Note: Descriptions are shown in the official language in which they were submitted.
~
, WO 96/09267 217 6 6 ~ 5 p~~S95/12060
1
PROCESS FOR PRODUCING IMPROVED
SULFUR-COATED UREA SLOW RELEASE FERTILIZERS
liAC.KGWOUNO OP TNVENTT_ON
s This invention is directed to an improved sulfur-coated
urea fertilizer. More particularly, the invention is directed
to an improved process for manufacturing sulfur-coated
controlled release fertilizers by coating sulfur-coated urea
with liquid monomers and polymerizing the monomers on the urea
io to provide a uniform coating having, ~n~ ~, improved
impact resistance. The specific monomers are diethylene
glycol-triethanolamine polyols and a diisocyanate such as 4,4-
diphenylmethane diisocyanate.
BAGKGROL1T1D OF THE INVENTT_ON
15 Sulfur-coated urea (SCU) slow release fertilizers
are known. In the customary process for the production of
sulfur-coated urea, granular urea of nominal size range, 1.7-
2.9mm, which has been preheated in a fluid bed heater to 160°F
to 180°F, is introduced into the front end of a rotating
2o horizontal cylindrical drum, nominally 12 feet in length and 5
1/2 feet in diameter. Lifting flights, or longitudinal
ledges, which are fastened to the inside wall of the drum and
evenly spaced around its circumference, lift and cascade the
urea granules as the drum rotates. As the cascading granules
25 pass through the drum, molten (290°F) sulfur is sprayed onto
the urea granules from a series of nozzles uniformly
positioned within the length of the drum. When a droplet of
molten sulfur contacts a granule, it quickly solidifies; and a
continuous coating of sulfur is formed on a urea granule when
3o a sufficient number of molten sulfur droplets have made
contact with it. In this randomized coating process the
granules are coated to an average target thickness of 40
microns (~) or about 13%-14% by weight sulfur-coating on the
urea. However, various random proportions of the granules,
35 which are discharging from the drum, have thin (<30;c), medium
(30~c-50~) and thick (>50~) sulfur-coating thickness.
Because of the inherent brittleness of the
crystalline solid sulfur-coating which forms on the granule,
SUBSTITUTE SHEET (RULE 26)
~
~ . WO 96/09267 217 6 6 0 5 p~~S95/12060
'- 2
and the thin, or even noncontinuous coating on many of the
granules, it is customary that some type of secondary outer
coating or sealant be spray applied onto the sulfur-coating
surface. Usually this is done in a second horizontal rotating
s drum in series with the sulfur-coating drum. Historically,
this sealant has been either a polymeric hydrocarbon,
petroleum-based wax, or a combination of high viscosity
polymeric paraffinic oil plus polyethylene, which is spray
applied as a hot melt liquid onto the hot, but solid sulfur-
to coating surface. Since the sealant melt will not solidify at
the 160°-180°F temperature of the sulfur-coated urea granules
onto which it is applied, the liquid sealant distributes
relatively uniformly onto all sulfur-coated granules by
flowing, or transferring, from one granule to the next as they
is cascade through the rotating secondary sealant coating drum.
These sealant coated sulfur-coated urea granules pass through
a fluid bed cooler, after they are discharged from the sealant
drum, wherein the sealant solidifies to a firm, but somewhat
tacky polymeric coating.
2o The characteristic tackiness, which is common to
these types of sealants, is offset by the application of a
powder material, often referred to as a conditioner, such as
talc, clay, silica, or diatomaceous earth. This conditioner
application usually is applied to the cooled, polymeric coated
25 sulfur-coated urea (PCSCU) in a third rotary drum in series
with the sulfur-coating and sealant coating drums. Products
of the above type are described in United States Patent Nos.
3,295,950; 3,342,577; 3,877,415; 3,903,333; 3,991,225 and
5,219,465.
3o Although these sulfur-coated urea products generally
have good slow release characteristics as manufactured, the
impact and abrasion resistance of the coating is low.
Mechanical degradation of the coating occurs as a consequence
of the handling, transportation, blending and application
35 operations, which results in significant deterioration of slow
release properties. Therefore, improved products still are
desired.
SUBSTfTUTE SHEET (RULE 26)
WO 96/09267 217 6 6 0 5 P~~S95/12060
'' 3
SUMMARY OF INVENTION
The present invention provides a process for
producing sulfur-coated slow release fertilizers which have
improved properties. According to the process of this
invention, the hot melt polymeric liquid sealant of the
customary process is replaced by specific liquid monomers,
which, when applied sequentially onto the surface of the hot
sulfur-coated urea granules, copolymerize to form a firm,
tack-free, water insoluble polymer coating sealant. This
io tack-free sealant process eliminates the need for a
conditioner application. The liquid monomers are
diisocyanates, such as MDI (4,4-diphenylmethane diisocyanate),
and a polyol mixture of DEG (diethylene glycol) and TEA
(triethanolamine). The TEA serves as both a reactive polyol
and a catalyst.
It was discovered that, surprisingly, the DEG-TEA
polyol when copolymerized with a diisocyanate gave
significantly better results than the customary hot melt
polymeric sealants, or other diisocyanate-polyol reaction
2o polymers, such as described in U.S. Patent No. 5,219465
(Goertz et al), or lower molecular weight EG (ethylene
glycol)-TEA polyol mixtures. The DEG:TEA mixture of polyols,
which are premixed at ratios from 2.5:1.0 to 4.5:1.0, and
preferably at a 3.5:1.0 ratio, are pumped from a hold tank
2s through separate pumps to separate fixed application nozzle
positions spaced uniformly through the length of the sealant
drum. The number of nozzle positions is determined by the
urea throughput rate and the percentage of reaction polymer
sealant to be applied to the SCU. The MDI is pumped from a
3o hold tank through separate pumps to fixed nozzle positions
located next to each polyol nozzle position. Usually one
nozzle set position is needed for up to each one percent of
polymer applied to the sulfur-coated urea granule. The amount
of liquid polyol(P) and MDI(M) which is pumped to each nozzle
35 set position is in a stoichiometric ratio of (P):(M) which is
consistent with optimum reaction polymerization to effect the
polymer sealant characteristics that result in a tack-free,
SUBSTITUTE SHEET (RULE 26)
WO 96109267 217 6 6 0 5 p~~S95/12060
'-- 4
durable, impact resistant surface. This (P):(M) ratio falls
within the range of 0.33:1 to 0.45:1 and preferably is 0.39:1.
It was discovered that the DEG polyol alone, or
mixtures of the DEG polyol, at relatively high concentrations,
s with other higher equivalent weight polyols with each
containing the stipulated TEA, gave significantly better
quality performance than the customary hctt melt polymeric
sealants, or other MDI-polyol reaction polymers, such as
described in the aforesaid Goertz '465 patent. Such
io comparisons are made at essentially equal sealant weight
percentage and sulfur coating weight percentage, commonly 2%
sealant and 13% sulfur coating on similarly sized granules.
THE DRAWTNCS ND DETAT D D CRTPTT(1N OF TNVENTT(~N
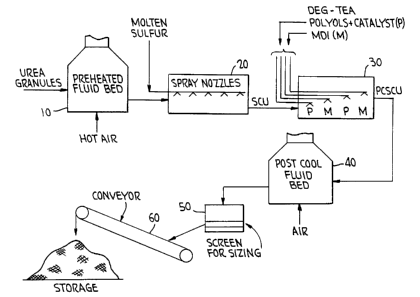
A flow diagram of the process of the present
~s invention is shown in the drawing. Referring to the drawing,
urea granules having a nominal size range of 2.3mm in a range
of 1.7 to 2.9mm are fed into a fluidized bed preheater 10
where the urea granules are heated with hot air. The
preheated urea granules, at a temperature in the range of 160°
2o to 180°F, then are fed into a rotating drum 20 where they are
spray coated by a series of spray nozzles which are spraying
molten sulfur at a temperature of approximately 280-310°F,
which is above sulfur solidification temperature of 235°F.
The sulfur-coated urea granules then are transferred from drum
2s 20 into a second rotating drum 30 containing a plurality of
fixed nozzle set positions, which simultaneously coat the SCU
granules with separately applied diethylene glycol-
triethanolamine polyol and diisocyanate, such as MDI. The
liquid DEG-TEA polyol mixture and the liquid diisocyanate
3o intermix react and, or polymerize, on the surface of the
sulfur-coated urea granules cascading within drum 30. After
polymerization the now polymer coated sulfur-coated urea
granules, at a temperature of about 170°F, are fed into a
fluid bed cooler 40 and are cooled with cool air to a
3s temperature in the range of 95°F to 115°F. After cooling, the
granules are fed through a screen 50 for sizing, and after
being sized, are transferred by conveyor 60 to storage.
