Note: Descriptions are shown in the official language in which they were submitted.
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
PIPERAZINE ETHYLAMIDE DERIVATIVES WITH 5-HT1A RECEPTOR ACTIVITY
Background of the Invention
US 5,126,346 and divisionals thereof describe (1-naphthyl)-4-alkylpiperazine
compounds disclosed as 5-HT 1 A receptor agonist and antagonist ligands that
decrease arterial blood pressure and heart rate.
ES 2027898 describes (2-methoxyphenyl)piperazine derivatives with S-HT1A
receptor activity which are secondary amide and secondary amine derivatives.
Summary to the Present Invention
In accordance with this invention are provided novel piperazine ethylamide
derivatives which are agonists and antagonists of the SHT 1 A receptor
subtype. By
virtue of their high binding affinity to the SHTIA receptor, compounds of the
present
invention are useful for the treatment of central nervous system (CNS)
disorders such
as depression, anxiety, panic, obsessive-compulsive disorder (4CD), sleep
disorders,
sexual dysfunction, alcohol and drug addiction, cognition enhancement,
Alzheimer's
disease, Parkinson's disease, obesity and migraine.
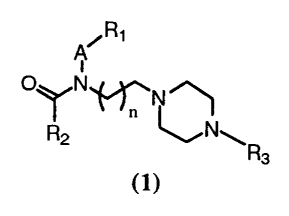
Compounds of the present invention are represented by the general formula (1),
~Ri
A
O~N~N
R2 n ~N~R
3
in which:
(1}
R1 is aryl, heteroaryl, cycloalkyl or heterocycloalkyl;
R2 is cycloalkyl, alkyl or N(R~RS);
R3 is aryl or heteroaryl;
R4 and RS are independently hydrogen, alkyl, cycloalkyl, heterocycloalkyl,
alkylcycloalkyl, alkylheterocycloalkyl, aryl or heteroaryl, or taken together
R4 and
RS form a heterocycloalkyl; provided that R4 and RS are not both hydrogen;
CA 02351860 2001-05-18
WO 00/35892 PCTNS99/29953
-2-
A is (CH2)m;
m is an integer from 1 to 4; and n is an integer from I to 3; or a
pharmaceutical salt
thereof.
S In preferred embodiments of the present invention, R~ is aryl, R2 is
cycloalkyl
and R~ is phenyl.
"Alkyl" as used herein means a branched or straight chain having from I to 6
carbon atoms and more preferably I to 3 carbon atoms. Exemplary alkyl groups
include methyl, ethyl, propyl, isopropyl, butyl, isobutyl, t-butyl, pentyl and
hexyl.
"Alkoxy" as used herein means an alkyl-O group in which the alkyl group is
as previously described. Exemplary alkoxy groups include methoxy, ethoxy,
n-propoxy, i-propoxy, n-butoxy, and t-butoxy.
"Aryl" as used herein means mono or bicyclic aromatic ring having from 6 to
10 carbon atoms. Monocyclic rings preferably have 6 members and bicyclic rings
preferably have 8, 9 or 1() membered ring structures. Exemplary aryl groups
include
phenyl and naphthyl. In some preferred embodiments aryl is phenyl, 1-naphthyl
or
2-naphthyl. The aryl group may be substituted with one or more substituents.
Substituted aryl groups preferably have one to three substituents.
"Cycloalkyl" as used herein means a monocycIic alkyl group having from 3 to
8 carbon atoms. In some preferred embodiments cycloalkyl may be substituted
with
from 1 to 3 substituents.
"Heterocycloalkyl" as used herein means a monocyclic alkyl group having
from 3 to 8 members including from 1 to 3 heteroatoms selected from N, O and
S. In
some preferred embodiments heterocycloalkyl may be substituted with 1 to 3
substituents.
Halogen, as used herein means fluorine, chlorine, iodine and bromine.
