Note: Descriptions are shown in the official language in which they were submitted.
CA 02397566 2002-07-15
WO 01/51589 PCT/USO1/01068
_1_
METHOD AND SYSTEM FOR CONTROLLING
A FLUID CATALYTIC CRACKER
BACKGROUND
This invention relates to the field of petroleum refining and in particular to
methods
and systems for controlling petroleum distillates produced in a distillation
tower.
Crude oil, also known as petroleum, is a complex mixture of hydrocarbons. The
component hydrocarbons are separated from one another to produce commercially
valuable
petroleum products. The various physical and chemical processing steps for
separating
crude oil into component hydrocarbons are collectively referred to as
"refining."
Because the component hydrocarbons differ in their volatility, an effective
technique for separating them from one another is fractional distillation. In
this technique,
heated petroleum is placed in a distillation tower having a temperature
gradient that
progressively decreases from a high at the base of the tower to a low at the
top of the tower.
Petroleum vapor, which consists of the component hydrocarbons in their vapor
phase, rises
through the distillation tower. As it rises, the petroleum vapor encounters
progressively
lower temperatures. When the petroleum vapor reaches a height at which the
temperature in
the distillation tower is equal to the condensation temperature of one of the
vapor's
components, that component condenses. A tray placed at that level of the tower
collects that
condensed hydrocarbon component.
The relative amounts of each petroleum fraction generated in the distillation
tower
depend, to a great extent, on the composition of the petroleum. A refiner's
control of the
composition of any particular variety of petroleum to be processed is limited.
Refiners
must often process equity crudes or those available on an attractive
commercial basis from
the short term or spot markets. Therefore, there is limited ability to
regulate the yields of
different petroleum fractions generated by distillation. In addition, demands
for light, clean
petroleum products normally exceed the quantity of these products appearing
naturally in
crude oils which are recoverable by distillation only.
At the dawn of the petroleum refining industry, the scarcity of electric
lighting made
clean-burning lamp oils such as kerosene the petroleum fraction in highest
demand.
However, with the advent of the internal combustion engine, the demand for
lighter
petroleum fractions, such as gasoline, increased significantly at the expense
of heavier
petroleum fractions, such as kerosene. In response, the petroleum refining
industry has
CA 02397566 2002-07-15
WO 01/51589 PCT/US01/01068
-2-
developed methods for increasing the gasoline yield obtained from a particular
variey of
petroleum. Catalytic cracking is one such method.
In catalytic cracking, a petroleum fraction heavier than gasoline, commonly
called
gasoil, is fed into a reaction chamber and vaporized into a hydrocarbon vapor
at an
extremely high temperature. Ceramic pellets containing catalytic agents are
introduced into
the hydrocarbon vapor. A high velocity vapor stream propels the ceramic
catalyst pellets
into the reaction chamber and disperses them throughout the hydrocarbon vapor.
The
catalyst carned by the pellets facilitates a chemical reaction in which the
hydrocarbon
molecules split, or "crack," into lighter molecules. These lighter molecules
pass to a
distillation tower called the main fractionator for recovery. Because the
vapor now contains
lighter hydrocarbon molecules, distillation of the vapor yields additional
gasoline and other
light hydrocarbons.
The extent to which hydrocarbon vapor is cracked into lighter molecules
depends, in
part, on the hydrocarbon composition supplied to the reaction chamber and on
the rate at
which it is supplied.
In conventional methods for controlling the operation of a catalytic cracker,
measurements of the gasoil composition and products from the main fractionator
are
obtained by periodically sampling the feed and product streams, and testing
the samples in
a laboratory. This, however, is costly and time-consuming. As a result, it is
difficult to
perform laboratory testing frequently enough to attain up-to-date values of
selected quality
properties of the feedstock and all the distillates which the cracked
hydrocarbon mixture
yields. Consequently, this known method results in poor control over the
catalytic cracking
process.
