Note: Descriptions are shown in the official language in which they were submitted.
- . CA 02240353 1998-06-11
DEVICE FOR COLLECTING A BLOOD SAMPLE
FROM A PLASTIC SEGMENT TUBE
BACKGROUND OF THE INVENTION
1. Field of the Invention. The present invention relates
generally to the field of devices for collecting blood samples. More
specifically, the present invention discloses a device for safely
piercing a plastic segment tube to release a blood sample into a
receptacle for subsequent testing.
2. Statement of the Problem. Donated blood is widely used for
transfusions to assist patients suffering trauma and during surgery. A
soft plastic bag called a blood collection bag is used for gathering
- blood from the donor. The blood collection bag is connected to a
flexible plastic tube and a needle at the distal end of the plastic tube is
penetrated into the donor's vein. Blood flows through the needle and
tube into the blood collection bag. After the desired quantity of blood
has been collected in the blood collection bag, the needle is
withdrawn and the tube is heat sealed into a series of segments
containing the donor's blood.
Prior to transfusion, each unit of blood must be tested to
ensure that it is compatible with the patient's blood type. This is
CA 02240353 1998-06-11
W~ 97/33149 PCT1US97103602
-2-
commonly referred to as a "type and cross-match" procedure. In
addition, donated blood is often tested for the presence of infectious
agents, such as hepatitis viruses and HIV. However, blood samples
cannot be obtained directly from the blood collection bag, because of
potential contamination of the blood that may occur from contact with
a syringe or pipette used to withdraw a sample.
As a result of this problem, the conventional approach has
been to heat seal a number of short segments of the plastic tube
leading from the donor's arm to the blood collection bag. These
sealed tube segments are commonly referred to as segment tubes,
pigtails, or segments. The segment tubes remain attached to the
blood collection bag, and are often folded into a group held together
with a rubber band. Blood is typically tested shortly after it has been
donated, and again immediately before transfusion. in both cases,
the laboratory technician simply removes one of the segment tubes
attached to the blood collection bag for testing. The customary
technique is to use a pair of surgical scissors to cut the segment tube
in half at the junction between the sedimented red blood cells and
plasma in the blood sample within the segment tube. The section of
.20 the segment tube containing the red blood cells is then squeezed to
force cells into a test tube for subsequent testing.
This current technique has a number of shortcoming and
potential hazards. The segment tube may be under internal pressure,
which can cause blood to spray outward when the segment tube is
cut. ~ This can expose the technician and work surfaces in the
laboratory to potential blood contamination. The scissors also
become contaminated with blood, and could cause transmission of
blood-borne infectious disease to health care workers, particularly if
the technician experiences an injury from sharp edges associated with
CA 02240353 1998-06-11
-3-
the scissors. The scissors are often reused without cleaning or
sterilization after cutting through a segment tube. This further
increases the dissemination of blood-borne microorganisms to work
surfaces and drawers where scissors are stored after use. The
surface of the donor blood bag can also become contaminated with
blood by laying the bag on contaminated work surfaces, or by
technicians touching the bag with blood-contaminated gloves or
hands. The blood-contaminated blood bag might then contaminate
other hospital environments, such as operating rooms and patient
areas. Again, this could potentially increase nosocomial and health
care worker infection rates from blood contamination (e.g.,
staphylococcal, streptococcal, hepatitis B and C infections). Finally,
failure to clean the scissors between samples could cause
subsequent blood samples to be contaminated with trace amounts of
blood from preceding samples. This can lead to inaccurate cross-
matching, with subsequent safety concerns for patients requiring
transfusions. Furthermore, this problem could unnecessarily increase
the time and cost for cross-matching and delay transfusion of blood to
patients in life-threatening emergencies.
A number of devices have been invented in the past for
,~_..;:
~.r~.:~
piercing segment tubes, including the following:
Inventor Patent No. Issue Date
Staebfer et al. 5,254,312 Oct. 19,_ 1993
McMorrow 4,176,451 Dec. 4, 1979
Minase et al. EPO Publ. 0350792 Jan. 17, 1990
"Introducing the SEG-SAFET"" Segment Processor", Alpha Scientific
Corp., Southeastern, PA (1995)
CA 02240353 2005-O1-18
WO 9T/33149 pCT~s9T~~~
-4-
"Directions for Using SegmentSampierT""," Gamma Biologicals, Inc.,
Houston, TX (Nov. 1994).
