Note: Descriptions are shown in the official language in which they were submitted.
CA 02402457 2002-09-06
WO 01/72353 PCT/GBO1/01371
NEEDLE ASSEMBLY AND SHEATH
& METHOD OF FILLING A DRUG DELIVERY DEVICE
Background
The present invention relates to drug delivery devices, and in particular to
ambulatory, drug delivery devices. Prefilled drug delivery devices are
desirable and
are-being adopted due to the significant advantages they provide over standard
drug
delivery devices, such as standard syringes that require filling by a health
care
worker immediately before administration. Benefits of such devices include
user
convenience, and decreased cost. Often, the user may administer the drug via
the
device. This can be done either within a physician's office, hospital, or at
home.
This results in an increased level of convenience for the user receiving the
dosage
and significant decrease in health care cost as a health care worker is not
necessarily
needed to administer the drug via the device. To prevent accidental needle
sticks
and contamination, the delivery needle associated with such a device typically
may
include a protective sheath that is removed immediately before use, or may be
designed so that the needle is retracted into a protective casing before use.
Prefilled drug or medication delivery devices must be filled with liquid drug
before they can be used. In most designs, the needle sheath shrouds and seals
the
needle-occupying compartment to prevent contamination. Sealing the needle
compartment from ambient also traps air bubbles within its drug-holding
compartment or drug reservoir. During priming or filling, air bubbles must be
allowed to escape. Otherwise, back pressure will cause incomplete filling of
the
drug reservoir in the device or will prevent filling altogether. Therefore, to
obtain
full and accurate filling in present drug delivery device designs, the needle
sheath
must be detached during priming or filling, and then reattached after the
priming is
completed.
During priming, when the sheath is removed, or otherwise not attached, the
needle can be exposed. Moreover, for accurate dosage, drug is typically filled
until
a small amount of drug is expelled through the needle. This ensures that 1)
the drug
CA 02402457 2002-09-06
WO 01/72353 PCT/GBO1/01371
reservoir is accurately filled and 2) that no air bubbles remain in the drug
reservoir.
The drug leakage, however, can be problematic as it can undesirably spill over
to
various components of the device, particularly if the needle end is exposed
inside
the device, e.g., retracted inside the device housing. In drug delivery
devices that
are designed to adhere to the skin surface during use, the leakage of drug
during
priming or filling is detrimental to the function of the device because the
liquid drug
may compromise the integrity of the adhesive.
Accordingly, there is a need to prevent the drug from spilling out of the
needle.
Moreover, there is a need for enabling priming or filling such a device while
the sheath remains attached to the device to enable air bubbles to escape
through the
sheath, but to prevent the drug from spilling out.
Moreover, there is a need for providing an improved needle sheath that
enables a drug delivery device to be filled without exposing the needle to the
external environment resulting in contamination of the needle and risk the
safety
and health of the user or health care worker.
A preferred aim is to provide an improved needle cover and sheath
that is capable of allowing air to pass therethrough but prevents the escape
of any
drug, thus enabling the drug to remain in the device and be delivered
accurately and
completely to the user.
There is still a further need to provide.an improved needle cover and sheath
that captures any liquid drug flowing out of the device during filling or
priming in a
manner that will not compromise the integrity of the adhesive layer of the
device.
The present invention provides new and useful needle assemblies, sheaths for
these, drug delivery systems including them and methods of use thereof.
Preferably
any one ox a combination of the above aims is met.
The present invention is drawn to a needle assembly, a sheath thereof, and a
method of filling or priming drug delivery device. The sheath is attachable to
the
needle assembly and allows fluid to flow out of the needle and capture any
liquid
expelled from the needle. Specifically, in one aspect the sheath comprises a
liquid
barrier that allows gas to escape but blocks passage of liquid therethrough.
2
CA 02402457 2002-09-06
WO 01/72353 PCT/GBO1/01371
Thus, according to one aspect of the invention, the needle assembly
comprises a needle holder and a sheath. A further aspect of the present
invention
comprises a needle sheath for receiving at least a portion of a length of
needle. The
internal cavity of the sheath is shaped to funnel and hold a volume of liquid
drug
that flows out of the needle tip during priming or filling. Another aspect of
the
invention is the sheath with a liquid barrier, which can comprise a
hydrophobic
membrane or the like.
