Note: Descriptions are shown in the official language in which they were submitted.
2~7~89
"ALUMINIZED STAINLESS STEEL
AND METHOD F~R PRODUCING SAME"
BACKGROUND OF THE INVENTION
This invention relates generally to chromium-
containing steels with aluminum coatings and more
particularly to an aluminized, AISI type 409, stainless
steel.
Chromium-containing steel, particularly stainless
steel, is used extensively in many industrial, residential,
automotive, and high temperature applications due to its
excellent ability to withstand corrosive and high
temperature oxidizing environments. One widespread use of
this steel is in automobile exhaust components such as
mufflers, tailpipes and catalytic converter shells. These
components must be able to withstand internal corrosion due
to high temperature gases and external corrosion due to
road salt.
The corrosion-resistant properties of stainless
steel are achieved by adding chromium to steel, optimally
about 10 wt.% or greater, although a lesser percentage may
be used. Chromium at this level forms, on the surface of
the steel, a passive film that is responsible for corrosion
resistance. At higher temperatures, chromium forms an
impervious oxide layer which slows down the oxidation
process.
It may conceivably appear to be desirable to add
aluminum to steel because, at higher temperatures, a steel
containing aluminum may be more oxidation-resistant than a
steel containing corresponding amounts of chromium. The
oxidation resistance of aluminum-containing steel is due to
the formation of aluminum-rich oxides at the surface of the
steel. Unfortunately, the addition to steel of aluminum in
20~1189
sufficient quantity to form an aluminum-rich oxide layer
causes manufacturing and mechanical problems.
Coating a chromium-containing steel substrate
with aluminum would be desirable in order to overcome these
S problems and would result in both (a) high temperature
oxidation resistance due to the aluminum coating and (b)
the beneficial passive film formed by the chromium
contained in the substrate. The superior corrosion
resistance of aluminum coated, AISI type 409 stainless
steel is described by Ando et al. of Nisshin Steel Co.,
NACE Annual Conference and Corrosion Show Paper Number 384
(March 1991).
There is another significant reason for coating
stainless steel with aluminum. After being exposed to the
atmosphere, especially a humid atmosphere, AISI type 409
stainless steel and similar stainless steels become
discolored and appear to have surface corrosion.
Therefore, although the steel is still protected from
oxidation by the tenacious chromium oxide layer, the steel
does not appear "stainless." As a result, coating the
stainless steel with aluminum is desired for aesthetic
purposes, especially when producing visible automotive
exhaust components, because many consumers would be
dissatisfied with a discolored product.
However, using a hot-dip process to coat
stainless steel with aluminum presents a problem. The
problem arises because stainless steel forms a tenacious,
chromium oxide, outer layer during an annealing step which
normally precedes hot dipping, and the chromium oxide outer
layer is not wettable to molten aluminum. Therefore, the
process usually results in uncoated spots and poor
adherence of the aluminum coating to the stainless steel
substrate. Poor adherence is reflected by flaking or
cracking of the coating during bending of the strip. U~
Pat. No. 4,675,214 to Kilbane et al. discloses a method for
207~189
enhancing the wettability of a chromium-containing steel by
a pure aluminum coatin~; the method involves controlling
the coating conditions.
Pre-plating the surface of the stainless steel
substrate with copper, nickel or iron-boron improves the
substrate's coatability with molten aluminum to a degree
which is commercially acceptable. One such process is
disclosed in U.S. Pat. No. 4,913,785 to Uchida et al.
However, this solution is undesirable due to the added time
and cost of performing such an intermediate coating or pre-
plating step.
SUMMARY OF THE INVENTION
The present invention is directed to a method ~or
coating a strip of chromium-containing steel, e.g.
stainless steel, with aluminum without the need for pre-
plating the steel substrate before coating the substrate
with aluminum, and to the product produced thereby.
In accordance with the present invention, a
chromium-containing, preannealed steel substrate in the
form of a strip (e.g., stainless steel strip) may be
continuously coated with aluminum by preferably first
subjecting the strip to a pre-cleaning step in which the
substrate is passed through a heated alkaline cleaning
bath, followed by rinsing and drying. The substrate is
then passed through a furnace or enclosed heating zone
wherein the strip is subjected to heating without flame
impingement, e.g. radiant heating. The dew point inside
the heating zone is controlled, preferably by passing
hydrogen and nitrogen gas through the heating zone and by
preventing the entry of oxygen gas into the heating zone.
A reducing atmosphere is thereby produced which will
substantially reduce the formation of chromium oxides and
other impurities on the surface of the chromium-containing
2071189
steel strip while the strip is in the heating zone. The
heating zone is kept at a controlled temperature,
preferably less than 800C, so as to substantially reduce
the growth of any chromium oxides which may possibly have
formed on the surface of the substrate.
After the substrate passes through the heating
zone, it is coated with aluminum by a hot-dip procedure,
wherein the strip is dipped in a molten aluminum bath. In
a preferred embodiment, the aluminum coating is an alloy
which contains about 10 wt.% silicon. After the strip
exits the bath, the thickness of the coating on the strip
may be controlled by a jet stream of inert gas such as
nitrogen. This step is preferably followed by steam
cooling which solidifies the coating and prevents its
transfer onto the rollers which direct the passage of the
strip downstream of the bath.
The method of the present invention produces an
aluminized, chromium-containing steel strip with a uniform
coating and acceptable adherence. There is a substantial
absence, on the coating, of spots reflecting an unwetted
substrate at the interface between the substrate and the
coating; no pre-plating step employing a pre-plating metal
wettable by molten aluminum is necessary. Heating the
steel to a low temperature in a controlled, reducing
atmosphere obviates the need for such pre-plating.
Other features and advantages are inherent in the
method and product claimed and disclosed or will become
apparent to those skilled in the art from the following
detailed description taken in conjunction with the appended
drawing and claims.
BRIEF DESCRIPTION OF THE DRAWING
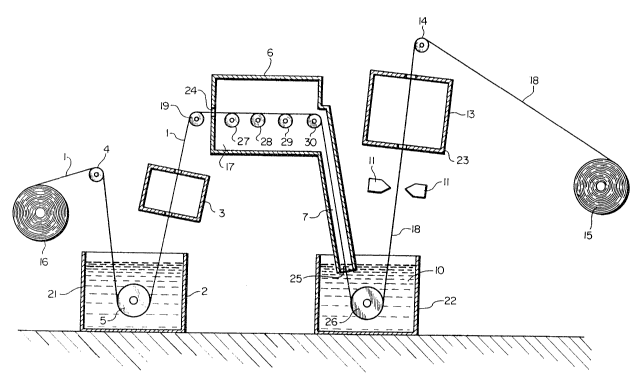
The sole figure is a schematic diagram
illustrating a method in accordance with an embodiment of
the present invention.
2 ~ 8 ~
DETAILED DESCRIPTION OF THE INVENTION
Referring to the drawing, a continuous, chromium-
containing, preannealed steel strip or substrate 1 is fed
from a coil 16 over a guide roller 4 into a pre-cleaning
bath 2. Following pre-cleaning, substrate 1 is subjected
to rinsing and drying in a rinsing and drying chamber 3.
The cleaned substrate l is fed over guide roller 19 and
then passed through an enclosed furnace or heating zone ~,
across supporting rollers 27 through 30. Furnace 6 has a
controlled temperature and dew point. After exiting
furnace 6 through a downstream furnace portion 7 or snout,
substrate 1 is coated with aluminum in a hot-dip coating
bath 10. The thickness of the coating is controlled by
nitrogen jet knives 11 and 11'. Finally, the aluminum-
coated strip is passed through a steam cooling zone 13,over a guide roller 14, and rewound into a coil 15.
The present method is applicable both to AISI
type 40g stainless steel substrates and to chromium-
containing steel substrates with other compositions. The
minimum chromium content of substrate 1 is, however, at
least 1 wt.%, preferably at least 2 wt.%, and most
preferably at least 10 wt.~. Greater than 10 wt.% chromium
achieves optimal corrosion resistance.
