Note: Descriptions are shown in the official language in which they were submitted.
- 1 2~2~
TIll,E:
C~YOGENIC AIR SEPARATION PROCESS AND APPARATUS
'l~'C~lIG~ArJ P'II~L~) OF '.~IE INVEN'rlC)N
This invention celates to the field of air separation processes
and particularly to a process and apparatus for the production of
nitrogen, oxygen and/or argon from air wherein liquefied air is used as
the heat exchange medium for the high pressure column condenser to
provide an energy efficient process.
BACXGRO~ND ART
Standard cryogenic air separation processes involve filtering
of feed air to remove particulate matter followed by compression of the
air to supply energy for separation. Generally the feed air stream is
then cooled and passed through absorbents to remove contaminants such as
carbon dioxide and water vapor. The resulting stream is subjected to
cryogenic distillation.
Cryogenic distillation or air separation includes feeding the
high pressure air into one or more separation columns which are operated
at cryogenic temperatures whereby the air components including oxygen,
nitrogen, argon, and the rare gases can be separated by distillation.
Cryogenic separation processes involving vapor and liquid
contact depend on the differences in vapor pressure for the respective
components. The component having the higher vapor pressure, meaning
that it is more volatile or lower boiling, has a tendency to concentrate
in the vapor phase. The component having the lower vapor pressure
meaning that it is less volatile or higher boiling tends to concentrate
in the liquid phase.
The separation process in which there is heating of a liquid
mixture to concentrate the volatile components in the vapor phase and
the less volatile components in the liquid phase defines distillation.
Partial condensation is a separation process in which a vapor mixture is
cooled to concentrate the volatile component or components in the vapor
phas~ and at the same time concentrate the less volatile component or
components in the liquid phase.
2 2 ~
A process which combines successive partial vaporizations and
condensations involving countercurrent treatment of the vapor in liquid
phases i5 called rectification or sometimes called continuous
distillation. The countercurrent contacting of the vapor and liquid
phases is adiabatic and can include integral or differential contact
between the phases.
Apparatus used to achieve separation processes utilizing the
principles of rectification to separate mixtures are often called
rectification columns, distillation columns, or fractionation columns.
When used herein and in the claims, the term "column"
designates a distillation or fractionation column or zone. It can also
be described as a contacting column or zone wherein liquid or vapor
phases are countercurrently contacted for purposes of separating a fluid
mixture. By way of example this would include contacting of the vapor
and liquid phases on a series of vertically spaced trays or plates which
are often perforated and corrugated and which extend crosswise of the
column, perpendicular to the central axis. In place of the trays or
plates there can be used packing elements to fill the column.
"Double column" as used herein refers to a higher pressure
column having its upper end in heat exchange relation with the lower end
of a lower pressure column.
The term "a standard air separation process or apparatus" as
used herein is meant to describe that process and apparatus as above
described as well as other air separation processes well known to those
skilled in the art.
As used herein and in the appended claims, the term "indirect
heat exchan~e" means the bringing of two fluid streams into heat
exchange relation without any physical contact or intermixing of the
fluids with each other.
Historically, nitrogen, oxygen and/or argon have been produced
by one of two ~asic process schemes including the single column process
and the double column process.
With respect to nitrogen, the single column process produces
good quality gaseous and liquid nitrogen at pressures of approximately
6-10 bar. The recovery of nitrogen is limited by the equilibrium at the
,
bottom of the column. Typically, the process can produce nitrogen at a
rate of approximately 50~60~ of the nitrogen in the initial air feed.
With the double column process, nitrogen is produced at
pressures of about 1-4 bar. It is more efficient than the single column
process, and approximately 90~ or more of nitrogen can be recovered from
the nitrogen present in the initial air feed. Typically the columns are
stacked with a condenser-reboiler separating the two columns. Since the
process produces nitrogen at relatively low pressures, further
compression of nitrogen is frequently needed adding to the cost of
production and use.
In the prior art double column process, air is separated by
cryogenic distillation or rectification to produce a nitrogen-rich
stream or fraction at the top of the high pressure column and
oxygen-rich stream or fraction at the bottom. The nitrogen-rich stream
is sent to the top of the low pressure column to provide the reflux for
this column. The bottom oxygen-rich stream is fed to the low pressure
column for further separation.
