Note: Descriptions are shown in the official language in which they were submitted.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98107806
POLYAMINE TRANSPORT INHIBITORS
FI : .D OF THE INVENTION
The present invention relates to a novel class of competitive inhibitors of
natural
polyamine transport in mammalian cells. The present invention is more
particularly
directed to low molecular weight, high-affinity, specific, impermeant, pure
antagonists of
polyamine transport of a structure different to that of endogenous polyamines.
The novel
inhibitors of the present invention exhibit an effect on cultured tumor cells
essentially
cytostatic, with minor non-specific effects. The present invention is also
directed to the use
of such novel inhibitors of polyamine transport to evaluate the antitumor
efficacy of
polyamine depletion strategies with minimal systemic cytotoxic effects or to
control and
treat disorders involving unrestrained cell proliferation and/or cell
differentiation wherein
polyamine transport is required.
BACKGROUND OF THE INVENTION
Natural polyamines such as putrescine (1,4-butane-diamine), spermidine (1V 3[-
aminopropyl]-1,4-diaminobutane) and spermine (N,N'-bis-[3-aminopropyl]-1,4-
butane-
diamine) play essential roles in the control of macromolecular synthesis and
growth
processes in eukaryotic cells. Cells maintain appropriate polyamine
concentrations
principally by de novo synthesis from amino acids wherein ornithine
decarboxylase
catalyzes conversion of ornithine to putrescine, which is then converted to
spermidine and
spermine. Most tissues also possess a specific plasma membrane transport
system allowing
for utilization of plasma sources of polyamines.
Inhibitors of polyamine biosynthesis such as a-difluoromethylornithine (DFMO),
which inhibits ornithine decarboxylase, cause an extensive depletion of
polyamines
followed by growth arrest in virtually all known mammalian cell types in
vitro. Since
tissues such as tumor cells and other transformed or rapidly proliferating
cells exhibit a
1
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
tissues such as tumor cells and other transformed or rapidly proliferating
cells exhibit a
high demand for polyamines, these properties have encouraged an extensive
assessment of
such inhibitors for the treatment of proliferative diseases, including several
types of tumors,
in experimental models and in clinical trials. Unforhinately, the antitumor
efficacy of such
inhibitors in vivo has been disappointing. The failure of DFMO to halt tumor
growth in
animal models has been clearly correlated with the elevated polyamine
transport activity
found in transformed cells. Indeed, decontamination of the gastrointestinal
tract, which is
the main vector of circulating polyamines through bacterial microflora
activity, along with
a polyamine-free diet, markedly potentiate the in vivo efficacy of DFMO
against tumor
progression. Moreover, mutant mouse leukemia cells deficient in polyamine
transport are
much more susceptible than the parental strain to growth inhibition by DFMO
treatment
in host animals. Besides, growth inhibition associated with DFMO-induced
polyamine
depletion in ZR-75-1 human breast cancer cells can be completely reversed by
concentrations of spermidine as low as 300 nM, i.e., such as those found in
human plasma
(Moulinoux, J.-P., Quemener, V., and Khan, N.A. 1991. Cell. Mol. Biol. 37: 773-
783;
Scalabrino, G. and Ferioli, M.E. 1981. Adv. Cancer Res. 36: 1-102; Bachrach,
U., 1989.
in The Physiology of Polyamines (Bachrach, U. and Heimer, Y.M., eds.) Vol. II,
pp. 235-
249, 2 vols, CRC Press, Boca Raton, FL). The striking efficiency of the
transport system
to salvage exogenous polyamines in DFMO-treated cells owes to its upregulation
consecutive to polyamine depletion (Seiler, N. and Dezeure, F. 1980. Int. J.
Biochem. 22:
211-218; Byers, T.L. and Pegg, A.E. 1990. J. Cell Physiol. 143: 460-467;
Lessard, M.,
Zhao, C., Singh, S.M. and Poulin, R. 1995. J. Biol. Chem. 270: 1685-1694;
Kalcinuma, Y.,
Hoshino, K., and Igarashi, K. 1988. Eur. J. Biochem. 176: 409-414). These data
reinforce
the view that cellular import of exogenous polyamines is the main factor
limiting the
efficacy of DFMO and other polyamine biosynthesis inhibitors as antitumor
agents in vivo
(Sarhan, S. Kntidgen, B., and Seiler, N. 1989. Anticancer Res. 9: 215-224;
Hessels, J.,
Kingma, A.W., Ferwerda, H., Keij, J., Van der Berg, G.A., and Muskiet, F.A.J.
1989. Int.
J. Cancer 43: 115-1166; Ask, A., Persson, L. and Heby, O. 1992. Cancer Lett.
66: 29-34;
Seiler, N., Sarhan, S., Grauffel, C., Jones, R., KnSdgen, B. and Moulinoux, J.-
P. 1990.
Cancer Res. 50: 5077-5083; Persson, L., Holm, I., Ask, A. and Heby, O. 1988.
Cancer Res.
48: 4807-4811).
2
- ---------- -
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
Depletion of intracellular polyamines in tumor cells ig thus a well-known
strategy
in anticancer therapies. However, it is now of common knowledge that depleting
intracellular polyamines generally enhances polyamine uptake. To date,
molecular
information on the carrier molecules of the mammalian polyamine transport
system is still
unavailable. A few attempts have been made previously to design specific
inhibitors of
polyamine transport. Based on the finding that paraquat (4,4'-bipyridine) is a
substrate of
the putrescine transport system (Smith, L.L. and Wyatt, I. 1981. Biochem.
Pharmacol. 20,
1053-10581; Rannels, D.E., Pegg, A.E., Clark, R.S. and Addison, J.L. 1985. Am.
J. Physiol.
249, E506-E513), a series of polypyridinium salts, including compounds with a
low K;
against putrescine uptake and low acute toxicity for mammalian cells have been
synthesized (Minchin, R.F., Martin, R.L., Summers, L.A. and Ilett, K.F. 1989.
Biochem.
J. 262, 391-395). However, it is unclear whether such compounds can
efficiently inhibit
polyamine transport or are accumulated intracellularly. A number of polyamine
analogs
are effective competitors of polyamine uptake while being themselves
substrates for
transport (Seiler, N. and Dezeure, F., 1990. Int. J. Biochem. Cell. Biol. 27:
425-442;
Bergeron, R.J., and Seligsohn, H.W. (1986) Bioinorg. Chem. 14: 345-355;
Porter, C.W.,
Bergeron, R.J. and Stolowich, N.J. 1982. Cancer Res. 42: 4072-4078; Porter,
C.W., Basu,
H.S., Feuerstein, B.G., Deen, D.F., Lubich, W.P., Bergeron, R.J., Samejima,
K., and
Marton, L.J. 1989. Cancer Res. 49: 5591-5597; Pegg, A.E., Wechter, R., Pakala,
R., and
Bergeron, R.J. 1989. J. Biol. Chem. 264: 11744-11749; Pegg, A.E., Nagarajan,
S., Naficy,
S. and Ganem, B. 1991. Biochem. J. 274: 176-171; Porter, C.W., Ganis, B.,
Libby, P.R. and
Bergeron, R.J. 1991. Cancer Res. 51: 3715-3720).
More recently, a high-molecular weight (Mr=25kD) spermine polymer has been
described by Aziz et al. in USP 5,456,908, as a competitive inhibitor of
polyamine
transport, with a K; in the 10-6M range. In this patent document are disclosed
two novel
classes of polyamine transport inhibitors of high molecular weight, namely
polymeric
conjugates of normally transported substances (TS) of the structure (TS)R, or
conjugates of
a polyamine and a protein or polypeptide (P) linked by known coupling agents
and
represented by (TS)-(P), wherein the repeating units of the polymer comprise
the targeted
polyamine. It is predictable that the inhibitors of Aziz et al. would be
difficult to eliminate
in vivo due to their high molecular weight and the high positive charge of the
polymers,
3
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
notwithstanding the risk of immunogenicity inherent to such high molecular
weight
inhibitors. The length of the polymers of Aziz et al. as well as their charge
would cause
their adsorption to the cellular surface, which bears negative charges due to
the presence
of glycoproteins, e.g. sialic acid. Poly-L-lysine, a commercially used
compound analogous
to high molecular weight polymers of polyamines by its positive charges, is
known to
promote a strong electrostatic interaction between the cell and its substrate,
as in the
induction of positive charges of gamma irradiation of synthetic polymers used
to produce
dishes for tissue culture. The polyamine transport inhibitors of Aziz et al.
present the
additional drawback of being highly cytotoxic. It is noteworthy that their
spermine polymer
is effective in decreasing contents of polyamines in cells even when not used
in
combination with DFMO and at concentrations much higher than those required to
block
polyamine uptake, which indicates inherent high toxicity of the compound
toward the cell
by a mechanism independent of polyamine transport per se. The cytotoxicity of
the
spermidine polymer of Aziz et al. is most probably explained by a non-specific
effect on
cellular physiology such as the cellular membrane. Although the authors
pretend to
demonstrate the specific action of the polymers with the fact that exogenous
spermidine
reverses the induced cytotoxicity, it is highly likely that competition
between spermidine
and the polymers modifies the electrostatic interaction with the negatively-
charged sites on
the cellular membrane is responsible for the effect. The results obtained by
Aziz et al.
indicate that at least part of the effect observed with high molecular weight
polymers is
non-specific (Aziz, S.M., Tofiq, S.F., Gosland, M.P., Olson, J.W. and
Gillespie, M.N.
1995. J. Pharmacol. Exp. Ther. 274, 181-196). The usefulness of this spermine
polymer
for specifically blocking polyamine accumulation is therefore uncertain in
view of its
marked cytotoxicity.
Cysteamine and aliphatic monoamines of similar chain length such a n-
butylamine
and n-pentylamine have a low but significant ability to antagonize putrescine
uptake
(Gordonsmith, R.H., Brooke-Taylor, S., Smith, L.L. and Cohen, G.M. 1983.
Biochem.
Pharmacol. 32, 431-437), although the mode of inhibition of these compounds
has not
been reported. The only polyamine-like structure known to interact non-
competitively with
the polyamine transport system is pentamidine, an aromatic diamidine (Jones,
H.E.,
4
----,----=-- - --- _ _ -~--_ _ _ __.... __. __ _._ ____-__
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
Blundell, G.K., Wyatt, I., John, R.A., Farr, S.J. and Richards, R.J. 1992.
Biochem.
Pharmacol. 43, 431-437), but the structural basis of its inhibitory activity
is not yet clear.
It follows that there still exists a need for effective polyamine transport
inhibitors
which, while inhibiting the transport of polyamines, will not be internalized
by the transport
system and will not be toxic to the cell. The availability of low molecular
weight inhibitors
of polyamine transport would provide for the possibility of better renal
elimination, as well
as lower risks of being immunogenic. The availability of high-affmity,
specific, but
impermeant antagonists of polyamine transport would also allow to evaluate the
antitumor
efficacy of polyamine depletion strategies in vivo with minimal systemic
cytotoxic effects.
There is much preclinical evidence supporting the hypothesis that the efficacy
of
the suicide inhibitor of ornithine decarboxylase, D,L-a-
difluoromethylornithine (DFMO
= Eflomithine) as a chemotherapeutic agent is limited by the enhanced ability
of tumor
cells to transport polyamines from plasma sources. Plasma polyamines are
partly derived
from various plasma sources (7, 12, 18, 58-60, 62, 70) and from the activity
of the
gastrointestinal microflora, which produces and excretes very high amounts of
putrescine
and cadaverine (1, 17, 45, 50, 62, 70), which can enter the general
circulation through the
enterohepatic pathway (6, 45). Other systemic contributions can also be
attributed to
polyamine excretion by peripheral tissues, including dying tumor cells (32,
35, 41, 42, 63,
64, 67, 79, 80). The enhanced uptake of polyamines by tumor cells results both
from the
increased polyamine transport activity that accompanies the malignant
phenotype (11, 43,
51, 68, 69), and from the effect of DFMO itself, which causes a compensatory
upregulation
of polyamine uptake across the plasma membrane (9, 10, 14-16, 22, 25, 29, 31,
38, 39, 43,
47, 48, 50, 57, 61). One possible strategy that could be used to overcome this
phenomenon
would be to administer a pure antagonist of polyamine transport, i.e. a drug
which binds
with high affmity to the polyamine transporter, but which cannot be
transported by this
membrane protein. While a need continues to exist for such compound, no such
compound
is yet available.
SUMMARY OF THE INVENTION
5
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
In accordance with the present invention, there is now provided polyamine
transport
inhibitors having a low molecular weight, less susceptible to immunogenicity
and to non-
specific interactions with the cellular membrane. These inhibitors have high
affinity, are
specific, impermeant, pure antagonists of polyamine transport in mammalian
cells while
exhibiting minimal cytotoxic effects.
There is thus provided in accordance with the present invention synthetic
derivatives of original polyamines. In some aspects, the original polyamine is
modified to
comprise an amido group immediately linked to a carbon atom of said original
polyamine,
said synthetic derivatives inhibiting the cellular uptake of natural
polyamines by
specifically binding cellular transporters for said natural polyamines.
Surprisingly, the
immediate vicinity of the amido group to the backbone of the original
polyamine preserves
the specificity of the derivative towards the transporter while conferring
thereto an
impermeant character, providing a true antagonist. In some embodiments, the
amido group
is located between two internal nitrogen atoms of the original polyamine. In
other
embodiments, the synthetic derivative comprises a dimer wherein monomers of
said dimer
are linked together by a spacer side chain anchored to the amido group of each
monomer.
Although natural polyamines, such as putrescine, spermine and spermidine, can
be
used as the original polyamine, other non-natural polyainines can be used as a
starting
material for the making of synthetic derivatives of the present invention.
Accordingly, a synthetic derivative comprising the following general formula:
R2 i12
RiHN ~---NIt-~CH~- CH-{CH) y N ~- NHR,
I
R3 C=0 R'3
I
NH
(
L
provides the structure of some embodiements of the invention, wherein R, and
R,'
independently represent a hydrogen atom or an alkyl group having 1 to 2 carbon
atoms, R2,
R2', or R3 and R3' independently represent a hydrogen atom or a methyl group,
w and z
6
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
independently represent an integer of 3 or 4, x represents an integer from 0
to n, y
represents an integer from 0 to n, n represents an integer from 3 to 6, the
sum of x and y
equals n, and L represents a hydrogen atom or a molecule which cannot be
captured by said
natural polyamine transporter. The side chain or linker L may be labeled and
be used as
a marker for a polyamine transporter. Furthermore, the side chain L can be
varied to
increase the affinity of the derivative for the transporter. The side chain L
may also become
a spacer molecule useful in the formation of a dimer. This spacer side chain
comprises a
linear hydrocarbon-containing backbone of 3 to 8 atoms. The backbone may
comprise
sulfur, oxygen, or nitrogen atoms.
In yet other embodiments, the original polyamine is spermine. Three
derivatives
have been obtained therefrom: N-(2-mercaptoethyl)spermine-5-carboxamide
(MESC), the
disulfide from thereof, namely 2,2'-dithiobis(N-ethyl-spermine-5-carboxamide)
(DESC),
and N-[2,2'-dithio(ethyl, l-aminoethyl)]spermine-5-carboxamide (DEASC).
It is an object of the invention to provide the use the above synthetic
derivatives for
inhibiting the activity of a natural polyamine transporter. In some
embodiements, the
method may be described as comprising the step of contacting said transporter
with an
inhibitory effective amount of said synthetic derivative. This inhibition
results in the
control of the treatment of disorders involving unrestrained cell
proliferation and/or
differentiation where control of polyamine transport is required, when used in
combination
with an inhibitor of polyamine synthesis such as DFMO.
It is further another object of the invention to provide a use of the non-
dimeric
derivatives as a marker for a polyamine transporter, which comprises the steps
of labeling
said synthetic derivative, binding to said transporter said labeled synthetic
derivative and
detecting said bound labeled marker as an indication of the presence of said
polyamine
transporter. The above sequence of steps results in the diagnosis of a
disorder involving
unrestrained cell proliferation and/or differentiation where control of
polyamine transport
is required.
It is also another object of the invention to provide a pharmaceutical
composition
for treating disorders wherein control of polyamine transport is required,
comprising
anyone of the above derivatives in adjunction with an acceptable
pharmaceutical carrier.
7
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
Preferably, this composition also comprises an inhibitor of polyamine
synthesis, such as
DFMO.
The applicants have unexpectedly discovered that the presence of a lateral
amido
group immediately linked to a carbon atom of the polyamine backbone of a
synthetic
derivative of an original polyamine confers impermeant properties to the so
derived
synthetic polyamine against the mammalian cell. It follows that the synthetic
polyamine
derivatives of the present invention, by exhibiting high affinity for diamine
and polyamine
transport systems, block the transport of natural polyamines by competing
therewith, while
in the same time acting as poor substrate for intracellular uptake. The
affinity of the
polyamine derivative for the transporter system is fu.rther enhanced by
increasing the length
of a side chain anchored to the amido group of the derivative. The best
affinity is achieved
by dimerizing the polyamine derivative with the aid of a spacer molecule
anchored at both
ends to the amido group of each monomer. The flexibility of the chemical
structure of the
inhibitors of the present invention permits better optimization of the
activity and affinity
than a simple polymeric structure such as (TS),,. For example, modifications
to the
polyamine backbone as taught by the present invention, such as methylation of
C 1 and C 12,
lowers the possibility of oxidation of the primary amides by the serum amine
oxidase,
which is present in mammalian sera. Additional modifications including
adjunction to the
lateral chain of alkylating groups that irreversibly modify residues that are
essential to the
activity of the polyamine transporter, such as carboxylic moieties of the
carrier protein, are
also contemplated in the present invention (Torossian, K., Audette, M., and
Poulin R.,
1996. Biochem. J. 319: 21-28). The inhibitory action of the derivatives of the
present
invention is thus enhanced. By diminishing the amount of active transporters,
additional
modifications to the side chain that can be of potential therapeutic interest
include the
incorporation of reactive groups to the side chain that would allow the
covalent
modification of residues in the polyamine transporter by the principle of
affinity labeling,
and its subsequent irreversible inactivation.
This finding clearly demonstrates that modification of the chemical structure
of the
lateral chain optimizes the affinity of the polyamine derivative without
augmenting to a
great extent the molecular weight thereof. This markedly contrasts with the
teachings of
Aziz et al. who make use of high molecular weight polymers. Moreover, the mode
of
8
CA 02304557 2007-11-09
action of the inhibitors herein proposed, clearly different to that of Aziz et
al. which relies upon
their inherent cytotoxicity, is a competitive inhibition of the polyamine
uptake.
Specific transport inhibition by polyamines dimerized via a side group on an
integral
atom of the polyamine chain is not limited to spermine or spermine-like
dimers, but can also be
obtained with dimers of triamines such as spermidine [N-(3-aminopropyl)-1,4-
diaminobutane],
sum-homospermidine [n-(4-aminobutyl)-1,4-diaminobutane] and sym-norspermidine
[N-(3-
aminopropyl)-1,3-diaminopropane]. We have synthesized novel spermidine, sym-
homospermidine and sym-norspermidine dimers cross-linked through aliphatic or
aromatic side
chains on the central secondary amino group. We have found that such compounds
act as
inhibitors of polyamine transport with a potency similar or greater than that
of dimeric
polyamine transport inhibitors with a spermine-like backbone such as 2,2'-
dithiobis[N-ethyl-
spermine 5-carboxamide (or DESC).
In some embodiments, one spermidine or sym-norspermidine chain is cross-linked
to a
second spermidine or sym-norspermidine chain with an N-alkyl spacer side group
anchored to
the secondary amino group of said spermidineor sym-norspermidine chain. In
particular
embodiments, said spacer side group is an aliphatic carbon chain or an
aromatic carbon chain.
In one aspect, the present invention provides a natural polyamine transporter
inhibitor
composition comprising a compound of formula (I) or (II) :
RIHN-l-CH2-}-N~CH2}-NHR1
\ / /
L
RIHN-i-CHZ-}-N~ CH2}-NHRI
\ / / (I)
I2 R I2 R2
RjHN-~Cx
~-NHR,
~-N-~C~ZN--~Cx
Iz I Iz Iz
L
IZ IZ IZ
RIHN--~C~X_N__~ c~N-{C~NHR1
1 I \I
R2 RZ R2
(II)
9
CA 02304557 2007-11-09
wherein L is :
--N ~-j
I\ / ;or
R, is H, methyl, ethyl or propyl; R2 is H or methyl;
x is greater than 2 and less than 5; y is greater than 2 and less than 5; i is
greater than 0 and less
than 5; j is greater than 0 and less than 5; and 1< i+ j<'7;
w is greater than two and less than 8 and
an acceptable pharmaceutical carrier.
