Note: Descriptions are shown in the official language in which they were submitted.
Le A 32 695-Foreign Countries Kri/Kr/W6/VO1/12/99
-1-
Substituted O-arvloxyalkvl N-aryl-carbamates
The invention relates to novel substituted O-aryloxyalkyl N-aryl-carbamates,
to
processes for their preparation and to their use as herbicides.
It is known that certain substituted O-aryloxyalkyl N-aryl-carbamates, such
as, for
example, the compounds O-[1-(3-trifluoromethyl-phenoxymethyl)-propylJ N-(2-
chloro-phenyl)-carbamate, O-[1-(3-trifluoromethyl-phenoxymethyl)-propylJ N-(2-
trifluoromethyl-phenyl)-carbamate and O-[1-(3-trifluoromethyl-phenoxymethyl)-
propylJ N-(3-trifluoromethyl-phenyl)-carbamate, have herbicidal properties
(cf.
US-A-S 099 059 and US-A-S 152 827). However, the herbicidal activity of these
known compounds is not always satisfactory.
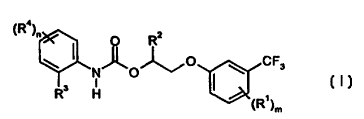
This invention, accordingly, provides the novel substituted O-aryloxyalkyl N-
aryl-
carbamates of the general formula (I),
~Re)~ O R2
O ~ CF3 (1)~
R3 H
~R~~m
in which
m represents the numbers 0, 1 or 2,
n represents the numbers 0, 1, 2, 3 or 4, with the proviso that in each
individual
case the sum of m and n is at least 1,
Rl represents cyano, halogen or in each case optionally cyano-, halogen- or C1-
C4-alkoxy-substituted alkyl or alkoxy having in each case 1 to 4 carbon
atoms,
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-2-
R2 represents optionally cyano-, halogen- or C1-C4-alkoxy-substituted alkyl
having 1 to 4 carbon atoms,
R3 represents cyano, fluorine, bromine, iodine or in each case optionally
cyano-,
halogen- or C1-C4-alkoxy-substituted alkyl, alkoxy or alkoxycarbonyl having
in each case up to 4 carbon atoms, and - in the event that m represents 1 or 2
-
also represents chlorine, and
R4 represents cyano, bromine, iodine or in each case optionally cyano-,
halogen-
or C1-C4-alkoxy-substituted alkyl, alkoxy or alkoxycarbonyl having in each
case up to 4 carbon atoms, and - in the event that m represents 1 or 2 - also
represents fluorine or chlorine,
except for the prior-art compound O-[1-(3-trifluoromethyl-phenoxymethyl)-
propyl]
N-(2-methoxy-5-methyl-phenyl)-carbamate - cf. US-A-5099059 and US-A-5152827,
in each case Table I, Compound No. 40 - which is excluded by disclaimer.
The compounds of the general formula (I) according to the invention contain an
asymmetrically substituted carbon atom and can therefore be present in various
enantiomeric (R- and S-configured forms) or diastereomeric forms. The
invention
relates both to the various possible individual enantiomeric or stereoisomeric
forms
of the compounds of the general formula (I) and to the mixtures of these
stereoisomeric compounds.
In the definitions, the hydrocarbon chains, such as alkyl- also in combination
with
heteroatoms, such as in alkoxy- are in each case straight-chain or branched.
The invention preferably provides compounds of the formula (I) in which
m represents the numbers 0, 1 or 2,
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-3-
n represents the numbers 0, 1, 2 or 3, with the proviso that in each
individual
case the sum of m and n is at least 1,
R1 represents cyano, fluorine, chlorine, bromine or in each case optionally
cyano-
fluorine-, chlorine-, methoxy- or ethoxy-substituted methyl, ethyl, n- or i-
propyl, n-, i-, s- or t-butyl, methoxy, ethoxy, n- or i-propoxy,
R2 represents in each case optionally cyano-, fluorine-, chlorine-, methoxy-
or
ethoxy-substituted methyl, ethyl, n- or i-propyl, n-, i-, s- or t-butyl,
R3 represents cyano, fluorine, bromine, iodine or in each case optionally
cyano-,
fluorine-, chlorine-, methoxy- or ethoxy-substituted methyl, ethyl, n- or i-
propyl, n-, i-, s- or t-butyl, methoxy, ethoxy, n- or i-propoxy,
methoxycarbonyl, ethoxycarbonyl, n- or i-propoxycarbonyl, and - in the event
that m represents 1 or 2 - also represents chlorine, and
R4 represents cyano, bromine, iodine or in each case optionally cyano-,
fluorine-,
chlorine-, methoxy- or ethoxy-substituted methyl, ethyl, n- or i-propyl, n-, i-
,
s- or t-butyl, methoxy, ethoxy, n- or i-propoxy, n-, i-, s- or t-butoxy,
methoxycarbonyl, ethoxycarbonyl, n- or i-propoxycarbonyl, and - in the event
that m represents 1 or 2 - also represents fluorine or chlorine,
except for the prior-art compound O-[1-(3-trifluoromethyl-phenoxymethyl)-
propyl]
N-(2-methoxy-5-methyl-phenyl)-carbamate - cf. US-A-5099059 and US-A-S 152827,
in each case Table I, Compound No.40 - which is excluded by disclaimer.