In the low pressure column the feed stream is further separated
by cryogenic distillation into an oxygen-rich stream or fraction at the
bottom and a nitrogen-rich stream or fraction at the top. The top
stream can then be recovered as nitrogen product. In the double column
arrangement, the high pressure column and the low pressure column are
thermally linked through the condenser-reboiler arrangement. Thus, in
the prior art double column process the nitrogen-rich fraction of the
high pressure column is condensed against the vaporizing o~ygen-rich
fraction of the low pressure column.
For a given pressure in the low pressure column, the pressure
of the air feed to the high pressure column is dictated by the
composition of the vaporizing oxygen-enriched stream, the temperature
difference o~ the high pressure column condenser and the low pressure
column reboiler, and to some extent the composition of the condensing
nitrogen-enriched stream which is relatively pure in nitrogen.
Other prior art process schemes are variations of the above
described single or double column process with additional features such
as an additional overhead condenser or bottom reboiler.
4 ~ 3
SU~A~Y OF THE INVENTION
The process of the invention can be utilized for the energy
efficient production of nitrogen, oxygen and argon.
Essentially, the invention lies in using vaporized and
liquefied air as the heating and cooling medium between the high
pressure and the low pressure columns. Formerly nitrogen has been used.
The invention will be explained in particular detail with
respect to nitrogen but it should be understood that the invention is
equally applicable to the production of o~ygen and argon. It will be
obvious to those skilled in the art ho~ to optimize temperature,
pressure and other operating conditions to optimize output of oxygen
and/or argon as primary product.
The particular advantage in the use of air for the heating and
cooling medium is that less energy is required to condense the air than
to condense a nitrogen rich stream. Since the main energy cost involves
compression of the gases, the lower pressure which is required to
condense air at a given temperature is less costly than to condense
nitrogen.
For example, nitrogen condenses at 7 bar pressure at -180C.
By contrast, only 6 bar pressure at -178C is required to condense air.
Thus the 2~ difference in temperature and the 1 bar pressure provides
the reduced energy expenditure in the invention process.
In prior art processes wherein nitrogen is used ~or the heating
and cooling medium ~etween the high pressure and low pressure columns,
it is necessary to compress the feed air to a higher feed air pressure
as required by the nitrogen. Thus, the primary energy savings come from
the reduced requirement for compression of the feed air.
The process of the invention makes possible the production of
high purity nitrogen to the extent of more than 90% of the nitrogen
contained in the initial feed air. It can be produced at a pressure
range within a~out 3 bar to about 15 bar. Both high pressure and low
pressure nitrogen can be produced. This can be done separately or
together. Moreover, the process i5 energy efficient compared with prior
ar~ processes.
2~2~
According to the invention process, feed air, which has been
treated to remove moisture and impurities such as CO2 and methane by
passage through ~olecular sieves, alumina, silica gel and the like is
compressed and fed to a heat exchanger to exchange heat with outgoing
products.
According to one embodiment, the feed air is split into two
fractions, one fraction being fed to the bottom of a high pressure
column and the other fraction being fed to a condenser/reboiler located
in the base of a low pressure column. Good re~sults have been obtained
by using equal fractions of feed air although other ratios can be used.
According to another embodiment, the feed air is split into
three fractions. Two of the feed air fractions are fed to the high
pressure column and the condenser/reboiler at the base of the low
pressure column as above described. The third air fraction is expanded
to provide plant cooling and then introduced into the low pressure
column for cryogenic separation.
The first feed air fraction is separated by cryogenic
distillation within the high pressure column into a first nitrogen~rich
vapor fraction and a first oxygen-rich liquid fraction. The oxygen
enriched liquid fraction is withdrawn from the base of the high pressure
column and sent to the low pressure column. The second feed air
fraction which is sent to the condenser/reboiler in the base of the low
pressure column is condensed by heat exchange with the oxygen-rich
liquid at the bottom of the low pressure column which is thereby
vaporized. The condensed liquefied air thus produced in the
condenser/reboiler is then fed to the top condenser of the high pressure
column where it is vaporized by indirect heat exchange with the first
nitrogen-rich vapor fraction produced in the high pressure column. This
causes the nitrogen to condense.
According to one embodiment, part of the condensed
nitrogen-rich fraction in the high pressure column is separated and fed
to the low pressure column to provide extra reflux. At the same time
the second feed air fraction which has been vaporized by indirect heat
exchange contact with nitrogen in the top condenser of the high pressure
column is then introduced into the low pressure column for cryogenic
separation.
Within the low pressure column, the second feed air fraction
6 ~ 3 ~ ~
along with a portion of the first oxygen-rich fraction from the high
pressure column are then separated into a second nitrogen-rich stream
and a second oxygen-rich stream.
According to another embodiment, a portion of the second
nitrogen-rich stream can be removed as high pressure nitrogen product
while the remaining portion is used to provide reflux for the low
pressure column.
According to another embodiment, a portion of the high pressure
nitrogen product can be expanded to provide plant cooling and added to
the low pressure nitrogen product stream.
The second oxygen-rich stream which falls to the bottom of the
low pressure column is vaporized by indirect heat exchange contact with
the incoming second feed air fraction which is thereby condensed. By
another embodiment, the second oxygen-rich fraction can also include a
third feed air fraction which has been expanded prior to being
introduced into the low pressure column.
A portion of the second oxygen-rich stream is fed to the
overhead condenser of the low pressure column where it is vaporized by
heat exchange contact with rising nitrogen which is thereby condensed.
The thus vaporized second oxygen-rich stream can be removed from the
overhead condenser as waste and warmed in subcoolers and in the heat
exchanger by indirect heat exchange with process streams and feed air.
If desired the waste oxygen can be expanded to provide plant
cooling. Alternately, the waste oxygen which has about 70% purity can
be utilized as product in applications where high purity oxygen is not
required.
Apparatus for the above described process are alsa provided.
The apparatus include, in combination, air compression means for
compressing air from an outside source, purification means for removing
carbon dioxide and water vapor from the air compressed by the air
compression means, and heat exchange means for cooling the compressed
air from the purification means to a cryogenic temperature. ~ first
distillation colu~n equipped with a top column or overhead
evaporator/condenser is included for cryogenic separation of a portion
of the feed air from the heat exchanger.
A second distillation column equipped with a top column
condenser and a bottom column reboiler is provided for separation by
fractionation of at least a portion of the cooled compressed feed air
after circulation through the bottom column reboiler of the second
distillation column and the top column condenser of the first
distillation column together with at least a portion of the oxygen-rich
liquid obtained from the first distillation column into a second
oxygen-rich fraction and a second nitrogen-rich fraction.
Means are provided for withdrawal of oxygen liquid at the base
of the second distillation column for introduction into the overhead
condenser of the second distillation column to provide indirect heat
exchange with vapors rising within the second distillation column.
Expansion means are provided for expansion of compressed air
prior to introduction in the second distillation column, of oxygen
withdrawn from the overhead condenser of the second distillation column,
and/or for expansion of nitrogen product to provide cooling.
BRIEF DESCRIPTION OF THE DRAWING
Figure 1 shows a schematic flow diagram of the process and
apparatus of the invention in which low pressure nitrogen is produced;
Figure 2 shows a schematic flow diagram of the process and
apparatus of the invention similar to Figure 1 except that air expansion
is provided in place of waste expansion;
Figure 3 shows a schematic flow diagram of the process and
apparatus of ~ho invention wherein high pressure and low pressure
nitrogen are produced; and,
Figure 4 shows a schematic flow diagram of the process and
apparatus of the invention similar to Figure 3 wherein pact of the high
pressure nitrogen is expanded to low pressure nitrogen.
DETAILED DESCRIPTION OF T~IE INVENTION
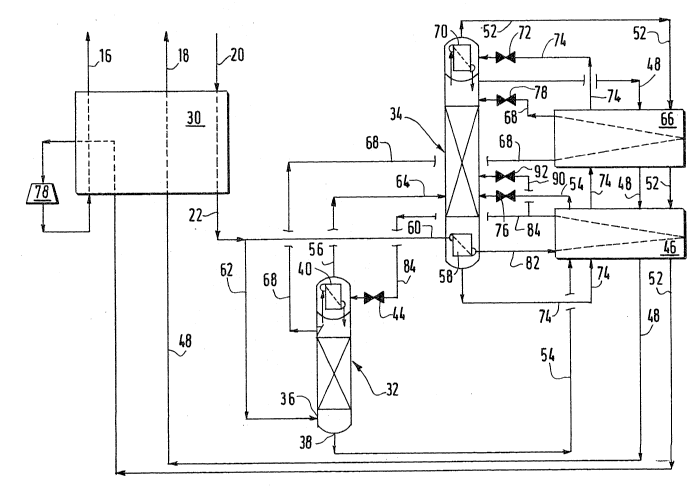
Referring now to the flow diagram of Figure 1, compressed feed
air free of impurities is introduced by means of conduit ~0 into a heat
8 ~ 3
exchanger 30. The air is preferably introduced into the heat exchanger
30 at a pressure in the range of about 5 bar to about 20 bar where the
temperature of the air is cooled to cryogenic temperature by indirect
heat exchange with outgoing waste and product streams.
