydrocarbon-d~luent m1xture and thereby further
reduces the vlscosity. The final mixture of heavy hydrocarbon product,
diluent, and ~issolved carbon dioxide then flows dlrectly to a pipeline ln
whlch lt cools by convection or conductlon to normal pipeline temperature,
,
;~ ~ or opt~onally is cooled to p~pel~ne temperature before flowing to the
pipeiine. The exact temperature and other cond~tions in the gas-liquid
contacting device are carefully selected and controlled so that no carbon
dioxide can ex~st as a gas at any po~nt in the p~peline. In genera;l, this
; 15 selected contact temperature will be higher than the maximum temperature of
; hydrocarbons flowing in the pipellne.
In one embodiment of the present ~nvent~on, wherein heavy hydrocarbon
products are recovered from a hydrocarbon-bearing format1On by an in situ
combustion process, the carbon diox~de utll~zed for f~nal v~scosity
reductlon as descrlbed above is recovered from the carbon dioxide-contaln1ng
combustion gas from the ~n s~tu combust~on process. In an alternate
~- embodiment of the present ~nvent~on, wherein heavy hydrocarbon products are
recovered from a hydrocarbon-bear~ng format~on by the injection of steam
into the format~on, the carbon diox~de utilized for final viscosity
reduction as descr~bed above is recovered from the carbon dioxide-containing
; flue gas of the steam generator wh~ch produces the steam for in~ection.
It is an ob~ect of the present ~nvent~on to enable the improved ,,
pipeline transportat~on of heavy hydrocarbon products wh~ch at ambient
temperatures possess fluid viscosities too high for pipel~ne transportation.
A feature of the ~nvention is the use of a miscible fluid such as
carbon dioxide which is mixed with the heavy hydrocarbon product along with
a hydrocarbon d~luent, so that the resulting reduced-viscosity mixture can -
be pumped through a pipeline. ,
2~ ~760
- .., .; .
-- 6 --
An advantage of the invent~on is that the use of a miscible fluid such
as carbon dioxide reduces the amount of diluent needed per unit votume of
heavy hydrocarbon product to yield a required mixture viscosity for p1peline
transportatlon. Another advantage is that the use of a miscible fluid
reduces the total volume of m~xture per un~t volume of heavy hydrocarbon
compared w~th a mixture of heavy hydrocarbon product and hydrocarbon diluent
alone having the same flu~d viscosity. Th~s results in reduced pipeline
pumping requirements and operat~ng costs for an existtng pipeline; alter-
nately a greater volume of heavy hydrocarbon product can be moved in the
pipel~ne for a given operat1ng cost. If a new pipeline is constructed
capital costs can be reduced by the process of the present invention for a
given flow of heavy hydrocarbon produtt transported.
BRIEF DESCRIPTION OF THE DRAWINGS
15 Fig. 1 is a schematic flow diagram illustrat1ng an existlng process for
the preparat~on of heavy hydrocarbon products for pipeline
transportatton.
Fig. 2 is a schematic flow d~agram ~llustrat~ng the alternatlve process
of the present invent~on for prepar~ng heavy hydrocarbon products
for pipeline transportation.
