Note: Descriptions are shown in the official language in which they were submitted.
CA 02248328 1998-09-22
FELD OF INVENTION
The present invention relates generally to steerable catheters and catheter procedures involving
functional devices, such as laser delivery devices and drug delivery devices. More particularly, the
invention relates to a steerable catheter and method of use, particularly adapted for laser-assisted
percutaneous transluminal revascularization (PTMR). The distal tip of the catheter for guiding a laser
delivery device, drug delivery device or other functional device extendable there through, is deflectable
CA 02248328 1998-09-22
.
in at least one given plane. The invention includes an automatic catheter tip alignment system for
maintaining constant relative positioning between the distal tip of the functional device and the distal tip
of the steerable catheter. The invention also includes a surface contact detection system to detect contact
with an interior body surface, such as the heart wall.
BACKGROUND OF INVENTION
In the treatment of heart disease, one method of improving myocardial blood supply is called
transmyocardial revascularization (TMR), the creation of channels in the myocardium of the heart. The
procedure using needles in a form of surgical "myocardial acupuncture" has been used clinically since the
1960s. Deckelbaum. L.I., Cardiovascular Applications of Laser Technology, Lasers in Surgery and
Medicine 15:315-341 (1994). The technique relieves ischemia by allowing blood to pass from the
ventricle through the channels either directly into other vessels communicating with the channels or into
myocardial sinusoids which connect to the myocardial microcirculation.
In the reptilian heart, perfusion occurs via communicating channels between the left ventricle and
the coronary arteries. Frazier, O.H., Myocardial Revascularization with Laser- Preliminary Findings,
Circulation, 1995; 92 [suppl II]:II-58-II-65. There is evidence of these communicating channels in the
developing human embryo. In the human heart, myocardial microanatomy involves the presence of
myocardial sinusoids. These sinusoidal communications vary in size and structure, but represent a
network of direct arterial-luminal, arterial-arterial, arterial-venous, and venous-luminal connections. This
vascular mesh forms an important source of myocardial blood supply in reptiles but its role in humans is
poorly understood.
Numerous surgical TMR studies have been performed, including early studies using needles to
perform myocardial acupuncture, or boring, to mechanically displace and/or remove tissue. Such studies
have involved surgically exposing the heart and sequentially inserting needles to form a number of
channels through the epicardium, myocardium, and endocardium to allow blood from the ventricle to
CA 02248328 1998-09-22
perfuse the channels. The early studies using needles showed that the newly created channels were
subject to acute thrombosis followed by organization and fibrosis of clots resulting in channel closure.
Interest in TMR using needles waned with the knowledge that such channels did not remain open.
However, interest in TMR procedures has recurred with the advent of medical lasers used to create TMR
channels. Histological evidence of patent, endothelium-lined tracts within laser-created channels shows
that the lumen of laser channels can become hemocompatible and resists occlusion. A thin zone of
charring occurs on the periphery of the laser-created channels through the well-known thermal effects of
optical radiation on cardiovascular tissue. Additionally, recent histological evidence shows probable new
vessel formation adjacent collagen occluded transmyocardial channels, thereby suggesting benefits from
TMR with or without the formation of channels which remain patent.
Surgical TMR procedures using laser energy have been described. U.S. Patent No.4,658,817
issued Apr. 21, 1987 to Hardy teaches a method and apparatus for surgical TMR using a CO2 laser
connected to an articulated arm having a handpiece attached thereto. The handpiece emits laser energy
from a single aperture and is moved around the surface of the heart to create the desired number of
channels. U.S. Patent No. 5,380,316 issued Jan. 10, 1995 to Aita et al. purports to teach the use of a
flexible lasing apparatus which is inserted into the open chest cavity in a surgical procedure. A lens at the
distal end of the flexible apparatus is used to focus laser energy, and the apparatus is moved about the
surface of the heart to create the desired number of channels.
The foregoing discussion relates to surgical procedures, i.e. procedures which access the heart
surgically, either via open heart surgery, or perhaps by minimally invasive surgical (MIS) methods if the
design and size of the distal ends of the hand pieces are suitable for use in an MIS site. However, since
TMR most often involves creating channels through the epidocardium into the lower left chamber of the
heart, it is desirable to create TMR channels in a percutaneous procedure, i.e. by extending a catheter
apparatus through the vasculature into the ventricle and creating the channels through endocardial
surfaces and into myocardium. Performing percutaneous TMR (PTMR) is desirable for a number of
CA 02248328 1998-09-22
reasons. Percutaneous catheter procedures are typically less traumatic to the patient compared to surgical
procedures. Adhesions between the pericardial sac and epicardium are elimin~te-l Percutaneous TMR
with a catheter apparatus also offers an alternative solution to persons who are not candidates for surgical
procedures.
Because TMR procedures generally involve creating a plurality of channels within the
myocardium, performing the procedure percutaneously requires the ability to steer a catheter apparatus
through the vasculature and maneuver the apparatus within the ventricle of the beating heart as rapidly as
possible to create the channels without subjecting the heart to the undue stress of a lengthy procedure.
Additionally, the ability to control and stabilize the catheter apparatus against the beating heart wall
while creating channels with a laser is desirable for percutaneous procedures to ensure creation of
channels as desired and to ensure that the laser is fired only within the myocardial tissue. TMR channels
should be spaced and grouped appropriately to achieve the desired result without weakening or rupturing
the heart muscle.
The early myocardial acupuncture procedures were not performed percutaneously. The Hardy
CO2 laser delivery system described above is rigid, relatively large, and not adaptable for pe~ laneous
use. The Aita '316 patent does not suggest a method for percutaneous use of the laser delivery device
described therein for surgical use.
U.S. Patent No. 5, 389,096 issued Feb. 14, 1995 to Aita et al. purports to teach one method of
percutaneous TMR using an elongated flexible lasing apparatus with control lines and a focusing lens
structure at the distal tip. The method uses pressure applied manually to attempt to stabilize the apparatus
against the wall of the heart.
Several patents describe the use of catheters within the ventricle for percutaneous treatment of
ventricular tachycardia. Such devices have a means to locate an arrhythmia site and ablate the site, at or
just below the ventricle surface, using an electrode device or laser energy. U.S. Patent No. 5,104,393
issued Apr. 14, 1992 to Isner teaches a catheter apparatus having a guiding Y-shaped sheath and guide
CA 02248328 1998-09-22
catheter assembly for introducing an optical fiber into the ventricle. Positioning is described to enable a
single burst of laser energy from a single aperture to ablate the site.
U.S. Patent Nos. S,255,679 issued Oct. 26, 1993 and S,465,717 issued Nov. 14, l99S
respectively to Imran and Imran et al., disclose non-laser, basket-shaped catheter apparatus for mapping
and/or ablation of arrhythmia sites within the ventricle. A pull cable is used to expand the basket portion
within the ventricle, and a plurality of electrodes on the arms of the basket are used for ablation. The
basket device is designed to place the electrodes on the ventricle wall.
U.S. Patent No. S,190,050 issued Mar. 2, 1993 to Nitzsche teaches a steerable catheter with a
handle and a tube, the distal tip of which may be selectively curved by controllably moving one of three
flat, sandwiched shims relative to the others by manipulation of a handle portion.
U.S. Patent No. S,358,479 issued Oct. 25, 1994 to Wilson, incorporated herein in its entirety by
reference, teaches another steerable catheter with a handle and an inner tube, the apparatus having a
single elongated, substantially flat shim spring mounted within the tip of the catheter tube, the shim
having at least one transverse or lateral twist which causes the tip of the catheter tube to assume a desired
curvature.
Drug therapies with angiogenic growth factors may expedite and/or augment collateral artery
development. U.S. Patent No. S,498,238 issued Mar. 12, 1996 to Shapland et al., discloses a method of
simultaneous angioplasty and drug delivery to localized portions of arteries. The patent teaches the use of
an expandable balloon end type catheter which can be filled with a drug-containing fluid and which is
allowed to permeate through a semi-permeable membrane of the balloon-tip end and thereby be delivered
directly to the surface of arteriosclerotic lesions on stenosed arteries.