Staebler et al. disclose a device for collecting a blood sample
5 from a segment tube. The main body of the device has a cup like
portion that is inserted into a test tube. The user then inserts a
segment tube into the cup like portion of the device and exerts a
downward farce to enable a piercing element (i.e., a blade or tance) to
puncture the segment tube, thereby allowing blood to flow from the
10 segment tube into the test tube. This device is marketed by
.,
innovative Laboratory Acrylics, Inc., of Brighton, Michigan, under the
name "LL.A. Safety Segment Slitter."
McMorrow discloses a segment tube cutter with a tapered
lower end 8 that is inserted into the test tube 6. A sharp spur 10 cuts
15 the segment tube 11 as it is inserted into the device.
Minase et al. disclose another example of a device for piercing
segment tubes. The tubular portion 2 of the device is inserted into a
test tube. A cutting edge or needle at the bottom of the tubular
portion pierces the segment tube as it is inserted. A hole 7 allows
20 blood to drain from the segment tube into the test tube.
The literature distributed by Alpha Scientific Corp. shows a
temporary receptacle for processing segment tubes that includes a
needle to puncture the segment tube.
The "SegmentSampler" device marketed by Gamma
25 Blologicals, Inc., is generally similar to that disclosed by Minase et al.
However, the lower tubular portion of the device is tapered to
accommodate a range of test tube diameters.
The prior art devices fail to address many of the technical and
30 safety issues associated with obtaining a blood sample from a
*Trademark
. CA 02240353 1998-06-11
-5-
segment tube. An idea( blood sampling device should address the
following concerns:
(a) The type and cross-match procedure is commonly pertormed
using any of several different test tubes diameters. It is important that
the device be able to accommodate different test tube diameters. In
particular, the device should not exert forces on the neck of the test
tube as the segment tube is punctured that might cause the test tube
to break.
(b) There are no accepted industry standards for the diameter and
thickness of the plastic tubing leading to the blood collection bag.
Therefore, the device should be able to accommodate different
segment tube diameters.
(c) Segment tubes are heat sealed using at least three different
heat-sealing devices that result in different shapes and thicknesses of
the heat-sealed ends of segment tubes. These variations are further
complicated by the fact that the sealed ends tend to have a major
dimension larger than the diameter of the remainder of the segment
tube. A device with a cylindrical opening to receive the segment tube
will fend not to provide a particularly good fit, and may not adequately
guide and the support the segment tube. The device should be able
to accommodate sealed ends having a wide range of dimensions
without exerting radial forces on the test tube.
(d) The segment tube should not be allowed to fold or buckle as it
is inserted into the device.
(e) The device should not have an opening that restricts insertion
of the segment tube to a particular orientation to accommodate the
flat sealed end of the segment tube.
(f) The device should minimize contact between the user's fingers
and the glass test tube.
CA 02240353 1998-06-11
WO 97/33149 PCT/US97/03602 ' '
-6-
(g) The device should prevent contact between the user's fingers
and the puncturing element within the device.
(h) After the segment tube has been punctured, the user should
not have direct contact with the punctured end of the segment tube to
minimize blood splatter and contamination. The device should retain
the punctured segment tube so that both can be discarded together.
(i) Considerable downward force may be necessary to puncture
the segment tube. The device should provide sufficient structural
support to maintain proper orientation for the puncturing element, and
to prevent the puncturing element from bending or being dislodged.
Q) If adhesive is used to bond the needle to the device, the
adhesive should not be permitted to plug the needle and thereby
interfere with drainage of bload from the segment tube through the
needle into the test tube.
(k) It is also important to minimize the dispersal of any blood
remaining in the device after the segment tube and device have been
discarded. Blood tends to remain within the needle and droplets of
blood accumulate at the bottom of the device. These droplets of
blood can easily become dislodged when the device is discarded and
contaminate the surrounding environment.
