Note: Descriptions are shown in the official language in which they were submitted.
CA 02262789 1999-02-08
wo ~8/0~333 PCT/US97/l3537
MALE IMPOTENCE DIAGNOSTIC ULTRASOUND SYSTEM
CROSS-REFERENCE TO RELATED APPLICATIONS
The subject matter of this application is related to the subject matter
of U.S. Provisional Application No. 60/023,959, filed August 9, 1996 and
U.S. Provisional Application No. 60/041,361, filed March 21, 1997, both of
which are incorporated herein by reference and priority to both of which is
claimed under 35 U.S.C. 119(e).
BACKGROUND OF THE INVENTION
1. Field of the Invention
The invention relates to an apparatus and method for diagnosing
erectile disfunction, and more particularly, for diagnosing vasculogenic
erectile disfunction by using Doppler frequency shift to measure penile
blood velocity.
2. Description of Related Art
Male impotence is defined as the chronic inability to attain and/or
maintain an erection of sufficient rigidity for sexual intercourse. This
problem affects approximately 10 million American men, with increasing
incidence in those of advanced age. Impotence is a source of great anxiety
~ for many and is the subject of many thousands of visits to physicians and
other medical professionals every year.
CA 02262789 1999-02-08
WO ~810~333 PCT/US97/13537
During a normal erection, neurochemical stimulation causes penile
arterial inflow to increase in the paired cavernosal arteries. The result is
increased blood flow into the corpora cavernosa. The subtunical venus
plexus is compressed against the tunica albuginea, and venous outflow is
reduced to trap blood in the corpora cavernosa. This combination of
increased inflow and decreased outflow results in vascular congestion of
the penis, tumescence, and rigidity sufficient for sexual intercourse. It is
believed that abnormal reduction of blood flow to the cavernosal arteries
and/or excess venal outflow, i.e. corporal venous leakage, are the primary
physical causes of impotence. These abnormal blood flow characteristics to
and from the cavernosal arteries can be caused by a number of factors, for
example atherosclerotic vascular disease, traumatic arterial occlusive
disease, or defective venoocclusive mechanisms.
By measuring blood velocity in the cavernosal arteries, duplex
ultrasonography has been used successfully to diagnose inadequate arterial
inflow and excessive outflow. Typically, a transducer emits ultrasound
energy and a receiver receives ultrasound energy reflected from the blood.
Electronic circuitry then measures frequency differences between the
transmitted and received ultrasound energy, to calculate blood velocity.
See, for example:
Bassiouny, et al., "Penile Duplex Sonography in the Diagnosis of
Venogenic Impotence," Jollrnal of Vascular Surgery, Vol. 13, No. 1,
pp. 75-83, January, 1991;
CA 02262789 1999-02-08
WO 98/06333 PCT/I~S97/13537
Cochlin, D.L., et al., Urogenital Ultrasound A Text Atlas pp. 257-
258;
Kisslo, J., et al., ed., Basic Doppler Echocardiography pp. 11-16 and
190;
Levine, L.A., et al., "Measurement of Venogenic Impotence Penile
Duplex Ultrasonography," Journal of Urology, 1990, 143: 211A;
Lue, T.F., et al., "Vasculogenic Impotence Evaluated by High-
Resolution Ultrasonography and Pulsed Doppler Analysis,"
Radiology, 1985, 155: 777-81;
Lue, T.F., et al., "Functional Evaluation of Penile Arteries with
Duplex Ultrasound in Vasodilator Induced Erection," Urol Clir
North Am 1989, 16: 799-806;
Meuleman, et al., "Assessment of Penile Blood Flow by Duplex
Ultrasonography in 44 Men with Normal Erectile Potency in
Different Phases of Erection," Journal of Urology, Vol. 147, pp. 51-56,
~anuary, 1992; and
Quam, J.P., et al., "Duplex and Color Doppler Sonographic
Evaluation of Venogenic Impotence," AJR, 1989; 159: 1141-7.
See also U.S. Patent No. 5,482,039 to Place. All of the above-listed
documents are incorporated herein by reference.
The general calculations involved in determining blood velocity
using Doppler shift are well known in the art. See, for example, U.S.
Patent No. 4,722,347 to Abrams and Hovland, which is incorporated herein
by reference. See also U.S. Patents Nos. 4,334,543, 4,485,821, 5,205,292, and
5,284,146, which are incorporated herein by reference. See also the
following documents, which are attached to and form a part of the above-
identified Provisional Application No. 60/023,959:
Advanced Technology Laboratories Ultramark 9 Ultrasound System
brochure, 1993.