A great deal of published scientific information concerning therapeutic agents is currently
available on the internet. One company, Annual Reviews is located at http://www.annurev.org. A list of
genetically engineered and/or naturally occurring drugs or other agents having pharmacological,
therapeutic, diagnostic or other utility is located at http://www.annurev.org/sup/im/imlS/imlSb.htm.
CA 02248328 1998-09-22
Additional scientific information is available at http://darwin.bio.uci.edu/~cchughes/index.htrn~.
Devices for effectuating drug injection have included non-articulating, viewing devices. U.S.
Patent No. 5,685,853 issued Nov. I l, 1997 to Bonnet teaches of a partially rigid endoscope to which is
attached an injection or aspiration cannula that is axially adjustable along the shaft of the endoscope.
U.S. Patent No. 5,261,889 issued Nov. 16, 1993 to Laine et al. teaches of an injection therapy
catheter that is insertable through a working channel in an endoscope for delivering fluid agents through
a hollow needle at the distal end of the catheter.
U.S. Patent No. 5,685,853 issued Nov. I l, 1997 to Bonnet teaches an injection device by means
of an injection cannula axially adjustable along an endoscope shaft. The injection cannula and guide tube
are axially adjustable relative to the endoscope shaft by means of a handle which can be operated with
one hand.
U.S. Patent No. 4,350,148 issued Sep. 21, 1982 to Sivak, Jr. et al. also teaches of a drug injector
device, in this case for treating esophageal varices. A flexible shafted endoscope has a conduit with distal
ended needle is inserted in the endoscope's biopsy channel for effectuating the treatment.
Prior devices also include viewing devices for cardiac interventional procedures. U.S. Patent
Nos. 4,784,133 issued Nov. 15, 1988 and 4,976,710 issued Dec. I 1, 1990, both to Mackin, both teach of
a flexible angioscope/bronchoscope device with an inflatable balloon structure for viewing
intravasculature structures. These flexible catheter devices include a ported working channel for
introduction of a working device and positioning of the working device at the viewing/treatment distal
end.
U.S. Patent No. 5,554,114 issued Sep. 10, 1996 to Wallace et al. teaches an infusion device with
preformed shape. An infusion guidewire or catheter is used for introduction of the device through a
selected path in a patient's vascular system. An elongated tubular diffusion body lies at the distal end of
an elongated tube, the diffusion portion having a plurality of infusion ports through which blood, drug,
diagnostic agent or other material can be delivered to the particular site in the vascular system.
CA 02248328 1998-09-22
.
U.S. Patent No. S,464,394 issued Nov. 7, 199S to Miller et al. teaches a multilumen percutaneous
angioscopy catheter which allows simultaneous irrigation and passage of an angioscope there through.
U.S. Patent No. 4,702,260 issued Oct. 27, 1987 and U.S. Patents No. 4,766,906 issued Aug. 30,
1988, both to Wang, teach bronchoscopic needle assemblies. The needle assemblies are especially
adapted for safe and efficacious collection of biopsy samples.
U.S. Patent No. 5,409,453 issued Apr. 25, 1995 to Lundquist et al. teaches a steerable medical
probe with stylets. The device is designed for reducing the mass of a body part, such as for biopsy
sampling or for removing prostatic tissue in the case of BPH. The torquable catheter has a control end
and a probe end, the probe end having a stylet guide means with a flexible tip and a tip directing means
extending from the control end to the flexible tip for changing the orientation of the central axis of the
stylet guide means for directing a flexible stylet outward through the stylet port and through intervening
tissue to targeted tissues.
U.S. Patent No. 5,571,151 issued Nov. S, 1996 to Gregory teaches a method for
contemporaneous application of laser energy and localized pharmacologic therapy. The method
comprises preparing a solution of a pharmacologic agent, inserting the catheter into the lumen, directing
the catheter to the site, transmitting visible light to the site, flowing the light transmissive liquid through
the catheter, viewing the site, transmitting laser energy through the liquid filled catheter to treat the site,
and introducing a flow of the pharmacologic agent in solution into the catheter for contemporaneous
discharge at the distal end into the lumen adjacent the site.
International Publication No. WO 92/10142 published June 25, 1992 by Pfizer Hospital Products
Group and Makower teaches a device and method for interstitial laser energy delivery. A catheter with
moveable needle system places one or more fiber optic elements and thermo-measuring devices through a
body passageway wall and into the bulk of an adjacent organ. The catheter is positioned adjacent to the
organ and the needles are extended to mechanically puncture the wall and move into the organ with the
fiber optic elements. The needle may be withdrawn into the catheter before delivery of laser energy or
CA 02248328 1998-09-22
remain in the organ to serve as an aspiration-irrigation vehicle. Lumens provided within the catheter for
carrying the hollow needles may likewise be used for aspiration or irrigation of the passageway. The
devices may also be used with a dilatation balloon, etc.
U.S. Patent 5,386,837 to Sterzer discloses an "electrochemotherapeutic" technique for treating
tumors in which high intensity electromagnetic force fields (including a laser) are applied to the body
after chemotherapy has been applied. This is intended to create large, transient pores in individual cells
of a superficially-seated tumor lesion located between individually mounted ceramic horn antennae by
non-invasively applying a highly directional beam of force-field shock of HF pulsed wave energy into the
cells, thus inducing the drug to enter the cells.
The use of superelastic and/or shape memory materials is widely known. Structure and Properties
of Ti-NI Alloys: Nitinol Devices & Components, Duerig et al., In Press, Titanium Handbook, ASM
(1994) In general, binary compositions of Nickel (Ni) and Titanium (Ti), yield alloys with shape memory
and superelastic properties. These alloys are comrnonly referred to as Ni-Ti, nitinol, and other industry
names. Their precise physical and other properties of interest are extremely sensitive to the precise Ni/Ti
ratio used. Generally, alloys with 49.0 to 50.7 atomic % of Ti are commercially available, with
superelastic alloys in the range of 49.0 to 49.4%, and shape memory alloys in the range of 49.7 to 50.7%.
Due to a rapid decrease in the ductility of the material, binary alloys with less than 49.4 at.% Ti are
generally unstable. In general, these types of materials exhibit hysteresis, defined as a phenomenon
exhibited by a system whose state depends on its previous history, and illustrated diagrammatically by
the familiar upper and lower curves which meet at the ends and define an area under the curves. In the
case of solid materials undergoing elastic hysteresis (as opposed to magnetic or electrical hysteresis), the
curves are related to stress necessary to cause deformation or otherwise overcome existing stress in pre-
stressed materials.
For the purposes of this disclosure, a distinction between superelastic materials and shape
memory materials is made. Sl~perelasticity refers to the highly exaggerated elasticity, or springback,
CA 02248328 1998-09-22
observed in many Ni-Ti alloys deformed at a specific temperature. The function of the material in many
of such cases is to store mechanical energy. Though limited to a rather small temperature range, these
alloys can deliver over 15 times the elastic motion of a spring steel, i.e., withstand a force up to 15 times
greater without permanent deformation. Shape memor,v materials will refer to those materials which can
be deformed, but which will freely recover their original shapes during heating, often utilizing electrical
resistivity, or which will develop a large recovery stress when recovery is prevented. With regard to the
present invention, it will be understood that the transition temperature of materials must, in general, be
somewhat above body temperature.
U.S. Patent No. 3,890,977 issued June 24, 1975 to Wilson teaches kinetic memory electrodes,
catheters and cannulae. These devices incorporate a material, such as a Ni-Ti alloy, having heat-activated
mechanical memory properties. The device is formed into an operative shape at a high temperature.