Substrate l is preannealed before it is subjected
to the invented method. Preannealing, wherein the steel is
heated to a suitable temperature, held for some period of
time, and then cooled at a suitable rate, gives the steel
the same properties as annealing done on-line. Annealing
softens the steel and can greatly increase the elongation
characteristics of the steel, from less than 3% for non-
annealed steel to possibly greater than 30% for annealed
steel. Annealing can decrease tensile strength from about
.~ .
2~71189
150 ksi for non-annealed steel to about 60 ksi for annealed
steel.
The chromium-containing steel substrate may also
contain any or all of the following: at least 0.25 wt.%
titanium, about 0.30 to about 1.0 wt.~ manganese, about
0.30 to about 1.0 wt.% silicon, and about 0.02 to about
0.08 wt.% carbon. The weight percentage of titanium is
generally at least six times the weight percentage of
carbon. Other elements that may be present in the
substrate include sulfur and phosphorus. The content of
sulfur, phosphorus and other residual elements should be
within the acceptable ranges given by AI5I designation.
Examples of useful chromium-containing steel
substrates are tabulated below:
15 Ingredient A B
chromium 11.0 wt.% 10.5 wt.% 12.5 wt.%
manganese 0.30 wt.% 1.00 wt.% 0.45 wt.%
carbon 0.04 wt.% 0.08 wt.% 0.05 wt.%
titanium 0.28 wt.% 0.48 wt.% 0.40 wt.%
phosphorus ~.03 wt.% .045 wt.% .007 wt.%
sulfur .003 wt.% .045 wt.% 0.02 wt.%
silicon 0.30 wt.% 1.00 wt.% 1.00 wt.%
iron essentially essentially essentially
the balance the balance the balance
In a preferred embodiment of the invention,
preannealed substrate 1 is not subjected to a pre-plating
step, employing a pre-plating metal wettable by aluminum,
upstream of bath 10.
Substrate 1 is subjected to pre-cleaning in order
to remove dirt, oil, and steel particles or fines which may
be present on the surface of substrate 1. Pre-cleaning
bath 2, contained in a container 21 having a guide roller
5 and either with or without scrubber brushes, may be an
alkaline composition containing about 2.5 to 3.5 wt.~ NaOH.
Bath 2 typically has a temperature of about 120F to about
2~71~89
180F (48C to 82C). Rinsing and drying zone 3 employs a
hot water rinse followed by hot air drying.
Furnace or enclosed heating zone 6 typically
employs radiant heating under controlled conditions. There
is, therefore, no flame impingement in furnace 6.
The dew point in heating zone 6 is controlled in
order to minimize the formation of ~a) chromium oxides and
(b) reaction products other than chromium oxides, on the
surface of the chromium-containing steel strip or substrate
1, while the strip is in heating zone 6. This is
accomplished by maintaining, in heating zone 6, a reducing
atmosphere which is substantially devoid of water vapor and
consists essentially of hydrogen and nitrogen gases, e.g.,
about 25 vol.% to about 50 vol.% hydrogen gas, with the
balance of the atmosphere being substantially nitrogen gas.
The hydrogen and nitrogen gases are substantially devoid of
water vapor and oxygen gas and are thus "dry" gases as they
enter heating zone 6. These dry gases may flow
continuously through heating zone 6, and this procedure
will reduce the relative amount of water vapor in heating
zone 6. The dew point is also controlled by preventin~
entry into heating zone 6 of oxygen gas, for example by
avoiding leaks.
In a preferred embodiment of the invention,
heatiny zone 6 has an upstream portion 17 (the main body of
the heating zone~ having an entry 24, and there is a
downstream furnace portion 7 (known as the furnace "snout"~
having an exit end 25. In this embodiment, the dew point
in upstream portion 17 is maintained at a temperature in
30 the range of about -30F to about -10F (-35C to -23C).
At entry 24 into upstream furnace portion 17l the dew point
is maintained in the range of about -20F to -10F (-29C
to -23C). The dew point in downstream furnace portion 7
is maintained at a temp~rature in the range of about -50F
35 to about -45F (-46C to -42C). The dew point is
20~1189
maintained at about -60F (-51C) at exit end 25 of
downstream portion 7.