Next the feed air is split into two fractions. Good results
have been obtained with equal fractions or streams of feed air but other
ratios can be used. The first fraction of the feed air is sent to the
high pressure column 32 through lines 22 and 62 and the remaining second
fcaction of feed air is sent to the reboiler 58 of the low pressure
column 34 through lines 22 and 60.
At the high pressure column 32 the pressure is preferably in
the range of about 5 bar to 20 bar.
The first feed air fraction is introduced into the lower part
of column 32 below the bottom distillation tray as indicated at 36.
Here, the first feed air fraction is separated into a first
nitrogen-rich vapor fraction which rises to the top of the column 32 and
a first oxygen-rich liquid fraction which falls to the bottom of the
column 32.
At least a portion of the first oxygen-rich liquid is
withdrawn from the bottom of the high pressure column at 38. It is
comprised of about 35% to about 40~ oxygen which is a'oout the same
proportion as for the prior art processes.
The first oxygen-rich liquid which is removed from the bottom
of the high pressure column 32 through line 54 is passed through
subcooler 46 where the temperature is further reduced by indirect heat
exchange with product nitrogen which exits from the upper part of the
low pressure column 34 through line 48 and with waste which exits
through line 52 from the overhead condenser/evaporator 70 of the low
pressure column 34.
The cooled first oxygen rich liquid from the subcooler q6
is then introduced into the low pressure column 34 above the bottom tray
after expansion through valve 76.
The second feed air fraction which enters the
condenser/reboiler 58 in the base of the low pressure column 34 is
condensed by indirect heat exchange with oxygen-rich liquid at the
r~ ~ ~
g
bottom of the low pressure column 34. This causes the second feed air
fraction to be condensed and the oxygen-rich liquid to be vaporized.
The condensed second feed air fraction leaves the
condenser/reboiler 58 of the low pressure column 34 via line 82 where it
enters subcooler 46. The liquefied air exits subcooler 46 via line 84
and expands through valve 44 into the condenser/reboiler 40 of the high
pressure column 32. If needed, a portion of the condensed second feed
air fraction can be introduced into the low pressure column 34 via line
90 after expansion through valve 92 to control the balance of air
between the high pressure and low pressure columns.
The first nitrogen-rich vapor fraction rises to the top of the
high pressure column 32 where it enters the condenser/reboiler 40. Here
the nitrogen vapor is brought into indirect heat exchange contact with
the condensed second feed air fraction which enters through valve 44
from the condenser/reboiler 5a of the low pressure column 34. This
causes the liquefied air to vaporize and the nitrogen vapor to be
condensed. As shown in Figures 3 and 4, part or all of the condensed
nitrogen portion is returned to the high pressure column 32 to provide
reflux as required.
Any nitrogen vapor which is not condensed by indirect heat
exchange with the condensed second feed air fraction can be recovered as
high pressure nitrogen by removal from the upper part of the high
pressure column 32 for example, through line 67 as shown in Figure 3.
Part of the condensed nitrogen can be sent to the low pressure
column 34 for extra reflux if the high pressure nitrogen flow is small
or not needed. This part of the condensed nitrogen is removed from the
upper part of the high pressure column 32 through line 68 as shown in
Fiqures 1 and 3. The condensed nitrogen is then passed through
subcooler 66 where it is brought into indirect heat exchange contact
with outgoing nitrogen product and waste. From the subcooler 66, the
condensed nitrogen passes through a continuation of line 68 and is
introduced into the low pressure column 34 after expansion through valve
78.
At the same time, the vaporized air exiting via line 56 from
the condenser/reboiler 40 at the top of the high pressure column 32 is
separated by introduction into the low pressure column 34 through line
64 at about the same level as for the introduction of the first
J~ ~
oxygen-rich liquid which enters through line 54.
The first oxygen-rich liquid withdrawn from the base of column
32 and the vaporized air withdrawn from the condenser/reboiler 40 at the
top of the high pressure column 32 through line 56 are further separated
within column 34 into a second nitrogen-rich vapor fraction and a second
oxygen-rich fraction.
The second nitrogen-rich vapor fraction rises to the top of the
low pressure column 34 while the second oxygen-rich fraction falls to
the bottom of the low pressure column 34.