In another aspect, the present invention provides a use of a compound of
formula (I) or
(II):
RIHN+H2}-N~CH2~-y NHR,
L
RIHN+H2~X-N-~ CH2NHRI 1~ Y
(I)
2 2 2
RIHN-~OTN --1- C~N~C~NHR,
I`I I
R2 R2 R2
L
2 2 2
RIHN-{C~NC~N--{ C~NHR~
\I \
R2 R2 R2 (II)
9a
CA 02304557 2007-11-09
for controlling or treating a disorder wherein control of polyamine transport
is required wherein
Lis:
;or
i j
Rl is H, methyl, ethyl or propyl; R2 is H or methyl;
x is greater than 2 and less than 5; y is greater than 2 and less than 5; i is
greater than 0 and less
than 5; j is greater than 0 and less than 5; and 1< i+ j< 7;
w is greater than two and less than 8.
In a further aspect, the present invention provides a use of a compound of
formula (I) or
(II):
RIHN+H2~x N+H2~-YNHRI
L
RIHN+H2Yx N-~ CH2NHR,
Y
(I)
2 2 2
RIHN-{-C~N---1-C1N-I-C-INHRI
\I / \/
R2 R2 R2
L
2 2 2
RjHN~C-}-N~C~N~C~NHR,
/ I I
R2 R2 R2 (II)
in the manufacture of a medicament for preventing or treating cancer wherein L
is :
9b
CA 02304557 2007-11-09
;or
Rl is H, methyl, ethyl or propyl; R2 is H or methyl;
x is greater than 2 and less than 5; y is greater than 2 and less than 5; i is
greater than 0 and less
than 5; j is greater than 0 and less than 5; and 1< i+ j< 7;
w is greater than 2 and less than 8.
In still a further aspect, the present invention provides a compound of
formula (I) or (II) :
R1 HN-{-CH2}-N-{-CH2~-YNHRI
L
RIHN+H2N~ CH2}-NHRI
/y
~X-
(I)
R2 R2 R2
R, HN C~ N I! ~ N C~ NHR1
/ X
~ / X ~ w ~I
i I RZ R2 R2
L
2 2 2
RIHN~C-}-N~C~N~C~NHR,
I/ I I R2 R2 R2 (II)
wherein L is :
9c
CA 02304557 2007-11-09
or
Rl is H, methyl, ethyl or propyl; R2 is H or methyl;
x is greater than 2 and less than 5; y is greater than 2 and less than 5; i is
greater than 0 and less
than 5; j is greater than 0 and less than 5; and 1< i+ j < 7;
w is greater than 2 and less than 8.
Other objects, advantages and features of the present invention will become
more
apparent upon reading of the following non restrictive description of
preferred embodiments
thereof, given by way of example only and with reference to the accompanying
drawings.
BRIEF DESCRIPTION OF THE DRAWINGS
In the appended drawings:
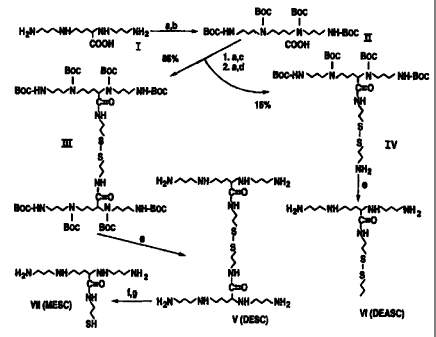
FIG 1 illustrates details of the synthesis of the compounds of the present
invention,
wherein a=triethylamine; b=di-tert-butyl dicarbonate; c=cyanuric chloride;
d=cystamine
dihydrochloride; e=3 N HCI; f--dithiothreitol: g=50 mM sodium phosphate in
aqueous solution
(pH=8.0); and wherein compound I is 5-carboxyspermine; compound II is tetra-
Boc-5-
carboxyspermine; compound III is 2,2'-dithiobis[N-ethyl-(N', N¾,1V8, N12)-
tetra-Boc-spermine-5-
carboxamide; compound IV is N-[2,2'-dithio(ethyl, 1'-aminoehtyl)]- N', N¾,1V8,
9d
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
N12-tetra-Boc-spermine-5-carboxamide; compound V is 2,2'-dithiobis(N-ethyl-
spermine-5-
carboxamide) octahydrochloride (DESC); compound VI is N-[2,2'-dithio(ethyl,l'-
aminoethyl)]-spermine-5-carboxamide pentahydrochloride (DEASC) and compound
VII
is N-(2-mercaptoethyl) spermine 5-carboxamide (MESC) tetrahydrochloride;
FIG 2 graphically illustrates the inhibition of ["C]spermine transport by
MESC,
DESC and DEASC in human ZR-75-1 breast cancer cells. The rate of spermine
uptake
was measured in ZR-75-1 cells grown as monolayers in 24-well culture plates in
the
presence of the indicated concentrations of DESC (0), MESC (0), and DEASC (0),
using
1,uM ['"C]spermine as substrate. Data are the mean SD of triplicate
determinations;
FIG 3 graphically illustrates the inhibition of ['H]spermidine uptake by
spermine
and DESC in ZR-75-1 cells. The rate of spermidine uptake was measured in ZR-75-
1 cells
grown as monolayers in 24-well culture plates in the presence of the indicated
concentrations of spermine (0) and DESC (0) using 3 M ['H]putrescine (A) or
1,uM
['H]spermidine (B) as substrate. Data are the mean SD of triplicate
determinations from
a representative experiment;
FIG 4 illustrates graphically the Lineweaver-Burke analysis of putrescine
transport
inhibition by DESC and DEASC in ZR-75-1 cells. The rate of ['H]putrescine
uptake was
determined in ZR-75-1 cell cultures with increasing concentrations of
substrate (A) in the
presence of 0 M DESC (0), 3 M DESC (40), 30 ,uM DESC (O) or 100 ,uM DESC (^)
or (B) in the presence of 0AM DEASC (0), 20 M DEASC (40), 50,uM DEASC (0) or
200 M DEASC (0);
FIG 5 illustrates graphically the structure of MESC thioether derivatives and
their
K; values with respect to spermidine uptake in CHO-Kl cells. The various
conjugates were
prepared from MESC as described supra, and structure and name of the
substituents are
given in the first two columns from the left, wherein R corresponds to the
group attached
to sulfur in MESC (structure VII, Fig. 1). The rate of spermidine uptake was
determined
in CHO-K1 cells in the presence of increasing concentrations of the various
MESC
derivatives, using 1 M ['H]spernudine as substrate. K; values are given as
the mean SD
of triplicate determinations from 2 to 3 experiments;
FIG 6 graphically represents the effect of DESC and MESC on the intracellular
accumulation of [3H]spermidine in ZR-75-1 cells, wherein at time 0 (A), 5,uM
~:.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
[3H]spermidine was added to ZR-75-1 cell cultures grown in 24-well plates (1
ml/well) in
the presence of 200 ,uM MESC (0), 50 M DESC (0) or 200 M DESC (0), and
accumulation of radio-labeled spermidine determined after the indicated
interval. Control
cells (0) received vehicle only. B, same as in A, except that 200 ,uM CHX was
added at
time 0 in the presence of 0(0), 50 (0) or 200 ,uM DESC (M). Data are the mean
SD of
triplicate determinations;
FIG 7 illustrates the effect of spermine, MESC, DESC and DEASC on ZR-75-1 cell
proliferation. Cells were incubated for 11 days in MEZR medium with the
indicated
concentration of spermine, DESC, MESC, or DEASC in the presence (shaded bars)
or
absence (plain bars) of 1 mM of aminoguanidine, and DNA content per culture
was then
determined. Data represent the mean SD of triplicate determinations;
FIG 8 represents the effect of DESC on the reversal of DFMO-induced growth
inhibition by exogenous spermidine in ZR-75-1 cells. Cells were incubated for
11 days in
SD medium with the indicated concentrations of spermidine in the presence of
50 M
DESC (0), 1mM DFMO (0), or the combination thereof (M), or in the absence of
drugs
(0). Data are the mean SD of triplicate cultures;
FIG 9 represents the chromatographic profile of DESC and its degradation
products
in IMEM or PBS. DESC (50 ,uM) was added to lml of IMEM containing 10% fetal
bovine
serum in the absence (A) or presence (B) of 1mM aminoguanidine, or 1 ml PBS
(C) in 24-
well culture plates in the absence of cells. Media were analyzed after 20
minutes (solid
lines) or 48 hours (dotted lines) of incubation at 37 C in 95% air: 5% C02,
water-saturated
atmosphere for amine composition by ion-pair reversed-phase HPLC as described
supra.
Peaks 1 and 2 are degradation products of DESC, whereas peak 3 is a minor
amount of
DEASC initially present in the DESC preparation. Note the disappearance of
peak 3
(DEASC) and the appearance of a shoulder (indicated by the arrow) at 42
minutes on the
48-hour profile in panel A; and
FIG 10 represents the time course of degradation of DESC in growth medium. At
time 0, 50 ,.cM DESC was added to 1m1 of IMEM in 24-well culture plates and
the content
in DESC (0), compound 1 (Comp 1, =) and compound 2 (Comp 2, 0) determined by
HPLC after the indicated incubation period at 37 C in a 5% CO, atmosphere.
Data
represent the mean of triplicate determinations from a representative
experiment.
11
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
FIG 11. Structures of putrescine, of the natural polyamines spermidine and
spermine, and of three cell-impermeant inhibitors of polyamine transport
(DESC, DEASC
and MESC).
FIG 12. Structure and scheme for the synthesis of unmethylated spermine
analogs
as polyamine transport inhibitors with a linker attached via amide bonds to
the polyamine
chains (BS-3, BS-4, BS-5 and BS-6 compounds). The method of synthesis is
described in
greater detail in Example 1.
FIG 13. Initial route of synthesis of terminal C-methylated, dimeric spermine
analogs as transport inhibitors with a linker attached via an alkyl bond to
the polyamine
chains (BMS-3, BMS-4, BMS-5 and BMS-6). The steps presented in this figure
describe
the complete route of synthesis leading to the precursor N', N, N", N/1-tetra
(Boc)-1, 12-
dimethylspermine-5-carbinol (XV).
FIG 14. The first step in the coupling of N', N~, 11', N'Z-tetra (Boc)-1, 12-
dimethylspermine-5-carbinol (XV) to the linker L(= a N-mono-FMOC-
diaminoalkane),
toward the synthesis of BMS compounds.
FIG 15. The second step in the coupling of N', NI, IV" N'2-tetra (Boc)-1, 12-
dimethylspermine-5-carbinol (XV) to the linker L(= aN-mono-FMOC-diaminoalkane)
toward the synthesis of BMS compounds.
FIG 16. The final step of the synthesis of BMS compounds (XX); the Boc-
protected, cross-linked 1, 12-dimethylspermine dimer is deprotected to
generate the BMS
compounds. BMS-3, BMS-4, BMS-5 and BMS-6 correspond to N, N-bis ([1, 12-
dimethyl-spermine]-5-methyl)-diaminoalkanes where the diaminoalkane linker is
1,3-
diaminopropane, 1,4-diaminobutane, 1,5-diaminopentane, and 1,6-diaminohexane,
respectively.
FIG 17A, 17B and 17C presents three classes of dimeric polyamine transport
inhibitors according to the site of attachment of the linker (L) to the
polyamine chain.
FIG 17A - C-linked dimeric analogs. R, is H, methyl, ethyl or propyl; R2 is H
or
methyl; R3 is an alkyl, amide, keto, ether, thioether, phosphono or sulfonyl
group; x is
greater than 2 and less than 5 (2 < x < 5), and the sum of y+z is greater than
or equal to 2
and less than or equal to 6 (2 s y + zs 6). The linker L is any chemical
structure covalently
linked to the R3 groups and which prevents the uptake of the analog.
12
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
FIG 17B - N-linked dimeric analogs. R, is H, methyl, ethyl or propyl; R2 is H
or
methyl; x is greater than 2 and less than 8(2 < x < 8), and w is greater than
2 and less than
7 (2 < w < 7). The linker L is any chemical structure covalently linked to one
internal
amino group of each polyamine chain and which prevents the uptake of the
analog.
FIG 17C - C-linked/N-linked mixed dimeric analogs. R, is H, methyl, ethyl or
propyl; R2 is H or methyl; x is greater than 2 and less than 5 (2 < x < 5),
the sum of y+z is
greater than or equal to 2 and less than or equal to 6 (2 s y + zs 6), and w
is greater than
2 and less than 8 (2 < w < 8). The linker L is any chemical structure
covalently linked to
one internal amino group of one polyamine chain and to the R3 of the other
polyamine
chain, and which prevents the uptake of the analog.
FIG 18. Initial route of synthesis of unmethylated,lV¾-alkylated dimeric
spermine
analogs (FIG 17B). Steps leading to the synthesis of the intermediate M-
benzyl, N8, N12-
di(CBZ)-spermine.
FIG 19. Final steps for the synthesis of unmethylated, N'-alkylated dimeric
spemiine analogs (FIG 17B, represented by type compound XXIX). For the
aliphatic linker
-(CH2)õ, 2 < n < 51.
FIG 20. Initial route of synthesis of terminal C-methylated, N'-alkylated
dimeric
spermine analogs (FIG 17B). Steps leading to the synthesis of the
interrnediate N`, N``'-bis
(1V [N-Boc-3-amino, 3-methylpropyl], N-[4-aminobutyl])-aw-diminoalkane. For
the
aliphatic linker-(CH2)õ, 2< n<51.
FIG 21. Final steps for the synthesis of terminal C-methylated, N4-alkylated
dimeric
spermine analogs (FIG 17B, represented by type compound XXXVIII). For the
aliphatic
linker-(CH2)õ, 2 < n < 51.
FIG 22. Initial route of synthesis of 1,12-dimethylspermine dimers cross-
linked
through N`-alkyl/5-alkyl attachments of the linker (FIG 17C). Steps leading to
the
synthesis of the intermediate N"([N-Boc-3-amino, 3-methylpropyl], N-[N-FMOC-4-
aminobutyl]), N-[5-(N', N`, N8, N12-tetra (Boc)-spermine)-methyl]-a, w-
diaminoalkane.
For the aliphatic linker -(CHZ)õ, 2< n< 51.
FIG 23. Intermediate route of synthesis of 1,12-dimethylspermine dimers cross-
linked through 1V -alkyl/5-alkyl attachments of the linker (FIG 17C). Steps
leading to the
synthesis of the intermediate N ([7V'-Boc-3-amino, 3-methylpropyl], N-[8-amino-
5-aza-
13
CA 02304557 2000-03-22
WO 99/54283 PCTIUS98/07806
octanoyl]), .N`''-[5-(N', N, N8, N'Z-tetra (Boc)-spermine)-methyl]-a, w-
diaminoalkane. For
the aliphatic linker -(CH2)õ, 2< n < 51.
FIG 24. Final route of synthesis of 1,12-dimethylspermine dimers cross linked
through 10-alkyl/5-alkyl attachments of the linker (FIG 17C represented by
type compound
XLV). For the aliphatic linker -(CHZ),-, 2< n < 51.
FIG 25 illustrates the structure of representative dimeric transport
inhibitors with
a triamine backbone that are included in the present invention. BABAC is N,N'-
bis(3-
aminopropyl), N, N'-bis(4-aminobutyl)cystamine, a dimeric spermidine
derivative with a
diethyl disulfide linker; (BNSpd-(n+2) (standing for bis(sym-
norspermidine)molecules with
a carbon chain length of n+2 atoms; 0<n<7) represents the general structure of
dimeric
sym-norspermidine-derived transport inhibitors with an aliphatic linker; BSpd-
(n+2)
(standing for bis(spemiidine) molecules with a carbon chain length of n+2
atoms; 0<n<7)
represents the general structure of dimeric spermidine-derived transport
inhibitors with an
aliphatic linker; TADAX is N,N,N;N'-tetrakis(3-aminopropyl) p-xylylenediamine,
a
dimeric sym-norspermidine derivative with a p-xylylenediamine linker.
FIG 26 illustrates the structure of other representaive dimeric transport
inhibitors
with a triamine backbone that are included in the present invention. BABA-
trans and
BABA-cis stands for the trans and cis isomers of N,N'-N,N'-bis(3-
aminopropyl),NN'-
bis(4-aminobutyl) derivatives of a,a'-diaminoalkenes, which are dimeric
derivatives of
either sym-homospermidine, sym-norspermidine or spermidine. BABA-yne stands
for
N,N'-N,N =bis(3-aminopropyl),NN'-bis(4-aminobutyl)derivatives of a,a'-
diaminoalkynes,
which are dimeric derivatives of either sym-homospermidine, sym-norspermidine
or
spermidine.
FIG 27 illustrates synthetic Scheme 1 used to obtain dimeric sym-norspermidine-
derived polyamine transport inhibitors (X) from a precursor cross-linking
diamine (VIII)
containing the linker (L), and typical examples of said linker.
Et,N=triethylamine.
FIG 28 illustrates synthetic Scheme 2 used to obtain dimeric sym-
homospermidine,
sym-norspermidine-, or spermidine-derived polyamine transport inhibitors (XIV)
from the
parent triamine (XI) cross-linked with a linker L via symmetrical N-alkylation
of the
secondary amino group of said triainine, and typical examples of said linker.
TrCl=trityl
chloride; Et2NH=diethylamine; DMF=dimethylformamide.
14
~;.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
FIG 29 illustrates synthetic Scheme 3 used to obtain dimeric sym-
homospermidine-,
sym-norspermidine-, or spermidine-derived polyamine transport inhibitors (XIV)
from the
trityl-protected parent triamine (XII) cross-linked with a linker L via
symmetrical
amidation of the secondary amino group of said triamine followed by reduction
of the
amide bonds, and typical examples of said linker. THF=tetrahydrofuran.
FIG 30 illustrates synthetic Scheme 4 used to obtain dimeric sym-
homospermidine-,
sym-norspermidine-, or spermidine-derived polyamine transport inhibitors
(XVIII) from
the parent triamine (XI) cross-linked via a diethyl disulfide linker L by
symmetrical
alkylation of the secondary amino group of said triamine. TFA=trifluoroacetyl.
FIG 31 illustrates a comparison between the structures of N,N'-bis(3-
aminopropyl),N,N'-bis(4-aminobutyl)cystamine (BABAC), a dimeric spermidine
derivative
with a diethyldisulfide linker, of its monomeric thiol form 1V (3-
aminopropyl),N-(4-
aminobutyl)cysteamine (AAC) and of 2,2'-dithiobis[N-ethyl-spermine 5-
carboxamide
(DESC).
FIG 32 [A-32C] represents the inhibition of [3H]putrescine (32A),
[3H]spermidine
(32B) and [14C]spermine uptake (32C) by spermine (SPM), DESC, BABAC and AAC in
human breast cancer cells ZR-75-1. Data represent the mean of triplicate
determinations
fro a representative experiment. Standard deviations, which did not exceed 10%
of the
mean value, were not shown for the sake of clarity.
FIG 33 represents the inhibition of [14C]spermine uptake by spermine (SPM),
DESC
and TADAX in ZR-75-1 human breast cancer cells. Data are the mean SD of
triplicate
determinations from a representative experiment.
FIG 34 represents the relative cytotoxicity of sym-norspermidine (NSpd), BABAC
and TADAX in ZR-75-1 human breast cancer cells. Data are the mean t SD of
triplicate
determinations from a representative experiment.
CA 02304557 2000-03-22
WO 99/54283 PCTIUS98/07806
DETAILED DESCRIPTION OF THE PREFERRED EMBODIMENTS
Materials and Methods:
Sym-norspermidine, ornithine dihydrochloride and other reagents for organic
syntheses were purchased from Aldrich (Milwaukee, WI) and Sigma (St. Louis,
MO).
Reversed phase silica gel liquid chromatography was performed with a
LichroprepTM RP-
18 C,$ silica gel column (40-63 M ; BDH, St. Laurent, Qc., Canada) using a
gradient of
CH3CN:MeOH:H20 (25:35:40 to 50:30:20) as eluent. Homogeneity of synthetic
products
was assessed by thin-layer chromatography performed on 0.20 mm F2M silica gel
60 plates
or 0.25 mm F2,4S RP-18 reversed phase silica gel plates (E. Merck, Darmstadt,
Germany).
FIR spectra were obtained on a Perkin-Elmer 1600 spectrophotometer (FTIR
series) and
were expressed in cm'. 'H and13C NMR spectra were recorded with a Bruker AC/F
300
(300 MHz);13C were recorded at 75.47 MHz. Chemical shifts (8 in ppm) were
referenced
to CDC13 (7.26 ppm for'H and 77.00 ppm for'3C). Mass spectra (MS) were
recorded at
the Mass Spectrometry Regional Center (University of Montreal, Montreal, Qc.,
Canada)
by fast atomic bombardment mass spectrometry (FABMS) or liquid secondary ion
mass
spectrometry (LSMIS), using a VG AutoSpecQTM and a Kratos MS50 TCTA,
respectively.
[2,3 'H(N]putrescine dihydrochloride (4.1 x 104 Cl/mol) and [1,8
jH(N)]spermidine
trihydrochloride (1.5 x 104 Cl/mol) were obtained from Dupont-New England
Nuclear
(Lachine, Qc., Canada). [5,8-'"C]spermine tetrahydrochloride (108 Cl/mol)) was
purchased
from Amersham (Arlington Heights, IL). DFMO was provided by the Marion Merrell
Dow
Research Institute (Cincinnati, OH). Fetal bovine serum (FBS) and CosmicTM
calf serum
were from Hyclone (Logan, UT). The heterobifunctional reagent 1( p-
azidosalicylamido)-
4-iodoacetamido)butane (ASIB) as obtained from Pierce (Rockford, IL). Lucifer
Yellow
(OY) iodoacetamide was purchased from Molecular Probes (Eugene, OR).
Putrescine
dihydrochloride, spermidine trihydrochloride, spermine tetrahydrochloride,
iodoacetamide,
5,5'-dithio(2-nitrobenzoic acid) and 3,4-diaminobenzoic acid as well as tissue
culture
reagents were purchased from Sigma. Ortho-phthaldialdehyde was purchased from
Fluka
(Ronkonkoma, NY) and other reagents for high-performance liquid chromatography
(HPLC) were from Fisher Scientific (Montreal, Qc., Canada) or Aldrich
(Milwaukee, WI).
16
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
Synthesis of 5-carboxyspermine (compound I)
Unless otherwise indicated, reactions were performed at room temperature.
Compound I of Figure 1, namely 5-carboxyspermine, was synthesized using a
known
scheme (Behr, J.P. 1989. J. Chem. Soc., Chem. Commun. 101-103). Briefly to a
stirred
solution of 10.0 g (59.3 mmol) of ornithine hydrochloride dissolved in 250 ml
MeOH were
added 18.0 g (197 mmol) of tetramethylammonium hydroxide. After dissolution of
ornithine salt, MeOH was evaporated, the mixture was then dissolved in 350 mL
of dry
dimethylformamide (HPLC grade; Aldrich, Milwaukee, WI) and the residual
ammonium
salt was filtrated, yielding ornithine as its free base. Following the
addition of acrylonitrile
(2.2. equivalents, 130.9 mmol), the mixture was stirred for 16 hours in the
dark to give 10.6
g (yield=74%) of crude Na,1Va-diethylcyanide ornithine, which was subsequently
used
without further purification. White solid: IR (film) v cm' 3372 (OH, acid),
2247 (CN); `H
NMR 8 (CDC13 300 MHz) 1.48 (m, 4H, CH2CHCOOH), 2.63 (m, 6H, 3 X CjjZN), 2.86
(2xt, J,=5.9 and J1 -2.7 Hz, 4H, 2 X CH2CN), 3.07 (t, J=7.2 Hz, 1 H, CIICOOH).
To
obtain 5-carboxyspermine KOH (2.7g, 48.0 mmol) was dissolved with vigorous
stirring in
8 ml of 95% (v/v) EtOH and 10.5 g (44.1 mmol) of 1V,1V6-diethylcyanide
omithine were
then added. The resulting mixture was placed under H2 at 40 psi in a Burgess-
Parr
hydrogenator, using 2.09 g (24.4 mmol) or Raney nickel as catalyst (Behr, J.P.
1989. J.
Chem. Soc., Chem. Commun. 101-103; Bergeron, R.J. and Garlich, J.R. 1984.
Synthesis:
782-784). After 22 hours, Raney nickel was removed by filtration, and the
solvent
evaporated in vacuo, yielding 16.07 g of crude 5-carboxyspermine potassium
salt. Yellow
oil; IR (film) v cm' 3363 (OH, acid), 2937 (NH2) no cyanide band; 'H NMR 8
(CDC13,, 300
MHz) 1.53 (m, 2H, CH2 NH2), 2.65 (m, (iH, C112NH), 3.09 (t, J=5.7 Hz, 1 H,
CJICOOH).
Synthesis of 2,2'-Dithiobis(N-Ethyl-Spermine-S-Carboxamide) (DESC) and 1V
[2,2J-
Dithio(Ethyl,l ! Aminoethyl)J spermine-5-Carboxamide (DEASC)
Amine protection of 5-carboxyspermine by tert-butyl carbonyl (Boc) groups was
performed as described (Ponnusamy, E., Fotadar, U., Spisni, A. and Fiat, D.
1986.
Syntheses: 48-49). To 16.0 g (65.0 mmol) of crude 5-carboxyspermine potassium
salt
dissolved in 1.5 L MeOH were added 9.64 ml of 10% (v/v) triethylamine and 54.3
g (4.4
17
~;.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
equivalents, 286 mmol) of di-tert-butyl dicarbonate. After stirring for 24
hours, solvent
was evaporated, 100-150 ml H20 were added and the resulting niixture was
chilled at 0 C.
After adjusting pH at 2.2 with 2 N HCI, the Boc-product was extracted with
ethyl acetate,
dried over anhydrous MgSO4 and purified by C,g reversed phase silica gel
chromatography,
yielding 3.3 g of pure tetra-Boc-5-carboxyspermine (Compound II, Fig. 1).
Light yellow
solid; IR (film) v cm'' 3356 (OH, acid), 1682 (C=O, amide); ' H NMR S(CDC13,
300
MHz) 1.32 (2 x s, 36 H, (CH3)3C from Boc-N), 1.90-1.40 (m, 9H, C11ZCH2N), 3.20-
2.90
(m, IOH, CjJ2N); M (for C31H58O1aN4) - 646.41;m/z (LSIMS) = 647.42 [(M+1)*].
Coupling
of tetra-Boc-5-carboxyspermine (compound II) to cystamine was then performed
in two
steps based on the method of Venkataraman (Venkataraman, K. 1979. Tetrahedr.
Lett. 32,
3037). To a solution of 1.15 g (1.78 mmol) of compound II in 20 ml dry acetone
was added
0.27 mL (1.1 eq, 1.96 mmol) of triethylamine (freshly distilled on KOH) and
361 mg (1.1
eq, 1.96 mmol) of cyanuric chloride and the reaction mixture stirred overnight
under N2 to
form the corresponding acid chloride. Cystamine dihydrochloride (241 mg; 1.07
mmol)
was then suspended in dry triethylamine and added to the acid chloride form of
compound
II, with the resulting triethylamine concentration being at z4-fold excess
relative to the
latter. After stirring for 12 hours, the residual triazine oxide was
filtrated, acetone was
evaporated and the product extracted with CHCl31 dried over anhydrous MgSO4
and
evaporated in vacuo. The crude compound was then purified by reversed-phase
C18 column
chromatography, yielding 0.682 g of 2,2'-dithiobis[N-ethyl-(N,,N4,N",N12-tetra-
Boc-5-
carboxamide (compound IV, Fig. 1). (m) Yellow oil; IR (film) v crn' 1693 (C=O,
amide);
'H NMR 8 (CDC13, 300 MHz) 1.38 (s, 36H, (CH3)3C), 1.59 (m, 8H, CH2CH2CH2),
2.53 (6,
.F=5.7 Hz, 1 H, CONHCH), 2.73 (t, JT---6.1 Hz, 2H, CH2S),3.11 (m, l OH,
CHZNH), 3.51 (m,
2H, NCJj2CH2S):M (for C.H124NN,oS2) = 1408.85; m/z (FABMS) = 1409.9 [(M+1)*].
Compound III (215 mg in MeOH) was then deprotected by addition of 1 ml of 3 N
HCI, bringing the pH from 6.0 to =0.5. After stirring vigorously for 15 hours,
the solvent
was dried out in vacuo and the resulting compound purified by cation exchange
chromatography with a Dowex 50W-X4 column (dry mesh; 100-200; Sigma) pre-
equilibrated with H20 and successively washed with H20,1 N HCI, 2 N HCI, 4 N
HCl and
6N HCI. Ninhydrin-positive fractions eluted with 6 N HCl were pooled and
evaporated in
vacuo, yielding 96 mg of pure 2,2'-dithiobis(N-ethyl-spermine-5-carboxamide)-
18
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
octahydrochloride (DESC, Compound V, Fig. 1. White sold; mp 75-78 C; bp 118
C, 'H
NMR 8 (CDC13, 300 MHz) 1.62 (m, 2H, CH2CHCONH), 1.97-1.80 (m, 6H, CHZCHZCH),
2.74 (t, J=6.2 Hz, 2H, CH2S), 2.92 (m, 10H, CH 111~), 3.46 (dt, J--7.1 Hz, 2H,
CHZCH2S),3.84 (t, J=7.0 Hz, 1 H, CjjCONH); M (for C Ij Fib)~= 608.96; m/z
(FABMS)= 609.4 (M*).
Compound IV was similarly deprotected to yield N-[2,2'-dithio(ethyl, 1'-
aminoethyl)]spermine-5-carboxamide (DEASC, Compound VI, Fig. 1). Yellow solid;
mp
50-54 C; bp 109 C. 1H NMR S(CDC13, 300 MHz) 1.89 (m,2H,Cli2 CHCONH), 2.10-
2.29 (m, 6H Cjj2CH2CH2), 3.04 (t,J=6.0 Hz, 2H, CONHCH2CH2S), 3.19 (t, .~--7.4
Hz, 2H,
SSCH2CH2HN2), 3.25 (m, 10H, CjjZ, NH), 3.51 (t, J=6.5 Hz, 2H, SSCH2Cj12HN),
3.78
(m, 2H, CONHCH2CHZS), 4.11 (t, J=6.7 Hz, 1H, CIJCONH). M (for C,5H41ONSS2) _
380.62; m/z (LSIMS) = 381.24.
Synthesis of N-(2-Mercaptoethyl)spermine-5-Carboxamide [MESC]
DESC was dissolved in 50 mM sodium phosphate bugger, pH 8.0, containing 250
mM dithiothreitol (DTT), and incubated for 30 minutes at 37 C in a water bath.
The
mixture was then loaded on a DowexTM 50W-X4 cation exchange column
equilibrated with
H20, and after washing with 5 column volumes each of 1 N HCl and 2 N HCI, the
free thiol
was eluted with 10 volumes of 4 N HCI, Amine-containing fractions, as
identified by
mixing 5,ul aliquots with 200 1 of an o-phthaldialdehyde solution (3.7 mM o-
phthaldialdehyde; 0.4 M boric acid, pH 10.4; 1% v/v MsOH; 0.45% v/v 2-
mercaptoethanol;
0.03% w/v Bri? 35) and hearing for 20 minutes at 37 C, were then pooled. The
amount
of N-(2-mercaptoethyl)-spermine-5-carboxamide [MESC] tetrahydrochloride
(compound
VII, Fig. 1) thus isolated was titrated for thiol equivalents with 5,5'-dithio-
bis-(2-
nitrobenzoic acid) (Jocelyn, P.C. 1987. Meth. Enzymol. 143, 44-67) using
either
cysteamine or DTT as standard. The yield of MESC using this procedure was
virtually
100%, based on the number of thiol equivalents determined with 5,5'-dithio-bis-
(2-
nitrobenzoic acid) and the expected number of thiol equivalents expected per
mass of
DESC. Finally, MESC purity was confirmed by ion-pair reversed-phase HPLC using
post-
column derivatization with o-phthaldialdehyde (Pegg, A.E., Wechter, R.,
Poulin, R.,
Woster, P.M. and Coward, J.K. 1989. Biochemistry 28: 8446-8453). 'NMR 8
(CDC13, 300
19
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
MHz) 1.91 (m, 2H, CHZCHCONH), 2.08-2.24 (m, 6H, CHZCI32CH2), 2.82 (t, J=6.3,
2H,
CON HCHZCIJ2SH), 3.22 (m, 10H, CIJ2NH, 3.56 (m, 2H, CONHCjJZ CH2 SH), 4.11 (t,
J=6.6, 1 H, CHCONH).
Synthesis of Thioether Adducts of MESC with lodoacetamides
To 1 ml of an extemporaneously prepared, DTT-free solution of MESC (20 mM in
H2) were added 50 l of 50 mM Tris-HC1(pH 7.0) and 105 l of a 40 mM solution
of
either iodoacetamide, LY iodoacetamide or ASIB in a light-protected
microcentrifuge tube,
and the mixture was incubated for 2 hours at 37 C. The extent of thiol
modification was
assessed by measuring the amount of thiol remaining at the end of the
incubation with 5,5'-
dithio-bis-(2-nitrobenzoic acid) as described above, and was determined to be
essentially
complete. Excess iodoacetamide was then inactivated by adding DTT to a final
concentration of 40 mM and incubating the solution for 2 hours at 37 C. The
resulting
solutions of MESC adduct was used without further purification for
[3H]spermidine uptake
assays conducted as described below. The effect of the respective DTT-
inactivated
iodoacetamide on spermidine transport was determined in parallel by incubating
cells with
the same reaction mixture from which MESC was omitted.
Cell Culture
Both ZR-75-1 human breast cancer cells and Chinese hamster ovary cells (CHO-
Kl) were obtained from the American Type Culture Collection (Rockville, MD).
ZR-75-1
cells were maintained in phenol red-free RPMI 1640 medium supplemented with
10% fetal
bovine serum, 2mM L-giutamine, 1 mM sodium pyruvate, 15 mM Hepes, 10 nM 17p-
estradiol, and antibiotics [MEZR medium] (Huber, M. and Poulin, R. 1995.
Cancer Res.,
55, 934-943). CHO-Kl cells were routinely grown in a-Minimal Essential Medium
supplemented with 10% CosmicTM calf serum in a 5% CO2 humid atmosphere at 37
C.
Even though the present invention has been described with a certain degree of
particularity, it is evident that many alternatives, modifications, and
variations will be
apparent to those skilled in the art in light of the following disclosure.
Accordingly, it is
intended that all such alternatives, modifications, and variations which fall
within the spirit
and the scope of the invention be embraced by the defined claims.
_...-,,.____.._.....__.__- - _._...._.~._
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
F,ILAMPL.E 1
EFFECT OF INHIBITORS ON CELL PROLIFERA_TION
For growth studies, ZR-75-1 cells were cultured in MEZR medium or in phenol
red-
free RPMI 1640 supplemented with 2mM L-glutamine, 1 mM sodium pyruvate, 15 mM
Hepes, antibiotics, 1 nM 17p-estradiol, 0.5 g of bovine insulin per ml and 5%
(v/v)
charcoal-treated fetal bovine serum (SD medium), as indicated in the text.
When
polyamines or polyamine analogs were added to serum-containing media, 1 mM
aminoguanidine was added to inhibit bovine serum amine oxidase (BSAO) activity
(Morgan, D.M.L. 1989. in The Physiology ofPolyamines (Bachrach, U., and
Heimer, Y.M.
eds) Vol. I, pp. 203-229, CRC Press, Boca Raton). The effect of the transport
inhibitors
on cell growth was measured by incubating ZR-75-1 cells for 11 days in medium
supplemented with antagonist, polyamines and/or 1 mM DFMO as indicated,
followed by
colorimetric determination of DNA content with 3,4-diaminobenzoic acid
(Simard, J.,
Dauvois, S., Haagensen, D.E., Levesque C., Mbrand, Y. and Labri6, F. 1990.
Endocrinology 126: 3223-3231). Medium was changed every other day in these
experiments because of the slow reaction of the compound with an unknown
component
present in the IMEM and RPMI 1640 medium formulation.
Polyamine Analysis
ZR-75-1 cells were plated in 100 mm culture dishes at 5 x 105 cells/dish in
MEZR
medium and grown for 5 days with medium changes every other day. Fresh MEZR
medium containing the indicated concentration of transport antagonist was then
added, plus
or minus 200 M cycloheximide (CHX), and cells were incubated for 1 or 6
hours.
Medium was then removed, cell monolayers rinsed twice with 10 ml of ice-cold
CaZ+/Mg2+-
free phosphate buffered-saline (PBS) (2.7 mM KCI; 1.5 mM KH2PO4; 8.1 mM
NaZHPO4;
137 mM NaCI), and harvested by centrifugation (2000 x g x 90 s at 4 C)
following a 5- to
7- minute-incubation with bovine trypsin/EDTA solution (0.05%/0.02%, w/v) in
Hanks'
Balanced Salt Solution (Huber, M. and Poulin, R. 1995. Cancer Res. 55; 934-
943). Cell
pellets were resuspended in 300 l of 10% (v/v) trichloroacetic acid or Tris-
DTT buffer (50
mM Tris/HCI, 0.1 mM EDTA, 5 mM DTT, pH 7.5) and stored at -20 C until further
analysis. For chromatographic analysis, samples were first quickly thawed and
incubated
21
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
for 15 minutes at 37 C. Trichloroacetic acid was then added to DTT-containing
samples
to a final concentration of 10% (wt/v). Samples were dispersed for 2 minutes
in a
sonicating water bath, and pelleted in a microcentrifuge for 5 minutes. The
trichloroacetic
acid-insoluble pellet was solubilized in 300-500 l of 1 N NaOH and used to
determine
protein content using bovine serum albumin (fraction V) as standard. Polyamine
contents
were then analyzed by ion pair reverse-phase HPLC with fluorometric detection
after
postcolumn derivatization with o-phthaldialdehyde as described (Pegg, A.E.,
Wechter, R.,
Poulin, R., Waster, P.M., and Coward, J.K. 1989. Biochemistry 28: 8446-8453;
Huber, M.,
and Poulin, R. 1996. Cancer Res., 55: 934-943). In this system, putrescine,
spermidine,
spermine, MESC and DEASC were resolved with retention times of 18.5, 31.0,
35.0, 36.5,
37.5, and 44.0 minutes respectively.
DESC stability
DESC stability was tested by incubating the compound dissolved (at 50 M) in
PBS
or in IMEM medium containing 10% (v/v) fetal bovine serum plus or minus 1 mM
aminoguanidine in a humid 5% CO2 atmosphere at 37 C and in the absence of
cells. At
indicated times, trichloroacetic acid was added to aliquots of this solution
to a final
concentration 10% (w/v) and the samples directly analyzed by HPLC as above.
Determination of Polyamine Uptake Activity
The rate of putrescine and spermidine transport was determined in ZR-75-1
cells
incubated in serum-free RPMI 1640 medium as described (Lessard, M., Zhao, C.,
Singh,
S.M. and Poulin, R. 1995. J. Biol. Chem. 270: 1685-1694), using [3H
]putrescine (30
Ci/mol) and [3H]spermidine (20 Ci/mol), respectively as substrates for a 20
minute-assay
period. Spermine uptake was similarly determined, using 1 M [14C]spermine (32
Ci/mol)
as substrate. Uptake activity was expressed per amount of DNA as
fluorometrically
determined using 3,4-diaminobenzoic acid (Simard, J., Dauvois, S., Haagensen,
D.E.,
Levesque, C., M6rand, Y. and Labrie, F. 1990. Endocrinology, 126: 3223-3231).
For the
determination of spermidine uptake activity in CHO-K1 cells, 80% confluent
cell
monolayers were rinsed twice with PBS and incubated for 20 minutes at 37 C in
400 l
of buffer A (20 mM Tris-HCI, pH 7.4; 0.42 mM CaC12; 0.41 mM MgSO4, 103 mM
NaCI;
22
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
5.7 mM KCI; 1.1 mM D-glucose) containing 5 M [3H]spermidine (20 Ci/mol). Cell
cultures were then washed twice with 1 ml PBS containing 5.7 mM sym-
norspermidine.
Cells were then lysed with 200- 1 aliquot of 1 N NaOH and incubated for 30
minutes at
60 C. After neutralization with 200 l of 1 N HCI, radioactivity was
determined from a
250- 1 of the cell lysate by scintillation counting. Uptake activity was
expressed per
amount of total cellular protein as detenrnined by the method of Bradford
(Bradford, M.M.
1976. Anal. Biochem. 72: 248-254). Non-specific binding of radioactive
substrate was
similarly determined in parallel for both cell lines after a 15 second-
incubation with 400
l of ice-cold uptake solution.