Then, at a low temperature below its transitional temperature, it is reformed into a shape for ease of
insertion into a guide catheter or the like or otherwise through a portion of a patient's vasculature or other
body lumen. When located in the organ or other desired region, those portions of the device constructed
using such shape memory materials are heated to above their transitional temperatures, using electrically
resistive elements, thereby returning the catheter to its original annealed anchoring or proper locating
shape. An important drawback of the Wilson apparatus is that heat must be applied to the catheter tip.
Complicated construction and electrical power distribution must be considered.
U.S. Patent No. 5,114,402 issued May 19, 1992 to McCoy teaches a maneuverable distal
apparatus with a temperature activated material of construction which, upon heating to a predetermined
position, will assume a predetermined, memorized shape, and which upon cooling, will assume a
different shape by action of a spring element urging the apparatus into the different shape.
U.S. Patent 4,846,171 issued July 11, 1989 to Kauphusman et al. teaches a transluminal
angioplasty catheter with a fiber advancing housing with a Hall effect sensor for control of a laser
irradiation source that cooperates with the fiber advance mechanism.
CA 02248328 1998-09-22
There is a need for steerable pel~;ul~lleous catheters, especially PTMR steerable catheters which
automatically maintain alignment of the distal end of the catheter with the distal end of a functional
device therein during catheter deflection movement relative to an interior body surface, particularly a
ventricular wall. Moreover, there is a need for a mechanism to detect interior body surface contact with
the distal tip of the catheter.
ADVANTAGES AND SUMMARY OF INVENTION
Thus, it is an advantage of the present invention to provide a steerable catheter and method of
use for percutaneous and other intra-vascular procedures, including but not limited to PTMR, or any
stimulation procedure.
The steerable percutaneous catheter of the present invention comprises a catheter jacket having
proximal and distal ends, and at least a first lumen, at least a first one functional device within the lumen
of the catheter jacket, the functional device having proximal and distal ends, a deflection mechanism at
the proximal end of the catheter, the deflection mechanism operatively attached to a deflector device at
the distal end of the catheter jacket, activation of the deflector device by movement of the deflection
mechanism deflects the distal end of the catheter jacket and the functional device therein; and a relative
movement compensation mechanism for maintaining alignment between the catheter jacket and the
functional device whereby movement of the deflecting mechanism causes simultaneous movement of the
relative compensation movement mechanism.
It is a further advantage of the present invention to provide a steerable catheter capable of being
guided into a heart chamber and used therein for creating a plurality of TMR channels controllably and
efficiently that can maintain tip alignment during motions of the distal section of the catheter. It is a
further advantage of the present invention to provide an elongated steerable catheter for placement within
a heart chamber, organ aperture or other body opening, the steerable catheter havin~ an inner tube
extending there through, the inner tube for guiding a laser delivery device or other functional device to
CA 02248328 1998-09-22
selected surfaces of the heart chamber, organ aperture or other body opening or body surface for laser or
other treatment thereon, particularly adapted for laser-assisted transluminal revascularization (TMR) and
further including a mechanism for maintaining a constant positional relationship between the translatable
laser delivery device or other functional device and the distal tip of the catheter during catheter tip
deflection.
It is yet a further advantage of the present invention to provide a percutaneous steerable catheter
which can be positioned securely into a selected position within the left ventricle, or other body opening
or cavity.
A further advantage of the present invention is to provide a steerable catheter with a tip
alignment system which m~int:~in~ precise alignment between the distal tip of the catheter and a laser
delivery device such as fiber, bundle, other functional devices being inserted there through, etc., as the
distal tip of the steerable catheter is deflected.
A further advantage of the present invention is to provide a steerable catheter which can be used
for detection of contact and maintenance of contact between the distal tip of the catheter and the heart
wall or other interior body structure or surface, such as during PTMR or stimulation.
A further advantage is to provide a steerable catheter with a handle portion at its proximal end
and a controllably deflectable portion at its distal end.
The elongated center or inner tube has a distal end, in the region where a curvature is to be
formed, and an anchor sleeve is slidably disposed over the center or inner tube. The anchor sleeve is
attached to the inside wall of the outer jacket and coupled to the distal end of the center or inner tube
with a bendable member which extends between the distal tip of the steerable catheter and the anchor
sleeve over the center or inner tube in a first embodiment. In a second embodiment of the center or inner
tube, a shim is attached to the sleeve and a coil on the proximal end and is positioned in a key hole
configuration. The distal section of the shim is attached to the distal tip of the catheter.
On the opposite the translation sleeve is a guide for a pull cable, the pull cable attached to the
CA 02248328 1998-09-22
distal end of the steerable catheter and extending through the guide to the handle. Thus, the translation
sleeve is maintained radially opposite the pull cable with the center or inner tube in between. An
alternate design includes a spring in which a pull wire is attached to the distal end of the catheter without
the shim.
An outer jacket has, in a preferred embodiment, distinct sections of different stiffness or
durometer. One or more distinct sections of material of differing stiffness or durometer can be used.
Junctions between the sections of different stiffness or durometer can be discrete and clearly defined, or
they can blend smoothly or gradually. A distal, more flexible portion is coupled to a proximal, stiffer
portion. The anchor sleeve is coupled to the outer jacket at or near the junction of two portions of the
outer jacket in a first embodiment of the catheter body. A second embodiment has the shim coupled to
the sleeve. Thus, the center tube moves freely through the anchor sleeve-
Adjacent the handle, the proximal outer jacket portion terminates at the catheter base. The pullcable extends through the catheter base, through a deflection housing tube, and terminat
es in a cable stop.
Rotation of a deflection knob threadably mounted onto the deflection housing tube will cause the pull
cable to be pulled backward, or the outer jacket to be pushed forward, relative to each other, thereby
inducing deflection of the distal end of the steerable catheter. Additionally, another degree of deflection
can be implemented by use of an additional deflection wire at the distal catheter tip that is controlled by a
knob or slide member on the catheter handle to achieve at least two degrees of deflection freedom at the
catheter tip.
The elongated steerable catheter is designed to be placed into the vasculature of the patient and
steered there-through until the distal tip is adjacent a selected portion of tissue, such as on an endocardial
surface within the left ventricle. Thus, the distal tip of a laser delivery device, such as an optical fiber or
fiber bundle or other functional energy delivery device such as radio frequency electrodes, microwave
cutters, ultrasound transmitters, mechanical coring devices, fluids jets and the like, can be extended
through the inner tube of the steerable catheter such that its distal tip comes into contact with the selected
CA 02248328 1998-09-22
surface structure for treatment thereon. With regard to PTMR therefore, the energy delivery device can
be controllably advanced through the steerable catheter for creating one or more PTMR channels.
Furthermore, with regard to non-laser PTMR, a cannula or trocar assembly may be extended through the
steerable catheter into the tissue of the ventricle, with or without use of a mechanical piercing tool.
In alternate embodiments, the steerable catheter of the present invention includes other
functional devices including but not limited to, fiber advance depth control mechanism, visualization
device, drug delivery apparatus, etc.
Numerous other advantages and features of the present invention will become readily apparent
from the following detailed description of the invention and the embodiments thereof, from the claims
and from the accompanying drawings.
BREF DESCRIPrION OF THE DRAWINGS
FIG. 1 is a representative isometric view of a preferred embodiment of the steerable catheter of
the present invention showing a handle having an actuator and deflective end portion.
FIG. 2A is a representative partial cutaway view of the deflectable end portion and anchor sleeve
of a preferred embodiment of the steerable catheter system of the present invention.
FIG. 2B is a representative isometric view of the distal end of a preferred embodiment of a laser
delivery device of the present invention.
FIG. 3 is a representative partial cutaway view of the deflectable end portion and anchor sleeve
of another preferred embodiment of the steerable catheter of the present invention.
