Note: Descriptions are shown in the official language in which they were submitted.
` 202830~
MEMBRANE PROCESS FOR ACID RECOVERY
This invention relates to the recovery of acid from
solutions by a membrane process, and, more particularly, to
a process for the recovery and concentration of acids in
5aqueous solutions by dialysis and electrodialysis.
BACKGROUND OF THE INVENTION
The recovery of acids from aqueous solutions by means of
membrane processes is well known. The many membrane
processes include dialysis and electrodialysis.
Dialysis is generally carried out in a multi-compartment
unit wherein dialyzate compartments are alternating with
diffusate compartments separated from each other by
suitable membranes. When acidic solutions are treated for
the recovery of a diffusate stream containing a major
portion of the acid, anionic membranes are used that
selectively allow the acid anion as well as protons to pass
from the dialyzate side to the diffusate side of the
membranes. The dialysis method for treating acidic
solutions has the main disadvantage of yielding an acid
product of a relatively low concentration because the
driving force is mainly determined by the acid
concentration gradient across the membranes. To obtain an
efficient production rate and a high acid concentration in
the product stream, large equipment with a high membrane
area is required.
` 202830~
_ 2
Electrodialysis is generally carried out in a unit that
comprises a multiplicity of alternating anionic and
cationic permselective membranes defining alternating
diluate and concentrate compartments between an anode
compartment containing an anode and a cathode compartment
containing a cathode. The electrodes are connected to a
source of direct electrical current. Electrodialysis is
more effective than dialysis for concentrating dilute acid
solutions, but has the disadvantage that, when the acid
solutions contain cations, the concentrated acid solution
often also contains undesirable concentrations of those
cations. Another disadvantage of electrodialysis is low
current efficiency due to proton leakage through the anion
exchange membranes. In order to improve the efficiency of
acid transfer, it is necessary to select membranes
carefully, as well as to operate at moderately high current
densities. Such high current densities, however, result in
a decreased selectivity of the acid removal.
The prior art contains many references on the use of either
dialysis or electrodialysis for the separation or
concentration of acids in solutions. In Jap. Kokai 53-
19171 (1978) there is described a method for separating and
recovering both acids and metals from acid and metal-
containing solutions. Solution is fed to a dialysis tank
for the removal of acid, and resulting solution is then
subjected to membrane electrolysis for the
electrodeposition of metals. The metal-reduced solution is
subjected to electrodialysis from which the concentrate,
i.e. solution concentrated in acids and metals, is fed to
2~2830~
_ 3
the membrane electrolysis tank and the dilute solution is
returned to the dialysis step. This appears to be one of
the few processes in which both dialysis and
electrodialysis are used.
SUMMARY OF THE I NVENT I ON
I have now found that the disadvantages of dialysis and
electrodialysis may be substantially obviated by using a
process for the recovery and concentration of acids from
aqueous acidic solutions which comprises dialysis
integrated with electrodialysis. The efficiency of the
process of the invention is much higher than obtained with
either dialysis or electrodialysis alone or when dialysis
and electrodialysis would be operated in series.
The process of the invention comprises feeding an aqueous
acid solution, that may contain at least one other
substance such as a metal ion in addition to hydrogen ions
(protons), to the dialyzate compartments of a dialysis
unit. The dialyzate, which substantially contains any
cations other than protons from the feed solution, is
removed from the process. The diffusate contains the major
portion of the acid from the feed solution and may contain
low concentrations of other substances. The diffusate is
fed into the diluate stream circulating through the diluate
compartments of an electrodialysis unit. A portion of the
circulating diluate is diverted to the diffusate
compartments of the dialysis unit. To increase the
concentration gradient of the acid anion across the
membranes in the dialysis unit, a quantity of an acid-
2û~3~Q
_~ 4
receiving solution may be added to the diluate portion that
is fed to the diffusate compartments.
A concentrate stream is circulated through the concentrate
compartments of the electrodialysis unit. By virtue of the
direct current applied between the electrodes, the
concentration of acid in the concentrate increases. A
portion of the circulating concentrate is withdrawn as the
concentrated acid product substantially free of any other
substance. To control the acid concentration of the
product at a desired level, a portion of the diffusate from
the dialysis may be fed into the circulating concentrate
stream, as necessary.