The invention relates in particular to compounds of the formula (I) in which
m represents the numbers 0 or 1,
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-4-
n represents the numbers 0, 1 or 2, with the proviso that in each individual
case
the sum of m and n is at least 1,
R1 represents cyano, fluorine, chlorine, bromine or in each case optionally
fluorine- or chlorine-substituted methyl, ethyl, methoxy or ethoxy,
R2 represents in each case optionally fluorine-, chlorine-, methoxy- or ethoxy-
substituted methyl, ethyl, n- or i-propyl,
R3 represents cyano, fluorine, bromine or in each case optionally fluorine-,
chlorine-, methoxy- or ethoxy-substituted methyl, ethyl, n- or i-propyl,
methoxy, ethoxy, n- or i-propoxy, methoxycarbonyl, ethoxycarbonyl, n- or i-
propoxycarbonyl, and - in the event that m represents 1 - also represents
chlorine, and
R4 represents cyano, bromine, iodine or in each case optionally fluorine-,
chlorine-, methoxy- or ethoxy-substituted methyl, ethyl, n- or i-propyl,
methoxy, ethoxy, n- or i-propoxy, methoxycarbonyl, ethoxycarbonyl, n- or i
propoxycarbonyl, and - in the event that m represents 1 - also represents
fluorine or chlorine,
except for the prior-art compound O-[1-(3-trifluoromethyl-phenoxymethyl)-
propyl]
N-(2-methoxy-5-methyl-phenyl)-carbamate - cf. US-A-5099059 and US-A-5152827,
in each case Table I, Compound No.40 - which is excluded by disclaimer.
The abovementioned general or preferred radical definitions apply both to the
end
products of the formula (I) and, correspondingly, to the starting materials or
intermediates required in each case for the preparation. These radical
definitions can
be combined with one another at will, i.e. including combinations between the
given
preferred ranges.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
' -S-
The novel substituted O-aryloxyalkyl N-aryl-carbamates of the general formula
(I)
exhibit strong and selective herbicidal activity.
Surprisingly, the novel substituted O-aryloxyalkyl N-aryl-carbamates of the
general
formula (>] have considerably higher herbicidal activity than the prior-art
compounds
O-[1-(3-trifluoromethyl-phenoxymethyl)-propyl] N-(2-chloro-phenyl)-carbamate,
O-[1-(3-trifluoromethyl-phenoxymethyl)-propyl] N-(2-trifluoromethyl-phenyl)-
carbamate and O-[1-(3-trifluoromethyl-phenoxymethyl)-propyl] N-(3-
trifluoromethyl-phenyl)-carbamate.
To a certain extent the compounds of the formula (I) also exhibit fungicidal
and
insecticidal activity.
Examples of the compounds of the general formula (I) according to the
invention are
listed in the groups below.
CA 02307344 2000-04-17
Le A 32 695-Forei~ Countries
-6-
Group 1
~R4~n
/ O CH3
\ ~ N~O~O ,~ CFs
3 H ~ /
here, n, R3 and R4 have, for example, the meanings given in the list below:
(position) (position) (position)
R3 (R4)n R3 (R4)n R3 (R4)n
CH3 (3) CH3 CH3 (4) CH3 CH3 (5) CH3
CH3 (6) CH3 CH3 (6) C2H5 CH3 (4,5)
~ (CH3)2
CH3 (3,5) CH3 (4,6) CH3 (4) CH3
(CH3)2 (CH3)2 (6) C2H5
CH3 (3) CF3 CH3 (4) CF3 CH3 (5) CF3
CH3 (3) Br CH3 (4) Br CH3 (5) Br
CH3 (3) CN CH3 (4) CN CH3 (5) CN
CH3 (3) OCH3 CH3 (4) OCH3 CH3 (5) OCH3
F (3) CH3 F (4) CH3 F (5) CH3
F (6) CH3 F (6) C2H5 F (4,5)
(CH3)2
F (3,5) F (4,6) F (4) CH3
(CH3)2 (CH3)2 (6) C2H5
F (3) CF3 F (4) CF3 F (5) CF
3
F (3) Br F (4) Br F (5) Br
F (3) CN F (4) CN F (5) CN
F (3) OCH3 F (4) OCH3 F (5) OCH
3
Br (3) CH3 Br (4) CH3 Br (5) CH3
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
7_
(position) (position) (position)
R3 (R4)n R3 (R4)n R3 (R4)n
Br (6) CH3 Br (6) C2H5 Br (4,5)
(CH3)2
Br (3,5) Br (4,6) Br
(4) CH3
(CH3)2 (CH3)2 (6) C2Hs
Br (3) CF3 Br (4) CF3 Br (5) CF3
Br (3) Br Br (4) Br Br (5) Br
Br (3) CN Br (4) CN Br (5) CN
Br (3) OCH3 Br (4) OCH3 Br (5) OCH
3
CF3 (3) CH3 CF3 (4) CH3 CF3 (5) CH3
CF3 (4) Br CF3 (4) OCH3 CF3 (4) CN
CF3 (3) CF3 CF3 (4) CF3 CF3 (5) CF3
OCH3 (3) CH3 OCH3 (4) CH3 OCH3 (5) OC2H5
OCH3 (6) CH3 OCH3 (6) C2H5 OCH3 (4,5)
~ (CH3)2
OCH3 (3,5) OCH3 (4,6) OCH3 (4) CH3
(CH3)2 (CH3)2 (6) C2Hs
OCH3 (3) CF3 OCH3 (4) CF3 OCH3 (5) CF3
OCH3 (3) Br OCH3 (4) Br OCH3 (5) Br
OCH3 (3) CN OCH3 (4) CN OCH3 (5) CN