A portion of the second oxygen-enriched liquid fraction at the
bottom of the low pressure column 34 is withdrawn through line 74 and
passed through a first subcooler 46. Here the second oxygen-enriched
liquid is further cooled by indirect heat exchange with nitrogen gas
removed from the upper part of the low pressure column 34 through line
48 and with the waste stream exiting through line 52 from the overhead
condenser 70 of the low pressure column 34.
The second oxygen-enriched liquid is passed by means of a
continuation of line 74 to a second subcooler 66 for further cooling
by indirect heat exchange with nitrogen gas removed from the top of the
high pressure column 32 through line 68 and with the waste oxygen stream
which exits from the overhead condenser 70 through line 52.
The resulting cooled second oxygen-rich liquid is passed
through an extension of line 74 where the liquid is introduced into the
overhead condenser 70 in the top of the low pressure column 34 after
expansion through a valve 72 to further cool the second oxygen enriched
stream.
A major part of the second nitrogen-rich stream is recovered as
nitrogen product from the upper part of the low pressure column 34
through line 48. The gaseous nitrogen stream is warmed by passage
through subcoolers 66 and 46 and heat exchanger 30 before exiting the
system.
The re~aining portion of the second nitrogen-rich stream within
the low pressure column 34 is condensed by heat exchange with the second
oxyqen-enrichèd liquid in the overhead evaporator/condenser 70 of the
low pressure column 34 which causes the second oxygen-enriched liquid to
11 2~ 3~3
be vaporized. ~he condensation of the nitrogen provides reflux for the
low pressure column 34. The vaporizing oxygen-enriched liquid exits
overhead evaporator/condenser 70 via line 52 and is subsequently warmed
by passage through subcoolers 66 and 46 and heat exchanger 30.
After warming in the heat exchanger 30, the waste oxygen stream
is passed through a turbo expander 78 where the stream can be expanded
to provide plant cooling.
It can seen that the above described process utilizes air as a
heating and cooling medium between the high pressure and low pressure
columns. Conventionally in prior art processes, the nitrogen-rich
stream has been used to transfer heat to the bottom of the low pressure
column. Keeping in mind that for a given nitrogen recovery, that is,
having the same composition of oxygen-rich stream, more energy is
required to condense the nitrogen-rich stream than to condense air.
What this means is that for a given nitrogen recovery, using air as the
heat transfer medium, the high pressure column can function at a lower
pressure than for conventional prior art processes. Also, for the same
pressure in the high pressure column, according to the invention
process, the low pressure column can function at a higher pressure.
Table 1 below shows the expected performance of the invention
process shown in Figure 1 and above described for the products of
nitrogen as product.
Table 1
Total Feed Air Flow Line 20 15462 Nm /h
Feed Air Pressure Line 20 10.2 bar abs.
Nitrogen Product Flow Line 48 10514 Nm /h
Nitrogen Pressure Line 18 5.5 bar abs.
Nitrogen Purity 18 vpm 02
Waste (Oxygen-Rich) Flow Line 52 4948 Nm3/h
Waste Pressure Line 16 1.3 bar abs.
Compressed Air Line 22 -160c
Column 32 10.2 bar abs.
Column 32 Top -170C
Column 32 Bottom -160C
Oxygen-Rich Liquid Line 38 -165.6C
Condensed Second Feed Air
Fraction Line 82 -167.5C
2 ~
12
Table I continued
Condensed Second Feed Air
Fraction Line 82 -167.5C
Condensed Second Feed Air
Fraction Line 84 -171C
Vaporized Second Feed Air
Fraction from Condenser/
Reboiler 40 Line 56 -172.6C
Nitrogen Exiting Column 32 Line 68 -170.6 C
Condensed Nitrogen Exiting
Subcooler 66 Line 6a -174.4C
Column 34 5.5. bar abs.
Oxygen-Rich Liquid from Column 34 Line 74 -168.8C
Oxygen-Rich Liquid Exiting from
Cooler 66 Line 74 -174.4C
Oxygen-Rich Liquid after
Expansion Valve 72 -179C
Nitrogen Product Exiting Column
34 Line 48 -177.6C
Nitrogen Product Exiting Column
34 Line 48 5 bar abs.
Oxygen Waste Stream from
Condenser 70 Line 52 -178.9C
When the embodiment shown in Figure 3 or Figure 4 is followed,
a feed air pressure of 21 bar abs. would produce a pressure of about 20
bar abs. within the high pressure column 32 and a pressure of about 14
bar abs. within the low pressure column 34.
Various modifications of the invention process and apparatus as
above described will be apparent to those skilled in the art and can be
resorted to without departing from the spirit and scope of the invention
as defined by the following appended claims.