CA 02351860 2001-05-18
WO 00/35892 PCTNS99/29953
-3-
"Heteroaryl" means 5 to 10 membered mono or bicyclic aromatic ring having
from 1 to 3 heteroatoms selected from N, O and S. Monocyclic rings preferably
have
or 6 members and bicyclic rings preferably have 8, 9 or 1() membered ring
5 structures. Exemplary heteroaryls include pyrrolyl, furyI, thienyl,
imidazolyl,
pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, pyridyl, pyrazinyl,
pyrimidinyl, indolyl, quinolyl, isoquinolyl and benzodioxanyl. Preferred
hetcroaryl
groups include thienyl, pyridyl, pyrimidinyl, indolyl, and benzodioxanyl. More
preferred are heteroaryl groups including 3-thienyl, 2-pyridyl, 2-pyrimidinyl,
indol-4-
yl and benzodioxan-S-yl. The heteroaryl group may be substituted with one or
more
substituents. Substituted heteroaryl groups preferably have from 1 to 3
substituents.
Suitable substituents include, unless otherwise noted, halogen, alkyl,
hydroxy,
alkoxy, amino, amido, nitro, alkylamino, alkylamido, perhaloalkyl,
carboxyalkyl,
carboxy, carbamide, dialkylamino and aryl.
Carbon number refers to the number of carbons in the carbon backbone and
does not include carbon atoms occurring in substituents such as an alkyl or
alkoxy
substituents.
Where terms are used in combination, the definition for each individual part
of the combination applies unless defined otherwise. For instance,
alkylcycloalkyl
means an alkyl-cycloalkyl group in which alkyl and cycloalkyl are as
previously
described.
Pharmaceutically acceptable salts are the acid addition salts which can be
formed from a compound of the above general formula and a pharmaceutically
acceptable acid such as phosphoric, sulfuric, hydrochloric, hydrobromic,
citric,
malefic, succinic, fumaric, acetic, lactic, nitric, sulfonic, p-toluene
sulfonic, methane
sulfonic acid, and the like.
The compounds of this invention contain a chiral center, providing for various
seteroisomeric forms of the compounds such as racemic mixtures as well as the
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
-4-
individual optical isomers. The individual isomers can be prepared directly or
by
asymmetric or stereospecific synthesis or by conventional separation of
optical
isomers from the racemic mixture.
S Compounds of the present invention may be prepared by those skilled in the
art of organic synthesis employing conventional methods which utilize readily
available reagents and starting materials. For example, reaction of known
arylpiperazines (A) with chloroacetonitrile and subsequent reduction with
lithium
aluminum hydride provides known and novel (4-aryl)piperazine-1-ethylamine
1(? derivatives (B). Treatment with acid chlorides provides the subsequent
amide (C),
which can be reduced to the amine (D) by the action of a reducing agents such
as
lithium aluminum hydride or borane. The amine (D) may then be again acylated
to
provide the required compounds (1) of the present invention.
HN~ H2N~N
~N n ~Nw
~ Rs Rs
A ~ B
r
N O N
A~ ~N~ ~ ~ ~N
R1 ~Nw R ~Nw
R3 R3
D
C
r
~R~
A
I
O \ In N N
\ R3
IS
(1)
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
-5-
The following non-limiting specific examples are included to illustrate the
synthetic
procedures used for preparing compounds of formula 1. In these examples, all
chemicals and intermediates are either commercially available or can be
prepared by
standard procedures found in the literature or are known to those skilled in
the art of
organic synthesis. Several preferred embodiments are described to illustrate
the
invention. However, it should be understood that the invention is not intended
to be
limited to the specific embodiments.
Compound 1
lU N-t2-f 4-(2-Methoxy_,uhenyl)-piperazin-1-yll-ethyll-benzamide
Benzoyl chloride (U.3U g, 2.12 mmol) was added dropwise at 0"C to a solution
of 1-(2-aminoethyl)-4-(2-methoxyphenyl)piperazinc (().5 g, 2.12 mmol) and
triethylamine (2 equivalents, 4.24 mmol) in dichloromethane (2() mL), and the
mixture stirred under nitrogen for 16 hours. The solution was evaporated,
water (St)
mL) added and the produce extracted into ethyl acetate (3 x 25 mL). The
combined
organics were washed with water (25 mL), brine (25 mL) and dried over
anhydrous
sodium sulfate. Filtration and concentration in vacuum afforded a white solid
(U.66
g, 94% yield). Treatment of the amide with an ethanolic solution of fumaric
acid
afforded the salt of the titled compound as a white solid.