OCH3 (3) OCH3 OCH3 (4) OCH3 OCH3 (5) OCH3
OCH3 (3) COOEt OCH3 (4) COOEt OCH3 (5) COOEt
Group 2
~R4~n
/ O CzHs
\ ~ N~O~O \ CF3
3 H ~ /
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-g-
Group 3
(R4)n
/ O CH(CH3)2
\ ~ N/\ ~O \ CFs
O
R3 H /
S here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 4
(R4)n
/ O CH3 CI
\ ~ N~O~O \ CF3
R3 H ~ /
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group S
(R4)n
/ O CzHs CI
\ ~ N~O~O \ CF3
R3 H ~ /
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-9-
Groin 6
(R4)n
/ O CH(CH3)z CI
\ ~ N/\ ~O \ CFs
O
R3 H /
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 7
(R4)n
/ O CH3
\ ~ N~O/~O \ CFs
R3 H ( /
CI
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 8
(R4)n
/ O CzHs
\ ~ N~O~O \ CF3
R3 H ~ /
1 S CI
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-10-
Group 9
(R4)n
O CH(CH3)z
\ ~ N~O~O \ CF3
3 H ~ /
CI
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Gro ~ 10
(R4 )n
/ O CH3
\ ~ N~O~O \ CF3
3 H ~ /
CI
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 11
(R4)n
/ O CzHs
\ ( N~O~O \ CF3
3 H ~ /
1 S CI
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-11-
Group 12
(R4 )n
/ O CH(CH3)z
\ ~ N~ ~O \ CFs
O
R3 H /
CI
S here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 13
(R4)n
/ O CH3
\ ~ N~O~O \ CF3
R3 H ~ /
CI
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 14
(R4 )n
/ O CzHs
\ ~ N~O~O \ CF3
R3 H ~ /
CI
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-12-
Group 15
(R4)n
/ O CH(CH3)z
\ ~ N~O~O \ CF3
H ~ /
CI
S here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 16
(R4)n
/ O CH3 F
\ ~ N~O~O \ CF3
H ~ /
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 17
(R4 )n
/ O C2H5 F
\ ~ N~O~O \ CF3
H ~ /
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-13-
Group 18
(R4)n
/ O CH(CH3)Z F
\ ~ N~O~O \ CF3
3 H ~ /
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 19
(R4)n
/ O CH3
\ ~ N~O~O \ CF3
3 H ~ /
F
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 20
(R4)n
/ O C2Hs
\ ~ N~O~O \ CF3
3 H ~ /
F
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-14-
Group 21
(R4)n
/ O CH(CH3)2
\ ~ N~O~O \ CF3
3 H ~ /
F
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 22
(R4)n
/ O CH3
\ ~ N~O~O \ CF3
3 H ~ /
F
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 23
(R4 )n
/ O C2Hs
\ ~ N~O~O \ CF3
3 H ~ /
F
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-15-
Group 24
(R4)n
/ O CH(CH3)2
\ ~ N~O~O \ CF3
3 H ~ /
F
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 25
(R4)n
/ O CH3
\ ~ N~O~O \ CF3
3 H ~ /
C
1~
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 26
(R4)n
/ O CzHs
\ ~ N~O~O \ CF3
3 H ~ /
F
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-16-
Group 27
(R4 )n
/ O CH(CH3)z
N~O~O ~ CF3
R3 H ( /
F
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 28
(R4)n
/ O CH3
N~O~O ~ CF3
3 H ~ /
CF3
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
Group 29
(R4)n
/ O CzHs
N~o~O ~ CF3
3 H ~ /
C F3
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-17-
Group 30
(R4)n
/ O CH(CH3)z
\ ~ N~O~O \ CF3
R3 H ~ /
CF3
here, n, R3 and R4 have, for example, the meanings given above in Group 1.
The novel substituted O-aryloxyalkyl N-aryl-carbamates of the general formula
(I)
are obtained when
(a) aryloxyalkyl chloroformates of the general formula (II)
O Rz
CI~O~O \ CF3
(R, )m
in which
m, R1 and R2 are as defined above,
are reacted with arylamines of the general formula (III)
(R4)n
i\
(nl)
/
NHz
Rs
in which
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-18-
n, R3 and R4 are as defined above,
if appropriate in the presence of a reaction auxiliary and if appropriate in
the presence
of a diluent,
or
(b) aryloxyalkanols of the general formula (N)
RZ
H~ ~O CF3
O ~ ~ (IV)
yR~ )m
in which
1 S m, R 1 and R2 are as defined above,
are reacted with aryl isocyanates of the general formula (V)
(R4)n
(~)
NCO
R3
in which
n, R3 and R4 are as defined above,
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-19-
if appropriate in the presence of a reaction auxiliary and if appropriate in
the presence
of a diluent.
