Note: Descriptions are shown in the official language in which they were submitted.
CA 02320941 2000-08-11
WO 99/41590 PCT/F199/00098
METHOD AND DEVICE FOR MEASURING THE AMOUNT OF COATING ON A
MOVING SUBSTRATE
The invention relates to a method for measuring the amount of
coating on a moving substrate, the method comprising measuring the amount
of at least one component in a coating by reflection measurement.
The invention also relates to a device for measuring the amount of
coating on a moving substrate, the device comprising a radiation source for
producing a light beam, a detector for measuring the reflected light beam and
means for processing a signal of the detector, the device being arranged to
measure the amount of at least one component in a coating.
fn a manufacturing process it is important to monitor the amount of
coating on a moving substrate, such as paper or cardboard web. Coating im-
proves the printing quality of paper, and the amount of coating should, if pos-
sible, be kept constant in one paper grade. The coating materials consist of
binders and coating pigments. Most common coating pigments used in coating
are kaolin, calcium carbonate and titanium dioxide. During a paper manufac-
turing process the amount of coating is in general continuously measured by
measuring devices moving in cross direction of the paper perpendicularly over
the paper web as the web moves forward.
US patent 5 455 422 describes a method in which the amount of
coating is measured by measuring, for example, the absorption peak of latex
at a wavelength of 2.30 micrometers and the absorption peak of clay at a
wavelength of 2.21 micrometers. Said patent further describes the measure-
ment of calcium carbonate by measuring the amount of backscattering at a
wavelength of 2.09 micrometers. However, for measuring the amount of cal-
cium carbonate said method is unreliable and inaccurate, since the method is
based on a weak cellulose absorption coverage effect caused by the coating
and dependent on multiple factors and not on the absorption caused by car-
bonate. The amount of calcium carbonate could also be determined, for ex-
ample, on the basis of kaolin measurement assuming that the ratio between
the amounts of kaolin and calcium carbonate in a coating is constant. How-
ever, this is not always the case, and problems are created particularly if
the
kaolin content is low i.e. below approximately 20% and the carbonate content
correspondingly high i.e. approximately 80%.
EP publication 0 332 018 shows a method in which the amount of
CA 02320941 2000-08-11
WO 99141590 ~ PCTlF199l00098
2
kaolin in paper is measured by transmission measurement, for example, at
wavelengths of about 1.4 and 2.2 micrometers. However, by transmission
measurement the portion of coating in the measurement result is very difficult
to determine. Furthermore, the portion of calcium carbonate has to be ap-
proximated as shown in the previous chapter.
GB publication 2 127 541 shows how transmission measurement is
used for measuring the amount of additives in paper. The publication de-
scribes how the amount of calcium carbonate is measured by measuring the
absorption peaks at wavelengths of 11.54 micrometers and 11.77 microme-
ters. The amount of coating cannot be measured by said method, since the
fillers in base paper are included in the results. Furthermore, the absorption
of
paper can be so high that measurement through paper is not possible. More-
over, i~~ ifs entirety, the accuracy of the measurement results is not good
enough.
!t is an object of the present invention to provide a method in which
the above drawbacks can be avoided.
The method of the invention is characterized by measuring the
amount of calcium carbonate by measuring ar7 absorption peak of calcium
carbonate at a wavelength of about 3.95 micrometers.
The device of the invention is further characterized by being ar-
ranged to measure the amount of calcium carbonate from the coating by
measuring an absorption peak of calcium carbonate at a wavelength of about
3.95 micrometers.
The essential idea of the invention is to measure the compcnents of
the coating by reflection measurement so as to measure the amount of cal-
cium carbonate by measuring the absorption peak of calcium carbonate at a
wavelength of about 3.95 micrometers and/or the amount of kaolin by meas-
uring the absorption peak of kaolin at a wavelength of about 2.7 micrometers.
The invention has the advantage that the amount of calcium car
bonate and kaolin can be measured accurately and reliably, simultaneously in
the same measurement or separately in different measurements. It is also
possible to measure the total amount of calcium carbonate and kaolin from
several applications on top of one another. The selectivity of the measurement
for both calcium carbonate and kaolin is very good.
In the following, the invention will be described in greater detail in
the accompanying drawings, in which
._..._a.___~._~~. _..._ ._.,._~._~.__._. ~ n....
CA 02320941 2000-08-11
WO 99/41590 PCT/FI99/00098
3
Figure 1 is a schematic view showing a measuring device of the in-
vention,
Figure 2 shows spectra of base paper and paper coated with cal-
cium carbonate, and
Figure 3 shows spectra of base paper and paper coated with kaolin.
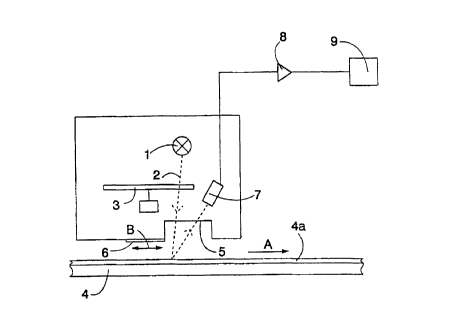
Figure 1 shows a measuring arrangement where radiation reflected
from an object is measured, i.e. the radiation source and the receiver are on
the same side of the object to be measured. Said measurement is referred to
as reflection measurement.
