Note: Descriptions are shown in the official language in which they were submitted.
2~9~736
FIELD OF THE INVENTION
This invantion relates to the addition of oxygen
scavengers to aqueous systems to reduce the corrosion of
metals that are in contact with the aqueous system, and
more particularly to the use of certain oxygen scavenging
compositions which comprise hydroxycarboxylic acids and
related compounds.
BACKGROUND OF THE INVENTION
The corrosion of metal surfaces which are in contact
with aqueous systems is a common problem. Corrosion may
occur in various locations including boiler feedwater
lines, heaters, steam lines, process tanks and return
lines. Dissolved oxygen in the aqueous system is often a
principal factor influencing the formation of the
corrosion, particularly where iron and steel are the
materials of construction. The corrosion of iron based
metals in conventional boiler systems is a well known
problem, and controlling the presence of oxygen in boiler
systems, particularly in the Eeedwater section has
received considerable attention. Oxygen removal may be
partially accomplished by either vacuum or thermal
deaeration, or both. Complete removal of oxygen cannot
be effected by these means, however, and further removal
by use of a chemical oxygen scavenging agent, such as
sodium sulfite, has been a customary practice.
Recently, the use of low pressure boilers (i.e.
those operating below about 150 psig) has been
increasingly supplanted by the use of boilers operating
at moderate pressure (i.e. operating between about 150
psig and about 600 psig) and high pressure (i.e.
operating above about 600 psig). As boiler operating
temperatures and pressures have increased, there has been
particular interest in the performance of chemical oxygen
20~0736
scavengers which are effective at: these operating
conditions. The use of sulfites at elevated temperatures
and pressures has been known to result in the formation
of sulfur dioxide and hydrogen sul~ide, which can be a
source of corrosion. Other scavenging agents such as
hydra~ine, hydroquinone, and cert;ain hydroxylamines have
been found to perform satisfactorily in some
circumstances. For example, U.S. Patent No. 4,278,635 to
Kerst discloses the use of various dihydroxy, diamino,
and amino hydroxy ben2enes and their lower alkyl
substituted derivatives, and particularly hydroquinone,
as corrosion control agents in boiler systems. U.~.
Patent No. 4,282,111 to Cuiba also relates to the use of
hydroquinone to reduce the oxygen content of an aqueous
medium. U.S. Patent No. 4,363,734 to Solvinsky discloses
the use of hydroquinone as a catalyst in combination with
dihydroxy acetone. Japanese Patent Publication No. SHO
51-93741 by Suzuki et al reports synergistic inhibition
of metallic corrosion by combinations of
dihydroxybenzenes (e.g. hydroquinone and methyl
hydroquinone) and various carboxylic acids in boiler
water systems. In other circumstances, the efficiency
with which the scavenging proceeds has not been optimal.
There is thus a continuing need for alternative oxygen
scavengers which can be effectively used at elevated
temperatures and pressures.
U.S. Patent No. 2,170,596 to Q~gqle describes
oxygen absorbing solutions using catalysts such as
various quinones together with reducing agents such as
reducing sugars like glucose U.S. Patent No. 1,g88,823
to Winning et al describe rust remover compositions which
comprise ammonium salt, an alkali salt of a water soluble
organic acid, sugar, and an inhibitor of corrosion such
as pyrogallol, hydroquinone, pyridine or quinoline.
209~736
--4--
Gluconic acid and its salts have been used for
corrosion control. U.S. Patent No. 2,529,178 to Nieland
et al discloses the use of gluconates such as sodium
gluconate to protect metals, particularly ferrous metals,
from corrosion in water systems. This patent describes
the formation of a protective layer on the metal surface
when certain amounts of the gluconate are added to the
water in the system.
U.S. Patent No. 3,589,859 to Foroulis discloses that
gluconate salts, particularly when used in combination
with a benzoate or a salicylate salt, inhibits the
oxidative corrosion in aerated cooling systems. U.S.
Patent No. 3,711,246 to Foroulis is directad to
inhibiting oxidative corrosion in an oxygen-containing
cooling water system using certain inorganic silicate
salts together with certain gluconate salts, optionally
with certain polyphosphates.