The needle holder is adapted to hold, or holds, a needle. The sheath has a
needle compartment that can accommodate the needle, and has a first opening
communicating with the needle compartment. The sheath is adapted to shroud at
least a portion of the needle. The liquid barrier is positioned in the first
opening and
blocks the needle compartment so that the liquid barrier allows gas to escape
through the first opening but prevent liquid from leaking through the first
opening.
According to another aspect of the invention, the sheath can have a second
1 S opening, which communicates with the needle compartment, through which a
portion of the needle holder is insertable into the sheath. The needle holder
can
have a projection dimensioned and configured to be inserted and fractionally
seated
into the second opening. The sheath can have a recess coaxially arranged with
the
second opening. The recess can seat the projection.
According to another aspect of the invention, the sheath can have a wall that
closes one end of the compartment. The needle is inserted into the compartment
by
piercing through that wall.
According to another aspect of the invention, the sheath can have a central
sheath portion housing the needle compartment and a flange portion extending
laterally outwardly from the central sheath portion. The central portion is
adapted
to be inse~-tPd into a needle passage formed in a base of a drug delivery
device while
the flange portion abuts against a liner abutting the base so-that the sheath
is
removable by removing the liner, which protects an adhesive layer.
According to another aspect of the invention, a support with an aperture is
provided to support the hydrophobic member substantially concentrically
positioned
over the aperture. At least the support can be fractionally engaged to a
surface of
3
CA 02402457 2002-09-06
WO 01/72353 PCT/GBO1/01371
the sheath surrounding the first opening. Alternatively, the support can be
seated in
the first opening, holding the hydrophobic member in the first opening.
According to another aspect of the invention, the sheath for a needle has a
central sheath portion adapted to be insertable through an opening of a drug
delivery
device and engage a portion of the needle, and a flange portion having a
dimension
larger than the central sheath portion and adapted to abut against an adhesive
protector of the drug delivery device. Removing the adhesive protector can
automatically remove the sheath from the needle in this embodiment. The
central
sheath portion and the flange portion can be monolithically formed of solid
material, such as rubber. This drug delivery device is a separate aspect.
Another aspect of the invention comprises a method of filling or priming a
drug delivery device having a delivery needle, a drug reservoir, and a filling
port.
The method can comprise providing the afore-described sheath and engaging the
sheath over the needle so that liquid expelled from the needle is confined
within the
1 S needle compartment, flowing drug through the filling port to fill the drug
reservoir
or priming the drug reservoir until the drug flows out through the needle. The
liquid barrier allows air bubbles to escape into ambient but traps the spilled
drug
within the needle compartment.
Brief Descri,~,ption of the Drawings
These and other features, aspects, and.advantages of the present invention
will become more apparent from the following description, appended claims, and
accompanying exemplary embodiments shown in the drawings, which are briefly
described below.
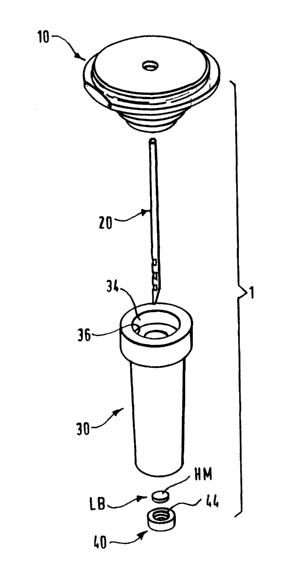
Fig. 1 is an exploded perspective view of a needle assembly having a sheath
accordin$,to the present invention.
Fig. 2 is a cross-sectional view of the needle assembly of Fig. 1.
Fig. 3 is another embodiment of the sheath according to the present
invention.
Fig. 4 is another embodiment of the sheath according to another aspect of the
present invention.
4
CA 02402457 2002-09-06
WO 01/72353 PCT/GBO1/01371
Detailed Description
Three exemplary embodiments, as shown in Figs. 1-4, serve to illustrate the
present invention. In this regard, same or.corresponding elements are labeled
with
S the same or similar reference numerals. Moreover, although references are
made
below to directions in describing the structure, they are made relative to the
drawings (as normally viewed) for convenience. The directions, such as left,
right,
upper, lower, etc., are not intended to be taken literally or limit the
present invention
in any form.
The needle assembly 1 comprises a needle holder 10, 10' for holding a
needle 20, and a needle sheath 30, 30'. The needle holder 10, 10' can be, for
instance, a conventional luer lock that can be detachably mounted to a syringe
or
injection device, or a hub or hub-like body that can be adapted for mounting
to a
drug delivery device, such as the type disclosed in U.S. Patent No. 5,858,001.