FIG. 4 is a representative exploded view of the internal assembly of a preferred embodiment of
the handle of the steerable catheter of the present invention using a rotatable differential screw
mechanism with an integrated functional device advance mechanism for achieving auto-alignment of the
distal tip of the catheter and functional device.
CA 02248328 1998-09-22
FIG. 5A is a representative sectional view of a preferred embodiment of the handle of the
steerable catheter of the present invention using a rotatable differential screw mechanism with an
integrated functional device advance mechanism for achieving auto-alignment of the distal tip of the
catheter and functional device in an un-deflected position.
FIG. SAA is an enlarged view of detail 5A.
FIG. SB is a representative section view of a preferred embodiment of the handle of the steerable
catheter of the present invention using a rotatable differential screw mechanism with an integrated
functional device advance mechanism for achieving auto-alignment of the distal tip of the catheter and
functional device in a deflected position.
FIG. SBB is an enlarged view of detail SB.
FIGS. 6A-6C are representative isometric cutaway views of the steerable catheter of the present
invention illustrating a preferred embodiment of the method of the present invention.
FIG. 6D is a partial cut-away view of a second embodiment of the handle using a rotatable
differential screw mechanism showing fiber advance and deflection components for achieving auto-
alignment of the fiber optical tip.
FIG. 6E is a cross-sectional view of a variation of the embodiment shown in FIGS. SC-E and 6A
of the handle device using an integrated rotatable differential screw with an integrated fiber advance
mechanism for achieving auto-alignment of the optical fiber tip.
FIG. 6F is a cross-sectional view of a variation of an O-ring sealing member for the optical fiber
for the design shown in FIGS. SC-E and 6A.
FIG. 7A is a representative isometric view of a drug delivery apparatus coupled to the proximal
end of the handle of the steerable catheter of the present invention.
FIG. 7B is a representative side view of an alternate embodiment of a laser delivery device
adapted for drug delivery of the steerable catheter of the present invention.
14
CA 02248328 l998-09-22
FIG. 8A is a representative schematic view of an alternate embodiment of the steerable catheter
of the present invention with a pressurized saline bag.
FIG. 8B is a representative schematic view of a preferred embodiment of the steerable catheter
with surface contact detection system of the present invention.
FIG. 9 is a representative perspective view of the steerable catheter of the present invention
within the left ventricle.
DETALED DESCRIPTION
FIG. 1 is a representative isometric view of a preferred embodiment of the steerable catheter 100
of the present invention showing a handle having an actuator 156 and deflective end portion 106 with the
distal tip of a functional device 102. A preferred embodiment of the catheter 100 has a handle 170 at its
proximal end 104 and a controllably deflectable end portion 106 at its distal end 108. The deflectable end
portion 106 is more flexible than the elongated catheter jacket 110, allowing the deflectable end portion
106 to develop a controlled bend with a small radius of curvature.
Components for effectuating multiple degrees of freedom of the distal tip of the catheter as well
as other features for steerable catheter systems are disclosed in U.S. Patent Application S.N. 08/833,352
entitled STEERABLE CATHETER by Giba et al. filed April 4, 1997, which is hereby incorporated by
reference in its entirety.
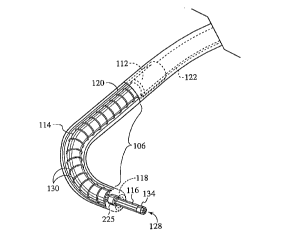
FIG. 2A is a representative partial cutaway view of the deflectable end portion 106 and anchor
sleeve 112 of a preferred embodiment of the steerable catheter 100 of the present invention. As will be
understood by the drawings and description herein, the curvature in the deflectable end portion 106 of the
inner tube 114 can be deflected as desired. The helical coil spring 130 can be constructed with varying
degrees of flexibility, and with any number of coils, such that the curvature can be moved closer to the
catheter tip 118 of the inner tube 114 or closer to the anchor sleeve 112. As will be understood,
increasing the tension in pull cable 122, attached at a location near the catheter tip 118, by retraction
CA 02248328 1998-09-22
thereof will cause deflection of the catheter tip 118 and the deflectable end portion 106 in a direction
essentially out of, and into and toward a position perpendicular to the plane of the shim 120. Continued
retraction of the pull cable 122 will cause continued deflection of the catheter tip 118 and deflection of
end portion 106 of the steerable catheter, with useful ranges of deflection between about 0 and about 180
degrees (U shape) to about 270 degrees (pig-tail shape), or more or less depending upon construction.
FIG. 2B is a representative isometric view of the distal end of a preferred embodiment of a laser
delivery device 116 of the present invention. Adjacent the distal end 128 of the optical fiber, fiber bundle
or other laser delivery device 116, a ring member 225 which has a greater diameter than fiber 116 is
attached so as to engage the catheter tip 118 and perform as an optional fiber displacement stop.
Placement of the ring 225 along optical fiber or fiber bundle 116 is selected to control depth, as shown by
length A, of created pathways into the myocardium. Radiopaque marker 134 is shown as well.
FIG. 3 is a representative partial cutaway view of the deflectable end portion 106 and anchor
sleeve 112 of another preferred embodiment of the steerable catheter 100 of the present invention. Pull
cable 122 is attached at a location near the tip 118 and extends through pull cable guide 103. The
deflectable end portion 106 is made out of a softer material than the proximal shaft catheter jacket 110.
As the pull cable 122 is pulled, a force is applied to the catheter tip 118 resulting in tip deflection as
shown in the phantom views. This design relies upon the flexibility of the spring 130 to provide the
necessary return force instead of a shim as in the design shown in FIG. 2A. The spring 130 in any of
these figures may be made of various materials known to those of skill in the art including, but not
limited to, stainless steel, tungsten, or even partially or completely constructed of one or more
superealstic and/or shape memory materials. Cross section of wire of the spring may be for example,
oval, round, rectangular or flat ribbon.
FIG. 4 is a representative exploded view of the internal assembly of a preferred embodiment of
the handle of the steerable catheter of the present invention using a rotatable differential screw
16
.. . ..
CA 02248328 1998-09-22
mechanism with an integrated functional device advance mechanism for achieving auto-alignment of the
distal tip of the catheter and functional device.
FIG. 5A is a representative sectional view of a preferred embodiment of the handle of the
steerable catheter of the present invention using a rotatable differential screw mechanism with an
integrated functional device advance mechanism for achieving auto-alignment of the distal tip of the
catheter and functional device in an un-deflected position. FIG. 5AA is an enlarged view of detail 5A.
FIG. 5B is a representative section view of a preferred embodiment of the handle of the steerable
catheter of the present invention using a rotatable differential screw mechanism with an integrated
functional device advance mechanism for achieving auto-alignment of the distal tip of the catheter and
functional device in a deflected position. FIG. 5BB is an enlarged view of detail 5B.
As seen in FIGS. 4 and 5, outer catheter jacket 110 terminates at its proximal end 126 and is
coupled to catheter base 162. Proximal hub 166 is contained within the catheter boot 164 and threads
over inside stepped and threaded portion 165 of catheter base 162. The proximal hub 166 is coupled to a
distal flange portion 153 of inner deflection knob 154. Deflection actuator 150 slides over catheter base
162 and has an external helical grooved portion 152 located distally on the deflection actuator 150. Two
pins 140 attached to inner deflection knob 154 engage helical groove 152, thus rotation of inner
deflection knob 154 about deflection actuator 150 translates into linear motion between inner deflection
knob 154 and deflection actuator 150. An actuator 156 (shown in two sections in FIG. 5) couples radially
around the inner deflection knob 154 and translates linearly with respect to inner deflection knob 154.
The actuator 156 engages inner deflection knob 154 at flange 158.