SUMMARY
The method of the invention overcomes deficiencies in the art by performing on-
line
measurements of the values of selected properties of one or more hydrocarbon
materials
involved in the catalytic process. In one instance, these are materials fed to
the catalyic
cracker, i.e. the input feedstock. In another instance, these materials
involved in the
catalytic process are at least one output fraction from the main fractionator
tower fed by a
fluid catalytic cracker. The results of these on-line measurements, together
with the desired
values of the selected properties of the materials being measured, are
provided to an
CA 02397566 2002-07-15
WO 01/51589 PCT/USO1/01068
-3-
automated controller. On the basis of these measurements, the controller
calculates values
of manipulated process variables to apply to the fluid catalytic cracker to
obtain output
fractions having the desired values of the selected properties. Because the
estimates are
performed on-line, a system incorporating the invention responds rapidly to
variations in
the measured materials.
In most practices of the invention, it is deemed preferable to secure both
gasoil feedstock measurements and output product measurements, and to
manipulate
process variables in response to both groups of measurements. However,
features of the
invention may extend to practices where only the gasoil feedstock is measured,
or where
only one or more output fractions are measured, and one or more manipulated
process
variables are adjusted in response to such measurement. The process variables
are adjusted
for feed-forward control in response to measurements of gasoil feedstock
material, and are
adjusted for feed-back control in response to measurements of product
material.
A preferred practice of the invention employs nuclear magnetic resonance
measurements in conjunction with values of selected material properties, e.g.
of the gasoil
feedstock or of one or more product fractions. NMR measurements do not rely on
optical or
infrared radiation, and are therefore not affected by high opacity.
Additionally, NMR
measurements are relatively stable as a function of temperature. Consequently,
reliable
measurements can be made across the broad range of temperatures of the gasoil
feedstock
and of the various product fractions produced in the main fractionator.
One illustrative practice of the invention includes the steps of imposing a
steady
magnetic field on a material involved in the catalytic process, for example on
a sample of a
hydrocarbon output fraction. With the steady magnetic field in place, a
nuclear magnetic
resonance (NMR) sensor imposes a transient magnetic field on the fraction and
measures its
response to the transient magnetic field. An NMR sensor generally does not
directly
provide values of the selected properties of the fraction. Instead, the NMR
sensor provides
the chemical composition of the fraction. For this reason, the method of the
invention
includes the step of ascertaining values of the properties of the measured
material on the
basis of its measured composition. These values are then used to selectively
control
process variables such as the gasoil flow rate into the fluid catalytic
cracker, the regenerator
catalyst slide valve position, reactor dense bed level, regenerator pressure,
feed preheat
temperature, air or oxygen injection rate, reactor/regenerator differential
pressure, wet gas
CA 02397566 2002-07-15
WO 01/51589 PCT/USO1/01068
-4-
compressor suction pressure, main fractionator product draw rates, steam
injection rates,
overhead reflux duty, overhead accumulator release, and slurry to steam
generators, thereby
generating a product fraction having desired values of selected quality
properties. The exact
set of process variables available for manipulation will depend upon the
specific
configuration of the individual fluid catalytic cracking unit. Examples of
these selected
quality properties include aromaticity, boiling point, flash point, cloud
point, viscosity, pour
point, API gravity, freeze point, octane, PIONA, RVP, and other chemical or
physical
properties.
The information obtained from the NMR measurements can, in accord with the
invention, be used to explicitly control both the conversion in a fluidized
catalytic cracking
unit, and the selectivity of the unit to the desired levels. This practice of
the invention
involves NMR measurements on materials directly involved in the catalytic
conversion, and
are made on-line and essentially in real time.
The desired product qualities are generally dictated by competitive market
forces,
environmental regulations and economic factors including feed and product
prices and
operating costs. They may also be constrained by specific characteristics of
the refinery
including equipment configuration and availability and operating constraints.
The method of the invention can thus provide real-time measurements of the
values
of selected properties of gasoil feed and of a variety of fractions produced
by the main
fractionator. These real-time measurements enable the control system of the
invention
promptly to adjust the operation of the catalytic cracker to compensate for
fluctuation in the
feedstock and/or in the output of the main fractionator. Because the method of
the
invention relies on NMR rather than on optical techniques, the accuracy of
these
measurements can be essentially independent of the opacity or the temperature
of the
measured material.