Figure 1 shows a measuring device comprising a radiation source 1
producing a light beam 2. The radiation source 1 may be, for example, a halo-
gen lamp or another suitable radiation source for producing an infrared beam.
The light beam 2 is directed through a filter 3. The filter 3 filters the
light so that
only the light that is essential for the measurement and that is at the right
wavelength band enters the measurement point. The filter 3 may be, for ex-
ample; a rotating filter disc including several interference filters or
another filter
solution known per se. The structure of the filter 3 is as such known for
those
skilled in the art, and is therefore not explained in greater detail in this
context.
After the filter 3 the light beam 2 is directed through a window 5 to a paper
or
cardboard web 4 moving in the paper machine. The window 5 can be made of
quartz glass or sapphire, for example. The paper or cardboard web 4 moves in
the direction of arrow A. A coating 4a is arranged on the surface of the paper
or cardboard web 4. Instead of the moving paper or cardboard web 4, the
moving substrate, the coatings of which are measured, may also be, for ex-
ample, a roll of a paper coating machine, a roll of a paper machine and/or
generally a surface of a metal plate. The device also comprises a reference
sample 6 which is moved at given intervals to the measurement point as indi-
cated by arrow B. The sample 6 operates as a reflection reference and the
measurement result obtained therefrom provides a picture of the condition of
the light source 1, the detector 7 and the window 5. In addition, reference
measurement can, if desired, be used to correct the actual measurement re-
sult.
The reflected light beam 2 is directed to a detector 7. From the de-
tector 7 the signals are directed through a preamplifier 8 to a computer 9 for
processing the measured data in a manner known per se. For the sake of
clarity, Figure 1 does not show the optics possibly needed to direct the light
CA 02320941 2000-08-11
WO 99/41590 PCT/FI99/00098
4
beam 2. The structure used for conductinglguiding the fight may be, for exam-
ple, visualizing optics, an optical fiber or a bundle of optical fibers.
in Figure 2, curve C shows a reflection spectrum of base paper and
curve D, indicated by a dashed line, shows the reflection spectrum of paper
coated with calcium carbonate. The wavelength ?~ in micrometers is on the
horizontal axis and the absorbency is on the vertical axis. When the spectra
were measured, an absorption peak E was unexpectedly found for calcium
carbonate at a wavelength range of 3.95 micrometers. By arranging the de-
vice according to Figure 1 to measure the absorption peak at a wavelength of
about 3.95 micrometers, the device can measure the amount of calcium car-
bonate.
Suitable reference wavelengths for measuring calcium carbonate
are, for example, 4.55 micrometers and/or 3.7 micrometers. The suitable ref-
erence wavelengths for kaolin are, in turn, for example 2.64 micrometers
andlor 2.8 micrometers. It should be noted that any reference wavelengths
close to the actual measurement peak can be used as reference wavelengths.
What is essential is that the absorbencies of base paper or coated paper are
equal or nearly equal at said wavelength range. The wavelength 3.7 mi-
crometers is particularly advantageous, since it can be used as reference also
for measuring the amount of water. It is advantageous to measure the amount
of water at a wavelength of about 3.175 micrometers.
The measurements performed show that the reflectivity increases in
the range over 6.3 micrometers, when the basis weight of carbonate was in-
creased. But in the range under 6.3 micrometers the reflectivity decreases,
when the basis weight of carbonate was increased, the absorption measure-
ment thus functioning reliably in this range.
)n Figure 3, curve F shows the reflection spectrum of base paper
and curve G, indicated by a dashed line, shows the reflection spectrum of kao-
lin coated paper. Figure 3 also shows the wavelength ?~ in micrometers on the
horizontal axis and the absorbency on the vertical axis. When the spectra
were measured, an absorption peak H was unexpectedly found for kaolin at a
wavelength range of about 2.7 micrometers. By arranging the device accord-
ing to Figure 1 to measure the absorption peak at a wavelength of about 2.7
micrometers, the amount of kaolin in the coating can be determined.
As for the properties affecting the signal-to-noise ratio of the radia-
tion source, the windows and the detectors and the price of the device, a
CA 02320941 2000-08-11
WO 99/41590 PCT/F199/00098
measurement wavelength of 3.95 micrometers is advantageous for measuring
the amount of calcium carbonate. Furthermore, the measurement of the
amount of carbonate at a wavelength range of 3.95 micrometers can be im
plemented in the same meter as the measurement of the absorption peak of
5 kaolin at 2.7 micrometers.
The drawing and the description associated thereto are merely in-
tended to illustrate the idea of the invention. As for the details the
invention
may vary within the scope of the appended claims.