The inner tube 114 is attached to handle 170 at coupling point 172 . A distal external, helical rib
or thread 174 on the handle 170 fits into and acts in cooperation with an operatively pitched and
contoured internal helical slot or groove 176 located proximally on the inner deflection knob 154. The
inner tube 114 continues proximally, sliding through a front tube 180 and terminates within a back tube
182. A bushing 184 is mounted on bushing pins 186 which extend through longitudinal slots 188 located
CA 02248328 1998-09-22
distally within handle 170 and extend into pin seats 190 located distally on front tube 180. Thus, as the
front tube 180 is moved linearly with respect to the handle 170, the bushing pins 186 move linearly
within slots 188.
Relative motion between the front tube 180 and the handle 170 is limited to linear motion; there
is no rotational motion between the front tube 180 and the handle 170 as such is prevented by the bushing
pins 186 which only slide linearly in slots 188. Similarly, axially and longitudinally extending ribs or
keys 192 located proximally and externally on catheter base 162 slide linearly within correspondingly
shaped linear grooves 194 located internally and distally on deflection actuator 150 opposite the external
helical groove 152, thus preventing rotational motion as between the catheter base 162 and the deflection
actuator 150. Finally, axially and longitudinally extending ribs or keys 196 located proximally and
externally on deflection actuator 150 slide linearly within correspondingly shaped linear grooves 198
located internally and distally on handle 170 at a point distal to slots 188, thus preventing rotational
motion as between the deflection actuator 150 and the handle 170.
Pull cable 122, such as shown in FIGS. 2A and 3, extends proximally from the catheter tip 118
through the catheter base 162 and through the deflection actuator 150, and terminates at pull cable stop
168. Pull cable 122 biases pull cable stop 168 against the proximal end 169 of deflection actuator 150.
The actuator 156 rotates around the bushing 184 and the entire assembly including the actuator
156, the bushing 184, bushing pins 186 seated in the front tube 180 along with the front tube 180, back
tube 182 and proximal assembly all translate linearly. Additionally, as the actuator 156 is rotated about a
central axis, the inner deflection knob 154 is co-operatively and simultaneously similarly rotated thus
effectuating linear translation of deflection actuator 150 and thereby increasing tension in pull cable 122.
To prevent the contractive forces on the pull cable 122 which deflect the catheter 100 and translate into
counter-rotational forces on the actuator 156 from actually causing the assembly to essentially "unwind",
therefore, bushing 184 is constructed with several detents 185 which compress between actuator 156 and
handle 170 distally. In a preferred embodiment of the bushing 184, therefore, the resilient detents 185 are
18
CA 02248328 1998-09-22
distributed around the bushing 184 so as to engage one or more correspondingly shaped grooves,
indentations within the proximal flange 157 on actuator 156.
Therefore, as the actuator 156 is rotated in a first direction so as to cause deflection of the
deflectable portion 106 of the catheter 100, engagement of the detents 185 of the bushing 184 within the
proximal flange 157 of the actuator 156 provides an indexed mechanism, which allows a tactile response
by the physician so as to control or at least be aware of the degree of deflection caused by said rotation
of the actuator 156. Furthermore, engagement of the detents 185 of the bushing 184 within the proximal
flange 157 of the actuator 156 prevents uncontrolled counter-rotation caused by the above described
contractive forces developed in the pull cable 122 of the deflected catheter 100. Upon intentional
counter-rotation by the physician, resilient detents 185 deform and allow rotation of the actuator 156 as
desired. Thus, bushing 184 is designed with resilient detents 185 which provide directionality, i.e., they
provide a certain degree of resistant to rotational forces on the actuator 156 intended to deflect the
catheter 100 but provide an increased resistance to counter-rotational forces, thereby providing an
indexed mechanism with tactile response upon rotation in either direction.
The handle 170 retains a portion of the back tube 182, the back tube 182 slidable through the
handle 170 and biased proximally by spring member 200; the spring member 200 is retained between
standing rib member 202 extending internally from handle 170 and distal flange 204 on back tube 182. A
sealing member 206 is placed between a proximal flange 208 on the front tube 180 and the distal flange
204 on the back tube 182. A depth stop 210 is threaded onto external helical threads 212 of back tube 182
extending proximally from handle 170. A Luer fitting 214 or other suitable coupling and sealing device is
useful for coupling a Touhy-Borst type fitting 216 to the back tube 182. An optical fiber, fiber bundle, or
other laser energy delivery device or other functional device may be coupled securely to the Touhy-Borst
type fitting 216 and be advanced through the back tube 182 and into the inner tube 114. A saline flush,
drug solution, visualization or other therapeutic agent containing fluid can be provided to the steerable
catheter via one branched arm 218 of fitting 216. In a preferred embodiment, it will be understood that
19
CA 02248328 1998-09-22
any back-flow preventer, check valve, blood seal, etc. with the necessary operative function and
suitability can be employed elsewhere on the steerable catheter 100 and will be included within the scope
of the present invention.
During a PTMR procedure using a steerable catheter as shown in FIG. 1, maintaining alignment
between the fiber tip 128 and catheter tip 118 is preferred for controlling channel depth in a heart wall.
The distal tip 128 and catheter tip 118 must manually be realigned during deflection in the steerable
catheter 100 as taught in U.S. Patent Application S.N. 08/833,352.
FIGS. 6A, 6B and 6C show an automatic tip alignment mechanism for a steerable catheter
system using a differential screw mechanism 020 within deflection knob 0156. The differential screw
member within the knob 0156 has two differing thread pitches where threads 0152 effectuate tip
deflection and threads 052 effectuate tip alignment compensation. When the deflection knob 0156 is
turned, a corresponding advancement or retraction of the catheter's outer jacket occurs causing handle
section 0172 to move in relation to the proximal region of center or inner tube 0114 and the optical fiber
thereby maintaining optical fiber alignment. FIGS. 6A and 6B show the sequential deflection of the
distal tip section as the deflection knob 0156 is turned. FIG. 6A shows the catheter distal section 0106
without fiber advance, FIG. 6B shows the distal section 0106 deflected and FIG. 6C shows the distal
section 0106 deflected with advancement of optical fiber tip 0128. The diaphragm valve 0188 acts as a
seal component to prevent saline solution, if used, from being emitted from the handle while still
allowing translation of the optical fiber. An optical fiber is inserted into the inner tube 0114 and the fiber
distal tip 0128 and catheter tip 0118 (as shown in FIG.1) are adjusted and aligned manually prior to use.
As the deflectable tip section 0106 is deflected as shown in FIG. 6B, the differential screw in deflection
knob 0156 causes relative motion of the catheter jacket 0110 and handle 0172 that is attached to the
optical fiber advance mechanism thereby maintaining the alignment between the fiber distal tip 0128 and
catheter tip 0118 with the improved auto-alignment knob incorporated in the handle 0172. FIG. 6C
shows distal section deflected with the fiber advanced using a ring-type knob 0175 which surrounds the
CA 02248328 1998-09-22
handle section 0172 and facilitates fiber advancement to a preset depth according to a setting made with
depth control knob 0180.
FIG. 6D shows a cross-sectional view of the deflection knob 0156 with the catheter base 0162.
The threads 0152 for effectuating deflection of the catheter's distal end 0118 are engaged by a pin 025
attached to the deflection knob 0156. The tip alignment compensation threads 052 inside proximal
section of the deflection knob 0156 are engaged by another pin 027 attached to the deflection housing
tube 0150. The pull wire 0122 is attached at a stop connected to the deflection housing tube 0150. When
the deflection knob 0156 is turned, the deflection housing tube 0150 translates over the catheter base
0162. The threads create linear translation compensation of the optical fiber distal tip 0128 as the
catheter tip 0118 is deflected.
FIG. 6E is a cross-sectional view of a variation of the embodiment shown in FIGs 6A-6D using
an integrated rotatable differential screw mechanism in deflection knob 0156 that further includes an
integrated fiber advance component thereby allowing a physician to maintain hand placement while
adjusting the amount of deflection by knob 0156 or while advancing an optical fiber. The design shown
in FIG.6E in cross-section further includes a fiber advance annular knob 0256 that slides over and rotates
with the deflection knob 0156. This sliding aspect is achieved by longitudinal slots 0252 in the outer
surface of the deflection knob 0156 and corresponding longitudinal slots in the annular knob 0256. The
fiber advance annular knob 0256 replaces the fiber advance knob 0175 shown in FIGs 6A-6C above.
