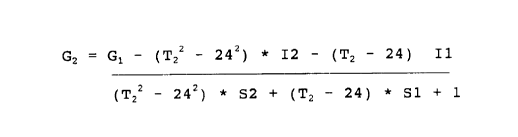Note: Descriptions are shown in the official language in which they were submitted.
CA 02236314 1998-04-29
METHOD AND APPARATUS FOR CORRECTING
AMBIENT TEMPERATURE EFFECT IN BIOSENSORS
Field of the Invention
The present invention relates to a biosensor, and, more
particularly, to a new and improved method and apparatus for
correcting ambient temperature effect in biosensors.
Description of the Prior Art
The quantitative determination of analytes in body fluids
is of great importance in the diagnoses and maintenance of
certain physiological abnormalities. For example lactate,
cholesterol and bilirubin should be monitored in certain indi-
viduals. In particular, the determination of glucose in body
fluids is of great importance to diabetic individuals who must
frequently check the level of glucose in their body fluids as
a means of regulating the glucose intake in their diets.
While the remainder of the disclosure herein will be directed
towards the determination of glucose, it is to be understood
that the procedure and apparatus of this invention can be used
for the determination of other analytes upon selection of the
appropriate enzyme. The ideal diagnostic device for the de-
tection of glucose in fluids must be simple, so as not to re-
quire a high degree of technical skill on the part of the
technician administering the test. In many cases, these tests
are administered by the patient which lends further emphasis
to the need for a test which is easy to carry out. Addition-
ally, such a device should be based upon elements which are
sufficiently stable to meet situations of prolonged storage.
CA 02236314 1998-04-29
2
Methods for determining analyte concentration in fluids
can be based on the electrochemical reaction between an enzyme
and the analyte specific to the enzyme and a mediator which
maintains the enzyme in its initial oxidation state. Suitable
redox erAZymes include oxidases, dehydrogenases, catalase and
peroxidase. For example, in the case where glucose is the
analyte, the reaction with glucose oxidase and oxygen is rep-
resented by equation (A).
gluCOSe t ~2 glucose oxidase (~o)) gluCOIlOlaCtOne + $
(A)
In a colorimetric assay, the released hydrogen peroxide,
in the presence of a peroxidase, causes a color change in a
redox indicator which color change is proportional to the
level of glucose in the test fluid. While colorimetric tests
can be made semi-quantitative by use of color charts for com-
parison of the color change of the redox indicator with the
color change obtained using test fluids of known glucose con-
centration, and can be rendered more highly quantitative by
reading the result with a spectrophotometric instrument, the
results are generally not. as accurate nor are they obtained as
quickly as those obtained using an electrochemical biosensor.
As used herein, the term biosensor is intended to refer to an
analytical device that responds selectively to analytes in an
appropriate sample and converts their concentration into an
electrical signal via a combination of a biological recogni-
tion signal and a physico-chemical transducer. Aside from its
greater accuracy, a biosensor is an instrument which generates
CA 02236314 1998-04-29
3
an electrical signal directly thereby facilitating a simpli-
fied design. Furthermore, a biosensor offers the advantage of
low material cost since a thin layer of chemicals is deposited
on the electrodes and little material is wasted.
H202 >O2 + 2H+ + 2e
(B)
The electron flow is then converted to the electrical signal
which directly correlates to the glucose concentration.
In the initial step of the reaction represented by equa-
tion {A), glucose present in the test sample converts the oxi-
dized fl.avin adenine dinucleotide (FAD) center of the enzyme
into its reduced form, (FADHZ). Because these redox centers
are essentially electrically insulated within the enzyme mole-
cule, direct electron transfer to the surface of a conven-
tional Electrode does nat occur to any measurable degree in
the absence of an unacceptably high overvoltage. An improve-
ment to this system involves the use of a nonphysiological re-
dox coupling between the electrode and the enzyme to shuttle
electrons between the {FADHZ) and the electrode. This is rep-
resented. by the following scheme in which the redox coupler,
typically referred to as a mediator, is represented by M:
Glucose + GO(FAD) --> gluconolactone + GO(FADHz)
GO ( FADH2 ) + 2MoX > GO ( FAD ) + 2Mred + 2H+
2Mr,~d > 2MoX + 2e ( at the electrode )
CA 02236314 1998-04-29
4
In this scheme, GO(FAD) represents the oxidized form of
glucose oxidase and GO(FADHZ) indicates its reduced form. The
mediating species Mred shuttles electrons from the reduced en-
zyme to the electrode thereby oxidizing the enzyme causing its
regeneration in situ which, of course, is desirable for rea-
sons of economy. The main purpose for using a mediator is to
reduce the working potential of the sensor. An ideal mediator
would be re-oxidized at the electrode at a low potential under
which impurity in the chemical layer and interfering sub-
stances in the sample would not be oxidized thereby minimizing
interference.