DETAILED DESCRIPTION
The present invention is directed to a method for
inhibiting or preventing corrosion of metal surfaces
which are in contact with aqueous systems containing
dissolved oxygen and more particularly to the use of
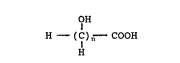
corrosion inhibiting oxygen scavengers comprising water-
soluble hydroxycarboxylic acids having the general
formula:
OH
H--( C ) n COOH
H
wherein n is either 5 or 6, the various lactone
derivatives of these acids and the water soluble salts of
said acids and lactones.
7 3 ~
--5--
The preferred oxygen scaven~ers of this invention
are gluconic acid and glucoheptonic acid or their water
soluble salts.
The method of this invention comprises adding to an
aqueous system which contains dissolved oxygen at least
one compound having the above general formula in an
amount effective to remove or control the presence of the
oxygen in the system.
The oxygen scavengers of this invention are suitable
for use in low, moderate or high pressure boiler systems,
and may also be utilized in other systems where aqueous
solutions containing dissolved air and/or oxygen are in
contact with metal surfaces. The precise dosage used in
each system may vary somewhat depending on the oxygen
content of the water as well as other characteristics of
the water system. In general, the dosages range from
0.001 ppm to 500 ppm, and are preferably in the range of
from 0.01 ppm to 100 ppm.
The oxygen scavengers of this invention may be used
as the sole oxygen scavenger for treating the aqueous
system, or other conventional oxygen scavengers such as
diethyl hydroxylamine, hydrazine, and the like, may be
used in combination therewith. In addition, the oxygen
scavengers of this invention may be used in combination
with other conventional water treating agents including,
but not limited to pH regulators, biocides, dispersants,
chelants, corrosion inhibitors, scale inhibitors,
polymeric agents, and the like, and mixtures thereof.
Without further elaboration, it is believed that one
of ordinary skill in the art, using the preceding
detailed description, can utili2e the present invention
to its fullest extsnt.
The following exarip-^~ are provided to illustrate
the invention in accorc - with the principles of this
2~90~3~
~j
invention, but are not to be construed as limiting the
invention in any way except as indicated in the appended
claims. All parts and percentages are by weight unless
otherwise indicated.
Example :L
This example demonstrates the oxygen scavenging
activity of the hydroxycarboxylic acids of this invention
under boiler sonditions at ~00 psig and 448F. During
the test, the boiler feedwater was saturated with
dissolved oxygen by continuous aeration. The
concentration of dissolved oxygen in the ~eedwater ranged
from 8.5 ppm to 9.5 ppm as 2- The boiler steam was
condensed through a heat exchanger producing a condensate
temperature of 55F. The condensate was then passed
through a chamber in which an oxygen probe was inserted
to monitor the concentration of dissolved oxygen. A
blank run (i.e. without an oxygen scavenger) was first
conducted until a constant oxygen reading was obtained.
once the initial dissolved oxygen reading had been
established, the oxygen scavenger being evaluated was fed
into the boiler. The reduction o~ the dissolved oxygen
in the condensate was then recorded. The boiler
feedwater contained 10 ppm total hardness (as CaC03).
Table 1 illustrates the extent of oxygen removal in the
stream condensate 30 minutes after the addition of the
oxygen scavenger. Sodium sulfite and hydrazine were
included in the evaluation for comparison purposes.
2~907~
--7--
~ ~ _____ ~
X o ~D ~ ~9 ~D ~r a: I ~
0~ ~1 a ~n co a~ ~D 1-'~
I t~
â ~ ~ _
~ .,., ~ ~ ~ ~, ~ ~, ~ I
~ ~ O O O O O O ¦ X
0~ __
C~ I
I
.~ .~ l` r~ I~ ~ o~ ~ I
~n ~ .~ ~ ~ ~ ~ ~ ~ I
U~ H ¦ la
. I
_ _ I
~a) Oo ~ o o o o o~
__ _ _ ~
~X~
~ ~ h rl
~ ~ ~ ~ ~Q)
~ ~ ~ ~ ~ q:~ ~
~A O O U ;:'
1~ ~ :~ ~ ~ a3
c) ~ ~ ~ ~ ~ ~1 u~
u~ u~ ~ ~ ~ ~ V ~1 ~
N N ~ ~ E~ U~ ,1
.~ S~ ~ rl .,1 .,
Q~
_ O O _ _ O O O
.,
In O