Thus, the "SegmentSampler" device marketed by Gamma
Biologicals, Inc:, has a number of shortcomings when compared
against the above list of desired features. In particular, the tapered
side walls of the SegmentSampler device create radial pressure if
used with smaller test tubes (e.g., 10 mm and 12 mm) that can cause
the test tube to break when a relatively small downward force is
exerted on the device. Also, the SegmentSampler device is not well
suited to receive segment tubes having a wide range of diameters
CA 02240353 1998-06-11
-7-
and shapes. Wider segment tubes and those with larger sealed ends
create an interference fit that can exert radial pressure on the wall of
the test tube and break the test tube when the user pushes downward
on the segment tube. This device also provides little structural
support for the needle. Hence, the segment tube can bend the
needle sideways, preventing puncture of the segment tube. The
segment tube could also buckle or fold upon itself without being
punctured.
The device disclosed by Staebler et al. has many of the same
shortcomings. In addition, this device uses a solid lancet to puncture
the segment tube that also plugs the opening in the segment tube,
and thus interFeres with the flow of blood into the test tube. Also, the
device requires that the flat end of the segment tube be inserted at a
predetermined orientation to allow the lancet to pierce the wall of the
segment tube.
3. Solution to the P~ot~lem. None of the prior art references
uncovered in the search show a device having the structure of the
present invention. In particular, the present device has a port for
..20 receiving the end of the segment tube that includes a plurality of
~~»y T . ~ ,
tapered ribs arranged in a radial pattern with slots interspersed
between each adjacent pair of ribs. This configuration allows the
device to handle a wide range of segment tube diameters and a wide
variance in the dimensions of sealed ends. The ribs guide and .
support the tubular portion of the segment tube so that it does not fold
or buckle, thereby enabling the segment tube to present onto the
puncturing element. Multiple slots allow the sealed end of the
segment tube to be inserted in any orientation. The ribs also help to
CA 02240353 1998-06-11
WO 97/33149 PCT/US97/03602
_$_
retain the segment tube after it has been punctured so that the device
and segment tube can be discarded together.
The segment tube is punctured by the needle above the level
of the test tube, and therefore never enters the test tube. As a result,
no radial forces are exerted on the test tube as the segment tube is
inserted into the device.
An annular recess in the bottom of the device accommodates a
wide range of test tube diameters without creating radial stresses that
might break the test tube. The annular recess contacts only the top
rim of the test tube and only a downward force is exerted on the rim of
the test tube when a segment tube is inserted into the device. The
lower portion of the device housing serves as a protective skirt
covering the rim of the test tube to protect the user's fingers if the test
tube breaks.
In addition, the needle is held firmly in place by a horizontal
divider within the device and a series of radial ribs within the lower
portion of the device. This additional structural support minimizes
deflection of the needle when the segment tube is inserted. The
lower ribs increase capillary attraction of blood that may remain at the
bottom of the device after the segment tube has been punctured, so
that blood droplets are less likely to contaminate the surrounding
environment after the test tube is removed and the device is
discarded.
- . CA 02240353 1998-06-11
-9-
SUMMARY OF THE INVENTION
This invention provides a device for collecting a blood sample
into a receptacle from a plastic segment tube, A cylindrical housing
contains a hollow needle that punctures the segment tube as it is
inserted into the upper port of the device. A series of ribs with
tapered media( edges are arranged in a radial pattern around the
needle within the upper port to guide and support the segment tube
as it is inserted. The ribs are separated by slots that also guide the
seated end of the segment tube. An annular recess around the lower
port of the device holds the rim of the receptacle and allows blood
released by the punctured segment tube to drain into the receptacle.
The annular recess accommodates a wide range of test tube
diameters, and exerts only a downward force on the rim of the
receptacle when a segment tube is inserted into the upper port of the
device.
A primary object of the present invention is to provide a device
for collecting a blood sample from a segment tube that can
accommodate a wide range of segment tube sizes, segment tube end
:: shapes, and test tube diameters.