Many compounds are useful as mediators due to their abil-
ity to accept electrons from the reduced enzyme and transfer
them to the electrode. Among the mediators known to be useful
as electron transfer agents in analytical determinations are
the substituted benzo- a.nd naphthoquinones disclosed in U.S.
Patent 4,746,607; the N-oxides, nitroso compounds, hydroxy-
lamines and oxines specifically disclosed in EP 0 354 441; the
flavins, phenazines, phenothiazines, indophenols, substituted
1,4-benzoquinones and indamins disclosed in EP 0 330 517 and
the phenazinium/phenoxaz.inium salts described in U.S. Patent
3,791,988. A comprehensive review of electrochemical media-
tors of biological redox systems can be found in Analytica
Clinica Acta. 140 (1982), Pp 1-18.
Among the more venerable mediators is hexacyanoferrate,
also known as ferricyanide, which is discussed by Schlapfer et
al in Clinical Chimica Acta., 57 (1974), Pp. 283-289. In U.S.
Patent 9,929,545 there is disclosed the use of a soluble fer-
ricyanid.e compound in combination with a soluble ferric com-
CA 02236314 1998-04-29
pound in a composition for enzymatically determining an ana-
lyte in a sample. Substituting the iron salt of ferricyanide
for oxygen in equation (A) provides:
Glucose + 2 Fe+++ ( CN ) 3 6 G°-> gluconolactone + 2 Fe++ ( CN ) 4
6
since the ferricyanide is reduced to ferrocyanide by its ac-
ceptance of electrons from the glucose oxidase enzyme.
Another way of expressing this reaction is by use of the
following equation (C):
Glucose + GO(FAD) --> Gluconolactone + GO(FADHZ)
GO ( FADHz ) + 2 FE ( CN3 ) 3 6 --> GO ( FAD ) + 2 FE ( CN ) 64 + 2H+
2 FE(CN)6' > 2 FE(CN)63 + 2e (at the electrode)
(C)
The electrons released are directly proportional to the amount
of glucose in the test fluid and can be related thereto by
measurement of the current which is produced upon the applica-
tion of a potential thereto. Oxidation of the ferrocyanide at
the anode renews the cycle.
Summary of the Invention
Important objects of: the present invention are to provide
a new and improved method and apparatus for correcting ambient
temperature effect in biosensors; to provide such method and
apparatus that eliminates or minimizes the ambient temperature
effect i.n analyte concentration value identified by a biosen-
CA 02236314 1998-04-29
6
sor; and to provide such method and apparatus that overcome
many of the disadvantages of prior art arrangements.
In brief, a method and apparatus are provided for cor-
recting ambient temperature effect in biosensors. An ambient
temperature value is measured. A sample is applied to the
biosensors, then a currE~nt generated in the test sample is
measured. An observed analyte concentration value is calcu-
lated from the current through a standard response curve. The
observed analyte concentration is then modified utilizing the
measured ambient temperature value to thereby increase the ac-
curacy of the analyte determination.
In accordance with a feature of the invention, the ana-
lyte concentration value is calculated by solving the follow-
ing equation:
G2 .- G1 - (T22 - 242) * I2 - (T2 - 24) * I1
(Tz2 - 242) * S2 + (Tz - 24) * S1 + 1
where G1 is said observed analyte concentration value, Tz is
said measured ambient temperature value and I1, I2, S1, and S2
are predetermined parameters.
Brief Description of the Drawing
The present invention together with the above and other
objects and advantages may best be understood from the follow-
ing detailed description of the preferred embodiments of the
invention illustrated in the drawings, wherein:
CA 02236314 1998-04-29
7
FIG. 1 is a block diagram representation of biosensor in
accordance with the present invention;
FIG. 2 is a flow chart illustrating logical steps per-
formed in accordance with the present invention of the method
correcting ambient temperature effect in biosensors by the
biosensor of FIG. 1.
