Note: Descriptions are shown in the official language in which they were submitted.
CA 02319230 2000-11-02
AK19821
BACKGROUND OF THE INVENTION
1. Field of Invention
This invention pertains to the extraction of valuable chemicals from dried
tall oil
soap.
2. Related Art
Tall oil soap is a by-product of the alkaline pulping of coniferous wood. Soap
consists of sodium salts of fatty acids, sodium salts of diterpenic (rosin)
acids,
free fatty acids, free rosin acids and unsaponifiable neutral compounds such
as
fatty alcohols, sterols, steryl esters and waxes. The physically-bound water
content of neat soap is 30 to 35%. The pH of as-is soap is typically in the
range
of 12 to 13, arising from the presence of entrained black liquor. In
industrial
practice, soap is routinely acidulated with sulphuric acid to produce crude
tall oil,
an article of commerce. See for example, Drew, J. and Propst, M., eds., "Tall
Oil", Pulp Chemical Association, New York, NY, 1981.
Sterols, in particular phytosterols, have been identified recently as the
source
material for the preparation of anti-cholesterolemic agents for humans.
During the past 50 years, numerous methods have been reported for the
isolation of sterols from tall oil soap. The general method involves the
extraction
of neat soap with a variety of organic solvents. The presence of entrained
black
liquor promotes and stabilizes the unwanted water-oil emulsion, which is known
to be extremely difficult to break. Consequently, the efficiency of solvent
extraction of neat soap for the isolation of sterols is greatly reduced.
Holmbom et al. teach in US Patent 3,965,085 the extraction of a mixture of
acetone-water soap slurry, with a water-immiscible solvent such as hexane. The
aqueous phase contains mainly sodium salts of fatty and resin acids. The
organic
phase contains mostly unsaponifiables including sterols. In US Patent
4,044,031,
Johansson et al. teach the dissolution of soap in a water-immiscible mixture
comprising hexane and acetone, extraction of the water-immiscible phase with
another solvent mixture comprising methanol, hexane, acetone and water, and
isolation of sterols from the methanolic phase by evaporative crystallization.
In
US Patent 5,770,749, Kutney et al. teach the use of a mixture of ketones,
hydrocarbons and water to extract sterols form soap. The hydrocarbon extract
is
further processed with methanol. The complexity of recovering the multi-
component spent solvent is very problematic in these cited processes.
2
CA 02319230 2000-11-02
AK19821
The object of the present invention is to provide an effective means to
circumvent
the problem of breaking complex water-oil emulsion as manifested in the
solvent
extraction of neat soap, and to eliminate the usage of multi-component organic
solvent for extraction with the attendant logistical problems of solvent
recovery.
SUMMARY OF THE INVENTION
The present invention affords an effective means to provide a sterol-enriched
extract from tall oil soap which can yield sterols upon subsequent
precipitation in
a water-organic solvent mixture. The preferred embodiment of the present
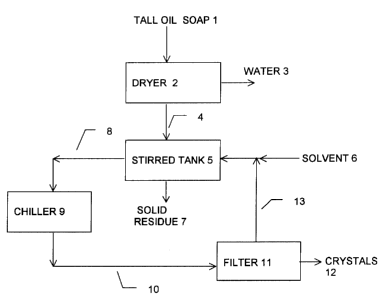
invention as given in Figure 1 comprises the following steps
a) Tall oil soap [1 ] is fed into a dryer [2] to remove water [3],
b) The dried tall oil soap solids [4] are transferred to a stirred tank [5].
c) A solvent such as acetone [6] is added to the stirred tank [5], which is
maintained at 40 to 50 degrees Celsius.
d) After a mixing time of about 30 minutes, the insoluble solids [7] are
allowed to settle and removed for disposal back to, for example, the kraft
pulp mill supplying the tall oil soap raw material. The supernatant [8] is
removed for transfer to a chilling vessel [9].
e) The supernatant [8] is chilled to about +3 to +4 degrees Celsius to effect
the formation of sterol crystals.
f) The slurry [10] is pumped through a filter [11 ] to recover the sterol
crystals
[12].
g) The filtrate [13] is recycled for use in mixture with fresh solvent [6].
The sterols in the said supernatant [8] may be recovered alternatively by
direct
precipitation by the addition of water to the supernatant [8]. In another
variant,
sterols in the said supernatant [8] may be further processed by known
distillation
methods to increase the concentration of sterols further prior to
precipitation of
sterols by known means in a mixture of water and solvent.
3
CA 02319230 2000-11-02
AK19821
EXAMPLES
The following example illustrate the preferred embodiment of the invention,
without limiting the broad scope of application:
Example 1
A sample of commercial tall oil soap was dried in an air-circulating oven at
105
degrees Celsius for 24 hours. The resulting soap solids was analyzed by a gas
chromatographic method to contain 45 mg total sterols per gram of dry soap.
Typically, the extraction of dried soap solids was conducted in a 250-ml
beaker
with a selected solvent at 45 degrees Celsius. The mixing time was 30 minutes.
The percent sterols recovered is calculated as the amount of sterols recovered
in
the dried extract from the original sterols present in the dried soap. The
test
results show that the extraction of dried soap with acetone provides the best
combination of small volume of extract, high concentration of sterols in
extract
and high recovery of sterols from starting dried soap.
Solvent Soap: Dried Extract
Solvent Wt % Dried Sterols, Sterols,
mg/g
Ratio Soap Recovered
Methanol 0.499 85.0 41 77
70% ethanol 0.496 78.5 50 87
+30%
methanol
Isopropanol0.500 32.1 95 68
Acetone 0.483 21.9 158 77
Methyl ethyl0.500 35.4 95 75
ketone
Ethyl acetate0.468 39.1 104 90
Toluene 0.250 12.1 62 17
Hexane 0.082 73.8 38 62
Diethyl 0.060 13.6 318 96
ether
Example 2
Dried soap sample used in the above example was extracted with acetone in the
same fashion under different operating conditions. The test results show that
the
extraction can be optimized by varying the extraction time, extraction
temperature, and soap:solvent ratio, in accordance with the dried soap
feedstock.
4
CA 02319230 2000-11-02
AK19821
Acetone is the Soap: Dried
Extract
solvent used Acetone Wt r6 Sterols,Sterols,
~
Ratio Dried mg/g Recovered
Soap
45 deg.C for 0.483 21.9 158 77
30
minutes
45 deg. C for 1.000 10.0 175 39
30
minutes
8.5 deg.C for 0.480 8.6 157 30
30
minutes
105 deg.C soxhlet0.462 51.7 103 100
extraction for
6
hours
CLAIMS
What is claimed is:
1. A process for the preparation of sterols from tall oil soap comprising the
steps of:
a) drying tall oil soap,
b) extracting the dried tall oil soap with a solvent, and
c) recovering sterols from the solvent extract.
2. A process according to claim 1 in which the drying of tall oil soap is made
above the prevailing boiling point of water.
3. A process according to claim 1 in which the extraction of dried soap is
made with a solvent at between 20 to 150 degrees Celsius.
4. A process according to claim 1 in which the solvent is substantially
miscible with water.
5. A process according to claim 1 in which the solvent is substantially
immiscible with water.
6. A process according to claim 1 in which the solvent is a mixture of
solvents.