Note: Descriptions are shown in the official language in which they were submitted.
2080370
Disposable Infusion Apparatus With Peristaltic Pump
Technical Field
The present invention relates to medical devices, and
more particularly, to an improved disposable infusion apparatus
and peristaltic pump for delivering intravenous drugs at a
controlled rate to a patient.
Backqround Art
It is often necessary to intravenously supply patients
with pharmaceutically active liquids over a long period of time
at a controlled rate. It is desirable that this be accomplished
while the patient is in an ambulatory state.
The prior art includes devices that employ a bag filled
with fluid medication that feeds by gravity through IV tubing
having drip or other controllers. It is difficult for a patient
to be ambulatory with a gravity fed infusion device and flow
control is very limited.
Another prior art infusion apparatus comprises an
elastic bladder forming a liquid container mounted in an
elongated cylindrical housing, a flow control valve, and tubing
for supply of the liquid to the patient. The elastic walls of
the bladder expand along the walls of the cylindrical housing
when filled with the liquid, and provide the pressure for
B
W O 91/16933 PC~r/US91/02930
2080370
expelling the liquid. The bladder is typically filled
by hand with a syringe which often requires an
inordinate amount of force. Another drawback is that
the bladder is forced to expand into an unnatural
5 elongated configuration along the housing walls as it
is filled. As a result of this unnatural
configuration, the pressure of the bladder varies
widely with the volume of liquid therein. Therefore,
in most cases this type of elastic infusion apparatus
10 does not have a reasonablv stable pressure and flow
rate over the infusion period.
Most of such devices either have a flow rate that
decreases with pressure, which decreases with volume,
or one that remains roughly constant until the end
15 where it surges. Attempts have been made to control
pressure and flow rates bv means of complicated and
expensive flow control valves and devices. Other
approaches have utilized exotic and expensive elastic
materials in an effort to control the pressures and
20 flow rates.
Another type of infusion apparatus emplovs a
peristaltic or other positive displacement pump which
is electrically driven. Programmable infusion pumps
have been provided having the capability for precise
25 tailoring of the fluid delivery rate parameters in
four different modes, namely, continuous,
intermittent, PCA (patient controlled analgesic) and
TPN (total parenteral nutrition). Originally such
programmable infusion pumps were large and not well
30 suited for ambulatory patients. They used complex and
expensive replacement pump cartridges to maintain
sterilitv. More recently, small proqrammable infusion
pumps have been available with disposable plastic
cartridges that engaae a peristaltic pump. However
b ; ~
.. . .
:. ~
WO 91/16933 PCI/US91/02930
20~0370
3--
such cartridges have been bulky and expensive and have
required excessive drive power in the pumps, leading
to rapid battery drain.
Accordingly, it would be desirable to provide an
5 improved disposable infusion apparatus and pump for
delivering intravenous drugs at a controlled rate to
an ambulatory patient.
Disclosure of Invention
It is therefore the primary object of the present
invention to provide an improved disposable infusion
apparatus and pump for delivering intravenous drugs at
a controlled rate to an ambulatory patient.
In accordance with our invention a disposable
15 infusion apparatus includes proximal, distal and
intermediate segments of IV tubing connected via
couplings. The intermediate segment has a length
sufficient for operative engagement with a pumpins
member of a peristaltic pump throughout a pumping
20 stroke thereof. The intermediate segment further has a
maximum Durometer of seventy-five on the Shore A
scale. The pumping member includes a plurality of
fingers mounted in side-by-side substantially parallel
relationship for individual reciprocation. Motor
25 driven cams individually reciprocate respective ones
of the fingers in a predetermined timed sequence so
that when the linearly disposed intermediate segment
is squeezed by the fingers, intravenous fluid in the
tubing will be pumped therethrough. The disposable
30 apparatus further includes a door connected to
opposite ends of the intermediate segment and which is
releasably loadable in a case to place the
intermediate segment into operative engagement with
the pumping member.