CA 02262789 1999-02-08
WO 98/06333 PCT/US97/13537
Advanced Technology Laboratories C7-4 HDI Broadband Curved
Array Scanhead brochure, 1993.
Advanced Technology Laboratories Ultramark 9 Digital Computed
Sonography System brochure.
Medasonics Neuroguard Transcranial Doppler Noninvasive Blood
Flow Monitoring System brochure.
Medasonics F3PA Doppler Fetal Pulse Detector Ultrasound
Stethoscope brochure.
One of the key variables necessary for proper velocity calculations is
the angle between the direction of transmitted/received ultrasound waves
and the blood flow path. To achieve accurate velocity determinations, this
angle, hereinafter called the angle of incidence or Doppler angle, must be
known accurately and/or maintained with some precision.
With conventional ultrasound devices, selection and precise
maintenance of this angle of incidence are difficult. Many ultrasound
devices suitable for measuring penile blood flow are handheld. An
important limitation of handheld devices is that the angle of incidence
varies unacceptably from reading to reading over time, or even during a
single reading. This variability is a result of the instability and imprecision
inherent with handheld devices. The urologist does not know what the
angle of incidence is, and even if a desired angle is known, achieving that
desired angle reproducibly on repeat measurements is very difficult during
a diagnostic procedure requiring many separate measurements. Even if
the relevant calculations eliminate the need to know the precise angle, e.g.
CA 02262789 1999-02-08
WO ~8/0~333 PCTIUS97tl3~37
by taking velocity ratios so as to effectively eliminate the angle variable,
the angle must be held constant. This instability represents a significant
disadvantage with previous ultrasonography devices, a disadvantage that
is heightened when taking measurements with respect to a readily
movable organ such as the penis.
A further disadvantage of prior art ultrasonography devices, and
particularly u~trasonography devices used for penile blood flow
measurement, lies in the current uncertainty in the medical community
concerning the proper timing of blood velocity measurements during a
diagnostic procedure. Typically, the urologist or other medical
professional takes an initial cavernosal artery flow velocity measurement.
Then, papaverine or another suitable vasodilating agent is injected or
transurethrally administered to induce erectile response. Further
ultrasonographic examination follows, with a series of velocity
measurements being taken over time after the vasodilator is introduced
and after subsequent self-stimulation by the patient. Some urologists
believe that if blood velocity increase is less than 25%, the patient is
probably impotent; if greater than 75%, the patient is probably healthy. For
increases between 25% and 75%, the urologist likely will perform
additional tests.
More specifically, the urologist measures peak systolic and end
diastolic velocities before injection and at 5,10, and 30 minutes after
application of the vasodilating agent. Normal peak systolic velocity is
CA 02262789 1999-02-08
WO ~8/0~333 PCT/US97/13537
considered by some medical professionals to be about 25 cm/sec. A
resistance index is calculated as the difference between the peak systolic
and end diastolic velocities divided by the peak systolic velocity, or
R.I. = (Peak systolic velocity - End diastolic velocity) / Peak systolic
velocity.
A resistance index approaching or equal to about 1 is considered to be
indicative of a normal result.
Choosing the exact timing of the post-vasodilation measurements
to achieve accurate diagnosis is the subject of the debate. For example,
slower but otherwise normal erectile response often occurs in males of
advanced age, so that premature ultrasonographic measurement might
result in a false positive diagnosis. A need has arisen, therefore, to
eliminate the uncertainties involved in examination timing.
SUMMARY OF THE INVENTION
To overcome the above and other disadvantages, an apparatus for
diagnosing male impotence according to the invention includes at least
one transducer for transmitting/receiving ultrasound or other suitable
energy to detect Doppler shift, for calculation of penile blood velocity. A
transducer housing supports the transducer in a substantially constant
orientation with respect to the penis, substantially fixing the angle of
CA 02262789 1999-02-08
WO 98t06333 PCT/US97/13537
incidence, that is, the angle between the transmitted/received ultrasound
or other energy and the blood velocity vector. An acoustic coupling film
can also be applied between the transducer and the penis, to enhance
transmission of the ultrasound energy to/from the penis.
A fixing device is mechanically coupled to the transducer housing
and at least partially surrounds the penis, to hold the transducer housing
substantially steady and in place during an impotence diagnostic
procedure. According to one embodiment, the fixing device includes a
pressure balloon supported by a disposable housing, optimizing support of
and frictional contact with the penis. According to another embodiment,
an elastic strap is used to achieve this purpose. Alternatively, an adhesive
tape mounts the transducer housing on the top surface of the penis. The
fixing device according to the invention allows the apparatus to be secured
to the penis for an extended period of time, allowing the angle of incidence
to be substantially fixed and allowing continuous measurement and
recording of data throughout an entire diagnostic procedure.
