Note: Descriptions are shown in the official language in which they were submitted.
2195619
"CARDIAC VALVULAR SUPPORT PROSTHESIS"
The present invention relates to a prosthesis
intended for the surgical correction of cardiac valvular
diseases and, more particularly, for the annuloplasty of
mitral and tricuspid valves.
Mitral and tricuspid valves are present at the
left and right atrio-ventricular junctions, respectively,
of the human heart. These valves open and close in
response to pressure gradient during each cardiac cycle of
relaxation and contraction. Their function is to prevent
the blood from flowing into atria from ventricles.
These valves consist of leaflets, an annulus
from which the leaflets stem and a complex consisting of
chordea and papillary muscles. The papillary muscles
originate from ventricular muscle mass and are attached to
the free margins of the leaflets through chordea. The size
of the leaflets is such that when the heart contracts, the
resulting increased blood pressure within the ventricle
cavity pushes the leaflets towards the atrial cavity.
During this process, the free margins of the leaflets come
in apposition to each other and close the atrial-
ventricular passage. The chordea and papillary musclecomplex holds them in this position throughout the state
of increased intraventricular pressure and prevents the
leaflets from bulging into and opening in the atrial
cavity. One of the conditions in which the mitral or
tricuspid valve can become functionally incompetent is
when the annulus become dilated, generally as a result of
acquired and/or degenerative diseases and disorders. Due
to the increased diameter of the annulus, the tips of the
valve cusps fail to meet each other during systolic
contraction. This non-closure of the valve allows the
219~619
-
blood to enter into atria from ventricles and renders them
incompetent.
There have been mainly two different approaches
in annuloplasty, i.e., re-modeling and narrowing of the
annulus. In remodeling annuloplasty, after excising the
excess tissue, a rigid metallic ring of appropriate size
is implanted in the annulus which restores the natural
shape of the annulus and reduces its diameter to a level
where the leaflets length becomes adequate enough to close
the valve. Although this technique has been applied
successfully for the last so many years, it has many
pitfalls. Unlike the natural annulus, the ring is rigid
and does not decrease in diameter during systolic
contractions or increase in diameter during diastolic
expansions. Because of its rigid nature, during systole,
the ring bulges into the left ventricular outflow tract,
causing a systolic anterior motion (SAM), a well
recognized and documented complication and thus giving
rise to obstruction to the blood flow. The rigid ring does
not allow the annulus to respond to the heamodynamic and
functional changes produced within the heart under
different physiological and pathological conditions. The
annulus is unable to contract and dilate. Consequently,
the sutures undergo stress and there is increased risk of
ring dehiscence.
In narrowing annuloplasty, a flexible purse-
string type of assembly is implanted at the annulus. The
annulus is made narrow by adjusting the length of the
string. In this type of repair, the natural shape and
configuration of the valve is lost leading to curling of
the leaflets and resulting in less than perfect repair and
valve function. Since the string used is non-stretchable,
the annulus does not dilate in high cardiac output states.
The SAM problem is not seen in this type of repair
2195619
provided excess tissue on posterior mitral cusp is
excised.
Recently, a semi-rigid annuloplasty ring has
been introduced. This ring is made of alternating strips
of metal and plastic and covered with fabric. This
configuration allows a certain amount of flexibility in
antero-posterior direction. The natural shape of the valve
is restored but the annulus is still unable to adjust to
high output blood flow conditions and there is no increase
in size as the age of the patient progresses.
It is therefore an object of the present
invention to overcome the above drawbacks and to provide
an improved support prosthesis which enables the annulus
to dilate in response to heamodynamic and functional
changes, increased blood flow and pressure, without
affecting the operativeness of the leaflets.
In accordance with the present invention, there
is provided a support prosthesis for a natural human heart
valve having an annulus of generally oval configuration
with a major axis and a minor axis and at least two
leaflets stemming from said annulus and each moving along
a naturally pre-ordained path during systolic contraction
or diastolic expansion. The support prosthesis of the
invention consists of an oblong, annular flexible member
of a size and shape to fit against the annulus, the member
having a longitudinal axis and being made of a
biocompatible material exhibiting elasticity only along
the longitudinal axis so as to permit dilatation of the
annulus along the major axis thereof, in response to
heamodynamic and functional changes, while preventing
dilatation of the annulus along the minor axis thereof so
2195619
that the path along which each leaflet travels remains
unaltered.
According to a preferred embodiment, the
biocompatible material is a fibrous biocompatible material
having fibers oriented in a manner to provide the
aforesaid elasticity. Preferably, such a fibrous
biocompatible material facilitates growth of endothelial
cells so that the member becomes embedded in endothelium,
thereby preventing clot formation; possible dehiscence of
the member is completely eliminated. An example of a
suitable material exhibiting these properties is a
modified form of polytetrafluoroethylene sold under the
trademark GORE-TEX. Suture stitches can pass through such
a material so that no track or recesses are left behind,
which can become sites for harboring infectious agents.
This material also allows one to produce a seamless and
jointless member, by either stamping or by simply cutting
the member out in the desired shapes and sizes. The shapes
are different for mitral and tricuspid valves. The member
is made in different sizes in order to meet the clinical
requirements in different individuals requiring different
cardiac output.
Since the member is made of a single material,
there is no possibility of material wear as seen with
other support prosthesis made of several materials and
having coverings.
According to another preferred embodiment, the
member is substantially flat and has two opposite planar
surfaces, thereby occupying minim~l intracardiac space and
volume. At least one of the surfaces is provided with
orientation markers allowing the surgeon to orient the
member while it is being placed in position.
-- 4
- 21gS519
Because of its pliable and flexible nature, the
support prosthesis of the inventlon does not produce a
systolic anterior motion of the mitral or tricuspid valve
and it conforms to the seat of implantation and adjoining
structures; in particular, the support prosthesis of the
invention adapts to the shape of the aortic root and
allows it to expand freely in response to heamodynamic and
functional changes within the aorta. Complete flexibility
allows natural contractibility of the annulus during
systole and eliminates stress on the sutures and
dehiscence of the member. The support prosthesis of the
invention does not interfer with the normal dynamic motion
of the mitral and tricuspid annulus during systole
contraction or diastolic expansion. The use of such a
support prosthesis prevents dilatation of the annulus
along its minor axis and thereby prevents non-closure of
the mitral and tricuspid valves during systolic
contraction. During diastolic expansion, the annulus can
dilate along its major axis in response to the
heamodynamic and functional changes in different
physiological and pathological states, while adequate
support is provided by the prosthesis to the annulus. The
support prosthesis according to the invention restores and
retains the physiological size and shape of the annulus
without rendering it stiff.
Further features and advantages of the invention
will become more readily apparent from the following
description of preferred embodiments as illustrated by way
of example in the accompanying drawings, in which:
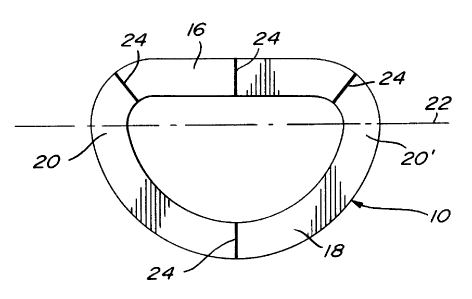
Fig. 1 is a top plan view of a support
prosthesis for a mitral valve, according to a preferred
embodiment of the invention;
Fig. 2 is an elevational view thereof;
- ~195613
Fig. 3 is a schematic sectional view of a
natural human heart illustrating the mitral valve fitted
with the support prosthesis of Fig. 1 shown in broken
lines;
Fig. 4 is a top plan view of a support
prosthesis for a tricuspid valve, according to a preferred
embodiment of the invention;
Fig. 5 is an elevational view thereof; and
Fig. 6 is a schematic sectional view of a
natural human heart illustrating the tricuspid valve
fitted with the support prosthesis of Fig. 4 shown in
broken lines.
The mitral prosthesis illustrated in Figs 1-3
consists of a substantially flat, oblong, annular member
10 having two opposite planar surfaces 12 and 14. The
member 10 which is seamless and jointless comprises a
rectilinear segment 16, a curved segment 18 and two end
portions 20,20'. It is made of a fibrous biocompatible
material which is flexible and has fibers oriented in a
manner such as to provide elasticity only along the
longitudinal axis 22. Due to such an elasticity, the end
portions 20,20' are extensible along the axis 22 whereas
the segments 16 and 18 remain in a fixed position relative
to another. Both surfaces 12 and 14 are provided with
orientation markers 24 allowing the surgeon to orient the
member 10 while it is being placed in position against the
annulus of a mitral valve.
