Note: Descriptions are shown in the official language in which they were submitted.
2 ~
,,~ ~
DUAL INDIFFERENT ELECTRODE
FIELD OF THE INVENTION
This invention relates broadly to the art of implantable
medical devices and, more particularly, to apparatus having dual
- indifferent electrodes which allow the implementation of an
implanted tetrapolar impedance system that requires only a bipolar
pacing lead. Such an apparatus finds use in a tetrapolar impedance
system that provides a stroke volume signal and a ventilatory
signal using any bipolar pacing lead. In a further aspect, the
dual indifferent electrode of the invention also facilitates a
tripolar impedance techni~ue using only a unipolar endocardial
lead.
~ACKGROUND OF THE INVENTION
The stroke volume of the heart has been recognized as
providing a useful signal to control the timing circuit of a
demand-type cardiac pacer. In such a system, the pacer pulse
generator will output stimulating pulses in accordance with the
physiologic demand indicated by stroke volume changes in the
patient's heart. In U.S. Patent 4,686,987 to Salo, et al.,
entitled "Biomedical Method and Apparatus for Controlling the
Administration of Therapy to a Patient in Response to Changes in
Physiologic Demand", a biomedical apparatus capable of sens~ng
changes in the heart's ventricular volume or stroke volume is
disclosed. The apparatus changes the operating performance of the
device as a function of stroke volume. The teachings of U.S.
Patent 4,686,987 are hereby incorporated by reference. Salo, et
al. teaches that a relatively low frequency signal (under 5 KHz) is
s
applied between spaced electrodes disposed in the heart. The
beating action of the heart serves to modulate the signal due to
changes in impedance being sensed between these or other electrodes
within the heart. The modulated carrier signal is processed to
remove R-Wave and other electrical artifacts and then demodulated
to remove the carrier frequency component, leaving an envelope
signal which is proportional to instantaneous ventricular volume.
This envelope signal then contains stroke volume and ventricular
volume information which can be used by the biomedical apparatus to
vary its operating parameters. For example, a current proportional
to changes in the stroke volume may be injected into the timing
circuit of a demand-type cardiac pacer pulse generator whereby the
interpulse interval of the pulse generator is varied as a function
of stroke volume.
A copending application assigned to the assignee of this
application having U.S. Patent Application Serial No. 07/664,461,
filed March 1, 1991, and entitled "Variation In Cardiac Chamber
Volume or Pressure as a Controlling Parameter", is also
incorporated herein by reference. The aforereferenced application
recognizes that the ventilatory signal also appears as a component
of the impedance signal. Because the intrathoracic pressure is
directly related to ventilation (i.e. pressure drops during
inspiration and increases during expiration), the amplitude of the
variation in intrathoracic pressure during a ventilatory cycle is
directly related to the depth of ventilation (i.e. respiration).
Patent Application Serial No. 07/664,461 provides an impedance
system for measurement of right ventricular (or atrial) volume or
a pressure transducer for measurement of right ventricular (or
atrial) pressure, a signal processing means to extract one of the
volume or pressure parameters on a beat-by-beat basis to thereby
. - .. ..
~ ~ f 4 ` i
yield a signal varying at the ventilatory rate and with a peak-to-
peak amplitude proportional to ventilatory depth.
Referring again to the Salo, et al. patent, for example, a
cardiac lead having two sensing electrodes and a stimulating
electrode is used. Often, in the case of a cardiac pacer
replacement, a bipolar lead having only two electrodes has
previously been implanted in the heart. In such cases, since it is
desirable to use the already implanted lead with a new pacemaker
system in the case of, for example, replacing a worn-out pacemaker,
the three electrode lead as used by Salo, et al. is often not
available. In such cases, only three electrodes are typically
available, namely, the pulse generator case or can, a lead ring on
the endocardial lead and a tip electrode on the endocardial lead.
Prior approaches to implementing an intracardiac impedance system
with only three electrodes available have used at least one
electrode as a simultaneous drive and sense electrode, since two
drive and two sense points are required. Such approaches have
several disadvantages.
One disadvantage of prior art techniques results from a high
current density region being sensed at the "common" electrode
(i.e., the electrode being used as both a drive and sense
electrode) making it very sensitive to local effects such as, for
example, mechanical motion. Another disadvantage of prior art
systems results from the interface impedance at the common
electrode which presents a large DC offset when sensed, yielding a
lower modulation index relative to that experienced with tetrapolar
impedance. Yet another drawback of prior art systems is that if
the common electrode is on the pacemaker lead, either the ring or
the tip, system performance will vary as a function of electrode
- 3 -
. ~ .
- , . : . ............................ ...
. ,, . ~ . . .. .