Using, for example, (1-(2-fluoro-5-trifluoromethyl-phenoxymethyl)-propyl]
S chloroformate and methyl 2-amino-4-ethyl-benzoate as starting materials, the
course
of the reaction in the process (a) according to the invention can be
illustrated by the
following scheme:
CzHS O CZHS CzHS
'f' C~~O~O ~ ~ CF3 ~ ~ O CZHS
NH / N~O~O ~ CF3
z F H
O OCH3 O OCH3 F
Using, for example, 2-(3,5-bis-trifluoromethyl-phenoxy)-1-methyl-ethanol and
2-bromo-6-methyl-phenyl isocyanate as starting materials, the course of the
reaction
of the process (b) according to the invention can be illustrated by the
following
scheme:
Br CH3 Br
H ~O ~ CF ~ O CH3
NCO \O I --~ ~ / N O~O ~ CF3
CH3 ~ H
CH3
CF3
CF3
The formula (II) provides a general definition of the aryloxyaryl
chloroformates to be
used as starting materials in the process (a) according to the invention for
preparing
compounds of the formula (I). In the formula (II), m, R1 and R2 each
preferably or in
particular have those meanings which have already been mentioned above in
connection with the description of the compounds of the formula (I) according
to the
invention as being preferred or as being particularly preferred for m, R1 and
R2 .
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-20-
The starting materials of the general formula (II) are known and/or can be
prepared
by processes known per se (cf. US-A-5099059, US-A-5152827, US-A-5399545).
The formula (III) provides a general definition of the arylamines further to
be used as
starting materials in the process (a) according to the invention. In the
formula (III), n,
R3 and R4 each preferably or in particular have those meanings which have
already
been mentioned above in connection with the description of the compounds of
the
formula (I) according to the invention as being preferred or as being
particularly
preferred for n, R3 and R4.
The starting materials of the general formula (III) are known chemicals for
synthesis.
The formula (IV) provides a general definition of the aryloxyalkanols to be
used as
starting materials in the process (b) according to the invention for preparing
1 S compounds of the formula (I). In the formula (N) m, R1 and R2 each
preferably or in
particular have those meanings which have already been mentioned above in
connection with the description of the compounds of the formula (I) according
to the
invention as being preferred or as being particularly preferred for m, R1 and
R2 .
The starting materials of the general formula (N) are known and/or can be
prepared
by processes known per se (cf. US-A-5099059, US-A-5152827, US-A-5399545).
The formula (V) provides a general definition of the aryl isocyanates further
to be
used as starting materials in the process (b) according to the invention. In
the formula
(V), n, R3 and R4 each preferably or in particular have those meanings which
have
already been mentioned above in connection with the description of the
compounds
of the formula (I) according to the invention as being preferred or as being
particularly preferred for n, R3 and R4.
The starting materials of the general formula (V) are known chemicals for
synthesis.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-21 -
Suitable reaction auxiliaries for the processes (a) and (b) are, in general,
the
customary inorganic or organic bases or acid acceptors. These preferably
include
alkali metal or alkaline earth metal acetates, amides, carbonates,
bicarbonates,
hydrides, hydroxides or alkoxides, such as, for example, sodium acetate,
potassium
S acetate or calcium acetate, lithium amide, sodium amide, potassium amide or
calcium
amide, sodium carbonate, potassium carbonate or calcium carbonate, sodium
bicarbonate, potassium bicarbonate or calcium bicarbonate, lithium hydride,
sodium
hydride, potassium hydride or calcium hydride, lithium hydroxide, sodium
hydroxide, potassium hydroxide or calcium hydroxide, sodium methoxide,
ethoxide,
n- or -i-propoxide, n-, i-, s- or -t-butoxide or potassium methoxide,
ethoxide, n- or
i-propoxide, n-, i-, s- or t-butoxide; furthenmore also basic organic nitrogen
compounds, such as, for example, trimethylamine, triethylamine,
tripropylamine,
tributylamine, ethyl-diisopropylamine, N,N-dimethyl-cyclohexylamine, dicyclo-
hexylamine, ethyl-dicyclohexylamine, N,N-dimethyl-aniline, N,N-dimethyl-benzyl-
1 S amine, pyridine, 2-methyl-, 3-methyl-, 4-methyl-, 2,4-dimethyl-, 2,6-
dimethyl-,
3,4-dimethyl- and 3,5-dimethyl-pyridine, 5-ethyl-2-methyl-pyridine,
4-dimethylamino-pyridine, N-methyl-piperidine, 1,4-diazabicyclo[2.2.2]-octane
(DABCO), 1,5-diazabicyclo[4.3.0]-non-S-ene (DBN), or 1,8-diazabicyclo[5.4.0]-
undec-7-ene (DBU).
Suitable diluents for carrying out the processes (a) and (b) according to the
invention
are especially inert organic solvents. These include, in particular,
aliphatic, alicyclic
or aromatic, optionally halogenated hydrocarbons, such as, for example,
benzine,
benzene, toluene, xylene, chlorobenzene, dichlorobenzene, petroleum ether,
hexane,
cyclohexane, dichloromethane, chloroform, carbon tetrachloride; ethers, such
as
diethyl ether, diisopropyl ether, dioxane, tetrahydrofuran or ethylene glycol
dimethyl
ether or ethylene glycol diethyl ether; ketones, such as acetone, butanone or
methyl
isobutyl ketone; nitrites, such as acetonitrile, propionitrile or
butyronitrile; amides,
such as N,N-dimethylformamide, N,N-dimethylacetamide, N-methyl-formanilide,
N-methyl-pyrrolidone or hexamethylphosphoric triamide; esters, such as methyl
acetate or ethyl acetate, sulphoxides, such as dimethyl sulphoxide, alcohols,
such as
methanol, ethanol, n- or i-propanol, ethylene glycol monomethyl ether,
ethylene
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-22-
glycol monoethyl ether, diethylene glycol monomethyl ether, diethylene glycol
monoethyl ether, mixtures thereof with water or pure water.