m.p. 136-138°C
Elemental Analysis for: C,"H=SN30: ().SC,HaO, 0.75H~0
Calculated: C, 64.29; H, 6.99; N, 10.22
Found: C, 64.08; H, 6.88; N, 10.()4
Compound 2
Cvclohexanecarboxvlic acid benzyl-~2-f 4-(2-methoxv-nhenvl)
piperazin-1-yll-ethyl?-amide
A solution of the amide (().279 g, 0.82 mmol) from example 1 in THF (3 mL)
was treated with the dropwise addition of 1 M BH3 THF (3 mL) and the resulting
mixture relluxed under a nitrogen atmosphere for 3 hours. The solution was
cooled
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
-6_
to 0°C, and the excess borane reagent destroyed by the careful addition
of 2N-HC1.
After stirring for 2 hours, the solution was made basic with 2N-NaOH and the
product extracted with EtOAc (3 x 25 mL). The combined organics were washed
with water (2 x 20 mL), brine (25 mL) and dried over anhydrous sodium sulfate.
Filtration and concentration in vacuum afforded the required amine as a light
yellow
oil (0.23 g , 88% yield).
Cyclohexanecarbonyl chloride (0.11 g, ().75 mmol) was added to a CH=Cl=
solution ( I () mL) of the amine 0.23 g , ().71 mmol) and tricthylamine (0.2
mL). After
stirring for 16 hours, the solvent was evaporated, water (25 mL) added and the
product extracted into EtOAc (3 x 2() mL). the combined organics were washed
with
water (20 mL), brine (20 mL) and dried over sodium sulfate. Filtration and
concentration gave a viscous yellow oil (0.29g) which was purified by dash
silica gel
chromatography to afford the required product (().24g, 80% yield). An
ethanolic
solution of the amide was treated with an ethanolic fumaric acid solution ( 1
eq) to
gave the fumarate salt of the titled compound as a white solid.
m . p. 144- I 46"C
Elemental Analysis for: C_,H3,N,0~ 1.OC,H;O,
Calculated: C, 67.49; H, 7.49; N, 7.62
Found: C, 67.50; H, 7.49; N, 7.63
Compound 3
Naphthalene-1-carboxylic acid(2-l4-(2-methoxy-phenyl)-
pinerazin-1-lrll-ethyl)-amide
1-Naphthoyl chloride (0.40 g, 2.12 mmol) was added dropwise at 0"C to a
solution of 1-(2-aminoethyl)-4-(2-methoxyphenyl)piperazine (().5 g, 2.12 mmol)
and
triethylamine (2 equivalents, 4.24 mmol) in dichloromethane (20 mL), and the
mixture stirred under nitrogen for 16 hours. The solution was evaporated,
water (50
mL) added and the product extracted into ethyl acetate (3 x 25 mL). The
combined
organics were washed with water (25 mL), brine (25 mL) and dried over
anhydrous
sodium sulfate. Filtration and concentration in vacuum afforded a white solid
(0.52
g, 65% yield). Treatment of the amide with an ethanolic solution of fumaric
acid
afforded the salt of the titled compound as a white solid.
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
-7_
m.p. 136-138°C
Elemental Analysis for: C=,H,,N30= 0.5C,H,0, 0.5H=O
Calculated: C, 68.40; H. 6.62; N, 9.20
Found: C, 68.34; H, 6.82; N, 9.08
Compound 4
Cyclohexanecarboxylic acid(2-14-(2-methoxy-nhenvl)-ninerazin-1-vll-ethvll-
na~hthalen-1 ylmethyl-amide
A solution of the amide (0.473 g, 1.21 mmol) from example 3 in THF (7 mL)
was treated with the dropwise addition of 1M BH3-THF (5 mL} and the resulting
mixture rel7uxed under a nitrogen atmosphere for 3 hours. The solution was
cooled
to 0"C, and the excess borane reagent destroyed by the careful addition of 2N-
HCI.
After stirring for 2 hours. the solution was made basic with 2N-NaOH and the
product extracted with EtOAc (3 x 25 mL). The combined organics were washed
with water (2 x 2(> mL), brine (25 mL) and dried over anhydrous sodium
sulfate.
Filtration and concentration in vacuum afforded the required amine as a light
yellow
oil (0.14 g , 31 ~lo yield).
Cyclohexanecarbonyl chloride (0.055 g, 0.37 mmol) was added to a CH,CI=
solution (7 mL) of the amine (0.14 g , 0.37 mmol) and triethylamine (0.1 mL).
After
stirring for 16 hours, the solvent was evaporated. water (25 mL) added and the
product extracted into EtOAc (3 x 20 mL). the combined organics were washed
with
water (20 mL), brine (2() mL) and dried over sodium sulfate. Filtration and
concentration gave a viscous yellow oil (().29g) which was purified by flash
silica gel
chromatography to afford the required product (().lg, 55% yield). An ethanolic
solution of the amide was treated with an ethanolic fumaric acid solution (1
eq) to
gave the fumarate salt of the titled compound as a white solid.
m.p. 204-206°C
Elemental Analysis for: C"H39N3O= 1.OC,H,O,
Calculated: C, 69.86; H, 7.20; N, 6.98
Found: C, 69.49; H, 7.21; N, 6.85
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
_g_
Compound 5
Naphthalene-2-carboxylic acidf 2-f 4-(2-methoxv-nhenvl)
piperazin-1-vll-ethyl)-amide
2-Naphthoyl chloride (0.40 g, 2.12 mmol) was added dropwise at 0°C to a
solution of 1-(2-aminoethyl)-4-(2-methoxyphenyl)piperazine (().5 g, 2.12 mmol)
and
triethylamine (2 equivalents, 4.24 mmol) in dichloromethane (1() mL}, and the
mixture stirred under nitrogen for 16 hours. The solution was evaporated,
water (50
mL) added and the product extracted into ethyl acetate (3 x 25 mL). The
combined
organics were washed with water (25 mL), brine (25 mL) and dried over
anhydrous
sodium sulfate. Filtration and concentration in vacuum afforded a white solid
(0.46
g, 57% yield). Treatment of the amide with an ethanolic solution of fumaric
acid
afforded the salt of the titled compound as a while solid.
m.p. 136-138°C
Elemental Anal sy'sfor: C_,H~,N,O= ().SC,HaO, 1.0H=O
Calculated: C, 67.08; H. 6.71; N, 9.03
Found: C, 67.29; H, 6.51; N, 8.92
Compound 6
Cvclohexanecarboxvlic acid (2 f4-(2-methoxy-phenyl)-ninerazin-1-vll-ethvl)-
naphthalen-2-ylmethyl-amide
A solution of the amide (0.406 g, 1.04 mmol) from example S in THF (6 mL)
was treated with the dropwise addition of 1 M BH,-THF (4 mL) and the resulting
mixture refluxed under a nitrogen atmosphere for 3 hours. The solution was
cooled
to 0°C, and the excess borane reagent destroyed by the careful addition
of 2N-HCI.
After stirring for 2 hours, the solution was made basic with 2N-NaOH and the
product extracted with EtOAc (3 x 25 mL). The combined organics were washed
with water (2 x 20 mL), brine (25 mL) and dried over anhydrous sodium sulfate.
Filtration and concentration in vacuum afforded the required amine as a light
yellow
oil (0.32 g , 82% yield).
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
_c)_
Cyclohexanecarbonyl chloride (U.125 g, U.85 mmol) was added to a CH,CI,
solution (lU mL) of the amine {U.32 g , 0.85 mmol) and triethylamine (U.25
mL).
After stirring for 16 hours, the solvent was evaporated> water (25 mL) added
and the
product extracted into EtOAc (3 x 2U mL). the combined organics were washed
with
water (2U mL), brine (2U mL) and dried over sodium sulfate. Filtration and
concentration gave a viscous yellow oil (U.29g) which was purified by dash
silica gel
chromatography to afford the required product (().19g, 467: yield). An
ethanolic
solution of the amide was treated with an ethanolic fumaric acid solution (U.5
eq) to
gave the fumarate salt of the titled compound as a white solid.
I(> m.p.141-143°C
Elemental Analysis for: C3,H3,,N30~ U.SC,HaO,
Calculated: C, 72.88; H, 7.76; N, 8.04
Found: C, 72.90; H, 7.60; N, 7.73
Compound 7
N-!2-f 4-!2-Methoxv-phenyl)-piperazin-1 yll-ethyll-
3-phenyl-~ropionamide
Hydrocinnamoyl chloride (U.357 g, 2.12 mmol) was added dropwise at
0°C to
a solution of 1-(2-aminoethyl)-4-(2-methoxyphenyl)piperazine (U.5 g, 2.I2
mmol)
and triethylamine (2 equivalents, 4.24 mmol) in dichloromethane (2() mL), and
the
mixture stirred under nitrogen for 16 hours. The solution was evaporated,
water (SU
mL) added and the product extracted into ethyl acetate (3 x 25 mL). The
combined
organics were washed with water (25 mL), brine (25 mL) and dried over
anhydrous
sodium sulfate. Filtration and concentration in vacuum afforded a white solid
(U.41
g, 53% yield). Treatment of the amide with an ethanolic solution of fumaric
acid
afforded the salt of the titled compound as a white solid.
m.p. 107-108°C
Elemental Analysis for: C==H_°N30~ U.SC,H,O, I.UH=O
3U Calculated: C, 64.99; H, 7.SU; N, 9.47
Found: C, 64.80; H, 7.11; N, 9.22
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
Compound 8
Cvclohexanecarboxylic acid-~2-f4-(2-methoxy_phenyl)-piperazin-1- Iy 1-ethyl)-
(3
phenyl-propel)-amide
A solution of the amide (0.36 g, 0.979 mmol) from example 7 in THF (5 mL)
was treated with the dropwise addition of 1 M BH,-THF (4 mL) and the resulting
mixture ret7uxed under a nitrogen atmosphere for 3 hours. The solution was
cooled
to 0°C, and the excess borane reagent destroyed by the careful addition
of 2N-HCI.
After stirring for 2 hours, the solution was made basic with 2N-NaOH and the
product extracted with EtOAc (3 x 25 mL). The combined organics were washed
with water (2 x 20 mL), brine (25 mL) and dried over anhydrous sodium sulfate.
Filtration and concentration in vacuum afforded the required amine as a light
yellow
oil (0.18 g , 52% yield).
IS Cyclohexanecarbonyl chloride (0.()75 g, ().S() mmol) was added to a CH~CI,
solution (7 mL) of the amine (().18 g , (>.5(> mmol) and triethylamine (().15
mL).
After stirring for 16 hours, the solvent was evaporated, water (25 mL) added
and the
product extracted into EtOAc (3 x 20 mL). the combined organics were washed
with
water (20 mL), brine (20 mL) and dried over sodium sulfate. Filtration and
concentration gave a viscous yellow oil (0.29g) which was purified by dash
silica gel
chromatography to afford the required product ((). I Sg, 659o yield). An
ethanolic
solution of the amide was treated with an ethanolic fumaric acid solution
(().S eq) to
gave the fumarate salt of the titled compound as a pale yellow colored solid.
Elemental Anal, sis for: C=~H"N30= 1.0C,H,0,
Calculated: C, 69.01; H, 7.67; N, 7. I 0
Found: C, 69.17; H, 7.86; N, 7.39
m.p. 103-105°C
Compounds of the present invention bind with very high affinity to the 5-
3() HT1A receptor and consequently, they are useful for the treatment of
primary
disorders of the central nervous system such as depression, anxiety and panic,
as well
as secondary attending problems such as sleep disorders and sexual
dysfunction.
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
Compounds of the present invention are also useful for other disorders of the
central
nervous system including alcohol and drug addiction, obesity and migraine.
Cognition enhancement may be achieved by use of compounds of the present
invention and neurodegenerative diseases such as Alzheimer's disease and
Parkinson's disease may be treated.
5-HT1A Receptor Binding Assay
High affinity for the serotonin 5-HTtA receptor was established by testing a
compound's ability to displace [~H] 8-OH-DPAT binding in CHO cells stably
transfected with the human SHT1A receptor. Stably transfected CHO cells are
grown
in DMEM containing 10% heat inactivated FBS and non-essential amino acids.
Cells
are scraped off the plate, transferred to centrifuge tubes, and washed twice
by
centrifugation (200() rpm for 1() min.. 4"C) in buffer (50 mM Tris pH 7.5).
The
resulting pellets are aliquoted and placed at -80"C. On the day of assay, the
cells are
IS thawed on ice and resuspended in buffer. The binding assay is performed in
a 96
well microtiter plate in a total volume of 25() ~L. Non-specific binding is
determined
in the presence of 10 mM S-HT, final ligand concentration is 1.5 nM. Following
a 30
minute incubation at room temperature, the reaction is terminated by the
addition of
ice cold buffer and rapid filtration through a GF/B filter presoaked for 3()
minutes in
0.5% PEI. Compounds are initially tested in a single point assay to determine
percent inhibition at I, 0.1, and 0.01 mM, and Ki values are determined for
the active
compounds.
5-HT1A Receptor Intrinsic Activity Assay
The intrinsic activity of compounds of the present invention was established
by testing the claimed compounds ability to reverse the stimulation of cyclic
adenosinemonophosphate (CAMP) in CHO cells stably transfected with the human
5-HT 1 A receptor.
Stably transfected CHO cells were grown in DMEM containing 10% heat
3() inactivated FBS and non-essential amino acids. The cells are plated at a
density of
x106 cells per well in a 24 well plate and incubated for 2 days in a CO,
incubator. On
the second day, the media is replaced with 0.5 mL treatment buffer (DMEM + 25
CA 02351860 2001-05-18
WO 00/35892 PGT/US99/29953
- 12-
mM HEPES, 5 mM theophylline, 1() ~M pargyline) and incubated for 1() minutes
at
37°C. Wells are treated with forskolin ( 1 wM final concentration)
followed
immediately by the test compound (0.1 and I t1M for initial screen) and
incubated for
an additional 10 minutes at 37°C. The reaction is terminated by removal
of the media
and addition of U.5 mL ice cold assay buffer (supplied in the RIA kit). Plates
are
stored at -2()°C prior to assessment of cAMP formation by RIA.
ECS° values are
determined for the active test compounds. Compounds shown to have no agonist
activities (Emax = 0 9~) are further analyzed for their ability to reverse
agonist
induced activity. In separate experiments, 6 concentrations of antagonist are
preincubated for 20 minutes prior to the addition of agonist and forskolin.
Cells are
harvested as described above. The cAMP kit is supplied by Amersham and the RIA
is performed as per kit instructions, and calculations of ICS" performed by
GraphPad
Prism.
Compound 5-HTlA binding CAMP
Ki (nM) Emax
Compound 2 1.8 36 % (EC;° = 4.2 nM)
Compound 4 26 88
Compound 6 1.2 (1% (IC;o = 24 nM)
2() Compound 8 20 77
Hence, compounds of the present invention exhibit high affinity for the
SHT1A receptor subtype and exhibit intrinsic activity as evidenced by their
ability to
reverse stimulation of cyclic adenosinemonophosphate (cAMP). Accordingly,
compounds of the present invention are useful for treatment of disorders of
the
central nervous system and may be administered to a patient suffering from one
or
more of said disorders. Treatment, as used herein, refers to alleviation or
amelioration
of symptoms of a particular disorder in a patient. In addition, compounds of
the
present invention may be administered as part of a treatment regime that
includes
other agents which act on the central nervous system. In some preferred
embodiments, compounds of the present invention are part of a combination
therapy
including a serotonin reuptake inhibitor. Serotonin reuptake inhibitors useful
in
CA 02351860 2001-05-18
WO 00/35892 PCT/US99/29953
- 13-
combination therapies of the present invention lluoxetine, lluvoxamine,
paroxetine,
sertraline and venlafaxine. Said agents may be administered at the same time,
where
they may be combined into a single dosage form, or at a different time, as
compounds
of the present invention, while still being part of the regime of the
combination
therapy.
Compounds of the invention may be administered to a patient either neat or
with a convention pharmaceutical carrier.
Applicable solid carriers can include one or more substances which may also
act as flavoring agents, lubricants, solubilizers, suspending agents, fillers,
glidants,
compression aids, binders or tablet-disintegrating agents or an encapsulating
material.
In powders, the carrier is a finely divided solid which is in admixture with
the finely
divided active ingredient. In tablets, the active ingredient is mixed with a
carrier
having the necessary compression properties in suitable proportions and
compacted in
the shape and size desired. The powders and tablets preferably contain up to
9971 oi'
the active ingredient. Suitable solid carriers include, for example, calcium
phosphate,
magnesium stearate, talc, sugars, lactose. dextrin, starch, gelatin,
cellulose, methyl
cellulose, sodium carboxymethyl cellulose, polyvinylpyrrolidine, low melting
waxes
and ion exchange resins.
Liquid carriers may be used in preparing solutions, suspensions, emulsions,
syrups and elixirs. The active ingredient of this invention can be dissolved
or
suspended in a pharmaceutically acceptable liquid carrier such as water, an
organic
solvent, a mixture of both or pharmaceutically acceptable oils or fat. The
liquid
carrier can contain other suitable pharmaceutical additives such as
solubilizers,
emulsifiers, buffers, preservatives, sweeteners, tlavoring agents, suspending
agents,
thickening agents, colors, viscosity regulators, stabilizers or osmo-
regulators. .
Suitable examples of liquid carriers for oral and parenteral administration
include
water (particularly containing additives as above e.g. cellulose derivatives,
preferably
sodium carboxymethyl cellulose solution), alcohols (including monohydric
alcohols
and polyhydric alcohols e.g. glycols) and their derivatives, and oils (e.g.
fractionated
coconut oil and arachis oil). For parenteral administration the carrier can
also be an
oily ester such as ethyl oleate and isopropyl myristate. Sterile liquid
carriers are used
in sterile liquid form compositions for parenteral administration.
CA 02351860 2001-05-18
WO 00/35892 PCT/US99l29953
- 14-
Liquid pharmaceutical compositions which are sterile solutions or suspensions
can be utilized by, for example, intramuscular, intraperitoneal or
subcutaneous
injection. Sterile solutions can also be administered intravenously. Oral
administration may be either liquid or solid composition form.
Preferably the pharmaceutical composition is in unit dosage form, e.g. as
tablets or capsules. In such form, the composition is sub-divided in unit dose
containing appropriate quantities of the active ingredient; the unit dosage
forms can
be packaged compositions, for example packeted powders, vials, ampoules,
prefilled
syringes or sachets containing liquids. The unit dosage form can be, for
example, a
capsule or tablet itself, or it can be the appropriate number of any such
compositions
in package form.
The therapeutically effective dosage to be used in the treatment of a specific
psychosis must be subjectively determined by the attending physician. The
variables
involved include the specific psychosis or state of anxiety and the size, age
and
1 S response pattern of the patient. The novel method of the invention for
treating
conditions related to or are affected by the 5-HT1A receptor comprise
administering
to warm-blooded animals, including humans, an effective amount of at least one
compound of Formula 1 and its non-toxic, pharmaceutically acceptable addition
salts.
The compounds may be administered orally, rectally, parenterally or topically
to the
2U skin and mucosa. The usual daily dose is depending on the specific
compound,
method of treatment and condition treated. The usual daily dose is U.U1 -
1()UU
mg/Kg for oral application, preferably U.5 - 5()U mg/Kg, and U.l - t(H> mg/Kg
for
parenteral application, preferably U.S - 50 mg/Kg.