Kinetic analyses
Kinetic analysis of polyamine transport was performed by determining uptake
activity in the presence of a 3 M [3H]putrescine or 1 M [3H]spermidine plus
increasing
concentrations of nonradioactive substrate. K'? K; and V. values were then
estimated by
Lineweaver-Burke analysis. For competitive inhibitors, K; values were also
estimated by
measuring uptake activity in the presence of logarithmically increasing
concentrations of
antagonist, and using the Cheng-Prusoff equation (Cheng, Y.-C. and Prusoff,
W.H. 1973.
Biochem. Pharmacol. 22: 3099-3108) by iterative curve fitting for a sigmoidal
curve. For
mixed competitive/noncompetitive inhibition, two methods were used to
calculate kinetic
constants. First, the equation
V_ ymBX
K
---~ 1 + ~ + 1 + i ;
S K1 K1
where v, s, and i are the transport velocity, substrate concentration and
inhibitor
concentration respectively, was used to calculate the inhibition constants for
inhibitor/carrier complex formation (K;) and carrier/inhibitor/substrate
complex formation
(K;') (Dixon, M. and Webb, E.C. 1976. Enzymes, 3rd Ed., Academic Press, San
Diego,
CA). Alternatively, the value of K; for a mixed competitor/non-competitor was
estimated
from the intersect of equations v" vs i at two different substrate
concentrations (Dixon, M.
and Webb, E.C. 1976. Enzymes, 3rd Ed., Academic Press, San Diego, CA).
23 --~-~-^--..._
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
Intracellular Accumulation
The time course of intracellular accumulation of spermidine in the presence of
transport antagonists was determined by incubating ZR-75-1 cells in 24-well
plates with
DESC (50 or 200 M) or MESC (200 M) in dissolved in MEZR medium containing 5
M [3H]spermidine in the presence or absence of cycloheximide (CHX, 200 M),
and
harvesting at the indicated times for the determination of intracellular
radioactive contents,
as described above for polyamine uptake assays.
Statistical Analysis
Statistical significance of differences between means was assessed by unpaired
Student's t-tests. Unless otherwise indicated, results are expressed as means
SD of
determinations from triplicate cell cultures.
Design and Synthesis of DESC, DEASC and MESC
The original rationale for synthesizing MESC (Compound VII of Fig. 1) was to
generate an affmity reagent with a thiol side chain that could be derivatized
with
fluorescent or radioactive sulfhydryl reagents to label the polyamine
transporter. The
precursor chosen for the synthesis, namely 5-carboxyspermine, has been
previously used
to prepare lipopolyamines for efficient DNA transfection (Behr, J.P. 1989. J.
Chem. Soc.
Chem. Commun. 101-103; Behr, J.P., Demeneix, B., Loeffler, J.-P. and Perez-
Mutul, J.
1989. Proc. Natl. Acad. Sci. USA 86: 6982-6986), and more recently, as a
photoaffinity
reagent to label the polyamine-binding site of casein kinase 2 (Leroy, D.,
Schmid, M., Behr,
J: P., Filhol, 0., Pares, S., Garin, J., Bourgarit, J.-J., Chambaz, E.M. and
Cochet, C. 1995
J. Biol. Chem. 270: 17400-17406). The scheme used to prepare MESC involved the
coupling of a cystamine bridge through amide bonds with two Boc-protected 5-
carboxyspermine molecules to form DESC after removal of the Boc groups
(Compound
V of Fig. 1), followed by reduction of the DESC disulfide bridge. A small
amount (10-
15%) of the mixed MESC-cysteamine disulfide (DEASC, Compound VI; Fig. 1) was
also
generated in the coupling process. Complete separation of DEASC from DESC on a
preparative basis proved to be difficult even using ion exchange
chromatography (data not
shown). Consequently, most DESC preparations contained a small amount (1-2%)
of
24
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
DEASC after reversed-phase liquid chromatography on C,? silica gel. DESC and
DEASC
were stable for months in aqueous solutions buffered at pH=7.0, whereas MESC
solutions
were supplemented with DTT to prevent oxidation.
Affinity of DESC, DEASC and MESC for the Mammalian Diamine and Polyamine
Transport
In order to evaluate the suitability of the spermine conjugates as prospective
affinity
ligands, their relative ability to inhibit putrescine and polyamine uptake was
evaluated. As
shown in Fig. 2, DESC was the most potent antagonist of [14C]spermine
transport in ZR-
75-1 cells, with a K; value about 5-fold and 16-fold lower than that of DEASC
and MESC,
respectively. The ability of spermine to compete against [3H]putrescine and
['H]spermidine
uptake was in fact only about 7-fold higher than that of DESC (Fig. 3). DESC
(Fig. 4A)
and MESC (data not shown) were pure competitive inhibitors of [3H]putrescine
uptake at
concentrations up to 100 and 200 M, respectively. On the other hand,
inhibition of
putrescine transport by DEASC belonged to a mixed competitive/non-competitive
type
(Fig. 4B). Table I summarizes the K; values determined for DESC, MESC and
DEASC
toward putrescine, spermidine and/or spermine uptake, in relation with the
mutual transport
interactions between the latter substrates. Notably, K; values of the three
spermine
conjugates with respect to putrescine uptake were 3-fold to 5-fold higher than
for spermine
uptake, unlike spermidine and spermine which both inhibited the uptake of
either substrate
with similar potency, and with a K; roughly equal to their K as substrate.
TABLE I
K, Values of Inhibition of Diamine and Polyamine Transport by
MESC, DESC and DEASC in ZR-75-1 Cells
K. or K; ( M)
Compound
Putrescine Spermidine Spermine
Putrescine 3.7 0.4 125 f 29' 0.23 f 0.138 b
Spermidine 0.23 0.058 0.49 0.158 0.37 t 0.09a
Spermine 0.33 0.02` ND 0.20 ~ 0.069
25 ---~r----
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
DESC 1.6f0.5b 2.7f 1. 1' 5.0t0.7b
MESC 22 f 3b ND 80 31b
DEASC 5.3f0.6(K)` ND 163d
4.1t0.5(K;')
Data annotated with a are from Lessard, M., Zhao, C., Singh, S.M., and Poulin,
R.
1995. J.Biol, 270: 1685-1694, b indicates data obtained with this work; mean
SD of
triplicate determinations from 2 to 4 different experiments; c corresponds to
values of
inhibition constants for carrier/inhibitor complex formation (K;) and for
carrier/inhibitor/putrescine complex formation (K;') assuming a mixed
competitive/non-
competitive model; mean SD of triplicate determinations at 3 three inhibitor
concentrations at two different substrate concentrations for a series of
increasing inhibitor
concentrations (Dixon, M, and Webb, E.C. 1976. Enzymes, 3rd Ed., Academic
Press, San
Diego, CA).
The relative potency of DESC and MESC as competitive inhibitors of polyamine
uptake was also evaluated in CHO-Kl cells, in which they respectively
exhibited K; values
of 0.92 0.15 and 33.6 f 7.2 M (Fig. 5).
E AMPLE 2
EFFECT OF SIDE CHAIN LENGTH AND SUBSTITUENTS ON
SPERMIDINE TRANSPORT INHIBITION BY MESC DERIVATIVES
The observation that MESC was a less potent inhibitor of diamine and polyamine
transport than DESC and DEASC suggested that the nature of the side chain
strongly
influences the interaction of these compounds with the carrier. The thiol side
chain of
MESC was thus derivatized with substituting groups of different sizes and
charges through
thioether linkage with three different iodoacetamides, namely LY
iodoacetamide, ASIB and
iodoacetamide itself, and the ability of the resulting complexes (MESC-LY,
MESC-ASIB,
and MESC-acetamide, respectively) to inhibit spermidine uptake was then
evaluated.
These studies were conducted using CHO-K1 cells. As shown in Fig. 5,
derivatization of
the thiol group of MESC did not significantly (P>0.10) increase the K; toward
spermidine
uptake for the three conjugates studied. In the case of MESC-ASIB, Ki values
might have
26
~:.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
been underestimated by partial inactivation of the polyamine carrier at the
assay
temperature, although the uptake reaction was conducted under subdued
lighting. Thus,
the results show that specific recognition of the spermine head of MESC can
accommodate
considerable variation in length, size, polarity or charge for the side chain
without
detrimental effect on its affinity for the polyamine carrier. Consequently,
inhibitors having
different side chains, while maintaining their inhibitory activity on
polyamine transport are
also encompassed by the present invention.
EXAMPLE 3
LACK OF PERMEATION OF DESC AND MESC
THROUGH THE POLYAMINE TRANSPORT SYSTEM
The ability of ZR-75-1 cells to accumulate DESC and MESC was determined.
Since DESC was eluted as a late, broad peak in the HPLC system used, DTT was
added
to cell extracts to reduce DESC to MESC and decrease the detection threshold.
Results are
shown in Table II. ZR-75-1 cells were incubated for 1 or 8 hours in MEZR
medium in the
presence of 50 or 200 M DESC or MESC prior to determination of polyamine
contents.
CHX was added at 200 M where indicated. Other details are provided under
"Materials
and Methods." Values are the mean f SD of triplicate determinations from 2
independent
experiments.
27
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
TABLE II
Intracellular Accumulation of DESC and MESC in ZR-75-1 Cells
Polyamine intracellular contents (nmoUmg protein)
Addition Time Spermidine Spermine DESC MESC
(h)
Control 1 0.69 t 0.08 8.22 0.48 - -
6 0.91f0.07' 9.16t0.13 - -
+50 MDESC 1 0.81f0.14 8.27f0.81 <0.01 <0.01
6 0.73 f 0.11 8.60 f 0.29 <0.01 <0.01
+200 gM DESC 1 0.79 0.11 8.77 0.79 <0.01 <0.01
6 0.76t0.11 8.66t0.26 0.12t0.01 <0.01
+200 gM DESC 1 0.75 0.04 9.57 0.31 <0.01 <0.01
+ CHX
6 0.70t0.03 9.55t0.13 0.10f0.01 <0.01
+ 50 M MESC 1 0.95 0.11 7.77 0.06 <0.01 <0.01
6 0.75f0.11 8.13t0.17 <0.01 <0.01
+ 200 M MESC 1 1.15 10.078 8.93 0.53 <0.01 0.020 0.005
6 0.81t0.15 8.32t0.43 <0.01 0.13f0.06
a Significantly different (P<0.5) from control value at time = 1 h. (?)
As shown in Table II, only trace amounts of DESC could be recovered in ZR-75-1
cells after a 6-hour incubation with 200 M DESC, but not with 50 M; DESC
could be
detected only after reduction with DTT. These levels represent only about 1.5%
of the
accumulation measured in ZR-75-1 cells under identical conditions for spermine
(Lessard,
M., Zhao, C., Singh, S.M. and Poulin, R., 1995. J. Biol. Chem. 270: 1685-
1694).
Moreover, inhibition of protein synthesis by cycloheximide (CHX), which is
known to
upregulate polyamine uptake by preventing the synthesis of a polyamine-induced
feedback
repressor of transport (Lessard, M., Zhao, C., Singh, S.M. and Poulin, R.
1995. J. Biol.
Chem. 270: 1685-1694; Mitchell, J.L.A., Diveley, R.R., Jr. and Bareyal-Leyser,
A. 1992.
Biochem. Biophys. Res. Commun. 186: 81-88), did not enhance DESC
internalization, in
marked contrast with its effect on spermidine accumulation under similar
conditions (Fig.
6B) (Lessard, B., Zhao, C., Singh, S.M. and Poulin, R., 1995. J. Biol. Chem.
270: 1685-
1694). Likewise, MESC was accumulated to measurable levels by ZR-75-1 cells
only
28
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
when present at 200 M (cf. Table II). Thus, neither DESC or MESC appear to be
used
as substrates for the polyamine transport system despite the high affinity of
the former
compound as an antagonist of diamine and polyamine uptake.
F.XAMPLE 4
EFFECT OF DESC AND MESC ON
INTRACELLULAR POLYAMINE ACCUMULATION
The time course of internalization of radio-labeled spermidine was determined
in
ZR-75-1 cells incubated for up to 6 hours in the presence of the impermeant
agonists. As
illustrated in Fig. 6A, steady-state [3H]spermidine accumulation in the
absence of
competitor abruptly reached a near plateau after about 1 hour, which results
from the
induction of feedback inhibition of polyamine transport (Lessard, M., Zhao,
C., Singh, S.M.
and Poulin, R., 1995. J. Biol. Chem. 270: 1685-1694). MESC and DESC decreased
the
initial rate of spermidine uptake according to their respective potency as
competitive
antagonists. Interestingly, spermidine accumulation in the presence of either
inhibitor
followed a pattern similar to that of control cells, i.e. a rapid phase during
the first 60
minutes, followed by a much slower rate of accumulation thereafter, which was
nearly
independent of antagonist concentration. This pattern suggests that even
cellular levels of
newly internalized spermidine as low as 20% of those found under control
conditions, e.g.,
in cells treated with 200 M DESC, may induce a near maximal degree of
feedback
repression of polyamine transport. Nevertheless, even a 40-fold excess of the
most potent
antagonist (i.e. 200 M DESC) only decreased net spermidine accumulation by
only 50%
after 6 hours. As previously observed ((Lessard, M., Zhao, C., Singh, S.M. and
Poulin, R.,
1995. J. Biol. Chem. 270: 1685-1694), CHX abolished the induction of feedback
transport
inhibition, resulting in a 4-fold increase in spermidine accumulation after 4
hours (Fig. 6B).
Protein synthesis inhibition also enhanced spermidine accumulation in DESC-
treated cells,
a finding consistent with the onset of substantial feedback transport
repression by
subthreshold levels of internalized substrate. Thus, in the absence of the
feedback
mechanism, the highest concentration of DESC tested (200 M) decreased net
[3H]spermidine accumulation by 80 to 85% after 6 hours and to a level lower
than that
found in control cells with a fully repressed uptake activity.
29
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
EXAMPLE 5
EFFECT OF DESC, DEASC AND MESC
ON CELL PRO IFERATION
Due to the analogy of the novel transport antagonists with spermine, it might
be
surmised that they would exhibit significant cytotoxicity like the parent
molecule. The
marked toxicity of low (<10'3M) spermine concentrations in biological media
mostly results
from catabolism by copper amine oxidases, which generates a dialdehyde,
acrolein and
H202 as deleterious products and can be iureversibly inhibited by carbonyl
reagents such
as aminoguanidine (Morgan, D.M.L. 1989. in The Physiology of Polyamines
(Bachrach,
U., and Heimer, Y.M. eds) Vol. I, pp. 203-229, CRC Press, Boca Raton). The
biocompatibility of DESC, MESC, and DEASC was thus evaluated during a long-
term (11-
day) incubation with ZR-75-1 cells grown in RPMI 1640 containing 10% (v/v) FBS
in the
absence and presence of 1 mM aminoguanidine. As shown in Fig. 7,
aminoguanidine alone
had a slight inhibitory effect on ZR-75-1 cell growth as previously observed
(Huber, M.
and Poulin, R. 1995. Cancer Res. 55: 934-943). Although DESC was only mildly
growth
inhibitory at 50 M, there was an abrupt, aminoguanidine-resistant increase in
toxicity at
200 M. In contrast, spermine was acutely cytotoxic at 50 M, an effect that
was only
partly prevented by aminoguanidine. MESC was considerably less toxic than its
dimer,
with a 35% decrease in cell growth at 200 M which was not blocked by
aminoguanidine.
On the other hand, 50 M DEASC caused a 20% inhibition of cell proliferation
which
could be completely prevented by the amine oxidase inhibitor. Thus, DESC, and
to a much
lesser degree, its thiol monomer MESC, are cytotoxic toward breast cancer
cells at high
concentrations through a mechanism that does not involve BSAO. Weak growth
inhibition
caused by the mixed MESC-cysteamine disulfide, however, apparently involved
degradation by a copper amine oxidase.
F.XAMPL.E 6
EFFECT OFDESC ON RESCUE OF DFMO-INDUCED
GROWTH INHIBITION BY EXOGENOUS SPERMIDINE.
Although DESC is indeed a potent antagonist of polyamine accumulation, the
slow
residual uptake that occurred even at a 40-fold molar excess of inhibitor
might be sufficient
to counteract polyamine depletion by inhibitors of polyamine biosynthesis.
This possibility
~:.
CA 02304557 2000-03-22
WO 99/54283 PCTIUS98/07806
was assessed by comparing the ability of DESC to prevent the reversal of DFMO-
induced
growth inhibition by increasing concentrations of exogenous spermidine. At
concentrations
superior to 0.3 M, spermidine inhibited ZR-75-1 cell proliferation by up to
20% (Fig. 8).
This effect could be due to an incomplete inhibition of BSAO by aminoguanidine
(Seiler,
N. 1987. in Inhibition ofPolyamine Metabolism. Biological Significance and
Basis for New
Therapies (McCann, P.P., Pegg, A.E. and Sjoerdsma, A. eds.), pp. 49-77,
Academic Press,
Orlando), since it was not observed in media supplemented with equine serum,
which does
not contain amine oxidase activity (Blaschko, H. and Hawes, R. 1959. J.
Physiol. 145: 124-
131), instead of FBS (data not shown). The approximately 50% growth inhibition
induced
by 1 mM DFMO after an 11-day incubation was completely reversed by as little
as 0.3 M
spermidine, whereas 0.1 M spermidine already restored growth of DFMO-treated
cells
to 78% of control value. However, addition of 50 M DESC was unable to prevent
the
reversal of DFMO-induced growth inhibition by spermidine, even at a
DESC:spermidine
ratio of 500. Essentially similar results were obtained using horse serum
instead of FBS,
or replacing RPMI 1640 medium, which contains 3.2 M reduced glutathione that
might
undergo thiol/disulfide exchange with DESC, with thiol-free IMEM (data not
shown).
F.XAMPLE 7
STABILITY OF DESC IN BIOLOGICAL MEDIA
The inability of DESC to block the biological effect of exogenous spermidine,
even
when present at large molar excesses, might have been caused by its
degradation in growth
media. To assess this hypothesis, DESC solutions (20 M) made in PBS or in
sterile
IMEM medium enriched with 10% (v/v) FBS were incubated for 20 minutes or 48
hours
under cell-free conditions at 37 C in a humid 5% CO2 atmosphere, and the
polyamine
analog was then analyzed by ion-pair reversed-phase HPLC. After 48 hours,
degradation
of DESC to two new amine-containing derivatives occurred in IMEM (Fig. 9A, B)
but not
in PBS (Fig. 9C), as evidenced by the appearance of a major (compound 1) and
minor
(compound 2) peaks of o-phthaldialdehyde-reactive material eluting earlier
than DESC.
Although aminoguanidine did not prevent DESC degradation to the two unknown
products,
it did prevent the degradation of a trace amount of DEASC (indicated as
compound 3)
initially present in the DESC preparations, thus confirming that DEASC can
indeed be a
31
CA 02304557 2000-03-22
WO 99/54283 PCTIUS98/07806
substrate of serum copper amine oxidase (Fig. 7). MESC could not be detected,
indicating
that DESC does not undergo reduction to MESC under conditions used for cell
culture.
Furthermore, the decomposition of DESC in IMEM showed an identical pattern in
the
presence or absence of FBS, which thus ruled out a serum component as being
responsible
for the degradation. Figure 10 shows that DESC was slowly degraded to
compounds 1 and
2. After 48 hours, i.e. the interval at which freshly made DESC-containing
media were
added to cell cultures in growth experiments, 40% of the DESC originally
present had been
decomposed by IMEM. Identical results were obtained using RPMI 1640 medium
instead
of IMEM. Thus, the present inventors propose that a component present in IMEM
and
RPMI 1640 medium, but not in PBS, must be responsible for the degradation of
DESC.
DESC, a novel type of spermine derivative, is shown to be endowed with high
affinity for the polyamine transport system while being highly resistant to
cellular uptake.
The combination of these two attributes confers unique characteristics to DESC
as a pure
competitive antagonist of polyamine uptake.
As compared with spermine, the higher Ki of MESC against putrescine,
spermidine
and spermine uptake could owe to the presence of an amide linkage, which
decreases the
basicity of the neighboring secondary amino group of the spermine head (pKa =
5.5 in
comparison with 8.9-9.8 for spermine) (Tabor, C.W. and Tabor, H. 1984. Ann.
Rev.
Biochem. 53: 749-790; Remy, J.-S., Kichler, A., Mordvinov, V., Schuber, F. and
Behr, J.-
P. 1995. Proc. Natl. Acad. Sci. USA 92: 1744-1748, and/or may cause steric
hindrance for
its interaction with the polyamine binding site (Bergeron, R.J. and Seligsohn,
H.W. 1986.
Bioinorg. Chem. 14: 345-355; Porter, C.W., Cavanaugh, P.F., Jr., Stolowich,
N., Ganis, B.,
Kelly, E., and Bergeron, R.J. 1985. Cancer Res. 45: 2050-2057). Despite the
particular
structural features of MESC as a ligand, its dimerization into DESC increased
by up to 20-
fold the affinity of the resulting structure for the polyamine transporter.
There is no
precedent for dimeric polyamine structures like DESC. Its overall design is
reminiscent
of that of 2-N-4-(1-azi-2,2,2; tri-fluoroethyl)benzoyl-1,3-bis(D-mannos-4-
yloxy)-2-
propylamine, an impermeant ligand which binds to the exofacial domain of
facilitative
glucose transporters and bears two symmetrical sugar moieties linked tail to
tail (Clark,
A.E. and Holman, G.D. 1990. Biochem. J. 269: 615-622). At least one mammalian
glucose
transporter, namely GLUT-1, exists as a tetrameric complex in its native form
(Hebert,
32
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
D.N. and Carruthers, A. 1992. J. Biol. Chem. 267:23829-23838; Gould, G.W. and
Holman,
G.D. 1993, Biochem. J. 295: 329-341). The stronger affinity of DESC relative
to MESC
could reflect a dyad symmetry in the organization of the transporter complex.
Alternatively, dimerization of MESC into DESC could impose conformational
constraints
(e.g. due to electrostatic repulsion) that would favor recognition of the
polyamine binding
site of the carrier by each of the symmetrical spermine moieties.
MESC thioethers as diverse in size as MESC-LY, MESC-ASIB, or MESC-
acetamide had K; values virtually identical to that of MESC, indicating that
the thiol group
of MESC does not specifically determine its lower affinity as a polyamine
transport
inhibitor as compared with DESC. These data suggest that additional bulk on
the side
chain has little influence on the interaction of MESC with the polyamine
transporter, in
agreement with the observation that large substituents attached to the distal
end of a spacer
of sufficient length do not notably decrease the affinity of spermidine as a
substrate for
uptake (Holley, J.O., Mather, A., Wheelhouse, R.T., Cullis, P.M., Hartley,
J.A., Bingham,
J.P., and Cohen, G.M. 1992. Cancer Res., 52: 4190-4195). Unexpectedly, the
MESC-
cysteamine mixed disulfide (DEASC) was found to block putrescine uptake as a
mixed
competitor/non-competitor, whereas MESC and DESC behaved like pure competitive
inhibitors of putrescine transport. Since the interaction of DESC or MESC with
the
polyamine transporter was strictly competitive, and because DEASC exhibits
higher
affinity than MESC as an inhibitor of diamine and polyamine transport, the
spermine head
and the cysteamine side chain of DEASC might be respectively responsible for
the
competitive and non-competitive components of its transport inhibition.
The biochemical properties of DESC clearly illustrate that the binding
affinity of
a compound can be dissociated from its ability to serve as a substrate for the
polyamine
transporter. The large size of DESC cannot be the main factor preventing its
intemalization
through the channel-like portion of the transporter since MESC was also
virtually
impermeant. Thus, the mere attachment of an amido side chain on the spermine
backbone
would appear to be responsible per se for the impaired internalization of MESC
and its
derivatives. Indeed, 1V¾-alkylated spermidine derivatives are far better
competitors of
spermidine uptake than their N-acyl counterparts in mouse leukemia cells, in
support of
the notion that charged secondary amino groups are important in the
interaction with the
33
CA 02304557 2000-03-22
WO 99/54283 PCTNS98/07806
polyamine carrier (Porter, C.W., Cavanaugh, P.F., Jr., Stolowich, N., Ganis,
B., Kelly, E.,
and Bergeron, R.J. 1985. Cancer Res. 45: 2050-2057). However, the latter
argument
cannot account for the fact that long-chain aliphatic a, w-diamines with at
least 6 to 7
methylene groups have an affinity comparable to that of spermidine (Lessard,
M., Zhao,
C., Singh, S.M. and Poulin, R., 1995. J. Biol. Chem. 270: 1685-1694, Bergeron,
R.J. and
Seligson, H.W. 1986. Bioinorg. Chem. 14: 345-355; Porter, C.W. and Bergeron,
R.J. 1983.
Science 219: 1083-1085; Minchin, R.F., Martin, R.L., Summers, L.A. and Ilett,
K.F., 1989.
Biochem. J. 262: 391-395; Gordonsmith, R.H., Brooke-Taylor, S., Smith, L.L.
and Cohen,
G.M. 1983. Biochem. Pharmacol. 32: 3701-3709). A more likely explanation for
the poor
affinity of polyamines bearing an acyl side chain might be the steric
hindrance due to the
amide group, which restricts the freedom of rotation around the adjacent
carbon and
nitrogen atoms. There are indications that cyclic or pseudocyclic
conformations of
polyamines stabilized by hydrogen bonds might be energetically favored for
recognition
and/or internalization of substrates of the polyamine transport system
(Lessard, M., Zhao,
C., Singh, S.M. and Poulin, R., 1995. J. Biol. Chem. 270: 1685-1694; Bergeron,
R.J. and
Seligsohn, H.W.1986. Bioinorg. Chem. 14: 345-355). The formation of such
folded
conformers would be impaired by the presence of an amide group next to the
polyamine
chain. In support of this hypothesis, chlorambucil-spermidine, which bears a N-
propyl
chlorambucil carboxamide side chain on the central nitrogen of spermidine, is
a good
substrate of the polyamine transport system, with a K. averaging that of
spermidine
(Holley, J.L., Mather, A., Wheelhouse, R.T, Cullis, P.M., Hartley, J.A.,
Bingham, J.P., and
Cohen, G.M. 1992. Cancer Res. 52: 4190-4195). In marked contrast, a spermidine
conjugate with a chlorambucil carboxamide side chain directly attached at the
C5 position
of the spermidine head is a very poor substrate of the polyamine uptake system
(Stark, P.A.,
Thrall, B.D., Meadows, G.G., and Abdel-Monam, M.M. 1992. J. Med. Chem. 35:
4264-
4269).
Although a 40-fold molar excess of DESC dramatically reduced the rate of
spermidine uptake in ZR-75-1 cells, slow but continuous spermidine
accumulation was still
observed in the presence of the inhibitor. The low rate of polyamine
internalization
observed even in the presence of a large excess of DESC, in addition to the
slow
34
----- -_----~---..,_._. ___ _-- __ . -----.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
decomposition of the inhibitor, may largely explain the complete inability of
DESC to
prevent polyamine-mediated prevention of growth inhibition by DFMO.
Since the affinity of MESC thioethers remains virtually unaffected relative to
the
unconjugated polyamine, MESC-ASIB might serve as a photoaffinity label to
detect
polyamine-binding proteins, including the polyamine carrier. Experiments are
currently
conducted with'ZSI-labeled MESC-ASIB to assess its usefulness as a probe to
identify the
mammalian polyamine transporter. A recent report has described the specific
labeling of
discrete plasma membrane proteins using125I-labeled N'-azidosalicylamido-
norspermine
and N'-azido-salicylamidoethylspermidine as photoaffinity reagents (Felschow,
D.M.,
MacDiannid, J., Bardos, T., Wu, R., Woster, P.M. and Porter, C.W. 1995. J.
Biol. Chem.
270: 28705-28711). However, these conjugates are internalized by mammalian
cells
(Felschow, D.M., MacDiarmid, J., Bardos, T., Wu, R., Woster, P.M. and Porter,
C.W.
1995. J. Biol. Chem. 270: 28705-28711), and MESC-ASIB or similar derivatives
could be
useful as a photoactivatable probes to exclude labeling of intracellular
proteins.
While not intending to be limited to any particular theory, the slow
degradation of
DESC observed in growth media, but not in PBS, was likely due to L-cystine,
which is
present at 100 and 200 M in IMEM or RPMI 1640 medium, respectively, through
the
formation of mixed disulfides with DESC. Nevertheless, the cytotoxicity of
high
concentrations of DESC and MESC is unlikely to be solely due to the formation
of such
adducts, since MESC was less toxic than DESC, despite the fact that the free
thiol group
of the former would make it more reactive toward L-cystine]. The present data
clearly
show that DESC has remarkably low toxicity in comparison with its homolog
spermine.
Thus, the basic features of this molecule, including its resistance to BSAO,
should be
useful for the design of potent transport inhibitors with minor non-specific
effects on cell
viability. The inherent structural features of DESC that confer its high
affinity and
resistance to uptake should thus provide a useful framework for the design of
potent
irreversible inhibitors of polyamine transport, which could incorporate an
alkylating group
such as that used in the design of specific suicide substrates of mammalian
glucose
transporters (Clark, A.E., and Holman, G.D. 1990. Biochem. J. 269: 615-622;
Lehmann,
J., and Scheuring, M. 1995. Carbohydrate Res. 276: 57-74)].
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
Polyamine derivatives (natural or synthetic) comprising sulfur in the side
chain have
been made, because they conducted to the formation of dimers simply by forming
a
disulfide bridge. By-products which are not dimers have also shown an
activity. However,
it will be readily apparent to those skilled in the art that compounds being
more stable than
those containing sulfur atoms are contemplated. Therefore, the side chains
used for
increasing the affinity of the derivatives for a polyamine transporter and/or
as substrates for
labeling molecules and/or as a spacer in the making of a dimer can be varied
to optimize
the characteristics of the derivatives of the present invention.
Any equivalent structures or modifications obtainable without departing from
the
teachings and the spirit of this invention are considered as part of the scope
thereof because
the invention is in no way limited to the particularly disclosed embodiments,
as reflected
in the appended claims.
EXAMPLE 8
SYNTHESIS AND EVALUATION OF SPERMINE DIMERS AS INHIBITORS
OF POLYAMINE TRANSPORT AND ENHANCERS OF EFI.ORNITHINE
ACTION IN T MQRCFLLS AND TUMO_R_-BEARiNG ANIMAi.S
Novel spermine analogs will be synthesized and evaluated as blockers of
transport
in tumor cells simultaneously treated with D,L-a-difluoromethylornithine (FMO
=
Eflomithine). These molecules are based on the overall design of a prototype,
2, 2'-
dithiobis(N-ethyl-spermine-5-carboxamide) (DESC). DESC has recently been
reported to
act as a competitive and potent antagonist of polyamine uptake in leukemia and
breast
cancer cells. DESC is proposed here to potentiate the chemotherapeutic
efficacy of DFMO.
While not intending to be limited to any particular theory, it is proposed
that such effect is
provided by preventing the replenishment of DFMO-treated tumor cells with
polyamines
from exogenous sources. Structural modifications to the molecule will improve
it to a
pharmacologically useful compound. These modifications include: [i] the
replacement of
the disulfide bridge with a fully reduced aliphatic chain to prevent its rapid
reaction with
biological thiols and disulfides and [ii] the addition of substituents to
prevent its oxidative
deamination by the ubiquitous plasma enzyme, serum amine oxidase.
Two types of DESC analogs will be synthesized, and characterized for their
ability
to inhibit polyamine transport and to enhance the therapeutic action of DFMO
in various
36
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
tumor cell types, including animal models. The first type of analogs will be
simply
obtained by substituting the original cystamine side chain of DESC with a,cj-
diamine
cross-linkers of varying length. The synthesis of these analogs will help in
the short term
to optimize the length of the cross-linker chain, and to rapidly evaluate
their relative ability
to potentiate DFMO action in vitro. The second type of analogs will be made
according
to a new route of synthesis to introduce methyl groups at the extremities of
the spermine-
like backbone, and will also incorporate alkylation instead of acylation of
the aliphatic,
a,cj-diamine cross-linker in order to improve their affinity for the polyamine
transport
system, their potency as antagonists of uptake and as enhancers of DFMO
therapeutic
action. The pharmacological evaluation of the second-type analogs will be
conducted in
a standard mouse model bearing L12101eukemia tumor cells treated with DFMO.
2,2'-dithiobis (N-ethyl-spermine-5-carboxamide) (DESC) and its thiol monomer,
N-(2-mercaptoethyl) spermine 5-carboxamide (MESC) (Fig. 11) have been
synthesized as
precursors of photoaffinity labeling probes of polyamine-binding proteins
(21).
Characterization of the potency of DESC and MESC to inhibit polyamine
transport
unexpectedly showed that DESC has -20-fold higher affinity than MESC for the
polyamine carrier. The marked difference in transport inhibitory potency
between MESC
and its dimer suggested that the carrier protein might have a dyadic symmetry,
and that the
conjugation of two spermine molecules through a cross-linking side chain could
markedly
enhance the interaction with the polyamine transporter. Moreover, neither DESC
nor
MESC is significantly internalized by human breast cancer cells or mouse
leukemia cells
at concentrations that saturate the polyamine carrier, indicating that they
are essentially
membrane-impermeant (21). The combination of high affinity and lack of carrier-
mediated
permeation of DESC provided the basis for a novel design of pure polyamine
transport
antagonists that could be used in combination with DFMO to enhance polyamine
depletion
in tumor cells exposed to physiological levels of exogenous polyarnines.
DESC was designed for biochemical use. It was found to degraded in
physiological
media due to thiol-disulfide reaction with compounds such as L-cystine. DESC
cannot
efficiently counteract the ability of exogenous spermidine to reverse DFMO-
induced
cytostasis in breast cancer cells as a result of this instability (21). DESC
is also subject to
attack by serum amine oxidase (SAO), an ubiquitous plasma enzyme which
oxidatively
37
CA 02304557 2006-11-22
deaminates aminopropyl groups, albeit to a much lesser degree than the parent
compound,
spenmine. Modifications that further improve the design of DESC analogs that
are part of
particular embodiments of the present invention are:
(1) To use chemically inert, aliphatic diainines as cross-linkers to conjugate
two
spermine-like moieties; and/or
(2) To introduce methyl groups on the terminal carbons of the spermine-like
backbones of the molecule. This latter modification will prevent/reduce the
oxidation of
spermine by SAO.
8.2) Synthesis and biochemical evaluation of unmethylated, stable DESC analoQs
DESC analogs are prepared with unmodified spermine backbones but different
side chain
lengths as lead compounds to guide us in the design of methylated analogs
described
herein. This series of compounds will be synthesized in order to:
(i) Perform a structure-function study in the short-term to deten:nine the
optimal length
of the cross-linker for inhibition of polyamine uptake.
Compounds VIIIa to VIIID (Fig. 12) will be rapidly.available in amounts
sufficient for in
vitro testing. A refinement to the originally proposed route of synthesis will
be the use of
FMOC-blocked dianu'ne precursors. One such diamine precursor is NHZ(CH),,NH2
where
n = 3 to 6. Instead of simultaneously coupling two spermine-like moieties to a
diamine
cross-linker, each amino group of the. diamine cross-linker was sequentially
amidated to
the spermine-Iike precursor with the 1V-FMOC-diamine, and then the other amine
group
was deprotected for the second amidation reaction. This sequential reaction
scheme
improves the purification of the spermine dimer from the spermine monoamide.
This was
difficult to achieve with the original method. The present method will improve
the yield
of desired product through a better control of the reaction stoichiometry.
The kinetic properties of these DESC analogs (abbreviated as BS-3, BS-4, BS-5
and
BS-6; Fig: 12), as inhibitors of polyamine transport will be determined by
uptake assays
of radiolabeled putrescine, spermidine and spermine, according to procedures
in Huber et
al. (1996), J. Biol. Chem., 271: 27556=27563. These structures are shown
below.
38
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
H Zh! NH ~'~..=~'~..~. NH ^~,...=~=,..~ NH 2
=Q
NH
i
(CM2 )n
NH
I
C-
H 2N N H N H NH 2
wherein n is 3, 4, 5 or 6.
(ii) Use of analogs to potentiate the effect of DFMO in the presence of
exogenous polyamines, which is the main criterion of pharmacological activity
for
polyamine transport inhibitors.
These compounds are expected to be stable under cell culture conditions in the
presence of aminoguanidine, a SAO inhibitor (13, 28, 40, 46, 49, 66, 67).
These polyamine
transport inhibitors will be evaluated using ZR-75-1 human breast cancer cells
and L1210
mouse leukemia cells. Briefly, the rate of cell proliferation will be
deterniined in ZR-75-1
and L 1210 cells grown in the presence or absence of DFMO (1 and 5 mM,
respectively),
and of the transport inhibitor candidate to be analyzed, in the presence of
increasing
concentrations of putrescine or spermidine. The ability of the transport
antagonist to
prevent the reversal of DFMO-induced growth inhibition by exogenous putrescine
or
spermidine will provide a valid measurement of the pharmacological potential
of these
compounds as enhancers of DFMO action in vivo. These studies will also include
(a) dose-
response experiments to evaluate the cytotoxicity of these analogs and the
optimal
concentration for their use as inhibitors of polyamine uptake, and (b)
measurement of the
uptake of the tcansport inhibitors during incubation with tumor cells by HPLC,
along with
their effect on polyamine pools.
Since the latter type of inhibitors will rapidly provide the first stable DESC
analogs
available, the thorough analysis of their biological properties with cultured
tumor cells will
be important to validate the concept of spermine dimers as polyamine transport
blockers.
Moreover, the structure-function relationships of this series will help in
refining the design
of the methylated analogs described in the following section.
39
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
8.3) Desigjy synthesis and evaluation of oxidatiQn_re, is~ tant' stable
methylated DESC
gnaloLys
While replacing the nature of the cross-linking chain is a rather
straightforward
modification, the second alteration required considerable changes in the
preparation scheme
originally used for DESC synthesis. The reduction step proposed to obtain a
1,12-
dimethylspermine 5-carboxyl methyl ester from a 1,4-bis(3-azidobutyramido)
omithine
methyl ester (Fig. 13, XIII) could not spare the ester group, resulting
instead into the
formation of 1,12-dimethylspermine 5-carbinol (Fig. 3, XIV). Changing the
nature of the
ester group did not improve the synthesis since steric hindrance problems
prevented
amidation of the amino groups of ornithine methyl ester the most proximal to
the ester
group. The nature of the proposed precursor was modified, and two 1,12-
dimethylsperrriine
5-methyl chains were conjugated to an a,cj-diamine cross-linker through
alkylation rather
than through amide bonds (Figs. 14 to 16). This modification represents an
improvement
over the original design, since direct alkylation will lead to compounds with
a higher
affinity for the polyamine transporter - and higher potency as transport
antagonists - as
compared with the more rigid acylated analogs, as previously shown for
spermidine analogs
(8, 19, 44, 52, 54, 56). The proposed scheme of synthesis, for which steps IX
to XIV have
already been realized, is provided in Figs. 13 to 16. This improved scheme
also includes
the use of mono-FMOC-protected diamines as building blocks for cross-linking
the
dimethylspermine-5-methyl precursors, as described above for the unmethylated
DESC
analogs. The resulting compounds are abbreviated as BMS-3, BMS-4, BMS-5 and
BMS-6
(Fig. 16; compounds XXa to Xxd).
HzN NH'..~ r NH2
NN
?'a
NH
I
t~~n
NH
1
CHt
H2N NH
"**~ '
NH NH2
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
wherein n is 3, 4, 5 or 6.
The in vitro evaluation of this series will be conducted for the unmethylated
DESC
analogs. The effectiveness of a combination of polyamine depletion with the
selected PA
transport antagonist as an antitumor strategy will then be assessed in vivo.
For this purpose,
an established experimental cancer model, namely L1210 mouse leukemia, will be
used to
evaluate the therapeutic potential of the candidate transport inhibitor. This
leukemia model
is an aggressive tumor type with a median host survival time of 9 days in the
absence of
treatment. Moreover, it is completely resistant to DFMO as a single tumor
agent in vivo
(albeit very sensitive in vitro), whereas PA transport deficiency and/or
reduction of
exogenous PA sources confers a striking ability to DFMO to extend survival
rates, with
complete cure being observed in Z75% of animals (1, 50).
Protocol 1- Toxicity will first be determined by single i.v. and i.p.
injections of
logarithmically increasing drug concentrations to mice and estimating the
LD50. Blood
samples will be taken at intervals to measure the plasma drug concentration by
ion pairing
reverse-phase HPLC (22, 23). Body weight and liquid consumption will also be
monitored
for 10 days, at the end of which period animals will be sacrificed to evaluate
the incidence
of liver and kidney damage. A similar experiment will be conducted by
dissolving the drug
in the drinking water with free access to the animals.