Detailed Description of the Preferred Embodiments
Having reference now to the drawings, in FIG. 1 there is
shown a block diagram representation of biosensor system des-
ignated as a whole by the reference character 100 and arranged
in accordance with principles of the present invention. Bio-
sensor system 100 includes a microprocessor 102 together with
an associated memory 104 for storing program and user data. A
meter function 106 coupled to biosensor 108 is operatively
controlled by the microprocessor 102 for recording test val-
ues, such as blood glucose test values. An ON/OFF input at a
line 110 responsive to the user ON/OFF input operation is cou-
pled to the microprocessor 102 for performing the blood test
sequence mode of biosensor system 100. A system features in-
put at a line 112 responsive to a user input operation is cou-
pled to the microprocessor 102 for selectively performing the
system features mode of biosensor 100. A signal input indi-
cated at a line 120 is coupled to the microprocessor 102 pro-
viding temperature information from a thermistor 122 in accor-
dance with the invention. Microprocessor 102 contains suit-
able programming to perform the methods of the invention as
illustrated in FIG. 2.
CA 02236314 1998-04-29
8
A display 150 is coupled to the microprocessor 102 for
displaying information to the user including test results. A
battery monitor function 160 is coupled to the microprocessor
102 for detecting a low or dead battery condition. An alarm
function 162 is coupled to the microprocessor 102 for detect-
ing predefined system conditions and for generating alarm in-
dications for the user of biosensor system 100. A data port
or communications interface 164 couples data to and from a
connected computer (not shown).
In accordance with ithe invention, to reduce the tempera-
ture bias, biosensor system 100 performs a temperature correc-
tion method of the preferred embodiment.
Referring to FIG. 2, logical steps performed in accor-
dance with the method for correcting ambient temperature ef-
fect in biosensors 108 by the biosensor processor 102 begin at
block 200. First ambient. temperature is measured as indicated
at a block 202 labeled MEASURE INSTRUMENT TEMPERATURE T2.
Then sensor current is measured as indicated at a block 204.
Next they measured current value is converted into an analyte
concentration value, such as glucose concentration value
(observed concentration),, as indicated at a block 206. Then
correction for temperature effect is performed in a final glu-
cose concentration calculation as indicated at a block 208.
The temperature corrected glucose concentration is calculated
utilizing the following equation:
G2 :- G1 - (T22 - 242) * I2 - (Tz - 24) * I1
(T22 - 24z) * S2 + (Tz - 24) * S1 + 1
CA 02236314 1998-04-29
9
where G1 is said observed analyte concentration value, Tz is
said measured ambient temperature value and I1, I2, S1, and S2
are predetermined paramet:ers . This completes the sequence as
indicated at a block 210.
Amperometric biosen~~ors 108 are known to be sensitive to
temperature. This temperature effect occurs because diffusion
of the rnediator to the working electrode is temperature de-
pendent. Diffusion typically induces a temperature effect of
1 - 2~ bias per degree centigrade. Therefore temperatures as
low as ll0°C would produce results with a bias of about -25~
and temperatures as high as 40°C would produce results with a
bias about +25~. The system 100 instrument provides results
between 0 to 50°C. The only available temperature measurement
comes from a thermistor inside the instrument. In order to
reduce the temperature bias it was necessary to develop a tem-
perature correction algorithm,
The temperature effect was determined experimentally by
biosensor system 100 whole blood glucose assay over the entire
glucose {50 to 600 mg/dL) and temperature range (10 to 40°C)
expected to be encountered. Actual blood glucose readings and
sample temperatures were measured. This was done for six dif-
ferent sensor 108 lots. When the " compound interest " tem-
perature correction method was used, several lots had percent
biases of -10~ to -13~ at the extreme temperatures. The for-
mula for the " compound interest " correction method is:
Equation 1
Gz = G1 * ( 1 + tc:/ 100 ) zs-T
CA 02236314 1998-04-29
l0
where G1 is the observed glucose concentration, tc is the tem-
perature coefficient determined experimentally and T is the
sample temperature.
The " compound interest " algorithm did not work well be-
cause the temperature coefficient, tc, changed with glucose
concentration. A " polynomial" correction algorithm was in-
vented to handle the varying temperature coefficient problem.
By using a polynomial correction algorithm, the percent bias
was limited to within +/- 10~ . The equation for the polyno-
mial correction method is described in Equation #2. The grand
sum of t:he absolute bias for both methods indicated that the
polynomial correction method had less overall bias. Also, at
the very extreme temperatures of 2 and 49°C, the polynomial
correction method had lower bias (below 13.50 where as the
compound interest method was as high as -25~.