The transducer or transducers are movably mounted on the
transducer housing, according to one embodiment, to allow lateral
adjustment of the direction of ultrasound transmission. According to
another embodiment the transducers are fixed within the housing. Using
multiple transducers allows simultaneous measurement, or at least
immediately sequential measurement, of penile blood flow in both
cavernosal arteries.
CA 02262789 l999-02-08
W O 98/0~33~ PCTrUS97/13537
The features of the invention summarized thus far can be used as
part of a larger diagnostic system according to the invention, including, for
example, monitors or other displays for continuously displaying velocity
data or other related data. Additionally, the larger system can be designed
to continuously record relevant data on a computer disk or a printer, for
example, for later review and analysis; real-time imaging is not necessary.
Both portable and desktop computer configurations are contemplated.
According to another aspect of the invention, a method of
diagnosing male impotence includes positioning at least one transducer
over the penis to transmit energy, such as ultrasound energy, into the
cavernosal arteries. The transducer is preferably supported by a housing,
either movable or fixed. The housing is secured to the penis with a fixing
device, such as a pressure balloon, elastic strap, tape, or equivalent. Once
the device is secured, blood velocity in the cavernosal arteries is measured
continuously, preferably before, during and after administration of a
vasodilating agent. According to one embodiment, the continuous
measurement data is saved to disk and/or continuously displayed on e.g. a
monitor screen.
To further aid diagnosis, the transducers can also be used to
measure penis diameter during the diagnostic procedure, according to the
invention. Graphical depictions of these penis diameter readings can
then be correlated and displayed in relation to the blood flow velocity
readings described earlier.
CA 02262789 1999-02-08
WO 98/06333 PCT/US97/13537
Embodiments of the invention provide significant advantages in
that they are office-based, relatively inexpensive, easy to use, relatively
independent of the operator, accurate over a wide range of velocities, and
able to make reproducible measurements to evaluate the effectiveness of
treatment over time.
BRIEF DESCRIPTION OF THE DRAWINGS
Embodiments of the invention will be described with reference to
the figures, in which like reference numerals denote like elements, and in
which:
Fig. 1 is an exploded perspective view of an impotence diagnostic
apparatus according to an embodiment of the invention;
Fig. 2 is a side view of the Fig. 1 apparatus;
Fig. 3 is an upper perspective view of an impotence diagnostic
apparatus according to an alternative embodiment of the invention;
Fig. 4 is a side view of the Fig. 3 apparatus;
Fig. 5 is a lower perspective view of the Fig. 3 apparatus;
Fig. 6 is a side view of an impotence diagnostic device according to
an alternative embodiment of the invention;
Fig. 7 is a perspective view of the Fig. 6 apparatus;
CA 02262789 1999-02-08
WO ~8/~6333 PCT/US97/13537
Fig. 8 is a top view of a transducer housing of an impotence
diagnostic apparatus according to an alternative embodiment of the
invention;
Fig. 9 is an end view of the Fig. 8 apparatus;
Fig. 10 is a top view of a transducer pair according to an
embodiment of the invention;
Fig. 11 is a top view of a transducer housing according to an
embodiment of the invention;
Fig. 12 is a side view of the Fig. 10 transducer pair;
Fig. 13 is a side view of the Fig. 11 transducer housing;
Fig. 14 is a perspective view of an alternative transducer housing
embodiment of the invention;
Fig. 15 is a perspective view of a transducer embodiment of the
invention;
Fig. 16 is a cross-sectional view of an impotence diagnostic apparatus
in a use position, according to an embodiment of the invention; and
Figs. 17-18 are views showing areas of ultrasonographic beam
intersection, according to an embodiment of the invention.
DETAILED DESCRIPTION OF PREFERRED EMBODIMENTS
As described earlier, one of the principal advantages according to
embodiments of the invention is that the elements used to
transmit/receive energy to detect Doppler frequency shift, e.g. ultrasonic
CA 02262789 1999-02-08
WO 98/06333 PCT/US97/13537
transducers according to one embodiment, are substantially fixed in
position with respect to the penis and to the cavernosal arteries. This
innovation in positioning defines and stabilizes the angle of incidence of
the energy on the blood flow path in the cavernosal arteries, allows precise
velocity measurements and, consequently, permits more accurate
diagnosis by the urologist or other medical professional. Additionally,
velocity measurements can be taken continuously, over an extended
period of time, eliminating uncertainty regarding the timing of velocity
measurements.