The process according to the present invention is
fundamentally different from that described in the Jap.
Kokai (supra). Not only does the present method not use
membrane electrolysis to deposit metals, but only the
diffusate, not both the dialyzate and the diffusate from
the dialysis, is further treated. Moreover, no means are
provided in the method described in the Jap. Kokai for
controlling the acid concentration at a desired level.
These and other differences will become apparent from the
detailed description of the invention.
Accordingly, there is provided a method for the recovery of
acids from aqueous acid solutions containing at least one
other dissolved substance comprising the steps of feeding
aqueous acid feed solution to a dialysis unit, said unit
comprising dialyzate compartments alternating with
` 2Q28300
diffusate compartments separated by acid anion permeable
membranes, said dissolved substance being a substance that
will not substantially transfer into the diffusate
compartments, and said feed solution being fed into said
dialyzate compartments forming a dialyzate; transferring
acid anions from said feed solution through said membranes
into said diffusate compartments to form a diffusate while
substantially preventing transfer of said dissolved
substance; withdrawing dialyzate reduced in acid anions and
substantially containing said dissolved substance from said
dialyzate compartments; withdrawing diffusate from said
diffusate compartments; passing withdrawn diffusate to an
electrodialysis unit comprising a multiplicity of
alternating anion and cation permselective membranes
defining alternating diluate compartments and concentrate
compartments arranged between an anode in an anode
compartment and a cathode in a cathode compartment, at
least a portion of said withdrawn diffusate being passed to
said diluate compartments; applying a direct electrical
current between said anode and said cathode; recirculating
a diluate stream through said diluate compartments; passing
a portion of said recirculating diluate stream to the
diffusate compartments of said dialysis unit; recirculating
a concentrate stream through said concentrate compartments,
said current causing said acid anions to pass from said
diluate stream into said concentrate stream; and
withdrawing a portion of said recirculating concentrate
stream as concentrated acid product.
` 2028300
It is an aspect of the present invention to provide an
efficient membrane process for recovering and concentrating
acid from aqueous acidic solutions containing at least one
other substance in addition to the acid.
It is another aspect to provide a process for the
concentration of acids by dialysis integrated with
electrodialysis to yield an acid product substantially free
from dissolved substances present in the feed.
BRIEF DESCRIPTION OF DRAWING
These and other aspects will become apparent from the
following detailed description with reference to the
accompanying drawing in which:
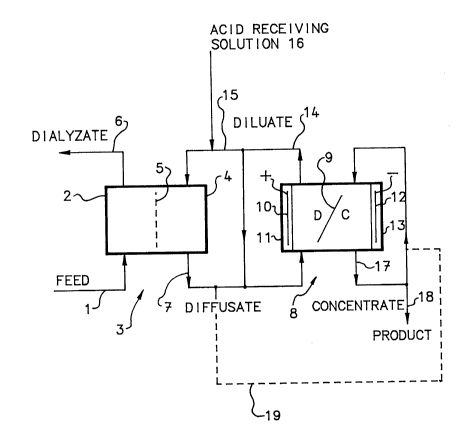
Figure 1 is a schematic flow diagram of the process
according to the invention.
DETAILED DESCRIPTION
The acid solutions that can be treated include aqueous
acidic solutions that comprise at least one acid chosen
from the group consisting of sulfuric acid, hydrochloric
acid and nitric acid, and at least one other dissolved
substance. The acid solutions may contain as little as
about 10 g/L acid but, practically, should contain at least
about 30 g/L acid. When nitric acid solutions are treated,
care must be taken that the acid concentrations does not
reach high values that may affect the membrane life through
oxidation. The at least one other dissolved substance
usually comprises cations in the acidic solution that are
associated with the acid anion, usually dependent on the
` 2~2~3~0
source of the acidic solution, but may also comprise others
present as non-anionic species such as, for example,
arsenical or antimonial species. The cations may include
cations of at least one metal chosen from the group
consisting of Zn, Cd, Ca, Mg, Na, K, Mn, Al, Fe, Cu, Ni and
Co. It is understood that other ions may be present.
Essentially the at least one other dissolved substance
includes those substances that will not substantially
transfer into the diffusate in the dialysis, as will be
described.
With reference now to Figure 1, aqueous acidic feed
solution 1 containing at least one acid of the above-
mentioned group, and which may contain amounts of cations
or other dissolved non-anionic substances, that will not
substantially transfer, is fed into the dialyzate
compartments 2 of a dialysis unit schematically and
generally indicated with 3. Dialysis unit 3 comprises
alternating dialyzate compartments 2 and diffusate
compartments 4 which are separated from each other by
suitable anionic membranes 5. For the sake of simplicity,
only one each of the dialyzate and diffusate compartments
and the membranes is shown. During dialysis, the acid will
be removed from the feed solution into a diffusate, while
the other substance(s) substantially remain in a dialyzate.
Suitable membranes are for example SelemionTM DMV and DSV
membranes, but other membranes giving similar results may
be used. Anionic membranes 5 are permeable for the acid
anions, which transfer through membranes 5 under the
driving force created by the concentration gradient of the
` ~28303
_ 8
anions across the membranes. Membranes 5 are substantially
impermeable t~ the at least one other substance. Protons,
however, easily transfer, due to their small size and high
mobility, thereby satisfying the requirement for electron
neutrality.
The at least one dissolved substance that might be present
in the diffusate is present in concentrations that will not
cause fouling of membranes.
Dialyzate 6 is removed from the dialyzate compartments 2 at
a rate substantially equal to the rate of addition of feed
1, which is in the range of about 1 to 5.0 L/h.m2. These
feed rates are about twice the commonly used rates for a
process using dialysis only. A solution, to be described,
is fed to the diffusate compartments 4. Diffusate 7 is
discharged from the diffusate compartments 4, and is passed
to the diluate compartments of an electrodialysis unit,
which is schematically indicated with 8. The dialysis is
carried out at ambient temperatures up to about 45C.
Electrodialysis unit 8 comprises a multiplicity of
alternating suitable anion and cation permselective
membranes, schematically indicated with 9. The alternating
membranes define alternating diluate and concentrate
compartments, schematically indicated with D and C,
respectively, arranged between an anode 10 in an anode
compartment 11 and a cathode 12 in a cathode compartment
13.
`` 21G283û0
g
Suitable cation permselective membranes are, for example,
strongly acidic membranes which have a membrane matrix of
a styrene di-vinyl benzene copolymer on a polyvinyl
chloride base and possess sulphonic acid radicals (R-SO3H)
as active groups. The active groups comprise 3-5
milli-equivalents per gram of dry resin. SelemionTM-type
membranes, such as SelemionTM CMR, CMV-A, CSV and CSR,
specially treated to be monovalent permselective, are
particularly suitable.
Suitable anion permselective membranes are, for example,
basic membranes with tertiary amine or quaternary ammonium
active groups, such as, for example, derived from
trimethylamine (for example, R-N(CH2)3.Cl), at 3-5
milli-equivalents per gram of dry resin, and having a
matrix of a styrene di-vinyl benzene copolymer on a
polyvinyl chloride base. SelemionTM ASV, and ASR membranes
and particularly SelemionTM AAV membranes are suitable. It
is understood that other membranes with similar properties
- are similarly suitable, and that the use of combinations of
other membranes may yield the desired results.
The anode 10 and the cathode 12 are made of suitable
materials, i.e., those that are compatible with the
generation of oxygen at the anode and the evolution of
hydrogen at the cathode. For example, anode 10 can be made
of platinum or platinum-coated titanium and cathode 12 of
platinum-coated titanium or stainless steel. A direct
electrical current is applied to the electrodes from a
source of electrical current (not shown).
2~283~
In the electrodialysis, the acid solution from the dialysis
step will be concentrated in a concentrate stream and the
diluate stream from the diluate compartments will
substantially contain the other substance(s) present in the
diffusate.
The diffusate 7 from diffusate compartments 4 is fed to the
diluate compartments D, and a diluate stream 14 is
withdrawn from the diluate compartments. Withdrawn diluate
14 is recirculated through the diluate compartments D, and
a portion 15 of the circulating diluate stream is passed to
the diffusate compartments 4 of dialysis unit 3. An
important feature of the present invention is that an
amount of acid-receiving solution 16, such as a dilute
acid, a salt solution, the salt being of the acid being
treated, or water may be added to the diluate portion 15
fed into diffusate compartments 4. Solution 16 is fed to
diffusate compartments 4 at a high rate in the range of
about 2 to 10 L/h.m2 of membrane surface area. The addition
of acid-receiving solution 16 decreases the anion (acid)
concentration in the diffusate compartments 4, and thereby
increases the concentration gradient across anionic
membranes 5 in the dialysis. The increased concentration
gradient enhances the efficiency of the dialysis and
thereby allows the use of a smaller dialysis unit. The
amount of acid-receiving solution added to the diluate
should be sufficient to increase the concentration gradient
in the dialysis.
3 0 ~
-- 11
A concentrate stream 17, i.e. a solution concentrated in
acid, is withdrawn from the concentrate compartments C,
preferably at a rate equal to the rate of the net water
transfer from the diluate to the concentrate during the
electrodialysis. Concentrate stream 17 is recirculated
through the concentrate compartments C, and a portion of
the circulating concentrate is withdrawn as product 18. If
desired, the acid concentration in the product 18 is
controlled, i.e. lowered, by feeding water (not shown) or
feeding a portion 19 of the diffusate 7 from dialysis 3
into the recirculating concentrate stream 17, as indicated
with the broken line. It is important to maintain
turbulent conditions in the concentrate and diluate
compartments. This can be done by passing solution through
the compartments at a sufficient rate, such as achieved by
the recycling of the diluate and the concentrate streams
to the respective compartments.
In the anode and cathode compartments 11 and 13, the
predominant reactions are oxygen and hydrogen evolution,
respectively. The anode and the cathode in their
respective compartments are rinsed with circulating rinse
solutions (not shown). If less control of conditions is
permissible, a common rinse solution may be circulated to
both the electrode compartments. The rinse solutions may
be chosen from water, dilute acid and acidified sodium
sulfate solution maintained at a pH in the range of about
0 to 4. Suitable acidic rinse solutions may contain sodium
sulfate in a concentration in the range of about 0.05 to
0.5 molar, or sulfuric acid in a concentration in the range
20283~
~~ 12
of from 0.01 to 1 molar. The rinse solutions are
circulated through the electrode compartments at rates
sufficient to give a differential pressure across the
membranes of less than about 150 kPa, preferably less than
about 50 kPa. A portion of the rinse solutions may be
removed from circulation and be replaced with a
`substantially equal portion of fresh solution. The flow
rates of the rinse solutions are generally in the range of
about 25 to 90 L/h.m2, and are preferably in the range of
about 40 to 80 L/h.mZ.
During electrodialysis, the acid anions (and protons) in
the feed to the diluate compartments D pass from the
diluate compartments D to the concentrate compartments C
through the anionic and cationic permselective membranes
respectively. The gases evolved at the electrodes are
carried from the cathode and anode compartments in the
rinse solutions.
The electrodialysis unit may be operated with solution
temperatures in the range of from just above the freezing
temperature of the solution to as high as about 60C, i.e.
from about 0C to 60C. At the higher temperatures, the
process is more efficient but the life of the membranes may
be reduced. The process is preferably operated with
solution temperatures in the range of from ambient
temperatures to about 50C.
Feed rates to the electrodialysis unit may be selected in
the range of about 2 to 40 L/h.m2 per membrane pair, the
`~ 13 ~2~3Q~
selected value being dependent on the concentration of
dissolved substance in the feed solution and the value of
the current density. The flow rate of solutions through
the concentrate and diluate compartments should provide
turbulent flow and the flows through the compartments
should be substantially balanced in order to maintain a
differential pressure across the membranes not exceeding
about 150 kPa, preferably less than 50 kPa.
The current applied to the electrodes is controlled to
maximize the current efficiency. The current is equivalent
to a current density (applied current per effective
membrane surface area) in the range of about 100 to 1000
A/m2, the particular value selected being chosen with
respect to the desired acid concentration in the diluate.
Below about 100 A/m2, the ionic transfer rate is low and
above about 1000 A/m2 heat generation may be excessive.
Preferably, the current density is chosen in the range of
about 500 to 1000 A/m2.
The invention will now be illustrated by the following non-
limitative examples.
Example 1
This example illustrates the use of dialysis by itself. A
dialysis unit was assembled consisting of 19 sheets of
Selemion DMV anionic membranes with a total effective
membrane area of 3970 cm2. A feed solution containing
hydrochloric acid and aluminum was fed into the dialyzate
compartments to separate the acid into a diffusate. Water
2~2~Q~
14
was fed into the diffusate compartments as acid-receiving
solution. The test conditions and results are given in
Table I.
Table I
Flow Rate g/L g/L
L/h.m2 HCl Al
Feed solution 0.83 105 14.8
Water 0.83
Diffusate 0.71 88 0.37
Dialyzate 0.95 26 12.7
The results show that a major portion of the acid is
separated in the diffusate, while the aluminum is
substantially retained in the dialyzate.
Example 2
Using the same unit as in Example 1, an acidic sulfate
solution derived from a hydrometallurgical production
process of zinc was similarly treated. The test conditions
and results are given in Table II.
Table II
Flow Rate g/L g/L g/L g/L
L/h.m2 H2SO4 Zn ____ Mn
Feed Solution 0.77234 20 7.9 1.1
Water 0.79
Diffusate 0.80182 0.4 0.130.03
Dialyzate 0.7645 19.8 7.9 1.1
2~283~
~_ 15
The results show that the diffusate contains a major
portion of the acid in the feed and is substantially free
of other substances, which are substantially retained in
the dialyzate.
Example 3
The membranes in the test unit of Example 1 were replaced
with Selemion DSV membranes and the test of Example 2 was
repeated with the same feed solution. The test data and
results are given in TabLe III.
Table III
Flow Rate g/L g/L g/L g/L
L/h.m2 H2SO4 Zn Mg Mn
Feed Solution 0.91234 20 7.9 1.1
Water 0.83 - - - -
Diffusate 0.76214 1.60.54 0.1
Dialyzate 0.9851 17 7.0 0.93
Example 4
Using the unit of Example 3 another acidic zinc solution
was similarly treated with the results as shown in Table
IV.
Table IV
Flow R2ate g/L g/L g/L g/L
L/h.m H2SO4 Zn Mg Mn
___ ____ ____ ____
Feed Solution 0.95 150 50 8 1.5
Water 1.0
Diffusate 0.95 120 2.0 0.31 0.06
Dialyzate 1.0 29 45.7 7.3 1.4
2~283~Q
_ 16
The results presented in Tables I, II, III and IV show that
acid can be selectively removed from a metal-containing
acid solution. However, the acid concentration in the
diffusate acid product was lower than that in the feed
solution. Moreover, at temperatures of 20C - 25C, feed
and water rates of 0.7 to 1 L/h.m2 had to be selected in
order to maximize both the acid recovery and the
concentration in the acid product.
Example 5
This example illustrates the use of electrodialysis by
itself to remove acid from a metal-containing solution. An
electrodialysis unit was assembled consisting of 10
membrane pairs with a total effective membrane area of 1720
cm2 for the 10 pairs. Selemion CMV-A specially surface-
treated cationic membranes, and Selemion AAV anionic
membranes were used. An acidic solution containing Zn, Mg
and Mn was treated at a feed rate of 43.4 L/h.m2.
The feed solution was circulated through the diluate
compartments at a linear velocity of 5 cm/sec. Water was
fed into the concentrate at 2.87 L/h.m2, the concentrate
being circulated through the concentrate compartments at a
linear of velocity of 5 cm/sec. A 20 g/L H2SO4 solution was
circulated through the electrode compartments as rinse
solution. The test was allowed to proceed for 24 hours.
Other test parameters and results are given in Table V.
2~283QQ
17
Table V
Current Density : 1,000 A/m2
Temperature : 40C
Solution Composition g/L
H2SO4 Zn Mg Mn
Feed Solution 100 5.1 6.6 1.5
Diluate 86 5.1 6.5 1.5
Concentrate 221 3.6 4.7 0.3
The results show that, while the degree of acid removal was
low, the acid concentration in the acid product
(concentrate) was much higher than that in the feed
solution. It can also be seen that the selectivity was
low, with significant metal losses to the acid product.
Thus, the use of electrodialysis alone to recover acid will
require more than one stage in order to maximize the
recovery, but significant metal concentrations would be
present in the acid product.
Example 6
This example illustrates the use of an integrated process
using dialysis and electrodialysis according to Figure 1 to
give a much more effective acid removal and recovery than
by using either a dialysis process or an electrodialysis
process by itself.
A feed solution containing 145 g/L H2SO4, 53 g/L Zn, 7.5 g/L
Mg and 1.5 g/L Mn was fed at a rate of 1.7 L/h.m2 to the
dialyzate compartments of the dialysis unit as used in
` ~132831~0 18
Example 1. Water was added to a portion taken from the
recirculating diluate stream from the electrodialysis unit
giving a solution containing 7.5 g/L H2S04, 1. 2 g/L Zn, 0.2
g/L Mg and 0.03 g/L Mn. The resulting solution was fed at
a rate of 4.8 L/h.m2 to the diffusate compartments of the
dialysis unit. The unit was operated at 24C. Acid was
removed from the feed solution giving a dialyzate
containing 38 g/L H2SO4, 41.2 g/L Zn, 5.8 g/L Mg, and 1.2
g/L Mn. The dialyzate was withdrawn at a rate of 2.2
L/h.m2. A diffusate stream was produced at a rate of 4.3
L/h.m2, and was found to contain 46 g/L H2S04, 1. 2 g/L Zn,
0.16 g/L Mg and 0.03 g/L Mn.
The diffusate was fed to the diluate compartments of the
electrodialysis unit, as used in Example 5, at a rate of
6.7 L/h.m2. The electrodialysis was operated at a current
density of 500 A/m2. The temperature was maintained at
45C. The diluate was withdrawn from circulation at a rate
of 5.6 L/h.m2, and was found to contain 11 g/L H2SO4, 1.3
g/L Zn, 0.2 g/L Mg, and 0.04 g/L Mn. An acid product
(concentrate) from the electrodialysis was withdrawn from
recirculating concentrate at the rate of 1.1 L/h.m2. The
acid product contained 220 g/L H2SO4, 0.7 g/L Zn, 0.1 g/L Mg
and 0.02 g/L Mn.
In a second test, a diffusate from the dialysis unit
containing 40.1 g/L H2S04 was fed at a rate of 11.6 L/h.m2
to the electrodialysis unit operated at 750 A/m2. Diluate,
was withdrawn from circulation at a rate of 10 L/h.m2 and
was found to contain 9.9 g/L H2SO4. Concentrate (acid
2~283~0
19
product) containing 250 g/L H2SO4 was withdrawn from
circulation at a rate of 1.5 L/h.m2.
Thus, the use of a combination of dialysis and
electrodialysis yielded an acid product which was high in
acid and low in dissolved metal content. Moreover, the
dialysis could be fed with the feed solution at a high
rate.
Example 7
This example illustrates how a combined dialysis-
electrodialysis process can be used to treat a solution
containing hydrochloric acid and aluminum for the effective
and selective recovery of acid substantially free of
aluminum.
A feed solution containing 105 g/L HCl and 14.8 g/L Al was
fed at a rate of 1.6 L/h.m2 to the dialysis unit as used in
Example 1. Water was added to a diluate from an
electrodialysis forming a solution containing 1.9 g/L HCl
and 0.3 g/L Al, which was fed to the diffusate compartments
of the dialysis unit at a rate of 4.7 L/h.m2. The dialysis
was carried out at 26C. A dialyzate was produced at a rate
of 2.3 L/h.m2, and was found to contain 24 g/L HCl and 9.7
g/L Al. A diffusate stream containing 28 g/L HCl and 0.4
g/L Al was produced at the rate of 4 L/h.m2. Diffusate was
fed to the electrodialysis unit, as used in Example 5, at
a rate of 6.5 L/h.m2. The electrodialysis unit was
operated at 500 A/m2. The temperature was controlled at
50C in the circulating diluate and concentrate streams.
2 ~ 3 0 ~
_ 20
Diluate was withdrawn from circulation at a rate of 5.0
L/h.m2, and was found to contain 2.6 g/L HCl and 0.44 g/L
Al. A concentrate (acid product) containing 118 g/L HCl
and 0.25 g/L Al was withdrawn from circulation at a rate of
l.S L/h.m2.
It is understood that changes and modifications may be made
in the invention without departing from the scope and
purview of the appended claims.