DETAILED DESCRIPTION OF THE INVENTION
The preparation of heavy hydrocarbon products such as heavy oil or
bitumen for p~peline transportat~on by m~x~ng the hydrocarbons with a
diluent which ~s common pract~ce ~n the heavy o~l product~on industry is
~11ustrated in Fig. 1. Heavy hydrocarbon products (here~nafter also called
heavy hydrocarbons) are typically produced from o~l-bearing formations by
thermal methods such as in s~tu combust~on or steam ~nject~on. Other
production methods also~are used although less frequently than these two
methods. Heavy hydrocarbons from a production well field flow through
line 1 are heated ~n heater 101 and flow through line 2 ~nto separator 102
from which gas is removed ~n l~ne 3 and some of the water produced with the
heavy hydrocarbons is removed ~n l~ne 4. Hydrocarbon diluent 6 is stored in
tank 103 and pumped by pump 104 through lines 7 and 8 and is mixed with the
~;," . ;,~ } ~ ,
2~76~
-- 7 --
heavy hydrocarbons ~n l~ne 5. The mixture flows through line 9 into
electrostatic dehydrator 105 where add~t~onal water is coalesced and removed
in line 10 and any diluent vapor formed flows through line 11. The
dehydrated and gas-free mixture of heavy hydrocarbons and diluent flows
through line 12 and into separator 106 where additional d~luent vap~r is
removed in line 13 ~s comb~ned w~th vapor ~n line 11 and combined stream
14 is condensed ln diluent condenser 107. Condensed diluent 15 flows into
separator 108 where noncondensible gases are removed ~n l~ne 16. Heavy
hydrocarbon-diluent mixture flows from separator 106 through line 17 and is
pumped via pump 109 through cooler 110 and l~ne 18. Condensed diluent from
separator 108 passes through line 19 is pumped through pump 111 and
line 20 is combined w~th add~t~onal diluent from line 21 and the combined
diluent stream 22 ~s comb~ned w~th heavy hydrocarbon-d~luent stream 18. The
resulting heavy hydrocarbon-diluent stream 23 flows into storage tank 112
from where it flows through l~ne 24 to pump 113 wh~ch pumps the final heavy
hydrocarbon-diluent stream through l~ne 25 ~nto the p~pel1ne for
transportation to a ref~nery. Th~s stream ~s typ~cally at about ambient
temperature and at a pipel~ne pressure between about 400 and 1200 psig. The
stream typically contains about 0.4 to 0.7 volumes of diluent per unit
2~ volume of the orig~nal heavy hydrocarbons to give a k~nematic viscosity ofless than 250 cent~stokes. Other d~luent/heavy hydrocarbon rat~os can be
used depending on the type of heavy hydrocarbons season of the year and
other factors.
In the present ~nvent~on the amount of diluent required to reduce the
vlscos~ty of heavy hydrocarbons to a certa~n level is decreased by the
addition of a miscible flu~d preferably carbon d~ox~de to the heavy
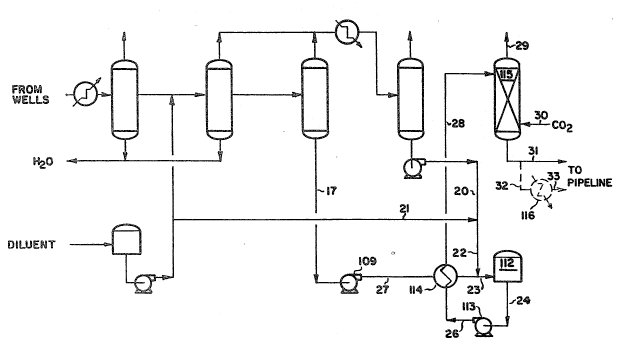
hydrocarbon-d~luent mixture. Referr~ng now to F~g. 2 heavy hydrocarbons
and diluent are processed in essentially the same manner as described above
with reference to F~g. 1 to provide diluent stream 22 and d~luent-heavy
30 hydrocarbon mixed stream 17. The total amount of diluent in streams 22 and -17 however is less for the present invent~on than for the process
described above for F~g. 1 as will be clear from the following discussion.
D~luent-heavy hydrocarbon stream 17 at a temperature of about 210 to 215F
is pumped by pump 109 through l~ne 27 is cooled in heat exchanger 114 is
.,.i:: . , . . . : .
~ 2~97~
,
mixed with dlluent stream 22 and the comblned stream 23 flows into storage
tank 112 at a temperature of about 90 to 110F. Mlxed stream 24 ls pumped
by pump 113 through llne 26 and heat exchanger 114 where lt ls heated by
lndlrect heat exchange w~th hot dlluent-heavy hydrocarbon stream 27 whlch ls
s inltlally at a temperature of about 210 to 215F. The now-heated stream 28is contacted wlth carbon dloxide stream 30 between about 80 to 217F
preferably 95 to 166F and between 400 and 1200 pslg ln fluld-fluld
contactor 115 where C~ dlssolves ~n the d~luent-heavy hydrocarbon mlxture
to yield a reduced-vlscosity stream 31 conta~nlng heavy hydrocarbons
dlluentj and C~ whlch flows to a plpeline for transportatlon to a
reflnery. Undlssolved gas flows from contactor 115 via 11ne 29.