Embodiments according to the invention can be used to measure
blood flow in a number of different areas and parts of the body. Flow can
be measured in blood vessels of the arms, legs, fingers, neck, or other areas,
for example. Thus, while preferred embodiments of the invention will be
described with respect to the penis and to procedures for diagnosing
impotence, the invention should not necessarily be construed as limited to
those embodiments.
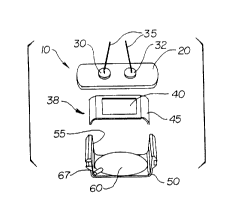
Turning to the figures, Figs. 1-2 illustrate an impotence diagnostic
probe apparatus 10 according to one embodiment of the invention. Probe
10 can be used as part of a larger impotence diagnostic system, as will be
described.
Probe 10 includes transducer housing 20 for supporting at least one
- transducer, such as transducers 30, 32, at a substantially fixed angle of
incidence. According to one embodiment, transducers 30, 32 transmit
. , , . . , , . . _
CA 02262789 1999-02-08
W O ~8/Of~33 PCTrUS97/13537
ultrasound energy, but any other transmit/receive scheme known to be
functional in a Doppler-shift detection environment can be used, for
example, other sonar-type or radar schemes.
Transducers 30, 32 (or their equivalents in non-ultrasound systems)
are set at respective angles with respect to the penis, to create a known
angular orientation with respect to the blood flowing in the cavernosal
arteries, that is, a known angle of incidence. Coaxial cables 35 or other
devices/systems connect transducers 30, 32 to electronics (not shown) that
direct the transmission and reception of the ultrasound signals, and
calculate and display Doppler frequency shift, blood velocity
measurements, and/or other data.
According to a preferred embodiment, each transducer 30, 32 is a
continuous wave, split-D transmitter/receiver. Each transducer is
effectively split into two separated sides, with one side continuously
transmitting and the other side continuously receiving. The split-D
configuration is particularly advantageous, because it eliminates many of
the transmitter-receiver angular alignments and other adjustments
necessary with otherwise separated transmitter/receiver pairs.
Transducers 30, 32 can also be used in what is known as an A mode,
using ultrasound or other energy to take various relevant distance
measurements. Thus, the changing diameter of the cavernosal arteries
can be measured and displayed, as can the diameter of the penis itself to
reflect changing penile tumescence. Visual confirmation of vessel
CA 02262789 1999-02-08
WO 98/06333 PCT/US97/13537
location is also provided. Measuring and displaying these values in
conjunction with blood velocity values over time provides the urologist
with an additional diagnostic tool. The A-mode measurement can also be
used for accurate alignment of the ultrasound (or other) beam with respect
to the cavernosal artery.
Of course, other transducer types and configurations are possible.
Single-transducer embodiments are contemplated, for example pulse-
wave piezoelectric devices in which a transmit mode is followed by a rest
mode, and then a reception mode for receiving the reflected energy. The
resulting combination of pulses requires the urologist to precisely set the
focus depth, a requirement that is somewhat disadvantageous for the
reasons described earlier. Whatever transducer arrangement is used, the
angle of incidence is mechanically defined and substantially fixed with
respect to the blood flow direction in the cavernosal arteries.
A continuous-wave ultrasound or other-type system with separate
transmit and receive transducers, one on top of the penis and one on the
bottom, is also contemplated. One particular example according to this
embodiment will be described below with respect to Fig. 10.
Two transducers 30, 32 are illustrated in Fig. 1, one for each
cavernosal artery. Using two transducers allows measurement, display
and comparison of blood flow velocity in both arteries, either sequentially
or simultaneously, both before and after vasodilation. Continuous peak
systolic and end diastolic velocities, as well as accelerations, can be
CA 02262789 1999-02-08
WO 98/06333 PCTIUS97/13537
measured/calculated and displayed with respect to both arteries.
Alternatively, of course, a single transducer or more than two transducers
also can be used, to measure flow in various vessels at various locations.
For example, an apparatus according to the invention also can be used to
take measurements with respect to the urinary tract.
Transducers 30, 32 preferably are off-the-shelf components available
on the open market from companies such as Blatek, Inc., of State College,
Pennsylvania. Such ultrasound transducers typically have concave lenses
in front, which focus the sound waves at the desired depth, here, in this
case, the depth of the cavernosal artery.
Supplemental lens(es) may also be used to bend the ultrasound
beam to the desired angle of incidence. According to one embodiment, the
supplemental lens(es) are disposed in the air gap that otherwise would be
created between the transducer and the patient's body, reducing the
amount of gel or other acoustic filler material needed there. In accordance
with Snell's law, the difference in the speed of sound between the lens~es)
and the patient's tissues affects the amount of bend in the ultrasound
beam as it passes into the tissues. Thus, the supplemental lens(es) allow
the angle between the housing of the transducer and the tissues to be
shallower than e.g. 60 degrees, providing a more compact overall
mechanism.
As mentioned above, an acoustic filler material is desirable for use
with embodiments of the invention. Because air is a relatively poor
14
CA 02262789 1999-02-08
WO 98/06333 PCT/US97/13537
conductor of ultrasonic energy, a filler material is generally needed to
direct the ultrasound into the patient's tissues with minimal attenuation.
One such material suitable for use according to the invention is a
non-invasive hydrogel skin pad from LecTec Corporation, Minnetonka,
Minnesota. As shown in Figs. 1-2, skin pad 38 includes film material 40
surrounded by border 45. According to a preferred embodiment, both the
top and bottom sides of film 40 and border 45 are sticky, to securely adhere
to the surface of the penis. Pad 38 preferably is a disposable, single-use
component, to promote cleanliness and reassure the patient that parts of
probe 10 in direct contact with the penis are sanitary/sterile.
Instead of or in addition to pad 38, it is also possible to use a
medium- or high-viscosity ultrasound transmission gel such as
OMNIGEL~) from Echo Ultrasound, Reedsville, Pennsylvania.
Once the urologist or other medical professional places pad 38 (or
other filler) directly on the top of the penis, transducer housing 20 is placed
on top, with transducers 30, 32 directly contacting film 40 for optimal
acoustic coupling. The medical professional then places a fixing device
around the lower surface of the penis to secure the transducer housing in
place over the penis. According to the embodiment of Figs. 1-2, the fixing
device comprises disposable lower housing 50, which is secured to
transducer housing 20 in a manner to be described.
Disposable lower housing 50 includes two upstanding sidewalls 55,
between which is supported disposable pressure balloon 60. Pressure
CA 02262789 1999-02-08
WO 98/0~333 PCT/US97/13537
balloon 60 receives the patient's penis and conforms to its shape,
expanding along the base of housing 50 and even up sidewalls 55 as
needed. As the penis increases in size during periods of erectile response,
pressure balloon 60 maintains the penis in substantially constant pressure
contact with transducers 30, 32, via film 40. Balloon 60 holds the penis in
position as it grows, such that side-to-side shifting is substantially
prevented. A substantially fixed relationship between the penis and
transducers 30, 32 thus is maintained.
According to one embodiment, balloon 60 has slots on opposite
ends thereof, to receive and accommodate sidewalls 55 of housing 50.
During assembly of the apparatus, balloon 60 is merely slipped into
position over sidewalls 55 for a secure and steady fit on lower housing 50.
Balloon 60 preferably forms a substantial U-shape against the base of
housing 50, riding up sidewalls 55, to center and comfortably hold the
penis within housing 50. Balloon 60 maintains substantially constant
pressure between the transducers and the penis, without occluding blood
flood.
According to one embodiment, balloon 60 includes balloon fill
valve 63 and constant pressure relief valve 67, to maintain a constant,
desired pressure in balloon 60. Alternatively, valves 63, 67 need not be
used, to eliminate the number of parts associated with balloon 60 and the
associated increased complexity and cost.
16
CA 02262789 1999-02-08
W O 98/06333 PCT~US97/13537
As shown in Fig. 2, lower housing 50, pad 38 and transducer
housing 20 are connected together by linking portions 70 of sidewalls 55.
Linking portions 70 include central recesses 73 and opposite tapered
portions 77, permitting easy insertion into and retainment within
corresponding slots (93, shown in Fig. 3) through transducer housing 20.
Linking portions 70 preferably are one piece with sidewalls 55.
Alternatively, transducer housing 20 and/or sidewalls 55 of lower housing
50 can include structure providing vertical adjustability with respect to
each other, to adjust the size of the space between lower housing 50 and
transducer housing 20. This structure can take the form of a toothed
section extending toward housing 20 on or near sidewalls 55, for example.
A corresponding section, in the form of a gear, ratchet wheel, and/or pawl,
for example, is supported by transducer housing 20 and matingly engages
the toothed section associated with sidewalls 55. This structure locks lower
housing 50 in a desired position with respect to transducer housing 20, yet
allows easy adjustability.
Housings 20, 50 and link members 70 preferably are formed of
injection-molded plastic, but of course other suitable materials, such as
nylon or various polymers or metals, for example, also can be used.