A system for practice of the invention includes an optimizer for specifying
desired
long-term values of selected fluid catalytic cracker operating variables that
produce
products of the desired qualities while optimizing economic operation of the
unit
considering among other things feed and operating cost and product values. The
optimizer
also takes into account specific characteristics of the refinery including
equipment
configuration and availability and operating constraints including product
production limits.
CA 02397566 2002-07-15
WO 01/51589 PCT/US01/01068
-S-
A system for practice of the invention further includes a sensor for
estimating the
values of selected product properties. These estimates, together with the
desired long term
values of selected operating variables specified by the optimizer, are
provided to a
controller. On the basis of the values from the sensor, desired product
properties values and
the long-term desired values from the optimizer, the controller determines
current values of
the operating variables needed to form products having the desired values of
the selected
properties, while optimizing economic performance of the refinery, and in
particular of the
catalytic cracker.
Practice of the invention also includes measuring, with nuclear magnetic
resonance,
parameters regarding the composition of at least one material or substance
directly involved
in a catalytic converter in a petrochemical process, and applying the measured
information
to a controller of the petrochemical process. The measured material or
substance can be the
feedstock, or an ingredient of the feedstock, and can be one or more
petrochemical product
fractions. A preferred practice performs both feedstock and product
measurements. The
controller determines, in response to the measured information and to further
target
information it stores or receives regarding desired parameters of the
petrochemical process,
including of materials produced or otherwise involved, control signals for
adjusting the
process to attain selected values of the measured information relative to the
target
information.
DESCRIPTION OF THE FIGURES
These and other features and advantages of the invention will be apparent from
the
following detailed description and the accompanying figures, in which:
FIG. 1 is a schematic block drawing of a multivariate distillation control
system
embodying features of the invention;
FIG. 2 is a schematic block drawing showing details of the distillation system
of
FIG. l; and
FIG. 3 is a schematic block drawing of another distillation system according
to the
invention.
CA 02397566 2002-07-15
WO 01/51589 PCT/US01101068
-6-
DESCRIPTION OF ILLUSTRATED EMBODMENTS
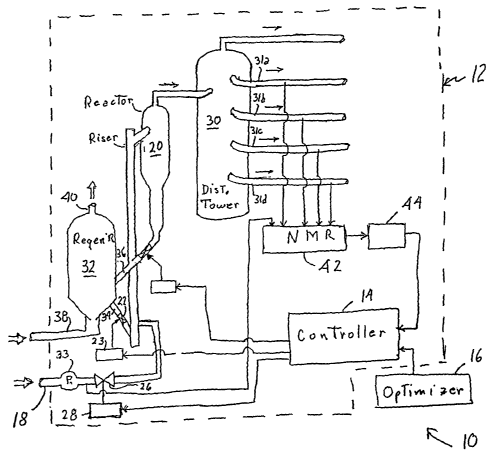
FIG. 1 shows a multivariate control system 10 incorporating principles of the
invention. The multivariate control system 10 includes a petrochemical
cracking system 12
that receives an input feedstock and that is in communication signal-wise with
a
multivariate controller 14. The output product of the cracking system 12
includes a plurality
of hydrocarbon products each characterized by values of selected properties.
These values,
which are represented in FIG. 1 by an output vector y that is fed back to the
multivariate
controller 14, are controlled variables for the multivariate control system
10.
The effectiveness of the illustrated controller 14 is enhanced by also
applying to it
an input vector z representing values of selected properties that characterize
the input gasoil
feedstock. The multivariate control system 10 includes an optimizer 16 for
generating
desired long-term values of selected operating variables that produce the
desired product
qualities, while optimizing economic operation . These desired values are
represented in
FIG. 1 by a setpoint vector r generated by the optimizer 16 and supplied to
the multivariate
controller 14. The elements of vector r are specified taking into account feed
and operating
cost, product economic values, and specific characteristics of the refinery
including
equipment configuration and availability and operating constraints including
product
production limits.
