Note: Descriptions are shown in the official language in which they were submitted.
CA 02298806 2000-02-15
r
A GAS CONCENTRATION METER AND INSULATING GLASS ASSEMBLY
AND METHOD THEREOF
- 5 Field of the Invention
This invention relates in general to a system for measuring a gas sealed
within an insulating glass assembly, and more particularly a gas concentration
meter
and method used for measuring the concentration of a gas within a sealed
cavity or
to window assembly and a means of providing access to the sealed cavity.
Background of the Invention
Due to the high cost of heating and cooling in most building structures, high
15 efficiency and energy saving means are desirable for fenestration systems
such as
windows, doors, curtain walls, skylights or the like. Typically such systems
include
sealed insulating glass units filled with an inert gas. Traditionally these
insulating
systems have had an insulating air space or vacuum between the panels, which
recently included a gas such as Argon to improve thermal performance. The
2o inclusion of the gas is to provide additional insulation or resistance to
thermal
conductivity. Argon is the gas predominantly used due to its high benefit/cost
ratio.
Over time the gas may escape or dissipate due to faulty seals of the like,
which negates the beneficial effects of the insulating windows and results in
2s increased energy costs. Due to the nature of the gases involved as
described above,
it is difficult to determine how much of the gas has escaped, and therefore
what
concentration of gas is left in the glass assembly. Furthermore if the gas
concentration is found lower than the promised or the pre-selected level, the
sealed
insulating glass unit cannot generally be replenished without destroying the
frame or
3o seal of the fenestration system.
CA 02298806 2000-02-15
-.5 ,
-2-
Several methods for measuring gas concentration in insulating glass units do
exist today. However, such methods require sampling and analysis of the gas in
the
insulating glass unit's cavity. The use of such methods is severely limited,
as the
sampling of gas within the cavity cannot be performed on an insulating glass
unit
" 5 once it is glazed in place. Once the insulating unit is glazed in place,
the edge seal
becomes inaccessible. Any attempt to carry out sampling of the insulating
glass unit
is made at the risk of damaging the edge seal, which will promote insulating
unit
failure in a very short period of time.
to Presently, buyers of gas-filled sealed units can only rely on the
supplier's
assuring words that the supplied units do contain insulating gas within the
insulating
cavity. As the fill gas is colorless and odorless, and inaccessible, there is
no means
of verifying such claims. Currently, insulating gas unit manufacturers may be
certified that they comply with National Standards regarding gas concentration
test
t5 requirements. Due to the lack of a more practical test method, the only
reliable
method that has been adopted by the Standard is gas chromatography. This
method,
however, requires sampling of gas in inaccessible cavity, and use of highly
specialized analytical equipment and technicians to operate under strict
laboratory
conditions .
Current method of determining gas concentration involves measuring of
Oxygen concentration in the cavity as it relates to presence of air. Other
methods
(under development) may be used to measure the concentration of a gas within
the
sealed units consist of applying a high voltage electrical current in the
range of
2s 10,000 to 20,000 volts across the glazing cavity (i.e. across the outside
surfaces of
the spaced glass).
Therefore, while the existing methods of determining gas concentration in
insulating glass units can be used for laboratory research and testing of non-
glazed
CA 02298806 2000-02-15
-3-
sealed units, these methods cannot be employed effectively for routine quality
control and field-testing purposes.
Furthermore, the ability to access and measure the level of gas within the
glass assembly would allow the installer to add more gas when required and
therefore maintain the insulating benefits of the glass.
Prior art glass assemblies have been devised to try and address the
aforenoted problems. For example, United States Patent No. 5,299,399 which
to issued on April 5, 1994 to the inventors Bruce A. Baier et al. and was
assigned to
Pella Corporation of Pella, Iowa. This patent relates to a dual glazing window
that
has a removable glazing panel, which includes a breather system that connects
the
air chamber between the glazing panels with the outside ambient air by routing
an
outwardly facing channel into the window rail.
United States Patent No. 2,880,475 issued on April 7, 1959, and relates to a
double glazed window unit suitable for use in a window, in which the space
between
the glass panels is evacuated to provide good insulating qualities and to
prevent the
formation of condensation due to temperature differences between the inner and
outer panels.
United States Patent No. 2,125,372 was issued on August 2, 1938 and
relates to a fitting for a glass sheet having a perforation and comprises of a
metal
connection plug having a threaded passage, which fits into the perforation for
2s sealing the glass sheet.
United States Patent No. 1,336,211 was issued on April 6, 1920 to Henry
Duffy of Longmont, Colorado and relates to an exteriorly threaded plug having
a
central port and a lateral passage with a valve seated in the passage, and
having a
3o transverse port.
CA 02298806 2000-02-15
-4-
Thus a gas concentration meter used for measuring the concentration of a gas
within a sealed insulating unit or glass assembly and method thereof is
desirable.
Furthermore a non-obtrusive means of providing access to the insulating cavity
to
s measure and/or replenish a gas within the cavity would be beneficial.
Summary of the Invention
An object of one aspect of the present invention is to provide a gas
Io concentration meter and improved insulating glass assembly and a method
thereof.
In accordance with one aspect of the present invention there is provided a
system for measuring the concentration of a gas within a sealed insulating
cavity
having an access means for communicating with the sealed insulating cavity, a
15 probing means for insertion through the access means to the gas and having
the
ability to sense the concentration of the gas, and a measuring means for
measuring
and displaying the concentration of the gas through the probing means.
Conveniently, the access means is further defined as an access port assembly
2o having a connecting means through the glazing spacer block and a stopping
means.
Preferrably, the connecting means spacer block may be defined as a
connecting tube being integrally molded to a spacer block for insertion
between the
glass panes of the insulating unit.
In accordance with another aspect of the present invention, the probing
means may be further defined as a pair of flexible electrodes which may be
separated by a predetermined distance to form a gap.
CA 02298806 2000-02-15
-5-
In accordance with still another aspect of the invention, the measuring means
may be defined as a meter having electronic circuitry to excite, control,
monitor the
excitation level, relate the excitation level to gas concentration through
calibration
means, and indicate or display the concentration level.
Another object of the present invention is to provide an improved method for
measuring the concentration of a gas in a gas filled sealed insulating glazing
assembly comprising of at least two spaced glass panes wherein the method
comprises the following steps:
to
a) inserting a probing means through an access means for communication with
the cavity of the gas filled insulating unit;
b) or placement of permanent probing means into the glazing cavity through the
edge seal of the insulating unit;
c) or placement of permanent probing means in a gas sampling device such as a
syringe or any other sampling container;
d) sensing the concentration of the gas with the probing means; and relaying
the
concentration of the gas to a measuring means for measuring the
concentration of the gas.
2s Advantages of the present invention are:
a) ability to measure actual gas concentration on site or at the plant,
b) ability to pressure equalize the glazing cavity in order to eliminate
3o glass distortion or breakage, and
CA 02298806 2000-02-15
-6-
c) ability to re-fill the insulated units with gas if gas-filling was deemed
inadequate, thereby reducing the number of gas-filled units that have
to be replaced.
Brief Description of the Drawings
A detailed description of the preferred embodiment is provided herein below
by way of example only and with reference to the following drawings, in which:
Figure 1 in a side cross-sectional view, illustrates a conventional insulating
glass assembly with the meter;
Figure 2 in a side cross-sectional view, illustrates the insulating glass
1s assembly in accordance with the preferred embodiment of the present
invention;
Figure 3 in a perspective view, illustrates the system in accordance with the
preferred embodiment of the present invention.
2o Figure 4 in a top plan view, illustrates the meter in accordance with the
preferred embodiment of the present invention.
Figure 5 in a cross-sectional view, illustrates the probing means being
inserted into the access means in accordance with the preferred embodiment of
the
25 present invention.
Figure 6 in a cross-sectional view, illustrates the probing means being
permanently installed into the glazing cavity in accordance with the preferred
embodiment of the present invention.
CA 02298806 2000-02-15
Figure 7 in a top plan view, illustrates the circuitry in accordance with the
preferred embodiment of the present invention.
In the drawings, preferred embodiments of the invention are illustrated by
s way of example. It is to be expressly understood that the description and
drawings
are only for the purpose of illustration and as an aid to understanding, and
are not
intended as a definition of the limits of the invention.
Detailed Description of the Preferred Embodiment
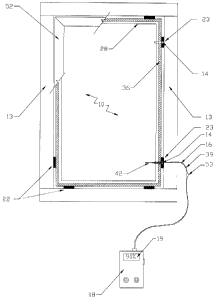
Referring to Figures 1 and 2, there is illustrated in a cross-sectional side
view, a system for measuring the concentration of a gas within a sealed
insulating
cavity 10 comprising of a sealed insulating glass units 11 , defined by two
glass
panes 12 separated by edge spacer 28 and edge seal 36, in accordance with the
~ s preferred embodiment of the present invention.
The system 10 includes an access means 14 for communicating with the
sealed cavity 32 of the sealed insulating glass units 11, a probing means 16
for
insertion through the access means 14 to the gas filled sealed cavity 32 and
for
2o sensing the concentration of the gas, and a measuring means 18 for
measuring and
displaying the concentration of the gas that is sensed through the probing
means 16.
The access means 14 may be further defined as an access port assembly 20,
whereby the access port assembly 20 can comprise of a connecting means spacer
2s block 23 and a stopping means 24. The connecting means spacer block 23 may
include a connecting tube 26 that may be integrally molded to a conventional
spacer
block 22. The connecting tube 26, permanently inserted and sealed in passage
30
made through the edge seal 36 and edge spacer 28, connects the cavity 32
between
the glass panes 12 to the outside environment. More particularly the passage
30
CA 02298806 2000-02-15
_g_
may be a 90-degree bore 34 made through the spacer block 23 which terminates
at
the connecting tube 26.
The stopping means 24 may be further defined as a resealable knock-out cap
s 37 which allows for temporary access to the cavity 32 via the passage 30 and
the
connecting tube 26. The spacer block 23 with connecting tube 26 may be made
from a variety of materials and may be employed in any number positioned at
any
location around the perimeter of the sealed insulating glass unit 11 being
used.
to Alternatively, system 10 includes a probing means 16 permanently
incorporated into the gas filled insulating glass unit 11 by means of
insertion
through the edge seal 36 for sampling and sensing the concentration of the
gas, and
a measuring means 18 for measuring and displaying the concentration of the gas
that
is sensed through the probing means 16.
is
The probing means 16 may be further defined as a pair of flexible electrodes
38. The flexible electrodes 38 (by way of example 26 Gage fine electrical
wires)
may be separated from one another by a pre-determined distance to form a gap
40.
More specifically the flexible electrodes 38 may be in the form of a ribbon
having
2o exposed tips 42 or sensing means at one end that may be placed within the
cavity
32, while the other end of the flexible electrodes 38 are connected to the
measuring
means 18 by means of electrical connector 53.
The flexible electrodes 38 may be made from a variety of materials and may
2s have different distances for the gap 40 as the capacities, calibration and
environment
for the flexible electrodes 38 can vary. The location and connection of the
flexible
electrodes 38 can also vary. More specifically, the flexible electrodes 38 may
be
integrated with the spacer block 23 so that the sensing means 42 of the
flexible
electrodes 42 extend from the connecting tube 26 of the spacer block 23 and
the
CA 02298806 2000-02-15
-9-
exposed end 39 of the flexible electrodes 38 can be connected into the
measuring
means 18.
The sealed insulating glass units 11 may be contained within a frame 13,
s such as a window or doorframe, and secured within the frame 13 by a
removable
glazing stop 52. For measuring gas concentration in glazing cavity 32 or gas
replenishing should the gas concentration within the insulating unit get too
low, the
glazing stop 52 may be removed to reveal the stopping means 24 located at the
edge
of the spacer block 23. The glazing cavity 32 may be accessed by removing the
stopping means 24. Following gas concentration measurement or gas
replenishment, the stopping means 24 can then be placed back and resealed.
Alternatively, if a permanent probing means 16 is employed, the glazing stop
52
may be removed to reveal the exposed end 39 of the flexible electrodes 38
which
can be connected into the measuring means 18 for sampling and measuring the
gas
15 concentration.
The measuring means 18 may be further defined as a meter 44 having
electronic circuitry 46. The meter 44 excites the sensing end 42 of the
probing
means 16, controls, monitors the excitation level, relates the excitation
level to gas
2o concentration through calibration means, and indicates or displays the gas
concentration level. Through the electronic circuitry 46, and more
specifically a
circuit board 50, an excitation signal is applied to the flexible electrodes
38 so as to
create an arc therebetween at the sensing means 42. The meter 44 may be
developed into a hand-held instrument, battery operated or an electronic
stationary
2s device. In general, meter 44 could be operated by a battery requiring one
or more
1.S.V AA size batteries. The concentration of the gas may be calculated
through
calibration and scaling circuit, and displayed on the meter 44 through a
LCD/LED
display 19 or through a variety of other visual display methods such as
digital,
graphic, analogue or interfaced with a computer for logging or process
control. The
3o concentration of the gas may also be indicated visually by a colour change
of
CA 02298806 2002-08-07
tl7
chemically sensitized medium or identifier or through a sound effect such as a
buzzer.
The meter 44 may be calibrated to read a variety of gases such as Argon,
Krypton or any mixture of inert gases. Calibration of the meter 44 is
conducted
using the type of gas to be tested and either standard hardware or software
such as
an embedded programmed chip
in operation, the method for measuring the concentration of a gas the
probing means 16 creates an arc within the sealed insulating cavity 32 having
electrical breakdown voltage proportonate to the concentration of the gas as
governed by Paschen's law described below.
Paschen's law essentially states that the breakdown voltage characteristics of
a
gap are a function (generany not linear) of the product of the gas pressure
and the
gap length. usually written as V = f (p b) where "p" ~s the pressure and "d"
~s the
gap distance. In actuality, the gas density should replace the pressure
Paschen's law states that the breakdown voltage mil remain wnstant as long
as the gas density and gap length remain constant. By way of example, the
breakdown voltage is a constant where the gap length is halved and the gas
density
is doubled. Therefore the breakdown voltage will be dependent on the gas
density
and on the type, shape and spacing of the flexible electrodes 38 at the
sensing
means 42. Knowing the breakdown voltage of the arc developed across the
electrodes 38 at the sensing end means 42 having known electrode properties,
the
gas concentration is thus determined.
Referring to Figure 3, the user would temporarily remove the glazing stop
52 to reveal the stopping means or knockout cap 24 located at the side of the
spacer
block 23. The stopping means 24 may be removed and the probing means ~6 or
CA 02298806 2000-02-15
-11-
flexible electrodes 38 may then be inserted into the passage 30. As the
flexible
electrodes 38 are flexible, they will bend around the 90-degree bore 34
through the
spacer block 23 and the connecting tube 26 to the cavity 32 located between
the
glass panes 12. Alternatively, in permanent electrode installation, the
electrodes 38
s extend outside of the edge spacer 28 of the sealed unit as previously
described.
Upon placing the flexible electrodes 38 within the cavity 32, the sensing end
42 is excited, so that arcing occurs across the gap 40 between the exposed
tips 42 of
the flexible electrodes 38 through the gas. The electronic circuitry 46 and
more
to specifically the circuit board 50 controls the excitation level through
circuit 54, and
measures the breakdown voltage and the current through the electrodes 38
across the
electrode gap 40 at the sensing end 42.
Once the breakdown voltage is stabilized through means of circuit 55, the
15 gas concentration can then be sampled and calculated by calibration circuit
56 from
the breakdown voltage relayed to the meter 44 by the flexible electrodes 38
and
other parameters as governed by Paschen's law and derived through laboratory
calibration. The results may be displayed on the meter through circuit 57 and
a
variety of display modes outlined above and in a variety of measurements such
as
2o percentage, parts per million etc.
The accuracy of the method and measurements may be improved by
repeating the excitation of the sensing means 42 of the electrodes 38 several
times
and averaging all of the measured individual breakdown voltage values.
Averaging
25 of the results may be done automatically by programming the circuit's
controller
chip.
Other variations and modifications of the invention are possible. All such
modifications or variations are believed to be within the sphere and scope of
the
3o invention as defined by the claims appended hereto.