The
needle holder 10, 10' contemplated according to the present invention thus can
be
any suitable structure, whether it be adapted for a syringe or custom design,
that can
hold a needle 20 and connect to a drug delivery mechanism.
In the illustrated first and second embodiments, the needle holder' 10, 10'
has
a central axial through hole 12 for passage of the needle 20. Although these
embodiments show the needle 20 extending completely through the needle holder
10, 10', the needle 20 need not extend completely through. The needle holder
10,
10' can be configured to work with a drug delivery mechanism 100, such as a
syringe, to deliver the drug from the upper side 10U of the needle holder 10,
10', as
shown in Fig. 2, such as through a port or channel formed in the needle holder
10,
10'.
In the first embodiment, Figs. 1-2, the needle holder 10 has a stepped portion
that cooperates with the opening of the sheath 30 to sealingly, but detachably
hold
the sheath 30. Specifically, the stepped portion is formed, for example, by a
smaller
dimensioned annular projection 14 extending axially downwardly. The projection
14 is dimensioned and configured to be inserted and fractionally seated into
an
5
CA 02402457 2002-09-06
WO 01/72353 PCT/GBO1/01371
opening of the sheath 30 or a twist type mounting mechanism, e.g., bayonet
mount,
threaded connection.
In the first and second embodiments, the sheath 30, 30' has a hollow and
elongated portion, and has a needle compartment 32 for receiving the needle
20.
The needle compartment is configured to funnel and hold any liquid expelled
from
the needle 20. In the first embodiment, the sheath 30 has an opening 34 with a
recess 36 at one end. The opening 34 communicates with the needle compartment
32.~ The recess 36 is dimensioned and configured to sealingly receive the
projection
14.
The second embodiment, Fig. 3, illustrates a needle assembly that is
particularly suitable for a drug delivery of the type disclosed in U.S. Patent
No.
5,858,001, the disclosure of which is incorporated herein by reference. In
this
embodiment, the needle holder 10' is substantially similar to that of the
first
embodiment, except that the sheath 30' has a different configuration. In the
second
embodiment, the needle holder 10' does not matingly and sealingly engage the
sheath 30', as occurs in the first embodiment. Instead, the needle 20 itself
punctures
through a closed upper end or upper wall 30U of the sheath 30'.
The sheath 30, 30' can be formed of any suitable material, but preferably is
made of rubber, such as a bromobutyl rubber, or any other suitable commercial
material.
According to one aspect of the present invention, the sheath 30, 30' has a
liquid barrier LB that lets gas, e.g., air bubbles, escape through the sheath
30, 30',
while blocking passage of drug therethrough. This is particularly advantageous
in a
drug delivery device, where the drug is filled through a separate drug filling
port. In
particular, during filling or priming, the drug can be accurately filled by
allowing
any trapped air bubbles to escape through the needle and through the sheath
30, 30'
but confine any liquid expelled from the needle within theneedle compartment
32.
As gas, e.g., air bubbles, can escape during the filling stage, no
backpressure is
developed to enable an accurate dosage.
According to the present invention, the liquid barrier LB comprises a
hydrophobic disc or membrane HM positioned inside the sheath 30, 30'. The
6
CA 02402457 2002-09-06
WO 01/72353 PCT/GBO1/01371
hydrophobic membrane HM is a material, such as a polymer with fine pores,
which
allows passage of air or gas thereacross or therethrough but repel liquid.
Examples
of suitable hydrophobic membrane HM are SUPOR, available from PALL
SPECIALTY MATERIAL, IV FILTER available from WHATMAN, and
IMMOBILON from MILLIPORE.
To accommodate the hydrophobic membrane HM, the sheath 30, 30' can
have an opening or aperture 38 that communicates with ambient and the needle
compartment 32. The hydrophobic membrane HM sealingly covers or blocks the
opening 38. For instance, the opening 38 can be formed at a lower end pf the
sheath, below the needle 20, as illustrated in Figs. 1-3. The hydrophobic
membrane
HM should be positioned away from the needle point so that it does not become.
punctured. If the sheath is made of rubber or relatively flexible or soft
material, it is
preferable to support the hydrophobic membrane HM with a relatively rigid
support
40, 40', which can be formed of a plastic, for example. Alternatively, the
sheath
could have a closed bottom with an opening. The sheath can be dimensioned to
sealingly seat the hydrophobic membrane HM over the opening.