Protocol 2- On day 0, mice will be injected with L1210 cells, with concomitant
treatment with DFMO or vehicle, plus or minus 2 different sublethal doses of
the transport
antagonist on a daily schedule. Oral, i.v. and i.p. routes will be compared
for the transport
antagonist. Survival will be evaluated for up to 120 days, with regular body
weight
measurements and blood sampling to deternune the steady-state plasma
concentrations of
inhibitor. L1210 cells are strongly immunogenic tuinors and cured animals
develop
extended immunity against this leukemia (1). Thus, to evaluate the curative
potential of
the drug combination, survivors will be rechallenged with L 1210 cells in the
absence of
treatment and survival monitored.
41
~..
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
EXAMPLE 9
The present example demonstrates the utility of the present invention with the
use
of compounds that are analogs of spermine that include two chains connected to
one
another through a linker. The linker molecule that attaches the two spermine
chains may
be any spacer chain that is capable of bridging the polyamine chains.
The two chains may attach to the linker at an internal C atom or an N group
within
the chain. It is also possible for one chain to be connected to the linker
through one of its
carbon molecules, while the second chain attaches to the linker molecule
through an N
group within its chain.
The general structure of compounds claimed include the following
characteristics:
(1) The central carbon chain of the spermine backbone can have between 3 and
7 methylene groups or carbon atoms. This is the range of central chain length
that can be
accommodated with good affinity by the mammalian polyamine transporter (81).
(2) Each methylene group of the polyamine chains can be modified by methyl
groups without compromising the ability of the inhibitor to interact with the
polyamine
transporter.
(3) The linkage between the polyamine chains and the spacer may comprise any
type of linkage compatible with a K, s20 M (relative to spermine) for the
resulting
inhibitor, such as direct alkyl substitution or ether group on the central
methylene groups
(Structure 1), or alkylation on the secondary amino (Structure 2) groups of
the polyamine
chain.
Structure 1
R2 R2 R2 R2 i2
RIHN---~-~-~NFi- +Y- C ~TN~ ~.- NHRy
R2 R2 R3 R2 R2
1
L
l
R2 R2 I3 i2 R2
RIHN~ ~N~~~C~T)-NH-( --- NHR,
R2 R2 R2 R2 R2
42
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
wherein R, is H, methyl, ethyl or propyl, R2 is H or methyl, x is greater than
two and less
than five (2<x<5), and the sum of y+z is greater than or equal to 2 and less
than or equal
to 6(2sy+zs6). R3 = Ch2, S, C=O or NH; 2< x <5; 2 s y+> s 6; L= a chemical
structure
(the linker) connecting covalently the two polyamine chains via alkyl, amide,
ether or
thioether bonds with a substituent group (R3) attached on a carbon atom
located between
the two most internal amino groups of the polyamine chain.
Structure 2
R2 R2 R2
R,HN~ ~ X -N ~-~--NH--(-~~ ~-- NHRi
R2 RZ R2
L
R` R2 R2
RIHN4~4x-NNH-{-C;-~ NHR.,
R; . R2 ` (R2
wherein R, is H, methyl, ethyl or propyl, R2 is H or methyl, x is greater than
two and less
than five (2<x<5), w is greater than 2 and less than 8 (2<x<8) and the sum of
y+z is greater
than or equal to 2 and less than or equal to 6(25 y+zs6).
The invention when a carbon of one chain is attached by a linker to the
nitrogen of
a second chain is represented in Structure 3.
Structure 3
RZ RR2 R2 R2
-~-N-NHRi
R,HN--` -~--N"' ( ~C-+~` I
T I R2 F 2 R3 R2 R2
(
L
R~ R2 R2
RiHN--~~~N-t 4~N~~ NHR,
F2 `RI2 R2
43
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
wherein R, is H, methyl, ethyl or propyl, R2 is H or methyl, x is greater than
two and less
than five (2<x<5), w is greater than 2 and less than 8 (2<x<8) and the sum of
y+z is greater
than or equal to 2 and less than or equal to 6(2sy+zs6). R3 = Ch2, S, C=O or
NH; 2< x
<5; 2 s y + Z s 6; L= a chemical structure (the linker) connecting covalently
the two
polyamine chains via alkyl, amide, ether or thioether bonds with a substituent
group (R3)
attached on a carbon atom located between the two most internal amino groups
of the
polyamine chain.
Alkylation can be preferred over amidation because the former allows a greater
flexibility to the polyamine chain to adopt the optimal conformation to
interact with the
polyamine transporter (81).
(4) The Linker (L) can be of any nature or chain length, as long as the total
mass
of the final structure does not exceed 3,000. These molecules may in other
embodiments
be described as having a total mass of between about 50 to about 2,500, or
about between
500 to about 1500 or about 1,000 as a total mass. By way of example, such
linkers may
comprise alkyl, ether, a thioether, amide, phosphono, keto, amine, or sulfonyl
groups or a
combination thereof.
The linker may comprise a carbon chain by a length of 2 to 50 carbons.
In some embodiments, the carbon chain will have a length of between 5 to about
carbons, or between 10 and 20 carbons, or in even other embodiments, the
carbon length
20 of 2 to about 15 or 12 carbons.
EXAMPLE 10
DIMERIC SPEIZIISIDINE DIMERS AS
POLYAMINE TRANSPORT INHIBITORS
25 In another family of synthetic derivitives of original polyamines, the
synthetic
original polyamine is either sym-norspermidine, sym-homospermidine, or
spermidine, and
is cross linked to a second original polyamineby a side group on the central
amino group
of said polyamines.
Various examples of such molecules, which by no means include all embodiments
made possible by this alternative design, are (A) spermidine dimers cross-
linked by an
a,a'-dialkyl disulfide side chain via N-alkyl bonds with the central amino
group of each
triamine chain as depicted for type compound I, NN'-bis(3-aminopropyl),N,N'-
bis(4-
aminobutyl)cystamine or BABAC (structure I, Fig. 25), (B) sym-norspermidine
dimers
44
CA 02304557 2000-03-22
WO 99/54283 PCTIUS98/07806
[BNSpd-(n+2), structure II, Fig. 25] or spermidine dimers [BSpd-(n+2),
structure III,
Fig. 25] cross-linked by an aliphatic chain with n+2 methylene groups (O<n<8)
via N-alkyl
bonds with the central amino group of each triamine chain, (C) sym-
norspermidine dimers
cross-linked by an aryl-containing side chain via N-alkyl bonds with the
central amino
group of each triamine chain as depicted for type compound IV, N,N,N',N'-
tetrakis(3-
aminopropyl) p-xylylenediamine or TADAX (structure IV, Fig. 25), or their sym-
homospermidine and spermidine-like homologs (not illustrated), (D) sym-
homospermidine,
sym-norspermidine or spermidine dimers cross-linked by an alkene side chain
via N-alkyl
bonds with the centralamino group of each triamine chain with either a
trans(BABA-trans,
structure V, Fig. 26) or a cis configuration around the single double bond
(BABA-cis,
structure VI, Fig. 26), and (E) sym-homospermidine, sym-norspermidine or
spermidine
dimers cross-linked by an alkyne side chain via N-alkyl bonds with the central
amino group
of each triamine chain as depicted for structure VII (BABA-yne, Fig. 26).
The general structure of the noval compounds claimed (Structure 4) include the
following characteristics:
(1) The two polyamine backbones are either spermidine [N- (3-aminopropyl)-
1,4-diaminobutane], sym-homospermidine [N- (4-aminobutyl)-1,4-diaminobutane]
or sym-
norspermidine [N - (3-aminopropyl)-1,3-diaminopropane].
(2) Each methylene group of the polyamine chain can be modified by methyl
groups without compromising the ability of the inhibitor to interact with the
polyamine
transporter.
(3) Each primary amino group of the polyamine chains can be modified by
methyl, ethyl or propyl groups without compromising the ability of the
inhibitor to interact
with the polyamine transporter.
(4) The central secondary amino groups of the two polyamine chains are
connected via 1V alkyl bonds by a linker (L) so that the resulting inhibitor
has a K; s 20 M
(relative to spermine).
(5) The linker (L) can be of any nature or chain length, as long as the total
final
structure does not exceed 3,000. These molecules may in other embodiments be
described
as having a total mass of about 50 to about 2,500, or about between 500 to
about 1,500, or
about 1,000 as a total mass. By way of example, such linkers may comprise
alkyl, aryl,
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
ether, thioether, disulfide, ainide, phosphono, keto, amine and sulfonyl
groups or a
combination thereof.
The general design and use of dimers of spermidine, sym-homospermidine and sym-
norspermidine corresponding to general Structure 4 comprise yet another aspect
of the
present invention.
For example, in some embodiements, these synthetic derivatives comprise a
structure of a first polyamine chain and a second polyamine chain according to
the
structure.
1 o ~2 R2
Fay~-111! N u imHRi
X R y
R2 2
L
R2 ~2
FtyHN 1 x N (c!) NHFil
R2 N t
Structure 4
wherein R, is H, methyl, ethyl or propyl, R2 is H or methyl, x is greater than
two and less
than five (2<x<5), y is greater than two and less than five (2<y<5), and L is
a chemical
structure (the linker) covalently connecting said second polyamine chain to
said second
chain through an alkyl bond, such as a a, c,), - diamine cross-linker.
In order to evaluate the effectiveness of dimeric triamines as polyamine
transport
inhibitors, we synthesized NN'-bis(3-aminopropyl),N,N'-bis(4-
aminobutyl)cystamine or
BABAC (structure 1, FIG 25; FIG 31), and its thiol form N-(3-aminopropyl),N-(4-
aminobutyl)cysteamine or AAC (FIG 31) according to Scheme 4 described above
(FIG 30),
using spermidine as a precursor polyamine. The ability of BABAC and AAC to
inhibit the
intracellular uptake of radio-labeled putrescine and spermidine in the ZR-75-1
human
46
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
breast cancer cell line was then determined according to standard published
procedures
(Lessard, et al. and Poulin, et al.). Briefly, ZR-75-1 cells were grown for
four days in
twenty-four well plates in RPMI 1640 medium supplemented with 10% fetal bovine
serum,
1 nM estradiol, 2mM L-glutamine, 1 mM sodium pyruvate, 15 mM Hepes and
antibiotics,
and specific uptake of [3H] putrescine, [3HJspermidine and [14C]spermine was
measured
as described (Lessard, et al.), using 20 M and 3 M substrate, respectively.
ZR-75-1 cells
are a convenient system to assess the potential of polyamine transport
inhibitors because
they exhibit elevated polyamine uptake activity (Lessard, et al.).
As shown in FIG 32, BABAC was a potent inhibitor of putrescine, spermidine and
spermine uptake in ZR-75-1 cells, with calculated apparent K; values of 0.15
M, 0.68 and
2.1 M, respectively. Its thiol monomer AAC was a less potent transporter
inhibitor (K;
0.54, 4.37 and 3.9 M, respectively, as expected from the potentiating effect
of
dimerization on polyamine transport inhibition. Although the potency of BABAC
to
inhibit putrescine and spermidine uptake was only slightly lower than that of
spermine (K;
of 0.0745 and 0.40 M, respectively, for spermine), it was clearly greater
than that of the
dimeric spermine-like inhibitor DESC (FIG 31), which inhibited putrescine,
spermidine
and spermine uptake with apparent K; values of 0.54, 5.6 and 7.5 M,
respectively. These
data clearly indicate that dimerization of a shorter polyamine chain (i.e.
spermidine relative
to spermine) can still generate potent polyamine transport inhibitors, and
that the nature of
the linker L as well as its site of covalent attachment on the polyamine
backbone can
strongly influence the potency of the dimer to inhibit polyamine uptake. A
comparison
between the structures of DESC and BABAC suggests that a N-alkyl type of
attachment
leads to superior properties of polyamine transport inhibition than an amide
linkage on a
methylene group of the polyamine backbone, and that this effect dominates over
the effect
of elongating the polyamine chains.
EXQMPLE 11
POLYAMINE TRANSPORT INHIBITION BY
SYM-NORSPERMIDINF. DIMERS
To further evaluate the validity of the design of dimeric triamines as
polyamine
transport inhibitors, we have assessed the ability of a sym-norspermidine
dimer, namely
47
~,.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
N,NN'N'-tetrakis(3-aminopropyl) p-xylylenediamine (TADAX) (FIG 25), to inhibit
polyamine uptake in ZR-75-1 cells.
TADAX (FIG 25) was synthesized according to the Scheme 2 described above
(FIG 26). Briefly, into a solution of norspermidine (2.55 g) and diethylamine
(8.0 mL) in
chloroform (100 mL) was added trityl chloride (10.87 g) portionwise. After
addition,
stirring was continued for 24 h. The reaction mixture was washed with water
and dried
over anhydrous potassium carbonate. The solution was concentrated under
reduced
pressure and the residue was re-crystallized from dichioromethane-methanol to
obtain
IV,N'-bis(trityl)norspermidine. 'H NMR (300 MHz, CHC13) 52.26 (m, 12H,
aromatic H),
7.12-7.30 (m, 18H, aromatic H), 2.36 (t, 4H, J = 8 Hz, 2 x CH2N), 2.18 (t, 4H,
J = 8 Hz,
2 x CH2N), 1.66 (m, 6H, 2 x CH2 and 2 xNHTr).
A mixture containing N',N' -bis(trityl)norspermidine (770 mg), a,a'-dibromo p-
xylene (166 mg), anhydrous Na2CO3 (573 mg), acetonitrile (45 ml) and DMF (3
drops) was
refluxed under nitrogen atmosphere for 3 days. Then, the mixture was filtered
while it was
still hot and the filtered solid was washed thoroughly with water and dried to
give
N,N,N',N'-tetrakis(N-trityl-3-aminopropyl) p-xylylene diamine. 'H NMR (300
MHz,
CHC13) S 7.24 (m, 24H, aromatic H), 6.92-7.34 (m, 36H, aromatic H), 3.42 (s,
4H, 2 X
CHZ), 2.42 (m, 8H, 4 X CH2N), 2.08 (m, 8H, 4 X CH2N), 1.64 (m, 12H, 4 X CH2
and 4 X
NHTr).
N,NN',N'-tetrakis(N-trityl-3-aminopropyl) p-xylylene diamine (374 mg) was
suspended in a solution of HC16 M (20 mL) and the mixture was refluxed for 24
hours.
The solid was removed by filtration and the aqueous phase concentrated to 2-3
mL by
rotary evaporation. Addition of ethanol into the concentrated solution
afforded the
hexahydrochloride salt of N,NN',N'-tetrakis(3-aminopropyl) P-xylylenediamine,
or
TADAX (FIG 25).
The ability of TADAX to inhibit (14C]spermine transport was next compared in
ZR-75-1 human breast cancer cells using 3 M substrate (FIG 32). In this
assay, spermine
has an apparent Kõ=0.6 M. It is clear that the p-xylylene norspermidine dimer
TADAX
(K; = 1.5 M) is a much more potent spermine transport inhibitor than the
prototypic
spermine dimer DESC (K; = 17 M), and was nearly as potent as the substrate
spermine
at interacting with the polyamine carrier in these cells. These data further
demonstrate the
48
`:.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
importance of the crosslinker L in the relative ability of triamine dimers to
inhibit
polyamine transport.
To further substantiate the usefulness of triamine dimers such as TADAX as
transport inhibitors, the cytotoxicity of TADAX and its ability to serve a
transported
substrate for the polyamine carrier were then evaluated. As shown in FIG 34,
TADAX was
remarkably non-toxic to ZR-75-1 cells (IC50>500 M), as compared to sym-
norspermidine
or the disulfide BABAC, which had lethal effects on ZR-75-1 cells with IC50
values of 6.4
and 17 M, respectively. While the cytotoxicity of sym-norspermidine has been
well
documented (Bergeron and Seligshon, Komori and Ohsugi, Porter and Bergeron), a
less
pronounced, but yet significant cytotoxicity of DESC has been found using ZR-
75-1 breast
cancer cells. That cytotoxicity may well be related to the reactivity of the
disulfide groups
of DESC and BABAC with biological thiols and disulfides found in growth media
and at
the cell surface. Thus, despite the presence of four 3-aminopropyl groups on
the TADAX
backbone, and its two sym-norspermidine-derived moieties, it is remarkably
inert toward
biological functions. This is consistent with the fact that no detectable
accumulation of the
compound was found in ZR-75-1 cells even after a 6-hour incubation, as
determined by ion
pairing-reverse phase high-pressure liquid chromatography of cell extracts
performed as
described (Lessard, et al.) (data not shown). These data suggest that the very
low
cytotoxicity of TADAX is related to its lack of uptake by ZR-75-1 cells.
Moreover, these
properties are those expected from an impermeant, pure antagonist of polyamine
transport
such as DESC, thus supporting the potential usefulness of TADAX and similar
triamine
dimers to preempt polyamine uptake by tumor cells.
Synthesis of Embodiments
(1) Unmethylated spermine analogs (FIG 17A): N', N', NY, N12-tetra Boc-5-
carboxyspermine (IV, FIG 12) is first prepared as described (88). If the
linker is going to
be amidated to the polyamine chain, the carboxyl group used as an acceptor is
activated
with cyanuric chloride (88), and conjugated with a N-mono-FMOC diaminoalkane
of the
desired length to generate the corresponding N', N~, N8, N'I-tetra-Boc-
spermine-5-N-(N-
FMOC-aminoalkyl) carboxamide (V, FIG 12). The FMOC group of the latter
compound
is removed with 20% piperdine/DMF, and the resulting N', N', N8, N'2-tetra-Boc-
spermine-
49
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
5(N-w-aminoalkyl) carboxamide (VI, FIG 12) is then reacted with the acid
chloride form
of N', Na, N8, N'1-tetra-Boc-5-carboxyspermine (IV, FIG 12). The latter
compound is then
deprotected with HC1/CH3COOH to obtain the corresponding Nx, N"-bis(spermine-5-
oyl)-
diaminoalkane, the desired transport inhibitor (VII, FIG 12). These compounds
are
symbolized as BS compounds, and BS-3, BS-4, BS-5 and BS-6 correspond to the
forms
where the diaminoalkane linker is 1,3-diaminopropane, 1, 4-diaminobutane, 1, 5-
diaminopentane and 1, 6-diaminohexane, respectively. If the spacer is going to
be
alkylated to the polyamine chain, the carboxyl group used as an acceptor in an
amidation
reaction is first reduced to an alcohol with LiAlH4. After protecting the
amine groups with
carbobenzoxy (CBZ) groups, the alcohol is then converted to a bromide with
PBr3. The
resulting CBX-protected spermine bromide is then reacted with a diamine spacer
with a 2:1
stoichiometry to generate the CBZ-protected spermine dimer. This dimer is
fmally
deprotected by catalytic hydrogenation with Pd/C (82) to generate the
urunethylated
spernune dimer (the transport inhibitor). If an ether linkage is desired, the
alcohol obtained
as above is then converted to an alkoxide with sodium metal, and then reacted
with an alkyl
dihalide (e.g. 1,3-diiodopropane) with a 2:1 stoichiometry to generate the CBZ-
protected
polyamine dimer, which is then deprotected as above to generate the
unprotected polyamine
dimer (the transport inhibitor). As an alternative precursor to ornithine, one
may use
instead 2-hydroxyputrescine, synthesized as described (83), and proceed with
carboxyethylation and catalytic hydrogenation as in the route using ornithine
as precursor,
to obtain 6-hydroxyspermine. The four amino groups of the latter are protected
with
carbobenzoxy groups, and the alcohol is converted into an alkyl halide or
alkoxide for
subsequent reaction with the spacer as described above.
(2) Methylated spermine analogs (FIG 17A): For example, omithine methylester
(X, FIG 13) is synthesized as described (89) and is diamidated with two
equivalents of 3-
azidobutyric acid (XII, FIG 13) using DCC/OHBt (90) to generate N', Na-bis (3-
azidobutyryl)-ornithine methylester (XIII, FIG 13). The latter is then reduced
using
BH3/THF (90) to obtain 1, 12-dimethylspermine-5 carbinol (XIV, FIG13). After
protection of all four amino groups with Boc groups (XV, FIG 13), the carbinol
group is
activated with PBr3 to generate 1, 12-dimethyl-N', Na, N", N'l-tetra (Boc)-5-
bromomethyl
spermine (XVI, FIG 14), and reacted with FMOC-NH-(CHZ)õNH2 (where 3n s 6) to
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
generate the corresponding 1, 12-dimethyl-N', N~, 1VB, NJ1-tetra (Boc)-
spermine-5 (N
methyl, N'''FMOC-diaminoalkane) (XVII, FIG14). After removing the FMOC group
with
piperidine/dimethylformamide (XVIII,FIG 14), the free amino group of compound
XVIII
is alkylated with one equivalent of compound XVI to generate a N", Nbis [ 1,
12-dimethyl-
N', N`,1V8, N1z-tetra (Boc)-spermine]-5-methyl)-diaminoalkane (XIX, FIG 15).