Figs. 3-5 show an alternative probe embodiment 10', in which the
fixing device for securing transducer housing 20 in place comprises strap
90, such as an elastic, paper, and/or cloth type strap, optionally with e.g.
Velcro, tape or other type fastening devices for achieving a snug fit around
CA 02262789 1999-02-08
WO 98/06333 PCT/US97/13537
the penis. Ends 97 of strap 90 pass through slots 93 in transducer housing
20, allowing easy adjustment into frictional engagement with the penis.
Although strap 90 according to this embodiment is potentially constrictive
and may not be as comfortable for the patient as the previously described
embodiment, it has the advantage of being simple to adjust and simple to
manufacture. It also is disposable, like housing 50 of the previous
embodiment. Pad 38 is also useable in connection with this embodiment,
but is not illustrated to simplify the drawings.
Other devices for securing transducer housing 20, or a similar
transducer support, are contemplated. For example, a double-sided
adhesive tape can be applied to the top side of the penis, without
necessarily extending to the underside of the penis, to hold the transducers
in place.
Whatever fixing device is used, it is desirable to position
transducer(s) 30, 32 to straddle the dorsal blood vessels extending along the
top of the penis, providing a relatively unobstructed ultrasound path to
the cavernosal arteries. To this end, transducer housing 20 can include a
central hinge. The two pivoting legs of housing 20 thus created permit the
transducers to be positioned as desired, for example at 10 o'clock and 2
o'clock positions, around the upper circumference of the penis. The angle
of incidence, however, remains fixed at e.g. 60~.
Figs. 6-7 show an alternative diagnostic probe 100, which includes a
transducer housing 20 that is similar or identical to the previous
18
CA 02262789 1999-02-08
wo g8~lC'~33 PCTrUS97/13537
embodiments. Probe 100, however, additionally includes spring
mechanism 110 to urge lower housing 50 into engagement with the penis,
as will now be described. Spring mechanism 110 includes compression
springs 130, connected to upstanding ends 120 of lower housing 50 via
connecting pieces 140. Springs 130 bias connecting piece 140 and
upstanding ends 120 vertically upward, urging lower housing 50 towards
transducer housing 20 to compress the penis therebetween. Springs 130 (as
well as strap 90 of the previous illustrated embodiment) are of course
governed by Hooke's law; thus the pressure between the transducer and
the penis changes depending on the size of the penis and consequent
displacement of springs 130.
Figs. 8-9 show an alternative transducer housing embodiment.
Transducer housing 170 defines an open frame and includes
longitudinally extending grooves 180, into which fit corresponding tongue
members 190 of individual transducers 200. Transducers 200 thus are
slidable within transducer housing 170 into a desired position with respect
to each cavernosal artery as needed, e.g. to adjust to a particular patient's
anatomy. Thus, the individual transducers are laterally adjustable, while
the housing maintains the angle of incidence substantially constant at
about 60~, or another desired angle.
Figs. 10-13 show a more detailed example of this type of structure.
Transducers 205 are supported by transducer housing 210, which includes
longitudinally extending rails 215. Transducers 205 include respective
19
CA 02262789 1999-02-08
WO 98106333 PCT/US97/13537
subhousings 220 with substantially rigid but bendable fingers 225, which
carry riders 230 for slidably engaging rails 215. Fingers 225 can be squeezed
together, substantially disengaging riders 230 from rails 215. Upon release,
fingers 225 snap back to their original positions, creating a friction fit
between riders 230 and rails 215. According to one embodiment (not
shown), rails 215 and riders 230 are formed with a series of mating ridges
or teeth extending along their lengths, to secure the transducers in a
substantially fixed lateral position in housing 210 yet readily permit
disengagement of riders 230 and rails 215 for lateral adjustment.
Transducer housing 210 also includes slots 213 for receiving upstanding
sidewalls 55 as shown in previous figures, for example. Housing 210 and
subhousing 220 can be made of a plastic-type material or the like.
Fig. 14 shows an alternative embodiment of transducer housing 210.
Transducer housing 210 includes a central bend 240 that is substantially
fixed to form a desired angle, e.g. 150~. Each "leg" of transducer housing
210 thus drops about 15~ below the horizontal or other reference plane.
Central bend 240 positions transducers 205 at about 10 o'clock and 2 o'clock
on the penis, avoiding interference with the dorsal blood vessels and also
reducing the potential "teeter-totter" effect present with a straight
transducer housing 210. The angle of incidence remains fixed at e.g. 60~.