The first and second embodiments both have the support 40, 40', configured
substantially cylindrically, but can have any suitable configuration. The
support 40,
40' has a central aperture 42, which provides a pathway for gas to escape
through
the hydrophobic membrane HM. In the first embodiment, the support 40 has a
recess 44 for sealingly seating the hydrophobic membrane HM. In the first
embodiment, the inner surface 32S of the sheath surrounding the opening 38, or
the
needle compartment 32, frictionally and sealingly engages the outer periphery
of the
support 40 to immobilize the support. That is, the support 40 with the
hydrophobic
membrane HM is inserted or pushed up into the opening 38 or the needle
compartment, and frictionally secured thereto.
In the second embodiment; the opening 38 is collinear with the needle
compartment, but has a larger dimension or diameter (in the case of a
cylindrical
support). The hydrophobic membrane HM is positioned to block the lower end of
the needle compartment 32, and is sealingly sandwiched between an upper side
40U
of the support 40' and a bottom surface 30B of the opening 38. The lower
portion
7
CA 02402457 2002-09-06
WO 01/72353 PCT/GBO1/01371
of the sheath 30' surrounding the opening 38 is dimensioned larger, forming a
flange portion 30F. The opening 38 can be formed in the flange portion 30F.
The
flange portion 30F also includes a lip 30L that extends radially inwardly at
the
entrance of the opening. The support 40'.is forced into the opening 38. The
lip 30L
helps to maintain the support 40' securely confined in the opening 38.
In the second embodiment, the sheath 30' is configured to protect an
adhesion layer 50 typically used for attaching a drug delivery device to a
drug
delivery site. That is, during priming or filling, overfilling the drug
reservoir, for
purposes of completely filling the sane removing any trapped air bubbles,
results in
spillage. The drug can undesirably wet the adhesion layer and make it less
effective
or completely ineffective. The sheath 30' funnels and holds excess liquid drug
that
flows out of the needle during pruning or filling. This design prevents
spillage and
protects the adhesion layer 50 from exposure to any liquid drug.
In the second embodiment, the liner 60 protecting the adhesion layer 50 can
be used to remove the sheath 30'. In this respect, the sheath 30' has a
central sheath
portion 30C that extends through a needle opening 70 formed through a base
portion B of the drug deliver device D, and shrouds the needle 20. The flange
portion 30F, which is dimensioned larger than the central sheath portion 30C,
abuts
against the underside of the base portion B. Pulling the liner 60 off the base
B
forces the sheath 30' to move outwardly off the base, removing the sheath 30'
from
the drug delivery device D.
The third embodiment, Fig. 4, is similar to the second embodiment, Fig. 3,
except that the sheath 30" is solid, with no needle accommodating compartment.
The sheath 30" has a central sheath portion 30C" that extends through the
needle
opening 70 formed through a base portion B of the drug deliver device D and
shrouds tl~e needle 20. The flange portion 30F", which is dimensioned larger
than
the central sheath portion 30C", abuts against the underside of the base
portion B.
Pulling the liner 60 off the base B forces the sheath 30" to move outwardly
off the
base, removing the sheath 30" from the drug delivery device D.
In the third embodiment, the needle pierces through the central sheath
portion 30" and blocks the needle, preventing the drug from spilling out of
the
8
CA 02402457 2002-09-06
WO 01/72353 PCT/GBO1/01371
needle. The sheath 30" also can be made of rubber, preferably a solid rubber
or the
like.
According to a method of filling or priming a drug delivery device, which
typically has a delivery needle, a drug reservoir, and a filling port,
according to the
present invention, the sheath is placed over the needle before filling take
place. If
the drug deliver device is provided with a prefilled cartridge, the device is
primed
while the sheath is placed over the needle. The drug reservoir is filled or
primed
until the drug spills out through the needle to remove air bubbles. The liquid
barrier
allows air bubbles to escape into ambient, but traps the spilled drug wit)~in
the
needle compartment.
Given the disclosure of the present invention, one versed in the art would
appreciate that there may be other embodiments and modifications within the
scope
and spirit of the present invention. Accordingly, all modifications attainable
by one
versed in the art from the present disclosure within the scope and spirit of
the
present invention are to be included as further embodiments of the present
invention. The scope of the present invention accordingly is to be defined as
set
forth in the appended claims.
9