Therefore, the polynomial correction method provided an
improvement over the " compound interest " correction method.
After running the glucose assay at different temperatures
the current response at each temperature was calculated
through the 24°C (sample temperature) standard response curve
to obtain the observed glucose concentration.
The observed glucose concentration and the sample tem-
perature were then used to calculate the corrected glucose
concentration using the following equation:
CA 02236314 1998-04-29
:L 1
Equation 2
GZ ._ G1 _ ( TZZ - 242 ) * I2 - ( T2 - 24 ) * I 1
(T22 - 24z) * S2 + (T2 - 24) * S1 + 1
where G1 is the observed glucose concentration, Tz is the sam-
ple temperature and I1, I2, S1, and S2 are the predetermined
coefficients. These coefficients were determined experimen-
tally. See the following exemplary procedure for details.
Table 1 shows an example of the temperature correction
results. Tz is the sample temperature. GR is the reference
glucose valve. I is the measured current. G1 is the observed
glucose concentration (without temperature correction). ~B is
the percent bias without: temperature correction. G2 is the
temperature corrected glucose concentration. ~B~ is the per-
cent bias after temperature correction.
The data shows the percent bias before and after the cor-
rection algorithm was applied. The algorithm and coefficients
were able to reduce the percent bias at the extreme tempera-
tures of 10 to 40°C to within +/-7~.
CA 02236314 1998-04-29
:12
EXAMPLE
Table 1: Temperature Correction for Lot C
I1 0.17706
I2 -0.0086
S1 0.01529
S2 0.00004
Lot C
Tz GR I G1 ~B Gz $B~
8.7 50 1024 38.3 -23.4 49.1 -1.8~
8.7 100 1484 78.6 -21.4 102.9 2.9~
8.7 200 2404 159.1 -20.5 210.6 5.3$
8.7 400 4243 320.1 -20.0 426.0 6.5~
8.7 600 6082 481.2 -19.8 641.4 6.9$
16.7 50 1109 45.7 -8.6~ 50.6 1.3~
16.7 100 1608 89.4 -10.6 100.4 0.4~
16.7 200 2606 176.8 -11.6 199.9 0.0~
16.7 400 4602 351.6 -12.1 398.9 -0.3$
16.7 600 6598 526.4 -12.3 597.9 -0.3$
23.9 50 1158 50.0 0.0~ 50.0 0.0~
23.9 100 1729 100.0 0.0$ 100.0 0.0~
23.9 200 2871 200.0 0.0~ 200.0 0.0$
23.9 400 5155 400.0 ~ 0.0~ 400.0 0.0~
23.9 600 7439 600.0 0.0~ 600.0 0.0~
30.6 50 1212 54.7 9.5~ 50.8 1.5~
30.6 100 1851 110.6 10.6 100.8 0.8~
30.6 200 3128 222.5 11.2 200.9 0.5~
30.6 400 5682 446.1 11.5 401.1 0.3~
30.6 600 8236 669.8 11.6 601.3 0.2~
38.2 50 1251 58.1 16.2 50.4 0.8$
38.2 100 2008 124.4 24.4 103.3 3.3~
38.2 200 3522 257.0 28.5 209.0 4.5~
38.2 400 6550 522.1 30.5 420.4 5.1~
38.2 600 9578 787.3 31.2 631.8 5.3~
The following describes an exemplary procedure used for
determining the temperature correction coefficients (I1, Iz.
S1, Sz i:n Equation 2). First venous heparinized whole blood
('45~ hematocrit) from a single donor was spiked close to dif-
ferent glucose concentrations (values determined by the Yellow
CA 02236314 1998-04-29
13
Springs Instrument, YSI, reference method and corrected for
any known sample interferences) and tested in system 100 in-
struments at different environmental chamber temperatures
(Table J., e.g. samples of 50 and 400 mg/dL glucose at 8.7,
16.7, 23.9, 30.6 and 38.2°CX.) The Yellow Springs Instrument
and method are described by Conrad et al., in the February
1989 " Journal of Pediatrics" Pages 281-287 and by Burmeister
et al., in " Analytical. Letters", 28(4), 581-592 (1995).
High relative humidity (65 to 85~) was maintained in the cham-
ber in order to prevent evaporative cooling, and the sample
was equilibrated to the chamber temperature; this way the tem-
perature effect would result only from the chemistry. The ac-
tual sample temperature was measured for each glucose spike.
To determine the sample temperature, a 0.0005" thermocouple
was inserted into a sensor without chemistry, and temperature
data was collected every second after the blood was added to
the sensor.