When carrying out the processes (a) and (b) according to the invention, the
reaction
temperatures can be varied within a relatively wide range. In general, the
processes
are carried out at temperatures between 0°C and 150°C,
preferably between 10°C and
120°C.
The processes (a) and (b) according to the invention are generally carried out
under
atmospheric pressure. However, it is also possible to carry out the processes
according to the invention under elevated or reduced pressure - in general
between
0.1 bar and 10 bar.
For carrying out the processes (a) and (b) according to the invention, the
starting
1 S materials are generally employed in approximately equimolar amounts.
However, it
is also possible to use a relatively large excess of in each case one of the
components.
The reaction is generally carned out in a suitable diluent, if appropriate in
the
presence of a reaction auxiliary, and the reaction mixture is generally
stirred for
several hours at the required temperature. Work-up is carried out by customary
methods (cf. the Preparation Examples).
The active compounds according to the invention can be used as defoliants,
desiccants, haulm killers and, especially, as weedkillers. By weeds, in the
broadest
sense, there are to be understood all plants which grow in locations where
they are
undesired. Whether the substances according to the invention act as total or
selective
herbicides depends essentially on the amount used.
The active compounds according to the invention can be used, for example, in
connection with the following plants:
Dicotyledonous weeds of the enera: Sinapis, Lepidium, Galium, Stellaria,
Matricaria, Anthemis, Galinsoga, Chenopodium, Urtica, Senecio, Amaranthus,
CA 02307344 2000-04-17
Le A 32 695-Forei~ Countries
- 23 -
Portulaca, Xanthium, Convolvulus, Ipomoea, Polygonum, Sesbania, Ambrosia,
Cirsium, Carduus, Sonchus, Solanum, Rorippa, Rotala, Lindernia, Lamium,
Veronica, Abutilon, Emex, Datura, Viola, Galeopsis, Papaver, Centaurea,
Trifolium,
Ranunculus and Taraxacum.
S
Dicotyledonous crops of the genera: Gossypium, Glycine, Beta, Daucus,
Phaseolus,
Pisum, Solanum, Linum, Ipomoea, Vicia, Nicotiana, Lycopersicon, Arachis,
Brassica, Lactuca, Cucumis and Cucurbita.
Monocotyledonous weeds of the enera: Echinochloa, Setaria, Panicum, Digitaria,
Phleum, Poa, Festuca, Eleusine, Brachiaria, Lolium, Bromus, Avena, Cyperus,
Sorghum, Agropyron, Cynodon, Monochoria, Fimbristylis, Sagittaria, Eleocharis,
Scirpus, Paspalum, Ischaemum, Sphenoclea, Dactyloctenium, Agrostis, Alopecurus
and Apera.
Monocotyledonous crops of the enera: Oryza, Zea, Triticum, Hordeum, Avena,
Secale, Sorghum, Panicum, Saccharum, Ananas, Asparagus and Allium.
However, the use of the active compounds according to the invention is in no
way
restricted to these genera, but also extends in the same manner to other
plants.
Depending on the concentration, the compounds are suitable for total weed
control,
for example on industrial terrain and rail tracks, and on paths and areas with
or
without tree stands. Equally, the compounds can be employed for controlling
weeds
in perennial crops, for example forests, ornamental tree plantings, orchards,
vineyards, citrus groves, nut orchards, banana plantations, coffee
plantations, tea
plantations, rubber plantations, oil palm plantations, cocoa plantations, soft
fruit
plantings and hopfields, in lawns, turf and pastures, and for selective weed
control in
annual crops.
The compounds of the formula (I) according to the invention are suitable in
particular
for the selective control of monocotyledonous and dicotyledonous weeds in
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-24-
monocotyledonous and dicotyledonous crops, both by the pre- and the post-
emergence method.
The active compounds can be converted into the customary formulations, such as
solutions, emulsions, wettable powders, suspensions, powders, dusts, pastes,
soluble
powders, granules, suspoemulsion concentrates, natural and synthetic materials
impregnated with active compound, and microencapsulations in polymeric
substances.
These formulations are produced in a known manner, for example by mixing the '
active compounds with extenders, that is liquid solvents and/or solid
carriers,
optionally with the use of surfactants, that is emulsifiers and/or dispersants
and/or
foam-formers.
If the extender used is water, it is also possible to use, for example,
organic solvents
as auxiliary solvents. Liquid solvents which are mainly suitable are:
aromatics such
as xylene, toluene, or alkylnaphthalenes, chlorinated aromatics and
chlorinated
aliphatic hydrocarbons such as chlorobenzenes, chloroethylenes or methylene
chloride, aliphatic hydrocarbons such as cyclohexane or paraffins, for example
petroleum fractions, mineral and vegetable oils, alcohols such as butanol or
glycol as
well as their ethers and esters, ketones such as acetone, methyl ethyl ketone,
methyl
isobutyl ketone or cyclohexanone, strongly polar solvents such as
dimethylformamide and dimethyl sulphoxide, and water.