The temperature in heating zone 6 is controlled
so as to substantially reduce the growth o~ any chromium
oxides which may possibly form on the surface of substrate
1. The temperature at entry 24 of upstream furnace portion
17 is maintained in the range of about 200F to about 300F
(93C to 149C). The maximum temperature in heating zone
6 is no greater than about 1570F to about 1670~F (854C to
910C).
Substrate 1 is heated in heating zone 6 to a
temperature in the range of about 1350F to about 1400F
(733C to 760C) and maintained at that temperature in
heating zone 6. The maximum temperature of substrate 1 in
heating zone 6 is no greater than about 1470F (800C).
The temperature of substrate 1 cools down to about 1250F
to about 1350F (677C to 733C) by the time it exits
downstream furnace portion 7.
The hot dip coating procedure of the present
invention may employ a coating metal consisting essentially
of aluminum. This is known as "Type 2" aluminum.
Alternatively, the adherent coating may be an alloy of
aluminum containing about 10 wt.% silicon, known as "Type
1" aluminum.
Coating bath 10, contained in container 22 and
employing guide roller 2~, commonly referred to as a sinker
roll, has a temperature which is maintained in the range of
about 1200F to about 1250F (648C to 677C). Substrate
l is immersed in coating bath 10 for a time of about 3
seconds to about 10 seconds.
Jet knives 11 and 11', which control the
thickness of the coating, direct nitrogen or other gas at
the coated strip. The thickness of the aluminum coating is
controlled to be in the range of about 10 microns to about
25 microns.
20711~9
The cooling of coated strip 18 is performed by
directing steam against coated strip 18 in steam cooling
zone 13 which is contained in housing 23. This cools
coated strip 18 to a temperature in the range of about
700F to about 900F (370C to 482C) before it reaches
roller 1~. Cooling solidifies the coating and prevents
transfer of the coating onto a roller 14 which directs the
passage of coated strip 18 downstream of housing 23.
Coated strip 18 is then wound into coil 15, for easy
transportation and storage.
~ metal product made from coil 15 includes a
chromium-containing steel substrate and an adherent, hot-
dip coating on the substrate. The coating consists
essentially of aluminum or an alloy of aluminum. The metal
product is characterized by the substantial absence, on the
coating, of spots reflecting an unwetted substrate at the
interface between the substrate and the coating. Such
spots are absent despite the absence of (a) a pre-plating
metal, wettable by molten aluminum, between the substrate
and the coating, and (b) any diffusion product of a pre-
plating metal wettable by molten aluminum, within the metal
product.
Pre-plating metals include at least one
ingredient such as copper, nickel or iron-boron, which are
conventionally used to provide a surface wettable by molten
aluminum. The diffusion product of the pre-plating metal
could be generated during heating in furnace 6 and would
include at least one of the following: (a) the pre-plating
metal, (b) an ingredient of the pre plating metal, (c) an
alloy of the pre-plating metal ingredient and aluminum, and
~d) an alloy of the pre-plating metal ingredient and an
ingredient of the steel substrate; none of these are
present in the metal product.
207~189
-- 10 --
The metal product is further characterized by
uniformity of coating and acceptable adherence of the
coating to the steel substrate.
The adherence of the coating to the steel
substrate is at least equal to the adherence exhibited by
the same aluminum coating where there is (a) a pre-plating
metal between the coating and the substrate or (b) a
diffusion product of a pre-plating metal within the metal
product.
10The metal product also exhibits corrosion
resistance at least equal to that of (a) the same metal
product having a pre-plating metal between the aluminum
coating and the steel substrate or of (b) the same metal
product having, within that product, a diffusion product of
the pre-plating metal.
The metal product may be in the form of an
automobile exhaust component, such as a muffler, a
tailpipe, a catalytic converter shell and other visible
automobile components.
20The foregoing detailed description has been given
for clearness of understanding only~ and no unnecessary
limitations should be understood therefrom, as
modifications will be obvious to those skilled in the art.