The latter
compound is then deprotected with HCUCH3COOH to finally obtain the methylated
spermine analog (BMS-3,BMS-4, BMS-5 and BMS-6), which are the desired
transport
inhibitors (XXa-d, FIG 15).
(3) Unmethylated,lV alkylated spermine analogs (FIG 17B): A symmetrical
dimer that can be made where the linker (L) bridges two polyamine derivative
chains
through one of the innermost, secondary nitrogens of each polyamine chain.
(a) N-benzyl-1,3-diaminopropane (XXI, FIG 18) is first obtained by
catalytic hydrogenation of 3-(benzylamino)propiononitrile with Raney nickel as
described (84).
(b) N-benzyl-1,3-diaminopropane is then N-alkylated with 3-
bromobutyronitrile to generate N'-benzyl, N3-(3-cyanopropyl)-1,3-
diaminopropane
(XXII, FIG 18) (85).
(c) N'-benzyl, 1V'-(3-cyanopropyl)-1,3; diaminopropane is protected
with a Boc group (86) to generate N'-benzy1,1V3-Boc, N3 (3-cyanopropyl)-1,3,-
diaminopropane (XXIII, FIG 18)
(d) N'-benzyl, AA-Boc, N'-(3-cyanopropyl)-1,3,-diaminopropane is
reduced to N'-benzyl, N-toc-spermidine ( XXIV, FIG 18) by catalytic
hydrogenation with Raney nickel (84).
(e) N'-benzyl, M-Boc-spermidine is then cyanoethylated with
acrylonitrile to generate N'-benzyl, Na-Boc, 1Vg-cyanoethyl- -spermidine, and
reduced to N'-benzyl, N -Boc-spermine by catalytic hydrogenation with Raney
nickel (84) (XXV, FIG 18).
(f) The two free amino groups of N'-benzyl, M-Boc-spermine are
protected with CBZ groups as described (87) to generate N'-benzyl,lV -Boc, N8,
N12-di(CBZ)-spermine (XXVI, FIG 18).
51
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
(g) N'-benzyl, N~-Boc, N8, N12-di(CBZ)-spermine is then deprotected to
N'-benzyl, N8, N12-di(CBZ)-spermine with trifluoroacetic acid as described
(87)
(XXVII, FIG 18).
(h) N'-benzyl, Ng, 0-di(CBZ)-spennine can then be cross-linked with
an a,w-dibromoalkane of the desired chain length to generate the corresponding
bis(N'-benzyl, N8, N'Z-di(CBZ)-spermine) dimer (XXVIII, FIG 19), which is then
deprotected by catalytic hydrogenation with Pd/C (87) to generate the
unmethylated, N-alkylated spermine dimer (the transport inhibitor) (XXIX, FIG
19).
(4) Methylated, N-alkylated spermine analogs (FIG 17B):
(a) The amino acid group of 3-aminobutyric acid is protected with Boc
as described (89), and the resulting N-Boc-3-aminobutyric acid ()XX, FIG 20)
is
condensed with N-FMOC-1, 4-diaminobutane using DCC/OHBt (88) to obtain N`-
(N-Boc-3-aminobutyryl), N;-FMOC-1, 4-diaminobutane (XXXI, FIG 20).
(b) N'-(N-Boc-3-aminobutyryl), N~-FMOC-1, 4-diaminobutane is then
reduced to N'-Boc-NB-FMOC-1-methylspermidine with BH3/THF (88) (XXXII,
FIG 20).
(c) Two equivalents ofN`-Boc-NB-FMOC-1-methylspermidine are then
M-alkylated with one equivalent of the a, w-diiodoalkane of the desired length
to
obtain the corresponding N", N-bis (N-[NBoc-3-amino, 3methylpropyl],1V [N-
FMOC-4-aminobutyl])-a, w-diaminoalkane (XXXIII, FIG 20).
(d) The FMOC groups of the resulting N`, N'-bis (1V [NBoc-3-amino,
3-methylpropyl], N-[N-FMOC-4-aminobutyl])-a,w-diaminoalkane are then
deprotected with 20% piperidine/DMF to yield the corresponding N", N'-bis (N-
[NBoc-3-amino,3-methyipropyl], N-[N-4-aminobutyl])-a,w-diaminoalkane
()COGV, FIG 20), which is then condensed with 3-azidobutyric acid, prepared as
described (88), using DCC/OHBt, to generate the corresponding N , N-bis(N-
[N'Boc-3-amino,3-methylpropyl],N-[N'-8-amino-5-aza-octanoyl])-a,w-
diaminoalkane.
(e) TheN`,N-bis(N-[N-Boc-3-amino,3-methylpropyl],N-[N-8-amino-
5-aza-octanoyl])-a, w-diaminoalkane is then reduced to the corresponding N, N"-
52
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
bis(N-[N-Boc-3-amino,3-methylpropyl], N-[N-8-amino-5-aza-octyl])-a,w-
diaminoalkane with BH3/THF (88) (XXXVII, FIG 21).
(f) The Boc groups of N`, N-bis (NV [N-Boc-3-amino, 3-methylpropyl],
N-[N-8-amino-5-aza-octyl])-a,w-diaminoalkane are then removed with
HC1/CH3COOH to generate the desired transport inhibitor, a N", N`'-bis (N-[3-
amino, 3-methyi-propyl],1V [8-amino-5-aza-octyl])-a, (j-diaminoalkane
(X)CXVIII,
FIG 21).
(5) 1,12-Dimethylated spermine dimers cross-linked through N-alkyl/C-alkyl
attachments of the linker (FIG 17C):
Dimeric polyamine transport inhibitors of a different type can be generated by
cross-linking one polyamine chain to a linker through a 1V alkyl bond as in
Examples 3 and
4 above, and the other polyamine chain via a C-linked anchor lying between the
two
innermost secondary amino groups as in Examples 1 and 2 above. Such compounds
(as
terminal C-methylated spermine analogs) can be obtained as follows:
(a) N'-Boc-Ng-FMOC-1-methylspermidine (XXXII, FIG 20), obtained
as described above (Example 4, steps a to b), is N"-alkylated using an w-
bromoalkylphthalimide of the desired length as described (92), to generate the
corresponding N'-Boc, N'-alkylphthalimide, Ng-FMOC-1-methylspermidine
(XXXIX, FIG 22).
(b) The phthalimide group of N"-Boc, N`-alkylphthalimide, N8-FMOC-1-
methylspermidine is removed with hydrazine in EtOH (88, 90) to generate the
corresponding N'-Boc, N4 ((a-aminoalkyl), N BFMOC-1-methylspermidine (XL,
FIG 22).
(c) The free amino group of N'Boc, N-(w-aminoalkyl), N-FMOC-1-
methylspermidine is then alkylated with N', IVO, N8, N12-tetra (Boc)-1, 12-
dimethyl-
5-bromomethylspermine prepared as described in Example 2 (XVI, FIG 14) to
obtain the corresponding N`([N-Boc-3-amino, 3-methylpropyl], NV [N-FMOC-4-
aminobutyl]), N"-[5-(N', N4, Ng, N12-tetra(Boc)-spermine)-methyl]a,w-
diaminoalkane (XLI, FIG 22).
53
CA 02304557 2000-03-22
WO 99/54283 PCTIUS98/07806
(d) The FMOC group of N`([N-Boc-3-amino, 3-methylpropyl], N-[N-
FMOC-4-aminobutyl]), N"-[5-(N', N', Na, N'Z-tetra (Boc)-spermine)-methyl]a,w-
diaminoalkane is then removed with 20% piperidine/DMF, and the resulting
A,1([N-Boc-3-amino, 3-methylpropyl], N-[4-aminobutyl]), NG'-[5-(N', N4, Ng,
N'2-
tetra (Boc)-spermine)-methyl]a, w-diaminoaUcane (XLII, FIG 23) is condensed
with
3-azidobutyric acid (XXX, FIG 21) as in Example 4 (step d) above, to generate
the
corresponding N([N-N-Boc-3-amino, 3-methylpropyl], N-[8-amino-5-aza-
octanoyl]), N"-[5-(N', N12-tetra (Boc)-spermine)-methyl]a, w-diaminoalkane
(XLIII, FIG 23).
(e) N(N-[N-Boc-3-methylpropyl],N-[8-amino-5-aza-octanoyl])X"-[5-
(N',11N',1V8, N'~-tetra (Boc)-spermine)-methyl]-a, w-diaminoalkane is then
reduced
to N(N-[N-Boc-3-amino, 3-methylpropyl], N-[8-amino-5-aza-octyl]), N-[a5-(N',
N',1V8, N'2-tetra (Boc)-spermine)-methyl]-a, w-diaminoalkane (XLIV, FIG 24)
with
BH3/'rHF as in Example 4 (step e) above, and the Boc groups of the resulting
compound are removed with HC1/CH3COOH to generate the corresponding N`(N-
[N-[3-amino, 3-methyl-propyl], N-[8-amino-5-aza-octyl]), N"-[5-(1, 12-dimethyl-
spermine)-methyl]-a,w-diaminoalkane (XLV, FIG 24), which is the desired
polyamine transport inhibitor.
(6) SCHEME 1(FIG 27): synthesis of sym-norspermidine dimers using total
cyanoethylation of aliphatic or aromatic diamines
This scheme is used to prepare symmetrical dimers of sym-norspermidine by
Michael addition via total cyanoethylation of hydrochloride salts of aliphatic
or aromatic
a,w-diamines. This route is the simplest one to generate polyamine dimers
since no amine
~ protection is necessary. The diamine hydrochloride of the desired nature and
chain length
(VIII, FIG 27) to be used as a crosslinker is stirred with a 4-fold excess of
acrylonitrile in
the presence of triethylamine (Et3N) in aqueous solution, yielding a N,N,N',N'-
tetrakis(cyanoethyl)-a,w-diamine (IX, FIG 27). Cyanoethyl groups are then
reduced with
NaBH4 in the presence of inethanolic CoC12 as a reducing agent, resulting in
the formation
of a sym-norspermidine dimer (X, FIG 27) with the desired linker L. The
compound is
54
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
obtained as its hexahydrochloride salt after alkaline extraction with
CHC13/NaZCO3 and
solubilization in HCI.
Various linkers can be used according to that scheme, including aliphatic a,w-
diaminoalkanes (preferably with a chain length greater than two and less than
ten) which
may incorporate one double bond in a cis or trans configuration, or one triple
bond, and
aromatic diamines where the amino groups are present as aminoalkyl
substituents at two
different positions of the aromatic cycle, such as the para configuration
shown in FIG 27.
(7) SCHEME 2 (FIG 28): synthesis of spermidine, sym-homospermidine and sym-
norspermidine dimers using direct alkylation of the central secondary amino
group
with a dibromoalkene or dibromoarene
Spermidine, sym-homospernZidine and sym-norspermidine dimers can be prepared
by protecting the primary amino groups of the precursor triamine with trityl
groups, and
directly alkylating the secondary amino group of the triamine with a
dibromoalkene, a
dibromoalkyne or a dibromoarene with the desired crosslinking carbon chain
(Zang and
Sadler). For this purpose, the primary amine groups of the triamine XI (FIG
28) are
reacted with trityl chloride in the presence of diethylamine in CHC13 to
obtain the N",1V-
bis(trityi) polyamine chain XiI (FIG 28). Two equivalents of the latter
compound are then
refluxed with the alkyl or aryl dibromide form of the desired linker L to
generate the trityl-
protected form of the desired triamine dimer (XIII, FIG 28). The trityl groups
of this dimer
are then removed by refluxing with 6 N HCL to obtain the hexahydrochloride
salt of the
polyamine transport inhibitor XIV (FIG 28). Dibromoalkanes tend to form
heterocyclic
by-products with the triamine precursor, and therefore the present scheme is
chiefly used
as a simply synthetic method to generate dimeric triamine-derived polyamine
transport
inhibitors with unsaturated or aromatic linkers.
(8) SCHEME 3 (FIG 29): synthesis of spermidine sym-homospermidine and sym-
norspermidine dimers by amidation with an acyl chloride followed by reduction
of
the amide groups
An alternative, even more versatile approach to generate spermidine, sym-
homospermidine and sym-norspermidine dimers as polyamine transport inhibitors
is to
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
crosslink the secondary amino groups of the triarnine chains, protected with
trityl groups
as in Scheme 2 (XII, FIG 28 and FIG 29), with the ap-diacyl chloride form of
the desired
linker L, and then reduce the amide bonds of the resulting molecules. For this
purpose, two
equivalents of the trityl-protected triamine precursor XII are reacted with
the diacyl
chloride of choice in the presence of triethylamine/CHC13 to obtain the
corresponding
diamide form of a trityl-protected triaminen dimer XV (FIG 29). The diamide is
then
reduced to its N-alkyl form with LiAlH4 in dry THF under nitrogen to obtain
the trityl-
protected form of the triamine dimer (XIII, FIG 28 and FIG 29). The primary
amine
groups are then freed with 6 N HCI as in Scheme 2 to generate the desired
dimeric
polyamine transport inhibitor XIV (FIG 28 and FIG 29).
This scheme is superior to Scheme 2 for dimerizing triamine molecules with
aliphatic linkers of the general structure (-(CH2)e,which tend to form
heterocycles with the
polyamine chain when reacted in their dihalide form.
(9) SCHEME 4 (FIG 30): synthesis of dimeric spermidine, sym-homospermidine
and sym-norspermidine disulfides
Synthesis of useful dimeric polyamine transport inhibitors is not limited to
those
containing aliphatic or aromatic carbon chains. Triamines can be dimerized via
N-alkyl
bonds with a linker L containing a disulfide bond, generating polyamine
transport
inhibitors with even higher potency than DESC, which is a dimeric spermine
disulfide
cross-linked through amide bonds with position C5 of each spermine skeleton.
The
following scheme allows the synthesis of dimeric triamines cross-linked via N-
alkyl bonds
with a diethyl disulfide chain.
The primary amino groups of the desired triamine XI (FIG 28 and FIG 30) are
first
protected with ethyl trifluoroacetate in aqueous acetonitrile to obtain the
bis-
trifluoroacetyl)-protected triamine XVI (FIG 30). The latter compound is then
reacted with
one equivalent with ethylene sulfide (Cohen, et al.), 1.2 equivalent of
diisopropylethylamine (Hunig's base) and 0.1 equivalent of CuCl in dry
acetonitrile in the
presence of 2,2'-dithiobis(benzothiazole) to catalyze the formation of the
disulfide from an
intermediate thiol (Brzezinska and Ternay), to generate the trifluoroacetyl-
protected
triamine dimeric disulfide XVII (FIG 30). Trifluoroacetyl groups are removed
from the
56
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
latter compound using MeOH/NH3 (4:1) at pH 11, yielding the free dimeric
triamine
disulfide XVIII (FIG 30). The latter disulfide can be further reduced to its
free thiol XIX
(FIG 30) with either dithiothreitol or tris(carboxyethyl)phosphine (Burns, et
al.).
57
CA 02304557 2006-11-22
REFERENCES
1. Ask, A.,L. Persson, and O. Heby. Increased survival of L1210 leukemic mice
by
prevention of the utilization of extracellular polyamines. Studies usmg a
polyamine-uptake mutant, antibiotics and a polyamine-deficient diet. Cancer
Lett.
66: 29-34, 1992.
2. Aziz, S. M., M. N. Gillespie, P. A. Crooks, S. F. Tofiq, C. P. Tsuboi, J.
W. Olson,
and M. P. Gosland. The potential of a novel polyamine transport inhibitor in
cancer
chemotherapy. J Pharmacol. Exper. Ther. 278: 185-192, 1996.
3. Aziz, S. 1VI., M. P. Gosland, P. A. Crooks, J. W. Olson, and M. N.
Gillespie. A
novel polymeric spermine conjugate inhibits polyamine transport in pulmonary
artery smooth muscle cells. J. Pharmacol. Exp.. Ther. 274: 181-196, 1995.
4. Bacchi, C. J., and P. P. IvlcCann. Parasitic protozoa and polyamines. In:
lnhibition
ofPolyamine Metabolism. Biological Significance.and Basis for New Therapies,
edited by P. P. McCann, A. E. Pegg and A. Sjoerdsma. Orlando, FL: Academic
Press, 1987, p. 317-344.
5. Bacchi, C. J., H. C. Nathan, A. B. Clarkson, Jr., E. J. Bienen, A. J.
Bitonti, P. P.:
McCann, and A. Sjoerdma. Effects of the ornithine decarboxylase inhibitors DL-
a-
difluoromethylorthine and a-nlonofluoromethyldehydroornithine methyl ester
alone
and in combination with suramin against Trypanosoma brucei 'brucei central
nervous system models. Am. J. Trop. Med. Hyg. 36: 46-52, 1987.
6. Bardocz, S. The role of basolateral polyamine uptake in intestinal
adaptation. In:
Polyamines in the Gastrointestinal Tract, edited by R. H. Dowling, U. R.
Folsch
and C. Loser. Dordrecht: Kluwer Academic Publ., 1992, p. 409-416.
7. Bardocz, S., G. Grant, D. S. Brown, J. C. Stewart, J. F. J. G. Koninkx, H.
G. C. J.
M. Hendriks, and A. Pusztai. Effect of .fasting and refeeding on basolateral
polyamine uptake and metabolism by the rat jejunum. In: Polyamines in the
Gastrointestinal Tract, edited by R. H. Dowling, U. R. Folsch and C. Loser.
Dordrecht: Kluwer Academic Publ., 1992, p. 435-445.
8. Bergeron, R. J.,, and H. W. Seligsohn. Hexahydropyrimidines as masked
spermidine vectors in drug delivery. Bioinorg. Chem. 14: 345=355,1986..
9. Byers, T. L., and A. E. Pegg. Regulation of polyamine transport in Chinese
hamster
ovary cells. J. Cell. Physiol. 143: 460-467, 1990..
58
CA 02304557 2000-03-22
WO 99/54283 PCTIUS98/07806
10. Chaney, J. E., K. Kobayashi, R. Goto, and G. A. Digenis. Tumor selective
enhancement of bioactivity uptake in mice treated with a-
difluoromethylornithine
prior to administration of'''C-putrescine. Life Sci. 32: 1237-1241, 1983.
11. Chang, B. K., P. R. Libby, R. J. Bergeron, and C. W. Porter. Modulation of
polyamine biosynthesis and transport by oncogene transfection. Biochem.
Biophys.
Res. Commun. 157:264-270, 1988.
12. Duranton, B., E. Nsi-Emvo, R. Schleiffer, F. Gossd, M. Galluser, and F.
Raul.
Suppression of preneoplastic changes in the intestine of rats fed low levels
of
polyamines. Cancer Res. 57: 573-575, 1997.
13. Frebort, I., and O. Adachi. Copper/quinone-containing amine oxidases, an
exciting
class of ubiquitous enzymes. J. Ferment. Bioeng. 80: 625-632, 1995
14. Gordonsmith, R. H., S. Brooke-Taylor, L. L. Smith, and G. M. Cohen.
Structural
requirements of compounds to inhibit pulmonary diamine accumulation. Biochem.
Pharmacol. 32: 3701-3709, 1983.
15. Hayashi, S., Y. Murakami, and S. Matfufuji. Ornithine decarboxylase
antizyme -
A novel type of regulatory protein. Trends Biochem. Sci. 21: 27-30, 1996.
16. He, Y., T. Suzyki, K. Kashiwagi, and K Igarashi. Antizyme delays the
restoration
by spermine of growth of polyamine-deficient cells through its negative
regulation
of polyamine transport. Biochem. Biophys. Res. Commun.. 203: 608-614, 1994.
17. Hessels, J., A. W. Kingma, H. Ferwerda, J. Keij, G. A. Van der Berg, and
F. A. J.
Muskiet. Microbial flora in the gastrointestinal tract abolishes cytostatic
effects of
a-difluoromethylomithine in vivo. Int. J. Cancer 43: 1155-1164, 1989.
18. Hinuma, K, M. Maghsoudloo, G. M. Murphy, and R. H. Dowling. Dietary and
intestinal polyamines in the rat: in vitro transport studies. In: Polyamines
in the
Gastrointestinal Tract, edited by R. H. Dowling, U. R. F6lsch and C. Lt3ser.
Dordrecht: Kluwer Academic Publ., 1992, p. 463-472.