Transducers 205 also preferably inc}ude arrows or sirnilar markings 245 to
indicate proper orientation of transducer housing 210 with respect to the
CA 02262789 1999-02-08
WO 98/06333 PCT/US97/13537
patient. Transducer housing 210 can be positioned, for example, such that
arrows 245 point away from the patient.
As shown in Fig. 15, according to one embodiment, transducers 205
include gap 250 for accommodating gel 255 or similar acoustically
conductive material, dispensed by a suitable dispenser 257. Gap 250 should
be completely filled with gel 255, to prevent air bubbles which block
transmission of ultrasound signals. Gel 255 can be applied before
transducer 205 is inserted into transducer housing 210, or afterwards.
Other aspects of the Figs. 10-15 embodiments are as shown and
discussed with respect to previously described embodiments.
Fig. 16 shows an alternative diagnostic probe embodiment 260,
which has separated transmit and receive transducers, as discussed earlier.
Probe 260 is of relatively simple construction, with one or two elastic straps
265 secured together at ends 270 by a tightening and securement device 280.
Straps 265 hold transducers 290, which according to one embodiment are
transmit transducers, in a desired, substantially fixed position on the top
surface of penis 300 relative to cavernosal arteries 310. Elastic straps 265
also hold transducers 320, e.g. receive transducers, in a substantially fixed
position on the lower surface of penis 300. According to one embodiment,
penis 300 is wrapped with a somewhat sticky cloth or cloth-like material
305 before bands 265 are tightened, to improve patient comfort and better
hold the bands and transducers in place. Elastic bands 265 provide
__ ._. " . _ , . ....
CA 02262789 1999-02-08
W O 98/06333 PCT~US97/13537
compression and expansion as needed to accommodate the changing size
of the penis.
Alternatively, of course, straps 265 can be used to hold just two
transducers, or even a single transducer, instead of the four separate
transducers illustrated in Fig. 16. Although significant mechanical angular
adjustment is necessary with this embodiment to achieve the desired
angle of incidence and optimal beam positioning, this embodiment is of
simple and low-cost construction.
Figs. 17-18 show transmit and receive transducer paths 330, 340,
associated with transmit and receive transducers 290, 320 of Fig. 16, for two
angles of incidence: 60 degrees and 72 degrees, respectively. Paths 330, 340
define intersection regions 350, which vary in size and positioning
depending upon the angle of incidence, and which are positioned to
intersect the cavernosal artery in which blood flow velocity is being
measured.
The angle of incidence depicted in Fig. 17, 60 degrees, is preferred.
As angle of incidence increases, Doppler shifting effect diminishes, to the
point where at 90 degrees, there is zero Doppler shift. As the angle of
incidence decreases, on the other hand, the sonographic or other energy
must travel through tissue for a greater distance before hitting the
intended target area. That travel is highly attenuative, and signal loss
ultimately occurs. 60 degrees has been found to be a preferable angle of
incidence to minimize these disadvantages.
CA 02262789 1999-02-08
W 0 ~8/0~33 PCTrUS97/13537
As indicated above, the electronics associated with the chosen probe
embodiment perform a wide variety of desired calculations and display
graphical representations of a wide variety of variables, as chosen by the
urologist or other medical professional. Control electronics components
useable according to the invention can be purchased from a variety of
companies. A graph of velocity versus time for both cavernosal arteries
can be displayed, either simultaneously or sequentially at the direction of
the medical professional, during the course of the procedure or afterwards.
Additionally, the velocity displays can be correlated with
electrocardiogram and/or pulse oximetry readings to depict variations in
blood velocity with respect to heartbeat. Penile diameter readings can also
be displayed and correlated.
The displayed graphical information can be in a number of different
formats on the display screen. For example, a spectral distribution
graphical mode displays a gray scale that represents all of the Doppler
frequencies. The greater the shift in frequency, the greater the distance
from the baseline on the display screen. In a mean frequency mode, the
statistical mean of the Doppler frequencies is displayed, for example as a
colored line. In the show index mode, pulsatility, resistance, spectral
broadening and heart rate values are displayed. The maximum frequency
mode displays maximum frequency shift, for example as a colored line.
Finally, the mode frequency graphical mode displays the single frequency
. . .. . . ...
CA 02262789 1999-02-08
WO 98/06333 PCT/US97/13537
that occurs most often during a sample, again as a colored line, for
example.
The spectral distribution, maximum frequency and mean frequency
displays can be used simultaneously to determine signal quality. When
adjusting the transducer(s) to measure velocity in the cavernosal arteries, a
maximum gray-scale (spectral distribution) signal should be displayed. If
the signal is of a good quality, the maximum frequency display and the
mean frequency display should follow the wave form displayed by the
spectral distribution. The mean frequency should be lower than the
maximum frequency, and the distance between the two should be
relatively constant.