Suitable solid carriers are: for example ammonium salts and ground natural
minerals
such as kaolins, clays, talc, chalk, quartz, attapulgite, montmorillonite or
diatomaceous earth, and ground synthetic minerals such as highly disperse
silica,
alumina and silicates; suitable solid carriers for granules are: for example
crushed
and fractionated natural rocks such as calcite, marble, pumice, sepiolite and
dolomite,
or else synthetic granules of inorganic and organic meals, and granules of
organic
material such as sawdust, coconut shells, maize cobs and tobacco stalks;
suitable
emulsifiers and/or foam formers are: for example non-ionic and anionic
emulsifiers,
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-25-
such as polyoxyethylene fatty acid esters, polyoxyethylene fatty alcohol
ethers, for
example alkylaryl polyglycol ethers, alkylsulphonates, alkyl sulphates,
arylsulphonates and protein hydrolysates; suitable dispersants are: for
example
lignin-sulphite waste liquors and methylcellulose.
S
Tackifiers such as carboxymethylcellulose and natural and synthetic polymers
in the
form of powders, granules or latexes such as gum arabic, polyvinyl alcohol and
polyvinyl acetate, or else natural phospholipids such as cephalins and
lecithins, and
synthetic phospholipids can be used in the formulations. Other possible
additives are
mineral and vegetable oils.
It is possible to use colorants such as inorganic pigments, for example iron
oxide,
titanium oxide and Prussian Blue, and organic dyestuffs such as alizarin
dyestuffs,
azo dyestuffs and metal phthalocyanine dyestuffs, and trace nutrients such as
salts of
1 S iron, manganese, boron, copper, cobalt, molybdenum and zinc.
The formulations generally comprise between 0.1 and 95 per cent by weight of
active
compound, preferably between 0.5 and 90%.
For controlling weeds, the active compounds according to the invention, as
such or in
the form of their formulations, can also be used as mixtures with known
herbicides,
readymixes or tank mixes being possible.
Possible components for the mixtures are known herbicides, such as, for
example,
acetochlor, acifluorfen(-sodium), aclonifen, alachlor, alloxydim(-sodium),
ametryne,
amidochlor, amidosulfuron, anilofos, asulam, atrazine, azafenidin,
azimsulfuron,
benazolin(-ethyl), benfuresate, bensulfuron(-methyl), bentazone, benzofenap,
benzoylprop(-ethyl), bialaphos, bifenox, bispyribac(-sodium), bromobutide,
bromofenoxim, bromoxynil, butachlor, butroxydim, butylate, cafenstrole,
caloxydim,
carbetamide, carfentrazone(-ethyl), chlomethoxyfen, chloramben, chloridazon,
chlorimuron(-ethyl), chlornitrofen, chlorsulfuron, chlortoluron, cinmethylin,
cinosulfuron, clethodim, clodinafop(-propargyl), clomazone, clomeprop,
clopyralid,
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-26-
clopyrasulfuron(-methyl), cloransulam(-methyl), cumyluron, cyanazine,
cycloate,
cyclosulfamuron, cycloxydim, cyhalofop(-butyl), 2,4-D, 2,4-DB, 2,4-DP,
desmedipham, di-allate, dicamba, diclofop(-methyl), diclosulam, diethatyl(-
ethyl),
difenzoquat, diflufenican, diflufenzopyr, dimefuron, dimepiperate,
dimethachlor,
S dimethametryn, dimethenamid, dimexyflam, dinitramine, diphenamid, diquat,
dithiopyr, diuron, dymrone, epoprodan, EPTC, esprocarb, ethalfluralin,
ethametsulfuron(-methyl), ethofumesate, ethoxyfen, ethoxysulfuron,
etobenzanid,
fenoxaprop(-P-ethyl), flamprop(-isopropyl), flamprop(-isopropyl-L),
flamprop(-methyl), flazasulfuron, fluazifop(-P-butyl), flumetsulam,
flumiclorac(-
pentyl), flumioxazin, flumipropyn, flumetsulam, fluometuron, fluorochloridone,
fluoroglycofen(-ethyl), flupoxam, flupropacil, flurpyrsulfuron(-methyl, -
sodium),
flurenol(-butyl), fluridone, fluroxypyr(-meptyl), flurprimidol, flurtamone,
fluthiacet(-
methyl), fluthiamide, fomesafen, glufosinate{-ammonium), glyphosate(-
isopropylammonium), halosafen, haloxyfop(-ethoxyethyl), haloxyfop(-P-methyl),
hexazinone, imazamethabenz(-methyl), imazamethapyr, imazamox, imazapyr,
imazaquin, imazethapyr, imazosulfuron, ioxynil, isopropalin, isoproturon,
isouron,
isoxaben, isoxaflutole, isoxapyrifop, lactofen, lenacil, linuron, MCPA, MCPP,
mefenacet, metamitron, metazachlor, methabenzthiazuron, metobenzuron,
metobromuron, (alpha-)metolachlor, metosulam, metoxuron, metribuzin,
metsulfuron(-methyl), molinate, monolinuron, naproanilide, napropamide,
neburon,
nicosulfuron, norflurazon, orbencarb, oryzalin, oxadiargyl, oxadiazon,
oxasulfuron,
oxaziclomefone, oxyfluorfen, paraquat, pelargonic acid, pendimethalin,
pentoxazone,
phenmedipham, piperophos, pretilachlor, primisulfuron(-methyl), prometryn,
propachlor, propanil, propaquizafop, propisochlor, propyzamide, prosulfocarb,
prosulfuron, pyraflufen(-ethyl), pyrazolate, pyrazosulfuron(-ethyl),
pyrazoxyfen,
pyribenzoxim, pyributicarb, pyridate, pyriminobac(-methyl), pyrithiobac(-
sodium),
quinchlorac, quinmerac, quinoclamine, quizalofop(-P-ethyl), quizalofop(-P-
tefuryl),
rimsulfuron, sethoxydim, simazine, simetryn, sulcotrione, sulfentrazone,
sulfometuron(-methyl), sulfosate, sulfosulfuron, tebutam, tebuthiuron,
terbuthylazine,
terbutryn, thenylchlor, thiafluamide, thiazopyr, thidiazimin, thifensulfuron(-
methyl),
thiobencarb, tiocarbazil, tralkoxydim, triallate, triasulfuron, tribenuron(-
methyl),
triclopyr, tridiphane, trifluralin and triflusulfuron.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
_2~_
A mixture with other known active compounds such as fungicides, insecticides,
acaricides, nematicides, bird repellants, plant nutrients and soil
conditioners, is also
possible.