Another object of the present invention is to provide a device
for collecting a blood sample from a segment tube that does not exert
radial forces on the test tube that might cause the test tube to break.
Another object of the present invention is to provide a device
for collecting a blood sample from a segment tube that guides and
supports the segment tube as it is inserted to prevent the segment
tube from folding or buckling.
CA 02240353 1998-06-11
WO 97/33149 PC'i'/1JS97/03602
-10-
Another object of the present invention is to provide a device
for collecting a blood sample from a segment tube that includes as a
protective skirt covering the rim of the test tube to protect the user's
fingers in case the test tube breaks.
Yet another object of the present invention is to provide a
device for collecting a blood sample from a segment tube that
includes sufficient structural support to prevent the needle from being
deflected by the segment tube.
These and other advantages, features, and objects of the
present invention will be more readily understood in view of the
following detailed description and the drawings.
CA 02240353 1998-06-11
_11 _
t3RIEF DESCRIPTION OF THE DRAWINGS
The present invention can be more readily understood in
conjunction with the accompanying drawings, in which:
FIG. 1 is a top perspective view of the present device 10.
FIG. 2 is a top view of the device 10.
FIG. 3 is a bottom perspective view of the device 10.
FIG. 4 is a bottom view of the device 10.
F1G. 5 is a side cross-sectional view of the device 10.
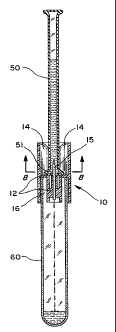
FIG. 6 is an exploded side elevational view of a segment tube
50, the device 10, and a test tube 60.
FIG. 7 is a side cross-sectional view of the device 10 on a test
tube 60 after a segment tube 50 has been inserted info the device 10.
FiG. 8 is a cross-sectional view of the device 10 and segment
tube 50 corresponding to FIG. 7 taken through a horizontal plane
extending through the needle 15 of the device 10 and the lower end of
the segment tube 50.
.."'.7
CA 02240353 1998-06-11
dV0 97/33149 PC'T/L1S97/03602 ~ '
-12-
DETAILED DESCRIPTION C3F THE INVENTION
Turning to FIG. 1, a top perspective view is shown of the entire
device 10. A corresponding top view is illustrated in F1G. 2. The
device 10 has a generally cylindrical housing 11 having an upper port
and a lower port. A bottom perspective view is provided in FIG. 3 and
a corresponding bottom view is provided in FIG. 4 showing the lower
port of the device 10. FIG. 5 is a side cross-sectional view of the
entire device 10. The housing 11 includes a series of vertical grooves
19 to provide a better grip for the user's fingers.
As illustrated in FIG. 6, the lower port of the device 10 is first
placed over a test tube 60 (or other receptacle) intended to receive
the blood sample. A segment tube 50 is then inserted into the upper
port of the device. A series of ribs 14 are arranged in a radial pattern
about a hollow needle 15 within the upper portion of the housing 11.
' The ribs 14 have tapered medial edges surrounding the needle 15
that define an unobstructed passageway leading downward from the
upper port to the needle 15. This vertical passageway has relatively
large cross-sectional dimensions at the upper port that progressively
reduce to smaller cross-sectional dimensions adjacent to the needle
15. In the preferred embodiment, the passageway is a tapered
vertical column having a generally circular cross-section with an
effective diameter adjacent to the needle 15 that results in a friction fit
with the tubular portion of the segment tube 50. Thus, the medial
edges of the ribs 14 serve to guide and support the tubular portion of
the segment tube 50 as it is inserted info the upper port of the device
10 and punctured by the needle 15. The ribs 14 also help to prevent
- CA 02240353 1998-06-11
-13-
the segment tube 50 from foiding or buckling, and help
to prevent
accidental contact by the user with the sharp point of
the needle 15.
Slots or spaces 13 between each pair of adjacent ribs
14 catch,
align, and guide the sealed end 51 of the segment tube
50 as it is
inserted so that the segment tube 50 is punctured by
the needle 15.
)n the preferred embodiment, the slots 13 are radiatly
arranged in
diametrically opposed pairs, so that the sealed end 51
of the segment
tube 50 can be inserted in any orientation about the
vertical axis and
yet engage one of the pair of slots 13, as shown in FiG.