Rotation of the optical fiber advance knob 0256 rotates the deflection knob 0156. Linear advancement of
the fiber advance knob 0256 alone without rotation of the deflection knob 0156 advances the optical fiber
without tip deflection due to the longitudinal slots 0252 in the deflection knob 0156 guiding longitudinal
slots in the fiber advance knob 0256. The advancement of the fiber is achieved through a fiber advance
collar 0275 that is attached to the advance slider. The fiber advance knob 0256 has a return spring 0276.
FIG. 6F is a cross-sectional view of an alternate design for the functional requirements of the
diaphragm valve 0188 sealing device for use with the catheter handle concepts shown in FIGs 6A-6E.
CA 02248328 1998-09-22
The sealing device is an O-ring 0250 that is disposed about a central member 0300 where the optical
fiber translates within the central member 0300. This central member 0300 is attached to the deflection
housing tube 0150. An annular extension member 0284 is attached to the advance slider 0184 shown in
FIG. 6B and slides along on the external side of O-ring 0250 to maintain the fluid seal. An equivalent
sealing member of O-ring 0250 is a quad seal. The O-ring seal operates comparable to a "syringe" type
device.
The following description of the mechanical operation of the steerable catheter 100 of the
present invention is intended for illustrative purposes only, and is not to be construed in any way as
limiting the scope of subject matter claimed herein. Reference is made to all of the figures.
As described above, the steerable catheter of the present invention has a tip deflection
mechanism as well as a functional device tip alignment mechanism. With regard to FIGS. 4 and 6A-6C,
rotation of the actuator 156 in a clockwise direction, i.e., as viewed from a proximal end, will effect
corresponding rotation of inner deflection knob 154. Since the actuator 156 and inner deflection knob
154 are rotated relative to the handle 170, and the catheter base 162 is keyed to the deflection actuator
150 by ribs 192 engaging grooves 194 along with the deflection actuator 150 being keyed to the handle
170 by ribs 196 sliding into grooves 198 thereby preventing rotational motion as between the handle 170,
the deflection actuator 150 and the catheter base 162, said clockwise rotation will cause proximal
translation of deflection actuator 150 by pins 140 riding in helical groove 152, as deflection actuator 150
is moved linearly in a proximal direction, tension in the pull cable 122 acts on the distal tip 118 of the
steerable catheter 100 and causes deflection thereof.
Operation of the automatic functional device tip alignment mechanism is based on a screw thread
pitch differential. Without the tip alignment feature of the present invention as deflection of the
deflectable portion 106 of the steerable catheter 100 occurs the orientation of the distal tip 118 of the
catheter 100 and the distal tip 102 of the functional device would be modified such that any pre-existing
alignment would be lost. The cause of this loss of alignment between the distal tip 118 of the catheter
22
CA 02248328 1998-09-22
100 and the distal tip 102 of the functional device upon deflection of the deflectable portion 106 is
caused by retraction of the pull cable 122, causing an apparent change in the length of the elongated
catheter jacket 110 and a displacement of any pre-existing alignment between the distal tip 118 of the
catheter 100 and the distal tip 102 of the functional device.
Therefore, to compensate for these alignment disrupting forces, screw threads having a
differential in pitch size are used. With reference to the drawings, as mentioned above, deflection of the
deflectable portion 106 of the catheter 100 is caused by clockwise rotation of the actuator 156 and inner
deflection knob 154. Said clockwise rotational motion of actuator 156 and inner deflection knob 154
causes distal linear translation of inner deflection knob 154, proximal hub 166 and catheter base 164
thereby causing compression of the outer catheter jacket 110 and proximal linear translation of deflection
actuator 150 and pull cable stop 168 thereby increasing tension in pull cable 122 and causing deflection
of the deflecting portion 106. Simultaneously, as will be apparent by an inspection of the drawings, as
inner deflection knob 154 is rotated clockwise by actuator 156, external helical thread 174 on the handle
170 engaged by internal helical groove 176 within inner deflection knob 154 causes simultaneous
translation of the handle 170, thus slightly moving the fiber 116 and thereby compensating for the
effective change in length of the outer catheter jacket 110 by maintaining alignment between the distal
tip 118 of the catheter 100 and the distal tip 128 of the laser energy delivery device 116.
In the case of laser assisted PTMR or other procedures, intervention occurs when an optical
fiber, fiber bundle or other laser energy delivery device 116 or other functional device is advanced
through the inner tube 114 of the steerable catheter and into the patient. Fiber advance is effected in one
of two ways - by manually urging in a distal direction either back flange 220 of depth stop 210 or
actuator 156. In either case, the fiber, fiber bundle or other laser energy delivery device 116 or other
functioanl device being held firmly in place at the proximal end by Touhy-Borst type fitting 216
advances distally along with the back tube 182 and the front tube 180, both sliding over the inner tube
114, the bushing pins 186 extending from the pin seats 190 in the front tube 180 contained by and riding
23
CA 02248328 1998-09-22
within the slots 188 located distally on the handle 170, thus placing the spring 200 into increased
compression. Retraction of the fiber 116 decreases the compressive forces on the spring 200.
In a preferred embodiment of the steerable catheter of the present invention, access port cover
plate 222, as shown in FIGS. l and 4 can be removed and any operative device, electrical contacts such
as thin coaxial or other electrical traces, leads, conductors, etc. can lead through at least the outer catheter
sheath and be utilized at any of various positions on the handle 170, elongated portion 110 or distal tip
118 of the steerable catheter 100 of the present invention. In particular, the distal tip 118 can be provided
with a positioning sensor or vic~ 1i7~ion device, for providing any of various signals from any of various
types of sensor or analyzer equipment, such as the ultrasound ranging methods and devices shown and
described in U.S. Patent Application Serial No. 08/852,977 filed 05/07/97 entitled ULTRASOUND
DEVICE FOR AXIAL RANGING which is hereby incorporated herein by reference in its entirety. In a
preferred embodiment, an annular ultrasound transducer is positioned distally on the distal tip 118 to
transmit ultrasound signals substantially perpendicular to tissue, the transducer further receiving
returning signals from the tissue to be treated.
U.S. application serial no. 08/773,872 entitled LASER MEANS ADAPTED FOR DRUG
DELIVERY filed December 27, 1996, and hereby incorporated by reference in its entirety, discloses a
drug delivery apparatus for dispensing a predetermined amount of one or more drugs in, near or around
the creation of one or more laser-created openings or channels, particularly PTMR channels and/or
stimulation pockets within myocardium or other stimulation zones to stimulate angiogenesis on or in
selected target surfaces in the body. The apparatus includes a laser delivery device such as an optical
fiber or fiber bundle having one or more conduits for transmitting drugs included as a part of the delivery
device. The conduit may comprise a space along a fiber optic cable between an outer jacket of the cable
and the fiber optic, or fiber optic bundle, and an aperture or array of apertures in the end of the cable
through which the drug escapes. The aperture or apertures can be replaced with a semi-permeable or
permeable membrane, strainer, set of leach holes, etc. Or the conduit may be one or more drug tubes
24
CA 02248328 1998-09-22
contained in the fiber bundle and the drug exits out of the target end surface of the cable. Or the conduit
may be one or more tubes between the fiber optic delivery means and the outer jacket. A piercing device
may be mounted on the target end of the laser delivery means, or an optical fiber with a pointed tip which
pierces the target area prior to applying the laser beam may be used. After or simultaneously with the
creation of a laser TMR channel or other opening the drug or drugs are transmitted through the conduit
directly into the TMR channel or other opening. The target surface may be mechanically pierced to
provide initial access to the target region of tissue, such as myocardium. The drug is dispensed by
manually or automatically activating an electric motor which actuates a piston element.