Table 2: Lot C Actual YS~I Glucose and Current Response
Sample
Temp. YSI Current Slope Intercept
8.7°C 54.2 1063
8.7°C 412.5 4358 9.20 564.6
16.7°C 54.9 1148
16.7°C 414.9 4750 9.98 610.2
23.9°C 55.7 1223
23.9°C 418 5359 11.42 587.1
30.6°C 49.3 1203
30.6°C 408.4 5787 12.77 573.7
38.2°C 51.6 1275
38.2°C 418.7 6833 15.14 493.8
CA 02236314 1998-04-29
14
Next, the current response at exactly 50, 100, 200, 400,
and 600 mg/dL glucose for each temperature was determined
through the curves using the slope and intercepts determined
in Table 2. Using these calculated current values the ob-
served cllucose concentration was determined through the 24°C
curve as provided in Table 3.
Table 3: Lot C - Current. Through the YSI 50 and 400 mg/dL
Curves and the Observed Glucose mg/dL Through the
24°C Curve
YS~I 23.9C Curve
Sample Reference Observed
Temperature C Glucose mg/dL Current Glucose mg/dL
8.7 50 1024 38.3
8.7 100 1484 78.6
8.7 2f0 2404 159.1
8.7 400 4243 320.1
8.7 600 6082 481.2
16.7 50 1109 45.7
16.7 100 1608 89.4
16.7 200 2606 176.8
16.7 400 4602 351.6
16.7 600 6598 526.4
23.9 50 1158 50.0
23.9 100 1729 100.0
23.9 200 2871 200.0
23.9 4G0 5155 400.0
23.9 6CI0 7439 600.0
30.6 ~i0 1212 57.7
30.6 1C10 1851 110.6
30.6 2C10 3128 222.5
30.6 4C10 5682 446.1
30.6 600 8236 669.8
38.2 'i0 1251 58.1
38.2 1C)0 2008 124.4
38.2 2C)0 3522 257.0
38.2 400 6550 522.1
38.2 600 9578 787.3
CA 02236314 1998-04-29
Next for each spike of blood, the observed glucose con-
centration (G1} was plotted against the sample temperature
(T2). The 2nd order polynomial curve was used to fit the plot
and the al and a2 constants for that level of glucose were ob-
tained as provided in Table 4. For example, a computer pro-
gram such as Slidewrite by Advanced Graphics Software Inc., or
any other equivalent curve fitting program can be used.
Table 4: Lot C - 2nd Order Polynomial Coefficients
50 100 200 400 600
Coefficient mg/dL mg/d1, mq/dL mg/dL mg/dL
a0 29.689 68.654 146.318 301.709 457.305
al 1.08071 1.06138 1.04494 1.00696 0.95187
a2 -0.00881 0.01035 0.04829 0.12417 0.20045
Corr.Coef.R 0.9990 1.000 0.9998 0.9996 0.9995
The al values obtained for the different levels of glu-
cose were plotted against. the glucose concentration. The data
was plotted using a linear fit, and the coefficients S1 (slope
of the linear fit) and I1 (intercept of the linear fit) were
generated. The Slidewrii:e program on a PC by Advanced Graph-
ics Software Inc., or any other equivalent curve fitting pro-
gram can be used.
The a2 values obtained for the different levels of glu-
cose were also plotted against the glucose concentration. The
data was. plotted using a linear fit, and the coefficients S2
(slope of the linear fit) and I2 (intercept of the linear fit)
were generated.
CA 02236314 2003-O1-08
is
To derive the algorithm: at each level of glucose, the
observed glucose concentration (G1) is related to the sample
temperature (T2) in a 2nd order polynomial relationship.
Or Equation 3 G1 = (T2z)*a2 + Tz*ai + a0
And at a sample temperature of 24°C, GZ (Corrected) - Gl
(Observed)
Or Equation 4 G2 = (24z)*a2 + 24*ai + a0
Subtracting equation (4) from equation (3) gives:
Equation 5 G1 - GZ = (Ta2 - 242) *a2 + (Ta - 24) *a1
From the linear plots generated at steps 4 and 5:
Equation 6 al - S1*GZ + I1
and
Equation 7 a2 = S2*GZ + I2
Combining equation (5), (6), and (7) gives equation (2).
While the present invention has,been described with ref-
erence to the details of the embodiments of the invention
shown in the drawing, these details are not intended to limit
the scope of the invention as claimed in the appended claims.