Fig. 3 illustrates the left atrium 25 (only the
bottom shown) of a normal human heart 26, which is
separated from the right ventricle 27 by the ventricular
_ 2195619
septum 28. Disposed at the bottom of the left atrium 25 is
the mitral valve 30 which comprises an annulus 32 of
generally oval configuration with a major axis 34 and a
minor axis 36, anterior and posterior leaflets 38,40
stemming from the annulus 32 and papillary muscles 42
attached to the free margins of the leaflets 38,40 through
chordea 44. Also shown are the aortic cusps 46. The mitral
prosthesis 10 has a size and shape to fit against the
annulus 32. As shown, the rectilinear segment 16 of the
prosthesis extends along a major portion of the annulus 32
from which stems the anterior leaflets 38, the curved
segment 18 extending along a major portion of the annulus
32 from which stems the posterior leaflet 40. The mitral
prosthesis 10 is secured to the annulus 32 by either
continuous mattress suturing or by interrupted sutures,
depending upon the discretion of the surgeon.
Since the end portions 20,20' of the mitral
prosthesis 10 are extensible along the longitudinal axis
22 coincident with the major axis 34 of the annulus 32,
the prosthesis 10 permits the annulus 32 to dilate along
its major axis 34 in response to the increased blood flow
and pressure in different physiological and pathological
states, thereby enabling the valve opening 48 to increase
in length so as to accommodate such an increased blood
flow. On the other hand, since the segments 16 and 18 of
the mitral prosthesis 10 remain in a fixed position
relative to one another, the prosthesis 10 prevents the
annulus 32 from dilating along its minor axis 36 so that
prevents the paths along which the leaflets 38,40 travel
remain unaltered. The full operativeness of the anterior
leaflet 38 and posterior leaflet 40 is thus retained
during increased blood flow and pressure.
35The tricuspid prosthesis illustrated in Figs 4-6
also consists of a substantially flat, annular member 50
2195619
-
having two opposite planar surfaces 52 and 54. The member
50 which is seamless and jointless comprises a rectilinear
segment 56, a slightly curved segment 58 and two end
portions 60,60'. Similarly to member 10, the member 50 is
made of a fibrous biocompatible material which is flexible
and has fibers oriented in a manner such as to provide
elasticity only along the longitudinal axis 62. Due to
such an elasticity, the end portions 60,60l are extensible
along the axis 62 whereas the segments 56 and 58 remain in
a fixed position relative to one another. Both surfaces 52
and 54 are provided with orientation markers 64 allowing
the surgeon to orient the member 50 while it is being
placed in position against the annulus of a tricuspid
valve.
Fig. 4 illustrates the base of the ventricular
part of the heart 26 with the atria and great vessels
removed. Reference numerals 66, 68 and 70 designate the
right atrium (only the bottom shown), aorta and right
ventricle, respectively. Disposed at the bottom of the
right atrium 66 is the tricuspid valve 72 which comprises
an annulus 74 of generally oval configuration with a major
axis 76 and a minor axis 78, and anterior, posterior and
septal leaflets 80,82,84 stemming from the annulus 74; the
papillary muscles and chordea which attach the papillary
muscles to the free margins of the leaflets 80,82 and 84
are not shown. The tricuspid prosthesis 50 has a size and
shape to fit against the annulus 74. As shown, the
rectilinear segment 56 of the prosthesis extends along a
major portion of the annulus 74 from which stem the
posterior and septal leaflets 82,84, the curved segment 58
extending along a major portion of the annulus 74 from
which stems the anterior leaflet 80. The tricuspid
prosthesis is secured to the annulus 74 by either
continuous mattress suturing or by interrupted sutures,
depending upon the discretion of the surgeon.
2195619
Since the end portions 60,60' of the tricuspid
prosthesis 50 are extensible along the longitudinal axis
62 coincident with the major axis 76 of the annulus 74,
the prosthesis 50 permits the annulus 74 to dilate along
its major axis 76 in response to the increased blood flow
and pressure in different physiological and pathological
states, thereby enabling the valve opening 86 to increase
in length so as to accommodate such an increased blood
flow. On the other hand, since the segments 56 and 58 of
the tricuspid prosthesis 50 remain in a fixed position
relation to one another, the prosthesis 50 prevents the
annulus 74 from dilating along its minor axis 78 and so
that the paths along which the leaflets 80,82,84 travel
remain unaltered. The full operativeness of the anterior
leaflet 80, posterior leaflet 82 and septal leaflet 84 is
thus retained during increased blood flow and pressure.