FIG. 7A is a representative isometric view of an alternate embodiment of the present invention
with a drug delivery apparatus 296 coupled to the proximal end 104 and a drug delivery needle 101
extending beyond the distal end of the steerable catheter 100 of the present invention. As shown, other
tools or functional devices may be attached to the handle 170 of the steerable catheter 100 of the present
invention for operation through the inner tube 114 in addition to the drug delivery or dispensing
apparatus 296. It will be understood, therefore, that such drug delivery or dispensing apparatus 296 can
be manually or automatically activated, can be adjustable or programmable to dispense individual
aliquots of a predetermined volume, at a predetermined or specified rate, as desired.
FIG. 7B is a representative side view of an alternate embodiment of a laser delivery device
adapted for drug delivery of the steerable catheter of the present invention. The laser delivery device 116
will be disposed within a drug conduit 105. The distal end 107 of the drug conduit 105 has a plurality of
(i.e. one or more) perforations 127 formed through the wall of drug conduit 105 allow drugs in space 121
to flow or otherwise be transmitted through drug conduit 105.
The distal end 128 of the laser delivery device 116 preferably extends past the distal end 107 of
the drug conduit 105. The laser delivery device 116 may consist of a single or bundle of individual
optical fibers.
Outer jacket 117, such as a thin plastic tubing material, surrounds the bundle of individual fibers
, 25
,
CA 02248328 1998-09-22
(as shown in FIG.7B), and thus, the combination of outer jacket 117 and drug conduit 105 defines an
interstitial drug channel 121 through which drugs can be conveyed through conduit 105 and out the
plurality of perforations 127. It will be understood that the outer jacket 117 is optional and may be
omitted. In such case, utilizing a single fiber mounted within conduit 105 will result in drug delivery
from the plurality of perforations 127 in essentially the same manner as described above. However, in the
case of a bundle of fibers without an outer jacket 117, drug solution or other substances will flow around
each of the individual fibers of the bundle, thus resulting in percolation of drug out of the drug conduit
105 at either or both the plurality of perforations 127 and the distal end of the fiber bundle.
In the embodiments shown, individual perforations 127 are spaced about outer jacket 117. It will
be understood that more or fewer perforations may be used, and perforations located at various axial
positions located adjacent the distal end 107 of the drug conduit 105.
In co-pending application serial no. entitled DRUG DELIVERY MODULE, filed
May 16, 1998, and hereby incorporated by reference in its entirety, teaches a drug delivery device with a
drug delivery needle for percutaneous catheter based procedures. The elongated portion of the device
comprises a single or multi-lumen flexible shaft for containing at least one drug delivery channel in a
drug delivery tube. A connector tube extends through a catheter mount and is sealed to a drug conduit.
The drug conduit extends through an elongated tubular portion of the catheter to the distal tip of the
elongated portion where the drug conduit connects to a piercing needle. The piercing needle end portion
has a bevel cut end tip or other operable tip for piercing tissue and delivering drug or other compound
there through. The drug delivery conduit with piercing needle is inserted through the working channel of
the device to treat the desired number of drug delivery tissue sites. The drug flow is communicated from
a reservoir through drug conduit and is dispensed through piercing needle subsequent to advance of
piercing needle through the distal tip of device. Drugs can be delivered to tissue via advanceable drug
conduits with piercing needle tips which pass through a working channel of the instrument.
26
CA 02248328 1998-09-22
FIG. 8A is a representative schematic view of an alternate embodiment of the present invention
with the steerable catheter and a pressurized saline bag. Reference is made to all the figures. During
percutaneous procedures, including PTMR, a saline drip or irrigation system 250 is employed. In the
practice of this invention the saline bag can be replaced with any fluid, in any type of reservoir or source.
Providing a slightly elevated pressure within the catheter keeps the inner tube 114 of the catheter 100,
within which the fiber, bundle or other laser energy delivery device 116 or other functional device 102
advances and retracts, lubricated. Additionally, blood from the ventricle or other portion of the
vasculature will not be able to advance past the tip 118 of the catheter 100 and foul or plug the inner tube
114.
During PTMR, saline or other solution flows from a pressurized saline bag 250 through an arm
218 of a Touhy-Borst fitting 216, through the inner tube 114 to the distal tip 118 of the catheter 100
operatively positioned or emplaced within the heart 252. Maintaining a small amount of saline flow
through the unrestricted system can readily be achieved. Additionally, a pressurized source may be
achieved by elevating the saline bag above operational height, utilizing a small peristaltic or other type of
pump to pump the fluid from the saline bag into the vasculature and ventricle, against ventricular
pressure, or other type of pump, etc., as may be necessary.
Thus, when the distal tip 118 of the catheter 100 is not in direct contact with an interior body
surface, such as the heart wall, a pressure drop would be present:
Pl - P2 (1)
If the distal tip of the catheter is pressed against the heart wall with sufficient pressure to stop the saline
or other fluid flow, the pressure throughout the flow path will be at essentially Pl, the pressure
differential Pl - P2 will decrease to zero.
FIG. 8B is a representative schematic view of a preferred embodiment of a steerable catheter
with heart wall or surface contact detection system of the present invention. By utilizing the system of
the present invention to detect saline or other fluid flow through the system, contact with an interior body
27
CA 02248328 1998-09-22
surface, such as the heart wall, can be determined accurately and dependably. Between the source of
pressurized saline solution 250 and the catheter handle 170, a flow restrictor 254 is emplaced. Such flow
restrictor 254 may, in preferred embodiments, consist of a restriction valve, a predetermined length of
narrow diameter tubing, or a Touchy Borst etc. Additionally, a pressure sensor 256 is utilized. If the
pressure drop across the flow resistor 254 is selected such that it is approximately equivalent to or greater
than the pressure drop across the catheter 100, then the pressure P3 at the sensor 256 can be expressed by
the following expression:
P3 ~ (P1~ P2) (RC~+ P2 (2)
RC + RR
where: P3 = pressure at the sensor 256;
P2 = pressure in the heart;
Pl = pressure in the saline bag;
RC = resistance to flow through the catheter; and
RR = resistance to flow through the flow restrictor.
When saline solution is flowing in the system, Pl is greater than P3. If the saline flow is
decreased due to application of pressure between the distal tip 118 of the catheter 100 and the heart wall,
P3 will increase. As the flow stops completely, P2 equals Pl and P3 also equals Pl. This system provides
the physician with a great deal of additional inforrnation. Contact between the distal tip 118 of the
catheter 100 and the heart wall can be determined by an increase in P3, as well as the amount of pressure
used between the distal tip 118 of the catheter 100 and the heart wall. As more pressure is applied to the
heart wall by the distal tip 118 of the catheter 100, P3 will increase until flow of saline through the
system stops and P3 becomes equal to Pl.
FIG. 9 is a representative perspective view of the steerable catheter 100 of the present invention
within the left ventricle 230. As indicated above and with regard to the figures, the present invention is
directed to catheter systems which are steered into and through parts of the body, such as into the left
ventricle, with and without the use of a guide catheter or other guide system. Guide catheter and guidance
28
CA 02248328 1998-09-22
systems are well known and may be used with the present invention, and therefore are included within
the scope of this invention. Typically, entry into the vasculature is made through the femoral artery. A
guide wire (not shown) is positioned within the left ventricle 230. The steerable catheter 100 is advanced
over the guide wire and into the left ventricle 230. The guide wire is retracted out of the steerable
catheter and the functional device, such as an optical fiber, is advanced into position with the steerable
catheter.