WO9t/16933 PCT/~iS91/02930
20~037~ _4_
~rief Description of Drawings
The above and other objects and advantages of the
present invention will become apparent from the
following description when read in conjunction with
5 the accompanying drawings wherein:
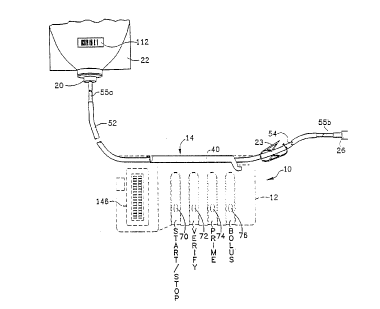
- Fig. l illustrates a programmable infusion system
in phantom lines with a preferred embodiment of our
disposable apparatus loaded therein. The disposable is
shown connected to conventional IV tubing segments, an
lO IV bag, a spike connector and a leur fitting. The IV
tubing segments are broken at various locations to
make this figure more compact.
Fiq. 2 is an enlarged fragmentary view of the
programmable infusion system with the disposable
l5 apparatus installed therein and showing its
relationship to a preferred embodiment of our
peristaltic pump.
Fig. 3 is a further enlarged view of the
peristaltic pump and the door of the disposable. The
20 IV tubing segments are not shown in this figure.
Fig. 4 is an enlarged view of the three seqments
of tubing that form part of the disposable.
Fig. 5 is an enlarged side elevation view of one
of the fingers of the peristaltic pump showing how it
25 squeezes shut the intermediate tubing segment of the
disposable.
Fig. 6 is a side elevation view of one of the cam
wheels of the peristaltic pump. The motion of the cam
wheel is illustrated in phantom lines.
Figs. 7-9 illustrate details of the disposable
door, tube clamp and IV tubing couplings.
W O 91/16933 PC~r/~S91/02930
2~80370
- Best Mode for Carrying Out the Invention
Referring to Fig. 1, a programmable infusion
system 10 is illustrated which is adapted to utilize
our disposable infusion apparatus and peristaltic
5 pump. It includes a compact, portable rectangular case
12. By way of example, the case may be made of
iniection molded plastic and may measure approximately
seven inches in length by approximatelv three and
one-half inches in width (right side in Fig. 1) by
10 approximately one inch in thickness (right side in
Fig. 1).
The preferred embodiment of our disposable IV
tubing apparatus 14 (Fig. 2) may be releasably loaded
or installed in a receptacle 16 (Fig. 2) in a long
15 side edge of the case 12. The proximal end disposable
IV tubing apparatus 14 is connected to a conventional
spike 20 (Fig. 1). The patient inserts the spike into
a conventional bag 22 of intravenous fluid in which
the desired medications are dissolved. The distal end
20 of the disposable IV tubing apparatus 14 is connected
to a conventional male leur fitting 26 which in turn
connects to a conventional IV catheter (not
illustrated). A disposable IV fluid conveying means
is a necessary requirement in an infusion system since
25 it ensures sterility. It also prevents residual
amounts of medication from one IV drug administration
from being inadvertentlY delivered when a new IV drug
administration commences.
Referring to Fig. 2, the preferred embodiment of
30 our peristaltic pump 30 is mounted inside the case 12
adjacent the receptacle 16. A pumping member 32 of
the pump engages a linearly disposed intermediate
segment 34 of the disposable IV tubing apparatus 14.
The pumping member 32 comprises nine individual
20~0370
--6--
fingers 32a which slide back and forth toward and away
from the intermediate IV tubing segment 34. The
fingers 32a are moved by corresponding cam wheels 36
(Fig. 6). A flexible boot 33 (Fig. 2) surrounds the
5 fingers and forms an interface between the fingers and
the intermediate segment 34 of IV tubing. The
peripheral edges of this boot are sealed to internal
walls of the case to protect the pump from
contamination. Each cam wheel 36 (Fig. 5) has a
10 splined mounting hole 37 therethrough which is offset
from the center of the wheel. Each finger 32a
comprises a rectangular block having a circular hole
in which a corresponding one of the cam wheels 36
rotates. The hole has a diameter slightly larger than
15 the outside diameter of the wheel so that the wheel
can rotate inside the hole and thereby pull the finger
back and forth. The motion of one of the cam wheels
is illustrated in phantom lines in Fig. 6.
The cam wheels 36 are mounted on a splined shaft
20 38 in progressive, offset alignment for individually
reciprocating respective ones of the fingers 32a in a
predetermined, timed sequence. The linearly disposed
segment 34 of IV tubing is progressively squeezed by
the fingers 32a so that intravenous fluid in the
25 tubing is pumped therethrough.
The intermediate IV tubing segment 34 is
preferably made of vinyl or silicone and has a maximum
Durometer of seventv-five measured on the Shore A
scale. By using a highly pliant, non-stiff resilient
30 flexible tubing segment of this type, it is possible
for the individual fingers 32a of the peristaltic pump
30 to each squeeze off and completely close the tubing
segment during one cycle of its respective cam wheel.
This squeezing off is illustrated in Fig. 5. This
~.
, ,~
20~0370
--7--
ensures a true positive displacement pump in which a
single rotation of the splined shaft 38 will cause a
predetermined volume of fluid to be pumped through the
disposable IV tubing apparatus 14. Having such a
5 non-stiff disposable IV tubing apparatus 14 also
ensures that less torque is required to rotate the
splined shaft 38, thereby resulting in an overall
reduction in energy consumption when the peristaltic
pump is electrically driven from battery power as
10 hereafter described.
The fingers 32a preferably have small teats 32b
(Fig. 5) projecting from the ends thereof. These
teats engage and squeeze the intermediate IV tubing
segment 34. It has been determined that these teats
15 ensure that the tubing segment 34 will be completely
squeezed off during each cycle of each finger. It is
important to understand that the intermediate tubing
segment 34 must have a minimum amount of stiffness and
resilience or else it will not open and close in a
20 manner that will permit it to function as a
peristaltic pump. Preferably the tubing segment 34
has a minimum Durometer of thirty-five measured on the
Shore A scale.
Referring again to Fig. 2, the programmable
25 infusion system 10 includes means for releasably
mounting the intermediate IV tubing segment 34
adjacent the pumping member 32. The intermediate
tubing segment 34 is mounted to a door 40 having a
hook-shaped member 40a (Fig. 3) at one end and a
30 compressible clasp 40b at the other end. The
hook-shaped member 40a of the door may be engaged by
the patient with a shoulder 16a (Fig. 2) located at
one end of the receptacle 16 in the case 12. The
other end of the door is then swung in and the clasp
~ i
~J,,
2~)370
-
--8--
40b snaps into the other end of the receptacle 16.
The intermediate IV tubing segment 34 is squeezed
between the individual fingers 32a and a compressible,
resilient pad 42 (Figs. 3 and 5) supported by the
5 innerside of the door 40. This pad mav be made of
polyurethane foam.
The shaft 38 (Fig. 3) which supports the cam
wheels 36 is journaled at opposite ends in ball
bearings 44. A gear 46 rigidly mounted on one end of
10 the shaft 38 meshes with another gear 48 (Fig. 2)
rigidly mounted on another shaft 50 of a DC motor
module 51 having an internal 141:1 gear reduction. In
other words, one-hundred and forty-one rotations of
the armature of the DC motor turns the shaft 50 one
15 revolution and thus the cam shaft 38 one revolution.
Referring to Fig. 4, the disposable IV tubing
apparatus 14 has a proximal tubing segment 52 and a
distal tubing segment 54 connected to opposite ends of
the intermediate IV tubing segment 34 by means of
20 couplings 56 and 58. These couplings are attached to
the underside of the door 40 as hereafter described in
conjunction with Fig. 9.
Referring to Fig. 4, the intermediate IV tubing
segment 34 preferably has an inside diameter of
25 approximately 0.125 inches. Silicone and VINYL tubing
can be commercially obtained having the desired
stiffness. The proximal segment 52 and the distal
segment 54 are each preferably made of clear polyvinyl
chloride (PVC) having an outside diameter of
30 approximately 0.140 inches and an inside diameter of
approximately .088 inches. This clear PVC tubing is
larger in both inside and outside diameter than the
conventional PVC tubing segments 55a and 55b (Fig. 1)
~,'
W O 91/16933 PCVr/~S91/02930 2o8o~7o
_g_
which connect the segments 52 and 54 to the spike 20
and male lure fitting 26, respectively.