In response to the set-point vector r and one or more differences between
desired
product qualities and the output vector y andlor changes in feedstock vector
z, the
multivariate controller 14 generates a vector of manipulated variables x that,
when applied
to the cracking system 12, adjusts the cracking system operation to change the
values of the
elements in the output vector y to approach the corresponding desired values.
FIG. 2 shows that the illustrated cracking system 12 includes an input
pipeline 18
leading to a reaction chamber 20. A pump 33 coupled to the input pipeline 18
propels a
gasoil feedstock mixture into the reaction chamber 20 through a control valve
26 that is
opened and shut by a valve actuator 28 under the control of the multivariate
controller 14.
The reaction chamber 20 is also in fluid communication with a regeneration
chamber 32 by
means of a catalyst feed pipe 34 and a catalyst removal pipe 36. This
regeneration chamber
32 has an air or oxygen intake 38 and an exhaust 40 for use with the catalyst
regeneration
process. A control valve 22 in the catalyst feed pipe 34 is controlled by an
actuator 23 that
responds to input control signals from the controller 14. Hot regenerated
ceramic catalyst
CA 02397566 2002-07-15
WO 01/51589 PCT/USO1/01068
particles from the regeneration chamber 32 are added, by way of the catalyst
feed pipe 34,
to the gasoil feedstock in the riser prior to delivery to the reactor chamber
20. The heat
from the catalyst particles causes the gasoil feedstock to vaporize and the
resultant high
velocity vapor stream carnes the catalyst particles into the reactor chamber
20.
The reaction chamber 20 is in fluid communication with the main fractionator
tower
30 of the type commonly found in petroleum refineries. Such a distillation
tower 30
typically includes a plurality of tower outputs 31 located at different
heights along the tower
30. Each output corresponds to a particular fraction distilled from the
petroleum input.
Outputs located at the lower portions of the tower 30 correspond to heavier
fractions, such
as heating oil or kerosene. Outputs located at the higher portions of the
tower 30 correspond
to lighter fractions, such as gasoline or naphtha . The number of tower
outputs affects the
computational burden sustained by the multivariate controller 14, and is not
the subject
matter of the invention. Hence, for the sake of clarity and ease of
exposition. FIG. 2 shows
only four tower outputs 31 a-d.
A nuclear magnetic resonance (NMR) sensor 42 is coupled to various selected
stages of the cracking system 12 to measure chemical properties of material at
those
selected stages in the process. The illustrated sensor 42 of FIG. 2 is coupled
to the input
pipeline 18 to sample the feedstock, and is coupled to each tower output 31 a-
d to sample
each product, or fraction, produced by the main fractionator tower 30.
Although the
cracking system 12 shown in Figure 2 has a single NMR sensor 42 responding,
for
example, on a time-shared basis to each of multiple inputs, it will be
understood that one or
more additional NMR sensors can be provided to respond to the sampled
materials. The
sensed information output from the NMR sensor 42 is applied to a calibrator 44
that
transforms it to a format suitable for the multivariate controller 14.
One preferred sensor employs the technology of the I/A Series~ Process NMR
equipment available from The Foxboro Company of Foxboro, Massachusetts,
however, a
wide variety of NMR sensors may be used for chemical analysis.
A suitable calibrator is preferably a chemometic modeling unit. A chemometic
modeling unit for the calibrator 44 and suitable for practice of the invention
is implemented
by a digital processor executing instructions for estimating values of
selected physical
properties on the basis of the measured chemistry of a sample, as determined
by an NMR
sensor. These instructions implement procedures that are well-known to those
of ordinary
CA 02397566 2002-07-15
WO 01/51589 PCT/USO1/01068
_g_
skill in the art. Such procedures include establishing look-up tables,
interpolating between
values in a look-up table, and implementing mathematical models for estimating
values of
the selected properties. FIG. 3 shows the calibrator 44 as a separate element
for clarity,
however, the calibrator 44 may be integrated into software anywhere in the
system.
Both the optimizer 16 and the multivariate controller 14 are preferably
implemented
as software instructions executed on a programmable digital processor. In one
practice,
these instructions are executed on a general purpose digital computer.