L~ r~
material, effective surface area, geometry and various other
electrode characteristics.
The method of the present invention uses tetrapolar impedance
techniques and overcomes the above described disadvantages of prior
art devices. since the present invention effectively implements a
tetrapolar impedance system that provides a stroke volume signal
using any bipolar pacing lead, the quality of the sensed stroke
volume signal equals that of a tetrapolar system using a pulse
generator can and a tripolar pacing lead. In a further aspect, the
signal sensed with the present invention contains a lower frequency
component due to ventilation. This component may be extracted as
it is related to tidal volume and may be used as another rate
controlling parameter.
The present invention also affords an advantage even when used
on a unipolar pacing lead. Although a tetrapolar method is not
possible for intra-cardiac use in such a case, the dual indifferent
method provided by the invention allows a tripolar technique. This
has the advantages of reduced motion artifact at the pacer can, as
well as a lower DC offset.
SUMMARY O~ THE INVENTION
This invention provides apparatus for use in a variable rate
pacer apparatus responsive to the metabolic needs of the patient.
In carrying out the instant invention and in accordance with a
first embodiment, an endocardial lead having first and second
spaced apart electrodes resides in a patient's heart. The first
electrode is a sensing electrode and the second electrode is a
carrier signal driving electrode. The lead has conductors coupling
a source of alternating current carrier signals of a predetermined
frequency to the second electrode. A third electrode is in
electrical contact with body tissues. The pacer can functions as
- 4 -
- : . . .
2 ~
a fourth electrode and has a plastic top wherein the third
electrode is located. Either the pacer can or the third electrode
may be coupled to the carrier signals and acts in cooperation with
the second electrode to form a pair of driving electrodes. The
first electrode and either the third electrode or the can may
comprise a sensing electrode pair. The sensing electrode pair is
coupled to a sense amplifier means for receiving and amplifying
modulated electrical signals developed across the sensing electrode
pair. A demodulator and filters circuit means for demodulating the
modulated carrier signal and recovering the modulating signal
therefrom is connected to the output of the sense amplifier means.
The modulating signal contains components proportional to
instantaneous stroke volume of the patient's heart and the
patient's ventilatory tidal volume, and the demodulator and filters
circuit develops control signals therefrom called stroke volume and
ventilation signals respectively. The control signals may be
applied individually or in combination to the pulse generator so as
to control the rate of stimulating pulses in accordance with a
predetermined rate control algorithm.
It is one object of the invention to provide an implanted
tetrapolar impedance system that requires only a bipolar
endocardial lead.
It is another ob;ect of the invention to provide an implanted
tripolar impedance system that requires only a unipolar endocardial
lead.
It is yet another object of the invention to provide a button
electrode electrically isolated from a pacemaker can and having a
surface area on the same order as that for a lead ring electrode of
an endocardial lead.
. . - , - . . , ~ , , - .
.:
It is yet another object of the invention to provide an
effective implementation of a tetrapolar impedance system that
provides a stroke volume signal using any bipolar pacing lead
wherein the ~uality and pulsatile morphology of the signal equals
5 that of a tetrapolar system using a pulse generator can and a
tripolar pacing lead as electrodes.
It is yet another object of the invention to provide an
effective implementation of a tetrapolar impedance system that
provides a ventilation signal free from can motion artifact using
any bipolar pacing lead.
Other objects, features and advantages of the invention will
become apparent to those skilled in the art through the
description, claims and drawings herein.
BRIEF DESCRIPTION OF THE DRAWINGS
Figure 1 schematically shows a pacer apparatus having a dual
indifferent electrode apparatus.
Figure 2 schematically shows one embodiment of a dual
indifferent electrical apparatus for use in an implantable heart
pacemaker in accordance with the invention.
Figure 3 schematically shows an alternate embodime~t of a dual
indifferent electrode apparatus as employed with a unipolar
endocardial lead.
Figure 4 schematically shows an alternative embodiment of a
dual indifferent electrode apparatus differing from Figure 2 in the
manner in which the electrodes are driven and used for sensing.
Figure 5 schematically shows an alternate embodiment of a dual
indifferent electrode apparatus employing a unipolar endocardial
lead but differing from the embodiment of Figure 3 in the manner
the electrodes are configured.
Figure 6 schematically shows an alternate embodiment of a dual
indifferent electrode apparatus for a pacer in which the drive
voltage is applied between the pacer can and a spot electrode.
DESCRIPTION OF THE PREFERRED E~BODIMENT
Referring to Figure 1 there is diagrammatically shown a side
view of a pacemaker apparatus 2 comprised of a conductive metal can
10 and an insulating top or header 11. Mounted in the top 11 and
isolated from the metal can 10 is a button electrode 12. Contained
within the can 10 is electronic circuit 100 which is explained in
more detail below and which comprises the dual indifferent
electrode circuitry.
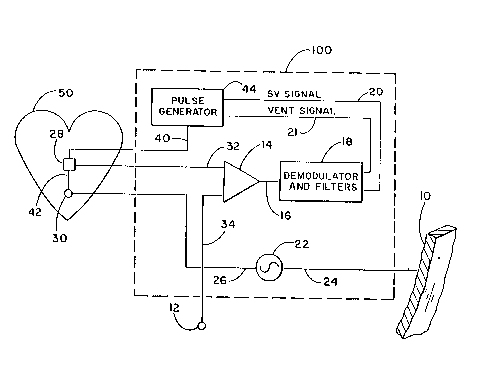
Now, referring to Figure 2, the circuit 100 is shown in more
detail. The can 10 is connected by lead 24 to an oscillator 22
which serves as a carrier current source. An endocardial lead 40
is connected to a pulse generator 44 which is contained within the
pacemaker 2. The lead 40 includes electrodes 28 and 30 located
within one of the chambers of the heart 50. Electrode 30 may be,
for example, a stimulating tip electrode on a catheter type lead
while electrode 28 may be, for example, a ring electrode.
Insulator lead body 42 mechanically supports electrodes 28 and 30.
The oscillator 22 is arranged to produce an alternating current
carrier signal at a frequency which is quite high compared to the
heart rate. Typically the carrier signal is in the range of from
about 500 to 20000 Hz. In ths arrangement of Figure 2, the carrier
signal is driven by electrode 30 through body tissues to the can
10. Button electrode 12 has a surface area typically on the same
order of magnitude as the surface area of ring electrode 28 and is
advantageously disposed on the plastic top 11 of the implantable
pacemaker 2. In the embodiment of Figure 2, the button electrode
12 is connected via lead 34 to a first input of a differential
f", r -if~
amplifier 14. Ring electrode 28 is also connected via lead 32 to
a second input of differential amplifier 14. The output of
differential amplifier 14 is carried via conductor 16 into
demodulator and filters circuit 18. The demodulator and filters
circuit 18 is connected by line 20 to the pulse generator. The
demodulator and filters circuit 18 may include signal processing
circuits as are shown in U.S. Patent 4,686,987, as well as
filtering mean~ to separate the higher frequency stroke volume
signal from the lower frequency ventilation signal as shown in
Patent Application Serial No. 07/664,461.
In operation, the pulse generator 44 provides stimulating
pulses to stimulating electrodes in a well known manner to pace the
heart. Electrodes 28 and 12 sense electrical impedance variations
in the thoracic cavity which may be due to the pumping action of
the heart or other physiological signals of interest. The signals
are fed into the differential sense amplifier 14 which provides a
differential signal to the demodulator and filters circuit 18. The
demodulator and filters circuit includes means for demodulating the
modulated carrier signal and recovering the modulating envelope
signal therefrom. The modulating signal is found to contain
frequency components proportional to the instantaneous stroke
volume of the patient's heart and to the instantaneous tidal volume
of the patient's ventilation. The demodulator and filters circuit
18 then provides control signals, SV SIGNAL 20 and VENT SIGNAL 21
to the pulse generator. The pulse generator responds to the
control signal by determining a rate at which the heart stimulating
pulses will be generated.
Now referring to Figure 3, an alternate embodiment of the
invention is shown as employed with a unipolar endocardial lead.
In this embodiment, it will be understood that circuit lOOA is
similar to circuit 100 except that it is modified to accommodate
unipolar pacing and sensing techniques. In this embodiment, the
can 10 is connected by lead 24 to the oscillator 22 which serves as
a carrier current source. The endocardial lead 4OA is connected to
a pulse generator 44 which is contained within the pacemaker 2.
Lead 45 connects the pulse generator to the can 10 which, in this
case, serves as a stimulating electrode. The lead 40A includes
electrode 30 located within one of the chambers of the heart 50.
Electrode 30 may be, for example, a tip electrode on a catheter
type lead. The oscillator 22 is arranged to produce an alternating
current carrier signal at a frequency which is quite high compared
to the heart rate. Typically the carrier signal is in the range of
from about 500 to 20000 Hz. The carrier signal is driven by
electrode 30 through body tissues to the can 10. Button electrode
12 has a surface area typically on the same order of magnitude as
the surface area of electrode 30 and is advantageously disposed on
the plastic top 11 of the implantable pacemaker 2. The button
electrode 12 is connected via lead 34 to a first input of a
differential amplifier 14. Tip electrode 30 is also connected via
lead 32A to a second input of differential amplifier 14. The
output of differential amplifier 14 is carried via conductor 16
into demodulator and filters circuit 18. The demodulator and
filters circuit 18 are connected by lines 20 and 21 to the pulse
generator. The circuit 18 is configured as described above with
reference to Figure 2.
In operation, the pulse generator 44 provides stimulating
pulses to stimulating electrodes in a well known manner to pace the
heart. Electrodes 30 and 12 sense stroke volume impedance signals
or other physiological signals of interest. The signals are fed
into the differential amplifier 14 which provides a differential
""' " '
Lr~ ri ~
signal to the circuit 18. The demodulator and filters circuit
operates as described above with reference to Figure 2.
ALTERNATIVE EMBODIMENTS
Figure 4 illustrates an alternative embodiment of the
invention, again involving a bipolar lead 42 as in the embodiment
of Figure 2 but instead of connecting the oscillator 22 between the
can 10 and the tip electrode 30, in the arrangement of Figure 4,
the oscillator 22 is connected between the tip electrode 30 and the
button electrode 12 disposed on the insulating plastic top or
header 11. Also, sensing takes place between the conductive metal
can 10 and the ring electrode 28 on the endocardial lead 42.
Specifically, the ring electrode 28 is connected by conductor 32 to
a first input of the differential sense amplifier 14. A conductor
52 connects the can 10 to the second input of this differential
sense amplifier. The oscillator 22 again produces an alternating
current carrier signal at a frequency which is far higher than the
patient's heart rate and, as indicated earlier, may fall into the
range of from about 500 to 20000 Hz.
The signals fed into the sense amplifier 14 reflect impedance
changes in the body tissue present between the location of the can
in the chest wall and the ring electrode 28 whiah would
typically be in the right atrium of the heart. This modulation is
removed by the demodulator and filters 18, again resulting in a
ventilatory signal component on line 21 and a stroke volume related
signal on line 20. These signals may be used individually or may
be combined together in a predetermined algorithm with the
resulting control signal ùsed to adjust the rate at which pulse
generator 44 emits pacing pulses to the tip electrode 30.
-- 10 --
:'
Figure 5 shows a further ,alternative embodiment of the
invention related to Figure 3 of the drawings in that it embodies
a unipolar lead 40A. Rather than connecting the high frequency
carrier signal oscillator 22 between the can 10 and the tip
electrode 30, in the arrangement of Figure 5, the carrier
oscillator 22 is connected between the spot electrode 12 on the
insulated header 11 of the pacemaker 2 and the tip electrode 30 of
the lead. When configured in this way, sensing occurs between the
can 10 and the tip electrode 30. That is to say, the first input
to the differential amplifier 14 is connected by a conductor 54 to
the metal can 10 housing the pacing circuitry while the conductor
32A joins the second input of the differential sense amplifier 14
to the tip electrode 30 of the monopolar pacing lead 40A.
The embodiments of Figures 4 and 5 thus provide alternative
ways of configuring the drive and sense electrodes but in most
respects, the operation of the pacer system is substantially the
same as is described in conjunction with the embodiments of Figures
2 and 3.
In the embodiment shown in Figure 6, the oscillator 22 is
coupled between the pacer can 10 and the button electrode 12.
Sensing occurs between the monopolar tip electrode 30 on the pacing
lead 42 and the button electrode 12. When a high frequency signal
which may range between 500 Hz and 20000 Hz is applied between the
can 10 and button electrode 12, the signal developed between
electrode and tip electrode 30 and applied to the sense amplifier
14 is found to be modulated primarily by ventilatory activity
rather than systolic activity. Thus, the output from the
demodulator and filter circuit 18 on line 21 is significantly more
dominating than the stroke volume signal presènt on line 20.
-- 1 1 --
... .
~ ` .
The invention has been described herein in considerable detail
in order to comply with the patent statutes and to provide those
skilled in the art with information needed to apply the novel
principles and to construct and use such specialized components as
are required. However, it is to be understood that the invention
can be carried out by specifically different equipment and devices.
For example, the control signal proportional to stroke volume
change may be used in conjunction with an implantable infusion pump
for administering such drugs as dobutamine, isoproterenol or
nitroprusside whereby stroke volume may be maintained at a desired
value. Alternatively, the demodulated waveform or control signal
may be used directly by other diagnostic equipment. By
appropriately utilizing the information derived from the
ventricular impedance, it would be possible to measure stroke
volume without having to resort to thermal dilution or other
techniques. Hence, various modifications, both as to the equipment
details and operating procedures can be effective without departing
from the scope of the invention itself.
What is claimed is:
- 12 -
: : .
.
:, : . .