The active compounds can be used as such, in the form of their formulations or
in the
use forms prepared therefrom by further dilution, such as ready-to-use
solutions,
suspensions, emulsions, powders, pastes and granules. They are used in the
customary manner, for example by watering, spraying, atomizing or spreading.
The active compounds according to the invention can be applied either before
or after
emergence of the plants. They can also be incorporated into the soil before
sowing.
The amount of active compound used can vary within a substantial range. It
depends
essentially on the nature of the desired effect. In general, the application
rates are
between 1 g and 10 kg of active compound per hectare of soil surface,
preferably
between 5 g and 5 kg per ha.
The preparation and use of the active compounds according to the invention can
be
seen from the examples which follow.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
_2g_
Preparation Examples:
Example 1
/ O C2Hs
HsC \ N~O/~O \ CF3
CH3 H ~ /
(Process (a))
At room temperature (approximately 20°C), 3.0 g (25 mMol) of 1-(3-
trifluoromethyl-
phenoxymethyl)-propyl chloroformate (racemic) are added with stirring to a
solution
of 2.5 g (10 mMol) of 2,3-dimethyl-aniline in 80 ml of toluene, and the
reaction
mixture is stirred at room temperature for 1 S hours. The mixture is
subsequently
washed with water and 1N hydrochloric acid, dried with sodium sulphate and
filtered. The solvent is carefully distilled off from the filtrate under water
pump
vacuum.
This gives 3.5 g (92% of theory) of O-[1-(3-trifluoromethyl-phenoxymethyl)-
propyl]
N-(2,3-dimethyl-phenyl)-carbamate (racemate) as a crystalline residue of
melting
point 76°C.
Analogously to Preparation Example 1 and in accordance with the general
description of the preparation process according to the invention, it is also
possible to
prepare, for example, the compounds of the formula (I) listed in Table 1
below.
~R4y 2
O R
N- -O v 0 ~ CF3 (I),
R3 H
~R~~m
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-29-
In all cases, they are racemates. The position of Rl is - if required - stated
such that
the CF3 group is in each case in the 3-position. The structures were confirmed
by
'H NMR spectra.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-30-
Table 1: Examples of compounds of the formula (I)
Ex.- (position) (position) Melting point
No. m R1 R2 R3 (R4) (~C)
2 0 C2H5 CH3 (4) CH3 58
3 0 C2H5 CH3 (5) CH3 102
4 0 C2H5 CH3 (6) CH3 61
0 C2H5 CH3 (4,5) (CH3)2
6 0 C2H5 CH3 (3,5) (CH3)2
7 0 C2H5 CH3 (3) Br
8 0 C2H5 CH3 (4) Br
9 0 C2H5 CH3 (5) Br
0 C2H5 CH3 (3) CN
11 0 C2H5 CH3 (4) CN
12 0 C2H5 CH3 (5) CN
13 0 C2H5 CH3 (5) CF3
14 0 C2H5 CH3 (3) OCH3
0 C2H5 CH3 (4) OCH3 (amorphous)
16 0 C2H5 CH3 (5) OCH3 (amorphous)
17 0 CZHS CH3 (3) COOC2H5
18 0 C2H5 CH3 (4) COOC2H5
19 0 C2H5 CH3 (5) COOC2H5
0 C2H5 F (3) CH3
21 0 C2H5 F (4) CH3 (amorphous)
22 0 C2H5 F (5) CH3 (amorphous)
23 0 C2H5 F (6) CH3
24 0 C2H5 F (4) Br
0 C2H5 F (3) CF3
26 0 C2H5 F (4) CF3
27 0 CZHS F (5) CF3
28 0 C2H5 F (6) CF3
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-31-
Ex.- (position)~ (position) Melting point
No. m R1 R2 R3 (R4 (C)
29 0 C2H5 F (5) OCH3
(6) CH3
30 0 C2H5 F (3) OCH3
31 0 C2H5 F (4) OCH3
32 0 C2H5 F (6) OCH3
33 0 C2H5 CF3 (6) Br
34 0 C2H5 CF3 (4) OCH3
35 0 C2H5 CF3 (4) Br
36 0 C2H5 CF3 (4) CN
37 0 C2H5 CF3 (3) CF3
38 0 CZHS CF3 (4) CF3
39 0 C2H5 CF3 (5) CF3
40 0 C2H5 OCH3 (3) CH3
41 0 C2H5 OCH3 (4) CH3
42 0 CZHS OCH3 (S) CH3
43 0 C2H5 OCH3 (S) COOC2H5
44 0 C2H5 CN (3) CH3
45 0 C2H5 CN (4) CH3
46 0 C2H5 CN (5) CH3
47 0 C2H5 COOC2H5 (3) CH3
48 0 C2H5 COOC2H5 (4) CH3
49 0 C2H5 COOC2H5 (5) CH3
50 0 C2H5 Br (3) CH3
S 1 0 C2H5 Br (4) CH3
52 0 C2H5 Br (S) CH3
53 0 C2H5 Br (6) CH3
54 0 CZHS Br (4) CF3
55 0 C2H5 OCH3 (4-)OCH3 (amorphous)