Optlonally the heavy hydrocarbon-dlluent-C~ mlxture is cooled to about
40 to 100F by flowing through line 32 cooler 116 and llne 33 to the
plpeline for transportatlon to a reflnery. The stream typlcally contalns
0-1 to 0.3 volumes of dlluent and 5 to 8 lbs. of C~ per unlt vclume of the
origlnal heavy hydrocarbonsj to g~ve a typ~cal k~nematlc viscoslty of less
than 250 centlstokes. Other diluent-C~-heavy hydrocarbon ratlos can be
used depending on the type of heavy hydrocarbons season of the year and
other factors.
EXAMPLE
The process performance of the present ~nvent~on ls compared with
existlng process technology ~n the followlng example.
Uslng exlstlng technology sùch as that d~sclosed ln prevlously-clted
art'cle by R. R. Capellng bltumen at 7 000 bb1/day ls mlxed with
3 850 bbl/day of condensate diluent to y~eld a b~tumen-dlluent mlxture of
10 850 bbl/day having a k~nematic v~scos~ty of about 175 centlstokes at
55F. Thls bltumen-dlluent mixture ls prepared and sent to a plpellne as
; lllustrated ln Fl;g. 1. For th~s example an 8-lnch pipeline having a length
of 100 miles ls assumed~for transport~ng the b~tumen-d~luent mlxture and a
4-lnch plpellne having the same length ls assumed ~or transporting the
dlluent from lts source to the blending process site. The bitumen-dlluent
mlxture ls pumped to the pipeline at 870 ps~g by transfer pump 113 of
Flg. 1 and pumped through the 100-mile p~pel~ne by a number of pumps spaced
,~
g
along the length of the p~peline. D~luent ~s pumped from its source to the
blending process site by a series of pumps spaced along the 100-mile pipe-
line. The net pumping horsepower requlred for the transfer pump product
pipeline pumps and d~luent p~pel~ne pumps for th~s example ~s summar1zed ~n
Table 1.
In the appl~cation of the present invention as shown ~n Fig. 2
7 000 bbl/day of bltumen ls mlxed wlth 1 540 bbl/day of condensate dlluent
and the m~xture 26 ls heated to 95F by ~nd~rect heat exchange with hot
bltumen-diluent stream 27 ~n heat exchanger 114. The heated mlxture 28 ls
contacted with C~ at about 870 pslg ~n flu~d-flu~d contactor 115 to
d~ssolve about 1 335 000 SCFD of C~ in the m~xture. The bitumen-diluent-
C~ mixture at about 870 ps~g and 95F flows through l~ne 31 at a rate of
9 240 bbl/day to the pipellne in which the mixture cools to pipel~ne
temperature during transportation to a refinery. Optionally the mixture
passes through line 32 to cooler 116 where ~t ~s cooled to about 40 to
55F pr1Or to flowing to the p~pel~ne through line 33. The net pumping
horsepower for the present invent~on is summarized and compared with the
ex1st~ng process ln Table 1.
- :'-,
~
. .: .
: : .
~-- 2~7~
.
- 10 -
TABLE 1
Comparison of Existing Process
With Present Invention
Existing Present
Process Invention -
:(Fig. 1) ~F~g. 2)
Bltumen Flow, bbl/day ~7,000 7,000
Diluent Flow, bbl/day 3,850 1,540
02 required, MSCFD -- 1,335
Total flow to pipeline, bbllday 10,850 9,240
V~scoslty at 55F, centistokes 175 175
Pump1ng requlrements, BHP
Transfer pump 486 414
P~peline product pumps 1,120 816
Plpellne diluent pumps 360 36
Total : 1,966 1,266
~;: 20
By utlllzlng the process of the present ~nvention, as ~llustrated by
this example, an equ~valent amount of b~tumen can be transported at
equ~val~nt plpel~ne cond1t~ons with a d~luent requ~rement 60% lower than
~25 that of the exlst~ng process. Th~s advantage of reduced d~luent requ~rement
:wlll become more ~mportant as d~luent avallab~ y decreases and dlluent
cost increases ~n future years. The example also illustrates that by
utllizing the present ~nvent~on, the total volume of flu~d which must be
pumped to transport a unlt volume of bitumen or other heavy hydrocarbons is
decreased, thus decreaslng the pumping costs for a given mixture viscosity,
p~pel~ne dlameter, and p~pel~ne length. In the example, the total mixture
volume requ~red in the present invent~on ~s 15% less than that of the
existing process, which reduces the total pump horsepower by 36%. The
overall economic advantage of the present invention wlll depend on a number
- 2~ ~71~
- 11 -
of factors including bitumen or heavy hydrocarbon properties diluent cost
C~ cost power cost and the s~ze and length of product and dlluent
pipelines.