In an actual prototype of our preferred
embodiment, the designed maximum delivery rate is
5 approximately three hundred milliliters per hour. The
minimum desiqned delivery rate is apProximatelv 0.1
milliliters per hour. The delivery resolution is
approximately 0.1 milliliters per hour for .01 through
99.9 milliliters per hour and approximately one
10 milliliter per hour for approximately one hundred to
three hundred milliliters per hour. In the prototype,
the desiqned maximum volume to be infused (VT~I) is
one thousand milliliters and the minimum volume to be
infused (VTBI) is approximately 0.1 milliliters. The
15 designed "keep vein open" (KVO) rate is approximately
one milliliter per hour for one through three hundred
milliliter per hour rates and approximately 0.1
through 0.99 milliliters per hour for 0.1 through 0.99
milliliter per hour rates.
Referring to Fig. 7, the door 40 hac a
rectangular aperture 90 formed adjacent the
hook-shaped member 40a. A resilient metal tubing
squeezer 92 has an upper end 92a which is tightly
received in the aperture 90 in the door 40. The
25 squeezer 92 has a pair of parallel downwardly
extending arms which terminate in coiled sections
92b. Referring to Fig. 8, when the disposable is
assembled, the intermediate segment 34 is squeezed
shut between the coiled sections 92b. When the door
30 40 is installed into the receptacle 16 of the case 12,
the hook-shaped member 40a is engaaed with the
shoulder 16a (Fig. 2). The other end of the door
having the clasp 40b is then swung toward the case.
As this happens, the coiled sections 92b of the tubing
W O 91/16933 PC~r/US91~02930
208037 ~ -ln-
squeezer 92 engage upstandinq projections 94 (Fig. 8)
in the receptacle. These projections are spaced so
that the coiled sections are displaced outwardly awav
from the intermediate tubing segment 34 thereb~7
5 unclamping the same. This is illustrated in phantom
lines in Fig. 8. The squeezér performs a very
important function. Namely, if the disposable IV
tubing apparatus 14 should be inadvertently removed
from the case 12 it will squeeze off the intermediate
10 tubing section 34 and prevent free flow of
intravenous fluid by gravity action.
Referring to Fig. 3, the clasp 40b consists of a
V-shaped element. The door 40 is preferably made of
injection molded plastic and the V-shaped clasp 40b is
15 compressible upon swinging the clasp into the
receptacle 16 in the case. This allows a wedqe-shaped
projection 96 on the clasp 40b to clear and snap into
engagement behind an L-shaped latch 98.
Further details of the disposable IV tubing
20 apparatus 14 are visible in Fig. 9. The coupling 56
is received in a slot in a bracket 102 which extends
from the underside of the exterior wall of the door 40
near the clasp 40b. The coupling 56 is solvent,
welded or bonded to the bracket 102. The couplinq 58
25 is similarly received in another recess formed in the
hook-shaped member 40a. Again the coupling 58 is
solvent bonded to the member 40a. A magnet 106 (Fig.
9) is attached to the bracket 102 and detected by a
Hall effect switch ln8 (Fig. 3) adjacent the
30 receptacle 16. The Hall effect switch is connected to
the micro-controller hereafter described so that the
output thereof will indicate whether or not a
disposable has been correctly loaded into the case 12.
WO 91/16933 PCr/l.'S91/02930
208037D
--11--
The peristaltic pump 30 is a single channel, linear
peristaltic pump having nine reciprocating ingers
32a. The intermediate IV tubing segment 34 is highly
flexible as previously indicated. In a preferred
5 embodiment of the disposable 14, the intermediate IV
tubing segment 34 is made of silicone and has an
inside diameter of approximately 0.125 inches.
Approximately 0.12 milliliters of intravenous fluid
are pumped through the intermediate IV tubing segment
10 34 for each single revolution of pump shaft 38. The
combined motor and gear reduction module 51 also
preferably includes a built-in motor encoder. One
suitable unit is the MICRO-MO Model 1624006S. It uses
TEFLON (Trademark) fixotropic lubricant. A
15 conventional Robert's clamp 23 (Fig. 1) may be
provided on the segment of conventional IV tubing 55b
for closing off the same.
While we have described preferred embodiments of
our improved disposable infusion apparatus and
20 peristaltic pump for use therewith, it should be
understood that modifications and adaptations thereof
will occur to persons skilled in the art. Therefore,
the protection afforded our invention should only be
limited in accordance with the scope of the following
25 claims.