However, a guide wire or guide catheter need not be used. Alternatively, the distal tip 118 and
deflectable end portion 106 of the steerable catheter 100 is inserted into the patient, extended over the
aortic arch 234 and prolapsed through the aortic valve 236 into the left ventricle 230. The steerable
catheter 100 can be guided into a selected position adjacent a selected surface 238, in this case a portion
of endocardium. As the actuator 156 is rotated, deflection of the deflectable portion 106 results in slight
modification of the dimension of the elongated portion 110 of the catheter 100, the modification
compensated for by the differential screw mechanism of the present invention. Furthermore, a wall
contact detection system provides wall contact and contact pressure information to the physician.
Thus, by sequential deflection the deflectable end portion 106 of the steerable catheter 100
and/or by rotation of the steerable catheter 100, extending the distal end 128 of a laser delivery device
116 or other functional device therethrough, delivering laser energy or performing other therapy,
visualization or diagnostic, and retracting the distal end 128 of the laser delivery device 116 or other
functional device back into the deflectable end portion 106, the steerable catheter 100 can treat a series of
individual, selected treatment points 240 of tissue such as endocardium. Such treatment points 240 would
typically be TMR channels or stimulation sites.
Alternatively, retro-lasing can be performed. This novel method includes the steps of advancing
the distal tip 128 of laser delivery device 116 a selected distance into the myocardium and then delivering
laser energy to create a TMR channel or other treatment site while simultaneously retracting the fiber,
laser delivery device 116 or other functional device. With this procedure, with regard to TMR especially,
29
CA 02248328 1998-09-22
inasmuch as laser energy is only delivered during retraction of the fiber, the possibility of advancing the
fiber too far and lasing through an epicardial surface is decreased, and the risks of complications arising
from such epicardial perforations, including but not limited to cardiac tamponade (a buildup of pressure
in the pericardial sac caused by the presence of an excess of fluid such as blood), proliferation of
adhesions between the epicardium and the pericardial sac (thereby preventing normal, frictionless
enclosure of the heart muscle within the pericardial sac), etc. are minimi7~-.A
The functional device or devices of the present invention includes those devices for treatment
and diagnosis of affected organs, tissues or interiors or interior surfaces of the body, including devices
configurable and extendable through one or more lumens within a steerable catheter, for example, energy
delivery devices, such as laser optical fiber elements, with or with out a piercing needle, laser wave
guides, radio frequency tissue ablation devices, microwave cutters, ultrasound transmitters, mechanical
coring devices, fluid jets, or drug delivery devices, with or without a piercing needle assembly.
Furthermore, adjunct use of ancillary drug delivery apparatus, blood seal device, depth stop
apparatus such as clamps, bushings, etc., visualization device, marker device as well as other hardware
and methodology will be considered within the scope of the present invention.
The alignment mechanism or tip alignment mechanism or automatic tip alignment mechanism of
the steerable catheter can be any relative movement compensation mechanism, including, but not limited
to, a screw mechanism, for example, a rotatable differential screw mechanism, gear, camrning or
threaded mechanism.
The present invention is intended for use with any medical laser. In particular, the Holmium or
excimer laser is particularly suited to the present invention. However, any suitable laser source, pulsed or
otherwise, could provide laser energy to the laser delivery device of the present invention for performing
the method of the present invention. Likewise, the steerable catheter and equipment, including laser
delivery device, referred to in the present document as well as that known and used in medicine and other
disciplines today and in the future, will be included in the scope of this disclosure. Such laser delivery
CA 02248328 1998-09-22
devices include, but are not limited to, individual optical fibers as well as bundles of fibers with and
without piercing tips and with or without firing tips or fiber ends having shaped or contoured end faces
for selectively diverging the laser beam or other laser energy diverging device, rods, mirrors
configurations and other laser delivery device with and without focusing lens and the like. It will also be
understood that the steerable catheter and method of the present invention as described herein including
the novel combination or use with of any conventional mechanism or method which are known to those
skilled in the art, are included within the scope of this invention.
Furthermore, with regard to non-laser TMR, a cannula or trocar assembly may be extended into
the tissue of the left ventricle, with or without use of a mechanical piercing tool.
For the purposes of the present invention and disclosure herein, the term "drug" or "drugs"
includes any and all drugs and therapeutic agents for use or useable within or on the body, including, but
not limited to the following, gene therapies, angiogenic agents, antibiotics, vaccines, function regulators,
anti-arrhythmic drugs, growth factors, anticoagulant antagonists, anticoagulants, anti-fibrinolytics,
platelet inhibitors, thrombolytics, antihistamines, anti-infl~mm~tory agents, immuno-suppressives,
receptor antagonists, adrenergic blockers, adrenergic stimulants, alpha/beta adrenergic blockers,
angiotensin converting enzyme inhibitors, angiotensin II receptor antagonists, anti-arrhythmics Group I,
Group II, Group III, Group IV, beta blockers, calcium channel blockers, diuretics, hypertensive
emergency agents, angiogenic agents, FGF-I, FGF-2, EGF, Vascular Endothelial Growth Factor (VEGF)
(preclinical), inotropic agents, patent ductus arteriosus therapy, Rauwolfia derivatives and combinations,
vasodilators, vasopressors, adjuncts, androgen inhibitors, antibiotic derivatives, anti-estrogens, anti-
metabolites, cytotoxic agents, enzyme inhibitors, horrnones, immuno-modulators, nitrogen mustard
derivatives, agents used in photodynamic therapy, such as photo-active or photo-labile compounds,
and/or other materials for performing functions including flushing and cooling, stim~ ting other
responses, detection, analysis, monitoring, visualization or control, etc., said solutions comprising waters,
saline and the like, solid and semi-solid materials, and in any forms including capsules and granules,
31
CA 02248328 1998-09-22
implants, etc.
The present invention includes the delivery of liquid, solid or semi-solid, time release
formulations, etc. It will be understood that there are additional drugs or therapeutic agents which may
become useful, such as agents directed at bone or implanted in semi-permeable sacs, radioisotopes, and
future gene therapies which are also included in the scope of this invention.
Active compounds which are given systemically have a normal therapeutic window which can be
expressed as mg of drug per kg of body weight. The amount of agent which is therapeutically acceptable
when adrninistering a drug locally can be approximated as mg of drug per kg of target treatment area (e.g.
organ weight), optimized accordingly with consideration of toxicity and mechanism of drug action.
Agents delivered to a specific site can achieve high local concentrations at the delivery point. Optimal
drug dose may scale differently when the drug is administered locally rather than systemically. Thus, the
amount of a given agent that should be delivered in order to achieve a therapeutic effect must be
optimized accordingly with consideration of toxicity levels (both locally and systemically), mechanism of
drug action, drug clearance mechanisms, and drug diffusion levels.
It will further be understood that while the present invention has been described for performing
TMR on endocardial surfaces in the left ventricle, the apparatus and methods described herein are
equally intended for use in any suitable procedure, including but not limited to procedures, where any
device need be extended through a guide catheter or the vasculature to an opening or other point within
the body for other medical procedures including one or more of the following, laser treatment, drug
delivery, visualization, biopsy, etc. "Stimulation", for example, is performed by using laser energy to
create zones or pockets, optionally interconnected at least initially by small channels ablated through the
tissue, for the introduction of blood born growth and healing factors and stimulated capillary growth
surrounding the lased zones or pockets to create an increased supply of oxygen to the tissue and thus a
revitalization of the heart muscle. Methods and apparatus for causing stimulation are more fully
32
CA 02248328 1998-09-22
described in co-pending U.S. Patent Application Serial No. 08/664,956 filed June 13, 1996, herein
incorporated by reference.
While the principles of the invention have been made clear in illustrative embodiments, there
will be imm~ tely obvious to those skilled in the art many modifications of structure, arrangement,
proportions, the elements, materials, and components used in the practice of the invention, and otherwise,
which are particularly adapted to specific environments and operative requirements without departing
from those principles. The appended claims are intended to cover and embrace any and all such
modifications, with the limits only of the true spirit and scope of the invention.
.