19. Halley, J. L., A. Mather, R. T. Wheelhouse, P. M. Cullis, J. A. Hartley,
J. P.
Bingham, and G. M. Cohen. Targeting of tumor cells and DNA by a chlorambucil-
spermidine conjugate. Cancer Res. 52: 4190-4195, 1992.
20. Hold, Y., P. J. Schechter, and L. J. Marton. Phase I-II clinical trial
with alpha-
difluoromethylomithine- an inhibitor of polyamine biosynthesis. Eur. J. Cancer
Clin. Oncol. 23: 1 103 -1107, 1987.
21. Huber, M., J. Pelletier, K. Torossian, P. Dionne, I. Gamache, R. Charest-
Gaudreault, M. Audette, and R. Poulin. 2,2'-Dithiobis(N-ethyl-spermine-5-
carboxamide) is a high affinity, membrane-impermeant antagonist of the
mammalian polyamine transport system. J. Biol. Chem. 271: 27556-27563, 1996.
59
~:.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
22. Huber, M., and R. Poulin. Antiproliferative effect of spermine depletion
by N-
cyclohexyl-1,3-diaminopropane in human breast cancer cells. Cancer Res. 55:
934-
943, 1995.
23. Huber, M., and R. Poulin. Permissive role of polyamines in the cooperative
action
of estrogens and insulin or insulin-like growth factor I on human breast
cancer cell
growth. J. Clin. Endorcinol. Metab. 81: 113-123, 1996.
24. Jtlnne, T., L. Alhonen, and P. Leinonen. Polyamines: from molecular
biology to
clinical applications. Ann. Med. 23: 241-259, 1991.
25. Kakinuma, Y., K. Hoshino, and K. Igarashi. Characterization of the
inducible
polyamine transporter in bovine lymphocytes. Eur. J. Biochem. 176: 409-414,
1988.
26. Kanter, P. M.., G A. Bullard, and J. M. King. Preclinical toxicologic
evaluation of
DENSPM (N', N"-diethylnorspermine) in rats and dogs. Anti-Cancer Drugs 5:
448-456, 1994.
27. Kohn, E. C., E. Reed, G. Sarosy, M. Christian, C. J. Link;, K. Cole, W. D.
Figg, P.
A. Davis, J. Jacob, B. Goldspiel, and L. A. Liotta. Clinical investigation of
a
cytostatic calcium influx inhibitor in patients with refractory cancers.
Cancer Res.
56: 563-568, 1996.
28. Lakanen, J. R., J. K. Coward, and A. E. Pegg. a-Methyl polyamines:
metabolically
stable spermidine and spermine mimics capable of supporting growth in cells
depleted of polyamines. J. Med Chem. 35: 724-734, 1992.
29. Lessard, M., C. Zhao, S. M. Singh, and R. Poulin. Hormonal and feedback
regulation of putrescine and spermidine transport in human breast cancer
cells. J.
Biol. Chem. 270: 1685-1694, 1995.
30. Love, R. R., P. P. Carbone, A. K. Verma, D. Gilmore, P. Carey, K. D.
Tutsch, M.
Pomplun, and G. Wilding. Randomized phase I chemoprevention dose-seeking
study of a-difluoromethylornithine. J. Natl. Cancer Inst. 85 732-737, 1993.
31. Marton, L. J., and A. E. Pegg. Polyamines as targets for therapeutic
intervention.
Ann. Rev. Pharmacol. Toxicol. 35: 55-91,1995.
32. Mausumoto, T., and O. Suzuki. Polyamines as markers of malignancy. In: The
Physiology ofPolyamines, edited by U. Bachrach and Y. M. Heimer. Boca Raton,
FL: CRC Press, 1989, p. 219-234.
33. McCann, P. P., and A. E. Pegg. Ornithine decarboxylase as an enzyme target
for
therapy. Pharmac. Ther. 54:195-212,1992.
CA 02304557 2000-03-22
WO 99/54283 PCTIUS98/07806
34. McCannn, P. P., and A. J. Bitonti. An overview of inhibition of polyamine
metabolism and the consequent effects on cell proliferation in mammalian cells
and
parasitic protozoa. In: Polyamines in the Gastrointestinal Tract, edited by R.
H.
Dowling, U. R. Fblsch and C. Ldser. Dordrecht: Klawer Academic Publ., 1992, p.
143-153.
35. McCormack, S. A., and L. R. Johnson. Putrescine uptake and release by
colon
cancer cells. Am. J. Physiol. 256: G868-G877, 1989.
36. Meyskens, F. L., Jr., and E. W. Gerner. Development of
difluoromethylomithine
as chemoprevention agent for the management of colon cancer. J. Cell.
Biochem.:
126-131, 1995.
37. Minchin, R. F., R. L. Martin, L, A. Summers, and K. F. Ilett. Inhibition
of
putrescine uptake by polypyridinium quatemary salts in B 16 melanoma cells
treated
with difluoromethylornithine. Biochem. J. 262: 391-395, 1989.
38. Mitchell, J. L. A., R. R. Diveley, Jr., and A. Bareyal-Leyser. Feedback
repression
of polyamine uptake into mammalian cells require active protein synthesis.
Biochem Biophys. Res. Commun. 186: 81-88, 1992.
39. Mitchell, J. L. A., G. G. Judd, A. Bareyal-Leyscr, and S. Y. Ling.
Feedback
repression of polyamine transport is mediated by antizyme in mammalian tissue-
culture cells. Biochem. J. 299. 19-22, 1994.
40. Morgan, D. M. L. Polyamine oxidases and oxidized polyamines. In: The
Physiology
of Polyamines, edited by U. Bachrach and Y. M. Heimer. Boca Raton, FL: CRC
Press, 1989, p, 203-229.
41. Moulinoux; J.-P., V. Quemener, B. Cipolla, P. Guill6, R. Havouis, C.
Martin, B.
Lobel, and N. Seiler. The growth of MAT-LyLu rat prostatic adenocarcinoma can
be prevented in vivo by polyamine deprivation. J. Urol. 146: 1408-1412, 1991.
42. Moulinoux, J.-P., V. Quemener, and N. A. Khan. Biological significance of
circulating polyamines in oncology. Cell. Mol. Biol. 37: 773-783, 1991.
43. Nicolet:, T., J.-L. Scemama, L. Pradayrol, P. Berth6lemy, C. Seva, and N.
Vaysse.
Putrescine and spermidine uptake is regulated by proliferation and
dexamethasone
treatment in AR4-2J cells. Int. J. Cancer 49: 577-581, 1991.
44. O'Sullivan, M. C., D. M. Dalrymple, and Q. B. Zhou. Inhibiting effects of
spermidine derivatives on Trypanosoma cruzi trypanothione reductase. J. Enzym.
Inhib. 11: 97-114, 1996.
45. Osborne, D. L., and E. R. Scidel. Gastrointestinal luminal polyamines:
cellular
accumulation and enterohepatic circulation. Am. J. Physiol. 258: G576-G584,
1990.
61
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
46. Parchment, R. E., A. Lewellyn, D. Swartzendruber, and G. B. Pierce. Serum
amine
oxidase activity contributes to crisis in mouse embryo cell lines. Proc. Natl.
Acad.
Sci. USA 87: 4340-4344, 1990.
47. Pegg, A. E., P. P. McCann, and A. Sjocrdsma. Inhibition of polyamine
biosynthesis and function as an approach to drug design. In: Enzymes as
Targetsfor
Drug Design, edited by M. G. Palfreyman, P. P. McCann, P. P. Lovenberg, W.
Temple, J. G. Temple and A. Sjoerdsma. Orlando: Academic Press, 1989, p. 159-
183.
48. Pegg, A. E., R. Poulin, and J. K. Coward. Use of aminopropyltransferase
inhibitors
and of non-metabolizable analogs to study polyamine regulation and function.
Int.
J. Biochem. Cell. Biol. 27: 425-442, 1995.
49. Pegg, A. E., R. Wechter, R Poulin, P. M. Woster, and J. K. Coward. Effect
of S-
adenosyl-1,12-diamino-3-thio-9-azadodecane, a multisubstrate adduct inhibitor
of
spermine synthase, on polyamine metabolism in mammalian cells. Biochemistry
28: 844t-8453, 1989.
50. Persson, L., I. Holm, A. Ask, and O. Heby. Curative effect of DL-2-
difluoromethylomithine on mice bearing mutant L12101eukemia cells deficient in
polyamine uptake. Cancer Res. 48: 4807-4811, 1988.
51. Pohjanpelto, P. Putrescine transport is greatly increased in human
fibroblasts
initiated to proliferate. J. Cell Biol. 68: 512-520, 1976.
52. Porter, C. W., R. J. Bergeron, and N. J. Stolowich. Biological properties
of N4-
spermidine derivatives and their potential in anticancer therapy. Cancer Res.
42:
4072-4078, 1982.
53. Porter, C. W., R. J. Benacki, J. Miller, and R. J. Bergeron. Antitumor
activity of
N',N'-bis(ethyl)norspermine against human melanoma xenografts and possible
biochemical correlates of drug action. Cancer Res. 53: 581-586, 1993.
54. Porter, C. W., P. F. Cavanaugh, Jr., N. Stolowich, B. Ganis, E. Kelly, and
R J.
Bergeron. Biological properties of N`- and N',1V$-spermidine derivatives in
cultured
L12101eukemia cells. Cancer Res. 45: 2050-2057, 1985.
55. Porter, C. W., B. Ganis, Y. Rustum, C. Wrzosek, D. L. Kramer, and R. J.
Bergeron.
Collateral sensitivity of human melanoma multidrug-resistant variants to the
polyamine analogue, N',N"diethylnorspermine. Cancer Res. 54: 5917-5924, 1994.
56. Porter, C. W., J. Miller, and R. J. Bergeron. Aliphalic chain length
specificity of
the polyamine transport system in ascites L 1210 leukemia cells. Cancer Res.
44:
126-128, 1984.
62
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
57. Porter, C. W., U. Regenass, and R. J. Bergeron. Polyamine inhibitors and
analogues as potential anticancer agents. In: Polyamines in the
Gastrointestinal
Tract, edited by R. H. Dowling, U. R. F61sch and C. Ldser. Dordrecht: Kluwer
Academic Publ., 1992, p. 301-322.
58. Pusztai, A., S. W. B. Ewen, G. Grant, D. S. Brown, W. J. Peumans, E. J. M.
Van
Damme, and S. Bardocz. Stimulation of growth and polyamine accretion in the
small intestine and pancreas by lectins and trypsin inhibitors. In: Polyamines
in the
Gastrointestinal Tract, edited by R. H. Dowling, U. R. F61sch and C. Loser.
Dordrecht: Kluwer Academic Publ., 1992, p. 473-483.
59. Quemener, V., Y. Blanchard, L. Chamaillard, L. Havouis, B. Cipolla, and J.-
P.
Moulinoux. Polyamine deprivation: a new tool in cancer treatment Anticancer.
Res.14: 443-448, 1994.
60. Quemener, V., J. P. Moulinoux, C. Bergeron, F. Darcel, B. Cipolla, A.
Denais, R.
Havouis, C. Martin, Ad N. Seiler. Tumour growth inhibition by polyamine
deprivation. In: Polyamines in the Gastrointestinal Tract, edited by R. H.
Dowling,
U. R. F61sch and C. LSser. Dordrecht: Kluwer Academic Publ., 1992, p. 375-385.
61. Rinehart, C. A., and K. Y. Chen. Characterization of the polyamine
transport
system in mouse neuroblastoma cells. Effects of sodium and system A amino
acids. J. Biol. Chem. 259: 4750-4756, 1984.
62. Sarhan, S., B. Knbdgen, and N. Seiler. The gastrointestinal tract as
polyamine
source for tumor growth. Anticancer Res. 9: 215-224, 1989.
63. Scalabrino, G., and M. E. Ferioli. Polyamines in mammalian tumors. II Adv.
Cancer Res. 36: 1-102, 1981.
64. Scalabrino, G., and M. E. Feroli. Polyamines in mammalian tumors. I. Adv:
Cancer
Res. 35: 151-268, 1981.
65. Schechter, P. J., J. L. R. Barlow, and A. Sjoerdma. Clinical aspects of
inhibition of
omithine decarboxylase with emphasis on therapeutic mals of eflornithine
(DFMIO) in cancer and protozoan diseases. In: Inhibition of Polyamine
Metabolism. Biological Signfficance and Basis for New Therapies, edited by P.
P.
McCann, A. E. Pegg and A. Sjoerdsma. Orlando, FL: Academic Press, 1987,
p. 345-364.
66. Seiler, N. Acetylation and interconversion of the polyamines. In: The
Physiology
of Polyamines, edited by U. Bachrach and Y. M. Heimer. Boca Raton, FL: CRC
Press, 1989, p. 159-176.
67. Seiler, N. Polyamine catabolism and elimination by the vertebrate
organism. In:
Polyamines in the Gastrointestinal Tract, edited by R. H. Dowling, U. R.
F61sch
and C. L'oser. Dordrecht: Kluwer Academic Publ., 1992, p. 65-85.
63
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
68. Seiler, N., J. G. Delcros, and J. P. Moulinoux. Polyamine transport in
maminalian
cells. An update. Int. J. Biochem. Cell. Biol. 28: 843-861, 1996.
69. Seiler, N., and F. Dezeure. Polyamine transport in mammalian cells. Int J.
Biochem. 22: 211-218, 1990.
70. Seiler, N., S. Sarhan, C. Grauffel, R. Jones, B. Knddgen, and J.-P.
Moulinoux.
Endogenous and exogenous polyamines in support of tumor growth. Cancer Res.
50: 5077-5083, 1990.
71. Sjoerdsma, A. Suicide enzyme inhibitors as potential drugs. Clin.
Pharmacol.
Therap. 30: 3-22, 19 81.
72. Sjoerdsma, A., J. A. Golden, P. J. Schechter, J. L. R. Barlow, and D. V.
Santi.
Successful treatment of lethal protozoal infections with the ornithine
decarboxylase
inhibitor, a-difluoromethylornithine. Trans. Ass. Am. Physic. 97:: 70-79,
1984.
73. Sjoerdsma, A., and P. J. Schechter. Chemotherapeutic implications of
polyamine
biosynthesis inhibition. Cli. Pharmacol. Therap. 35: 287-300, 1984.
74. Steele, V. E., R C. Moon, R. A. Lubet, C. J. Grubbs, B. S. Reddy, M.
Wargovich,
D. L. McCormick, M. A. Pereira, J. A. Crowell, D. Bagheri, C. C. Sigman, C. W.
Boone, and G. I. Kelloff. Preclinical efficacy evaluation of potential
chemopreventive agents in animal carcinogenesis: methods and results from the
NCI Chemoprevention Drug Development Program. J. Cell. Biochem. (suppl.) 20:
32-54, 1994.
75. Sunkara, P. S., S. B. Baylin, and G. D. Luk. Inhibitors of polyamine
biosynthesis:
cellular and in vivo effects on tumor proliferation. In: Inhibition of
Polyamine
Metabolism Biological Significance and Basis for New Therapies, edited by P.
P.
McCann, A. E. Pegg and A. Sjoerdsma. Orlando: Academic Press, 1987, p. 121-
138.
76. Talpaz, M., K. Nishioka, and J. Gutterman. Clinical studies of
a-difluoromethylornithine and a-interferon combination in cancer patients. In:
The
Physiology of Polyamines, edited by U. Bachrach and Y. M. Heimer, Boca Raton,
FL: CRC Press, 1989, p. 287-292.
77. Tanaka, T., T. Kojima, A. Hara, H. Sawada, and H. Mori. Chemoprevention of
oral
carcinogenesis by DL-a-difluoromethylornithine, an ornithine decarboxylase
inhibitor: dose-dependent reduction in 4-nitroquinoline 1-oxide-induced tongue
neoplasms in rats. Cancer Res. 53: 772-776, 1993.
78. Tempero, M. A., K. Nishioka, K. Knott, and R. K Zetterman. Chemoprevention
of mouse colon tumors with difluoromethylomithine during and after carcinogen
treatment. Cancer Res. 49: 5793-5797, 1989.
64
~:.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
79. Tjandrawinata, R. R., and C. V. Byus. Regulation of the efflux of
putrescine and
cadaverine from rapidly growing cultured RAW 264 cells by extracellular
putrescine. Biochem. J. 305: 291-299, 1995.
80. Tjandrawinata, R. R., L. Hawel, and C. V. Byus. Regulation of putreseine
export
in lipopolysaccharide or IFN-gamma-activated murine monocytic-leukemic RAW
264 Cells. J. Immunol. 152: 3039-3052, 1994.
81. Li, Y., MacKerell, A.D., Jr., Egorin, M.J., Ballesteros, M.F. Rosen, M.F.,
Rosen,
M., Wu, Y.-Y., Blamble, D.A., and Callery, P.S. (1997) Cancer Res., 57,234-
239.
82. Bergeron, R.J., Ludin, C., Muller, R., Smith, R.E., and Phanstiel, 0., IV.
(1997) J.
Org. Chem. 62, 3285-3290.
83. Tobari, J., and Tchen, T.T. (1983) Meth. Enzymol. 94, 431-433.
84. Bergeron, R.J., and Garlich, J.R. (1984) Synthesis 782-784.
85. Bergeron, R.J., Garlich, J.R., and Stolowich, N.J. (1984) J. Org. Chem.
49, 2997.
86. Itoh, M., Hagiwara, D., and Kamya, T. (1977) Bull. Chem. Soc. Jpn. 50, 718-
721.
87. O'Sullivan, M.C., Dairymple, D.M., and Zhou, Q.B., (1996) J. Enzym. Inhib.
11,
97-114.
88. Huber, M., J. Pelletier, K. Torossian, P. Dionne, I. Gamache, R. Charest-
Gaudreault, M. Audette, and R. Poulin. (1996) J Biol. Chem. 271, 27556-27563.
89. Stark, P. A., B. D. Thrall, G. G. Meadows, and M. M. Abdel-Monem. (1992)
J.
Med. Chem. 35, 4264-4269.
90. Lakanen, J.R., J.K. Coward, and A. E. Pegg (1992) J. Med. Chem. 35, 724-
734.
91. U.S. Patent No. 5,456,908 - Aziz, et al.
92. Itoh, M., D. Hagiwara, and T. Kamiya (1997) Bull. Chem. Soc. Jpn. 50 718-
72 1.
93. Felschow, D.M., J. MacDiarmid, T. Bardos, R. Wu, P. M. Woster, and C. W.
Porter
(1995) "Photoaffinity labeling of a cell surface polyamine inding protein."
270 48
28705-28711.
94. Bergeron, R.J. and H.W. Seligsohn (1986) Hexahydropyrimidines as masked
spermidine vectors in drug delivery. Bioinorg. Chem. 14:345-355.
95. Brzezinska, E., and A.L. Ternay, Jr. (1994) Disulfides. 1. syntheses using
2,2'-
dithiobis(benzothiazole). J. Org. Chem. 59:8239-8244.
CA 02304557 2000-03-22
WO 99/54283 PCT/US98/07806
96. Bums, J.A., J.C. Butler, J. Moran, and G.M. Whitesides (1991) Selective
reduction
of disulfides by tris(2-carboxyethyl)phosphine. J. Org. Chem. 56:2648-2650.
97. Cohen, G.M., P.M. Cullis, J.A. Hartley, A. Mather, M.C.R. Symons, and R.T.
Wheelhouse (1992) Targeting of cytotoxic agents by polyamines: synthesis of a
chlorambucil-spermidine conjugate. J. Chem. Soc. Chem. Commun. 293-300.
98. Komori, T., and Y. Ohsugi (1991) Norspermidine inhibits LPs-induced
immunoglobulin production in an FCS-independent mechanism different from
spermidine and spermine. Int. J. Pharmacol. 13:67-73.
99. Lessard, M., C. Zhao, S.M. Singh, and R. Poulin (1995) Hormonal and
feedback
regulation of putrescine and spermidine transport in human breast cancer
cells. J.
Biol. Chem. 270:1685-1694.
100. Porter, C.W., and R.J. Bergeron (1983) Spermidine requirement for cell
proliferation in eukaryotic cells: structural specificity and quantitation.
Science
219:1083-1085.
101. Poulin, R., C. Zhao, S. Verma, R. Charest-Gaudreault, and M. Audette
(1998)
Dependence of mammalian putrescine and spermidine transport on membrane
potential: identification of an amiloride binding site on the putrescine
carrier.
Biochem. J. 330:1283-1291.
102. Zang, E., and P.J. Sadler (1997) Synthesis of hexamine ligands by using
trityl as
an N-blocking agent. Synthetic Communications 27:3145-3150.
66
,..
.._._.._.~,~,._- _