8. In
addition, the ribs 14 and slots 13 guide the segment
tube 50 into a
vertical position if it is initially inserted at a tilt.
A floor or divider 12 separates the upper port of the
device 10
from the lower port. The base of the hollow needle 15
is held by and
extends upward through the divider 12, thereby providing
a
passageway to allow blood to drain from the punctured
segment tube
50 through the lower port of the device and into the
receptacle 60.
The sharp upper point of the needle 15 remains shielded
within the
housing 11 to prevent accidental contact by the user
with the point of
the needle 15. A sleeve 18 extends upward from the divider
12 to
_ 20 support the lower portion of the needle 15 and thereby
prevent
-' bending or buckling, as shown in FIG. 5. It should also
be expressly
understood that other means could be substituted for
puncturing the
segment tube 50. Far example, a solid needle, sharp spur,
or blade
could be used with a separate conduit through the divider
12 to allow
. 25 blood to drain into the receptacle 60.
The lower port includes an annular recess 16 that receives
the
rim 61 of the test tube 60. The width of this annular
recess 16 can be
made quite substantial to accommodate a wide range of
test tube
diameters. The lower portion of the cylindrical housing
11 serves as a
CA 02240353 1998-06-11
WO 97/33149 i'CT/US97/03602
-14-
skirt covering the upper portion of the test tube. This provides support
to prevent the device 10 from accidentally flipping or sliding off the
test tube 60. The lower portion of the housing 11 also helps to protect
the user's fingers and hand from sharp edges in the event the test
tube 60 breaks. It should be expressly understood that other means
could be used to temporarily mount the device 10 on the test tube rim
61. For example, a circular recess or mechanical fasteners could be
employed to attach the device 10 to a test tube 60.
The base of the needle 15 is surrounded by a series of lower
ribs 17 arranged in a radial pattern on the underside of the divider 12.
The exposed surface area of the lower ribs 17 adjacent to the base of
the needle 15 provides capillary attraction for any remaining droplets
of blood after the test tube 60 is removed, and thereby reduces the
risk of contamination to the surrounding area. Furthermore, the lower
ribs 17 protrude below the base of the needle 15, as shown in FIG. 3,
and prevent the user's hand or fingers from accidentally coming into
contact with the base of the needle 15.
In the preferred embodiment, the needle 15 extends upward
from the center of the divider 12 along the vertical axis of the housing
11. The annular recess 16 is also centered about this common
vertical axis. As the segment tube 50 is inserted into the upper port of
the device 10, the slots 13 guide and support the sealed end 51 of the
segment tube 50 so that it is punctured by the needle 15. Axial
alignment of the upper port, needle 15, and annular recess 16 ,
ensures that only downward forces of any significant magnitude are
exerted on the rim 61 of the test tube 60. It should also be noted that
the segment tube 50 is punctured by the needle 15 above the level of
the test tube 60, as shown in F1G. 7. The segment tube 50 never
enters the test tube 60. As a result, no radial forces are exerted on
CA 02240353 1998-06-11
-15-
the test tube 60 as the segment tube 50 is inserted into the device 10.
This feature allows a wide range of test tube diameters to be used
without concern of whether the segment tube 50 (or its sealed end 51 )
will fit into the test tube 60.
After the segment tube 50 has been punctured, blood drains
from the segment tube 50 through the hollow needle 15 into the
receptacle 60, as shown in FIG. 7. The device 10 is then removed
from the receptacle 60, and the device 10 and segment tube 50 are
discarded together. As previously mentioned, the medial edges of the
ribs 14 create a friction fit with the tubular portion of the segment tube
50. The needle 15 also tends to retain the punctured segment tube
i
50. These frictional forces help to keep the device 10 and segment
tube 50 together when they are discarded, and thereby minimize
contamination of the surrounding area.
The above disclosure sets forth a number of embodiments of
the present invention. Other arrangements or embodiments, not
precisely set forth, could be, practiced under the teachings of the
present invention and as set forth in the following claims.