Although the probe according to the invention can be placed at any
location on the shaft of the penis, it is preferably placed as close as possibleto the base of the penis during the diagnostic procedure, allowing full
accessibility for vasodilator introduction or self-stimulation. This
placement also allows for duplex scanning to be conducted in the
remainder of the penis, to look for damage within the penis as needed.
Embodiments of the invention, therefore, provide optimum penile
accessibility. Placement at the base also provides greater stability. Proper
initial placement can also be verified by using headphones or external
speakers to broadcast the "whooshing" sound detected as the ultrasound
intersects the cavernosal arteries.
24
CA 02262789 1999-02-08
WO 98/0~333 PCT/US97/13537
For the typical patient, it is fair to assume that the cavernosal
arteries run
parallel to the surface of the penis and actual blood flow velocities can be
determined. Even in the irregular case, where the cavernosal arteries
deviate from true parallel with the surface, the medical professional can
make an accurate diagnosis, according to embodiments of the invention.
Because the angle of incidence is held substantially constant during the
time of the diagnostic procedure, impotence or health can be diagnosed
with reference to velocity ratios, i.e. the percentage increase in blood
velocity over time, eliminating the need to know the angle of incidence
ultra-precisely. Relative velocity calculations are independent of the angle
of incidence, but only when the angle is kept constant by mechanical
fixturing during the diagnostic procedure as with embodiments of the
invention.
CA 02262789 1999-02-08
W O 98/06333 PCTrUS97/13537
Acoustic Output Levels
According to one continuous-wave model, the following holds true:
Max. Value W0 fc ZSP X 6,Y 6 EPD
l5MA3 78 (+22.6/-14.0) 1.6 (+0.72/-0.29) 7.85 MHz llrrrn 1.0(=0.12/-0), .76x.38cm
mW/om2 nUV 0.8(+0.10/0)
r~n
The values in parenthesis are measurement errors calculated by
adding worst case error values for hydrophone, measurement, calculation
and position uncertainties, using the "sum of squares" method.
Velocity Ranges
Maximum range of velocities detectable at maximum Doppler
frequency display range:
+ 32kHz Doppler shift at 0~: + 3 meters/second (+ 7 cm/s)
+ 32kHz Doppler shift at 30~: + 3.5 meters/second (+ 40 cm/s)
Explanation of Terms
Max. Value The maximum values given above were derated from
intensities measured in water. For the derated intensities,
tissue attenuation is taken into account. Tissue
attenuation is estimated at 0.3 dB / cm x MHz.
Attenuation is calculated for the focal depth, i.e. the point
of highest ultrasound intensity (Zsp). The spatial peak
temporal average intensity (ISPTA) measured in water is
26
SUBSTITUTE SHEET (RULE 26)
CA 02262789 l999-02-08
W 098/Q~333 PCTrUS97/13537
decreased by the loss due to attenuation which leads to the
derated value (ISPTA3)
W0 This value represents the overall acoustic power delivered
by a transducer and is calculated by integrating the output
intensity over the complete ultrasound beam.
fc The center frequency of the transducer. This value is
calculated from a Fourier analysis of the spectrum
produced by a transducer and can differ slightly from the
actual working frequency.
ZsP This is the axial distance that is used to calculate the
aerated intensity. It is the distance from the transducer
surface at which the peak value for the derated intensity
occurred.
X 6, Y-6 The dimensions of the ultrasound beam measured at the
distance of ZsP. The values indicate the width of the beam
with boundaries defined by the acoustic intensity being 6
dB less than the maximum intensity.
CA 02262789 1999-02-08
W O 98106333 rCT~US97/13537
EBD Entrance beam dimensions. The dimensions of the
ultrasound beam at the surface of the transducer.
Transducers have a "split-D" configuration; transmitting
and receiving elements each have the form of a half-circle.
Numbers represent diameter and radius of the half-circle.
While the invention has been described with respect to preferred
embodiments, the invention is not intended to be limited to those
embodiments. Various modifications and changes will be readily apparent
to those of ordinary skill upon reading the application. Many elements of
the various embodiments described above can be mixed and matched. For
example, pad 38 can be used with the embodiment of Figs. 8-9, even
though such use is not specifically described in the text. Film 40 and
border 45 can also be applied separately instead of in a one-piece pad.
Further, as referenced earlier, embodiments of the invention have
application to other areas of the body and other diagnostic procedures.
Various other modifications and changes may occur to those skilled in the
art without departing from the spirit and scope of the invention as defined
in the following claims.
28