Bitumen and heavy oil are often produced from hydrocarbon-bearing
formations or reservo~rs by thermal recovery methods includ~ng in situ
combust~on talso known as f~reflood~ng) and steam ~njection ~also known as
steam flooding). In both cases the heat from in situ combustion or steam
injection reduces the v~scos~ty of the heavy hydrocarbons in the reservoir
thereby caus~ng flow toward producer wells from which the heavy hydrocarbons
are produced.
In the in s~tu combust~on process the heavy hydrocarbons are ignited
at the bottom of an ~njection well and an oxygen-contain~ng gas is ~njected
to sustain combust~on. The heat and product gases from the combustion cause
the heavy hydrocarbons to flow to adjacent product~on wells from which
heavy hydrocarbons and combustion gases are produced. The heavy hydro-
carbons are then treated and m~xed w~th d~luent as earl~er described for
transportation by p~pel~ne. The combust~on gases from this process conta~n
light hydrocarbons steam carbon d~ox~de and other components. Carbon
d~oxide ~s recovered from th~s combustlon gas for ~ntroduction into the
heavy hydrocarbon-diluent mixture for further viscosity reduction as
d~sclosed in the present ~nvent~on. There are a number of known methods for
recover~ng C~ from such a combust~on gas and these include for example
chem~cal react~on w~th amine-based solvent solut~ons physical d~ssolution
under pressure by methanol or ether-based solvents or phys~cal adsorption
processes. For cases in which the 'n s~tu recovery process uses pure oxygen
for combust~on the produeed combustlon gas may be su~f~ciently h~gh ~n C~
content to be used directly in contactor 115 of Fig. 2.
In the steam inject~on method steam ~s produced ~n boilers by the
combustion of natural gas produced gas or other hydrocarbon fuels and is
in~ected into the reservoir which heats the heavy hydrocarbons such that
they flow to adjacent product~on wells from wh~ch heavy hydrocarbons and
product gases are recovered. In an alternate mode of operat~on steam is
injected into a format~on through an injector well for a specified perlod of
time. At the end of th~s period steam injection is discont~nued and heavy
2Q~ 9~
- 12 -
hydrocarbons flow into this well and are recovered. In the present ~nven-
tion, carbon dioxide is recovered from the combustlon flue gases from the
steam boilers for introduction into the heavy hydrocarbon-dlluent mixture
for further viscosity reducti:on. C~ can be recovered from the steam
boller combustion flue gases by means of the known processes described
above.
Thus the process of the present invent~on can be integrated with eithsr
of these two thermal methods of produc~ng heavy hydrocarbons by recoverlng
the required carbon diox~de from the byproduct combustlon gases produced ~n
these thermal production methods. As discussed earlier, th~s recovered C~
is ~ntroduced ~nto the heavy hydrocarbon-d~luent m~xtures under controlled
condlt~ons such that no C~ is present as a gas while the heavy
hydrocarbon-d~luent-C~ m~xture flows through the p1peline.
Whlle carbon dioxide is preferred for use in the present invent~on,
other components wh~ch are gaseous at amb~ent pressure and temperature can
be used. These ~nclude methane, ethane, propane, butane, and other low-
boillng components wh~ch are present for example in the gases produced w~th
the heavy hydrocarbon product ~n the two recovery methods d~scussed above,
or are present ~n natural gas. M~xtures of carbon diox~de w~th these other
components can also be used as the misc~ble flu~d of the present ~nventlon.