However,
particularly to meet demanding performance requirements, the optimizer 16 and
the
multivariate controller 14 can be implemented in hardware, for example, with
application
specific integrated circuits, in software, or in a combination of hardware and
software. A
suitable optimizer and multivariate controller for practice of the invention
are sold by
Simulation Sciences of Brea, California, USA under the trade designations
ROMeoTM and
ConnoisseurTM, respectively. The specific implementation details of the
multivariate
controller 14 and the optimizer 16 are within the purview of one of ordinary
skill in the art
1 S and do not affect the scope of this invention.
In operation of the system shown in FIGS. 1 and 2, the pump 33 propels gasoil
feedstock through the control valve 26 and into the reaction chamber 20. At
the same time,
small hot pellets of catalyst material are added through the catalyst feed
pipe 34 and the
slide valve 22, from the regeneration chamber 32. The heat from the catalyst
particles
causes the gasoil feedstock to vaporize and the resultant high velocity vapor
stream propels
the catalyst particles into the reaction chamber 20. The catalyst is typically
a ceramic
material, such as zeolyte. However, other catalysts are known and used in
connection with
fluid catalytic cracking. The presence of the catalyst within the reaction
chamber 20
facilitates breaking the covalent bonds of the molecules in the gasoil mixture
to produce
lower molecular weight hydrocarbons.
Heat is produced by burning carbon off catalyst in the regenerator 32. This
heat is
transferred to the reactor via the catalyst feed pipe 34 to heat the gasoil
feed to sufficient
temperature to catalytically crack the gasoil to lighter molecules. This
results in the
generation of a vaporous mixture of numerous hydrocarbon species having
generally lower
molecular weights than the hydrocarbon species present in the feedstock. This
vaporous
mixture of low molecular weight hydrocarbons flows into the main fractionator
30 for
CA 02397566 2002-07-15
WO 01/51589 PCT/USO1/01068
-9-
separation into a plurality of products. These products exit the main
fractionator 30 through
a plurality of tower outputs, four of which are shown in FIG. 2.
A byproduct of the cracking process is the deposition of carbon on the
catalyst
pellets. These carbon deposits suppress the interaction between the catalyst
and the gasoil
vapor, thereby reducing the effectiveness of the catalyst. As a result,
catalyst pellets are
continuously drawn off the reaction chamber 20, blown through the catalyst
removal pipe
36, and returned to the regeneration chamber 32. Air or oxygen from the intake
38 is
injected to burn carbon off the catalyst pellets to renew catalytic activity
and to generate
heat to sustain the cracking process. The gaseous byproducts of this
regeneration process
are vented from the regeneration chamber through the exhaust 40.
As noted above, the illustrated NMR sensor samples the feedstock and samples
the
products from the main fractionator 30. The NMR sensor 42 imposes a steady
magnetic
field on each such sample to align the magnetic dipole moments associated with
the
molecules in the sample. With the steady magnetic field in place, the NMR
sensor 42
imposes a transient magnetic field having a direction different from, and
preferably
orthogonal to, that of the steady magnetic field. This transient magnetic
field temporarily
aligns the dipoles from the sample in a direction other than that in which the
static magnetic
field aligns dipoles. When the transient magnetic field is turned off, the
dipoles in the
sample spring back into the alignment imposed upon them by the steady magnetic
field. As
they do so, the dipoles generate a RF signal. The rate at which a particular
dipole springs
back to alignment with the steady magnetic field, and hence the frequency of
the resulting
RF signal, is characteristic of the sample's molecular structure. The
resultant RF spectrum is
responsive to, and hence provides a way of determining, the chemical
composition of the
sample.
The NMR sensor thus provides information on the chemical composition of
materials or substances directly involved in the process stream of the
cracking system 12.