SUBSTITUTE SHEET (RULE 26)
WO 96/09267 217 6 6 0 ~ p~~S95/12060
'-' 5
Preferred compositions of the reaction polymer
coated SCU (PCSCU) products for the most common size granule
used in commercial fertilizer blends for turfgrass
applications, which is designated "regular size,~~ nominally
s 2.4mm in a size range from l.7mm to 3.Omm, are given below:
TA8L8 1
Percentage
A B
1o Granular Urea (substrate) 85.000 85.000
Sulfur (coating) 12.800 12.800
Polyols(P) (reactant)
a.l DEG (low E.W.) .394 .197
a.2 Polyester polyol --
.421
1s (high E.W.)
b. TEA .112 .102
c. Pigment (optional) .030 .030
MDI(M) (reactant) 1.364 1.150
Plasticizer (optional) .300 .30o
2o Total PCSCU Product 100.000 100.000
As shown in Table 1, most preferred formulation A contains as
the polyol a mixture of DEG and TEA, whereas, formulation B
has part of the polyol mixture of formula A replaced with a
high equivalent weight polyester polyol containing an
zs appropriate level of TEA.
The ratio of polyol mixture (P) to the diisocyanate
(M) and the ratio of DEG, or DEG plus polyester polyol, to the
TEA catalyst polyol are shown in Table 2.
TAHhE 2
30 $~,;
a. ~ (reaction 0.393 0.652
M stoichiometry) 1.000 1.000
b. ~yols (mixtures) 3-55
35 TEA 1 1
The improved impact resistance of the PCSCU product
of this invention is measured by decreased percentage release
of urea in: 1) a 2-hour 20°C water immersion test of the
4o PCSCU granules, which have been subjected to a 25 foot drop
impact test, and 2) thereafter continuing the water immersion
SUBSTITUTE SHEET (RULE 26)
~
~ _ WO 96/09267 ~ 5 PCTIUS95/12060
6
test described in 1) for a period of seven days at 30°C. In
the following table this quality performance characteristic of
the preferred formulation is compared with other process
formulations of essentially the same sulfur and sealant weight
s percentage compositions:
TABh$ 3
Full Bcale Plant Production Products
Sulfur and Sealant at 13% and 2%
io Sealant-Type Cumulative % Urea Released in
Wa I m ai nn ~A t~r~r ~ ft Dz'O~)
2 hrs at 7 Days at
20°C 30°C
15 MDI & DEG/TEA
Reaction Polymer 6 3g
Polymeric Wax
(with conditioner) 24 61
Solvent Dispersed
Polymer 28 66
As apparent from Table 3, the sealant of the present
invention provides a substantial improvement over either a
2s polymeric wax sealant or a solvent dispersed sealant. It is
believed that this improved impact resistance is, apparently,
because the uniquely small molecular size and shape of the DEG
and TEA molecules, coupled with their high functionality, when
reacted in the aforementioned balance with high functionality
3o MDI, produce a highly crosslinked, rigid polyurethane sealant
coating with high impact strength and surface hardness
properties. Particularly, the TEA, a three-functional, amine
based self-catalytic polyol, when properly balanced with the
DEG, yields this extraordinary result when polymerized with
35 the MDI.
Further, the polymerization reaction among the
polyol and 1~I monomers ostensibly delays sufficiently to
facilitate the flow of these unreacted liquids over the
sulfur-coated granules. This flow of liquids onto all the
4o granules cascading in the rotating drum provides the intimate
SUBSTITUTE SHEET (RULE 26)
WO 96/09267 217 b b 0 5 p~~S95/12060
7
contact and mixing among monomer reactants, which is necessary
for the designed stoichiometric polymerization to occur that
results in an impact resistant polymer sealant. Moreover,
this reaction delay allows the liquids to spread to a uniform
s polymer sealant thickness among all the granules, irrespective
of the thickness of the sulfur coating on any granule; and,
therefore, all sulfur-coated granules have a protective
polymer sealant on their surface.
~It will be apparent to one skilled in the art that
1o various modifications can be made to the sulfur-coated urea
fertilizers within the scope of the aforesaid description.
Such modifications being within the ability of one skilled in
the art form a part of the present invention and are embraced
by the appended claims.
SUBSTITUTE SHEET (RULE 26)