56 0 C2H5 CH3 (6-)C2H5 (amorphous)
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-32-
Ex.- (position) (position) Melting point
No. m R1 R2 R3 (R4 (C
57 0 C2H5 OCH3 (5-)OCH3 (amorphous)
58 0 C2H5 OCH3 (3-)OCH3 (amorphous)
59 0 CH3 CH3 (4-)CH3 (amorphous)
60 0 CH3 CH3 (5-)CH3 (amorphous)
61 0 CH3 CH3 (4-)OCH3 (amorphous)
62 0 CH3 OCH3 (5-)OCH3 (amorphous)
63 0 CH3 OCH3 {3-)OCH3 (amorphous)
64 1 (6-)F C2H5 CH3 (3-)CH3 (amorphous)
65 1 (6-)F C2H5 CF3 (amorphous)
66 1 (2-)F C2H5 CH3 (3-)CH3 (amorphous)
67 1 (2-)F C2H5 Cl (3-)Cl (amorphous)
68 1 (2-)F C2H5 CF3 (amorphous)
69 1 (2-)F C2H5 Cl (3-)Cl
(np = 1.5263)
70 1 (2-)F C2H5 CH3 (3-)CH3 (amorphous)
71 1 (4-)F C2H5 CF3 (np = 1.4842)
72 1 (4-)F C2H5 CH3 (4-)F (amorphous)
73 1 (4-)Cl C2H5 CH3 (3-)CH3 (amorphous)
74 1 (4-)Cl C2H5 CH3 (4-)F 76C
75 1 (5-)CF3 C2H5 CH3 (4-)F 113C
76 1 (5-)CF3 C2H5 CH3 (3-)CH3 140C
77 0 C4H9-n CH3 (3-)CH3 92C
78 1 (4-)F C2H5 F (4-)F 40C
79 1 (4-)F C3H~-n F (4-)F 71C
80 1 (4-)F C3H~-n Cl (3-)Cl 81C
81 1 (4-)F C3H~-n CH3 (3-)CH3 73C
82 1 (6-)F C2H5 F (4-)F (np = 1.4876)
(4-)F
83 1 (2-)F C H F (np = 1.4874)
2 5
84 1 (5-)CF3 C2H5 CF3 (amorphous)
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-33-
Ex.- (position) (position) Melting point
No. m Rl R2 R3 R4) (C
85 0 CH3 F (S-)CH3 (amorphous)
86 0 CH3 CH3 (5-)OCH3 (amorphous)
87 0 CH3 F (4-)CH3 (amorphous)
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-34-
Example A
Pre-emergence test
Solvent: 5 parts by weight of acetone
Emulsifier: 1 part by weight of alkylaryl polyglycol ether
To prepare a suitable preparation of active compound, 1 part by weight of
active
compound is mixed with the stated amount of solvent, the stated amount of the
emulsifier is added and the concentrate is diluted with water to the desired
concentration.
Seeds of the test plants are sown in normal soil. After approximately 24
hours, the
soil is watered with the preparation of active compound such that the
particular
1 S amounts of active compound desired are applied per unit area. The
concentration of
the spray liquor is chosen so that the particular amounts of active compound
desired
are applied in 1000 1 of water/ha.
After three weeks, the degree of damage to the plants is rated in % damage in
comparison to the development of the untreated control.
The figures denote:
0 % - no effect (like untreated control)
100 % - total destruction
In this test, for example, the compounds of Preparation Examples 1, 2, 3 and 4
exhibit strong activity against weeds, and some of them are tolerated well by
crop
plants, such as, for example, maize, barley and Soya beans.
CA 02307344 2000-04-17
Le A 32 695-Foreign Countries
-35-
Example B
Post-emergence test
S Solvent: 5 parts by weight of acetone
Emulsifier: 1 part by weight of alkylaryl polyglycol ether
To prepare a suitable preparation of active compound, 1 part by weight of
active
compound is mixed with the stated amount of solvent, the stated amount of
emulsifier is added and the concentrate is diluted with water to the desired
concentration.
Test plants of a height of 5-15 cm are sprayed with the preparation of active
compound such that the particular amounts of active compound desired are
applied
1 S per unit area. The concentration of the spray liquor is chosen so that the
particular
amounts of active compound desired are applied in 1000 1 of water/ha.
After three weeks, the degree of damage to the plants is rated in % damage in
comparison to the development of the untreated control.
The figures denote:
0 % = no effect (like untreated control)
100 % = total destruction
In this test, for example, the compounds of Preparation Examples 1, 2 and 4
exhibit
strong activity against weeds, and some of them are tolerated well by crop
plants,
such as, for example, maize.
CA 02307344 2000-04-17