As illustrated, these include the feedstock and the fractions formed in the
main fractionator
30. It is known in the art to predict the corresponding values of selected
properties from this
measured composition of the sample. This operation of converting the measured
sample
composition to values of selected properties is carried out, in the embodiment
of FIG. 2, by
a calibrator 44 in communication with both the multivariate controller 14 and
the NMR
sensor 42. The input information to the calibrator 44 includes the chemical
composition of
CA 02397566 2002-07-15
WO 01/51589 PCT/USOI/01068
-10-
the samples as measured by the NMR sensor 42. The output of the calibrator 44
is a
corresponding set of measured values of the selected properties for each
measured sample.
These selected properties can include aromaticity, boiling point, flash point,
cloud point,
viscosity, pour point, API gravity, freeze point, octane, PIONA, RVP, or other
chemical or
physical properties.
In response to the data provided by the NMR sensor 42, together with other
conventional data, as from other sensors (not shown) of level, pressure,
pressure
differential, temperature, time, and flow rate as conventional and known in
the art, the
multivariate controller 14 determines values of the manipulated variables
which minimize
the difference between the desired values of the product qualities and the
values of the
corresponding selected product qualities, as provided by the NMR sensor 42.
Desired long-
term values of the operating variables as determined by the optimizer 16 are
also taken into
account by the controller 14. The controller 14 determines the values of the
manipulated
variables using methods known in the art. These methods include the use of a
look-up table
and the implementation of empirically derived mathematical models. These
manipulated
variables include the flow rate of gasoil feed into the reaction chamber,
regenerator catalyst
slide valve position, reactor dense bed level, regenerator pressure, feed
preheat temperature,
air or oxygen injection rate, reactor/regenerator differential pressure, wet
gas compressor
suction pressure, main fractionator product draw rates, steam injection rates,
overhead
reflux duty, overhead accumulator release, and slurry to steam generation. The
exact set of
process variables available for manipulation will depend upon the specific
configuration of
the individual fluid catalytic cranking unit.
Having determined the values of the manipulated variables, the multivariate
controller 14 transmits control signals to the actuator 28 to marginally open
or close the
control valve 26 to adjust the flow rate of the gasoil feed into the reaction
chamber 20.
Similar signals are sent to control the other manipulated variables.
Because the NMR sensor 42 and the calibrator 44 rapidly provide information
concerning the process stream to the multivariate controller 14, the
controller 14 will
readily respond to fluctuations in properties of the feedstock and of the
distillates. This
enables the controller 14 to change the settings associated with the catalytic
cracking
process to reduce any difference between measured properties and corresponding
desired
properties.
CA 02397566 2002-07-15
WO 01/51589 PCT/USO1/01068
-11-
FIG. 3 shows an NMR stage 42' that can be used in the system of FIG. 2 in
place of
the NMR 42 of that drawing. The illustrated NMR stage 42' has a set of
multiple NMR
sensors 42a, 42a, each connected to receive information regarding the
different product
sample. The inputs to each NMR sensor 42a, 42a, can be a separate tower output
31a,
S 31b... of FIG. 2. The output signals from the NMR sensors are applied to an
interface
selector 42b that operates in conjunction with the multivariate controller 14
of FIG. 2. The
illustrated interface selector 42b applies the output from any selected one
NMR sensor 42a
to a calibrator 44, typically the calibrator 44 of FIG. 2.
As also shown in FIG. 3, the illustrated NMR stage 42' has a further NMR
sensor
42c, the output of which is applied to the input selector 42b. The illustrated
NMR sensor
42c receives information from either of two feedstock samples, by way of
sample lines 43a
and 43b and a controllable selector 42d. The two input lines 43a and 43b
illustrate for
purposes of example only, that the invention can be practiced both with
measured
information from multiple products and with information from multiple
feedstock samples.
The illustrated NMR stage 42' further illustrates that the invention can be
practiced with one
or more NMR sensors that receive information from a single sample or from any
of two or
more material samples, depending on the optimum instrumentation strategy for a
particular
petrochemical conversion facility.
The system configurations described above illustrate the chemical cracking
techniques described herein. However, a wide variety of other configurations
can also take
advantage of these techniques.
Having described the invention, and a preferred embodiment thereof, what is
claimed as new and secured by Letters Patent is: