Note: Descriptions are shown in the official language in which they were submitted.
CA 02462320 2004-03-29
STEEL TUBE HIGHLY RESISTANT TO THE CRACKING DUE TO
TENSIONS IN A MEDIUM CONTAINING HYDROGEN SULFIDE AND
METHOD TO PRODUCE SUCH TUBE
FIELD OF THE INVENTION
The present invention relates to a steel product, particularly a
steel product such as a seamless steel tube with high mechanical
resistance and an excellent resistance to cracking caused by
tension in media containing hydrogen sulfide. This steel product is
characterized by a combination of a specific chemical composition,
a steel production process and a heat treatment of the material.
BACKGROUND OF THE INVENTION =
The presence of hydrogen sulfide (H2S) in the fluids of the oil
wells imposes severe restrictions to the use of steel tubes due to
their degradation resulting from a phenomenon known as Sulfide
Stress Cracking (SSC). The growing development of deep wells
containing hydrogen sulfide demands the use of high mechanical
resistance tubes and an excellent resistance to SSC.
The SSC phenomenon has been studied for many years, cf. G.
CA 02462320 2004-03-29
. .
2
Presouyre, I. Berstein, Acta Met. 27, 1979, p89 y cfr. D.
SponseIler, R. Garber y J. Straatmann, ASTM STP 792 Micon 82,
1982, p172.
In carbon steels, the inclusion of hydrogen, as a result of the
reaction between hydrogen sulfide and iron, causes a noticeable
reduction of the fracture toughness of the material. This reduction
becomes even more pronounced when the mechanical resistance of
the steel is higher., cf. Corrosion in the Petrochemical Industry,
ASM, p264 and cf. Internal Report CINI 748/94, C. Pampillo, 1994.
Due to economic reasons, mainly all research and development
works in this field are focused on high mechanical resistance and
low alloy carbon steels, cf. E. Anelli, L. Cariboni, F. Leone, A. Mascanzoni,
3rd Int.
Conf. on Steel Rolling, Japan, 1985, p637. and dr. M. Watkins, R.
Ayer,Corrosion 95, Paper 50.
The National Association of Corrosion Engineers (NACE), has
standardized some of the most used tests for this kind of materials
under Standard NACE TM0177.
In this Standard 4 tests are presented, among which is the so-
called Method D, it is an arrest type fractomechanical test. A
double cantilever beam type sample (DCB) is used to evaluate the
resistance to cracking propagation due to SSC and to quantify it
through the critical stress intensity factor in the medium (Klssc).
CA 02462320 2011-05-18
3
There is also the so called Method A, which consists in the application of a
tension load by means of a metallic ring to a test sample of the material to
be tested, submerged in a medium consisting in an aqueous saturated
solution. It is then determined whether the test sample is fractured. This
means that this method can obtain two results: positive or negative.
It can be noticed that Method A has been commonly used to evaluate SSC
resistance, although this test does not allow to obtain any property of the
material that could be used as a design parameter.
Application of Method D has spread in the last years due to its quantitative
character and the possibility of applying the results obtained to the design.
To carry out the present invention, the SSC resistance of several low-alloy
carbon steels has been determined by means of the application of the
DCB test. Likewise the effect of chemical composition and heat treatments
on steel behaviour when faced to the SSC phenomenon has been studied.
The steps and metallurgical characteristics necessary to meet the
mechanical requirements of this type of steels resistant to
CA 02462320 2011-05-18
4
corrosion have been extensively analyzed in the literature, cf. Sour Grade
C110, Liane Smith, lntetech, June 1999, private report.
Both the experience in the field and the laboratory tests have demonstrated
that there is a hardness limit above which the SSC phenomenon appears.
This fact represents a great dilemma, due to the need to combine high
resistance with relatively low hardness values. This means that this kind of
steels, in a stage after the heat treatment, must have a high relationship
between yield and tensile stress. This indicates that the most adequate steels
for those requirements are quenched and tempered steels.
The previous results have been deeply analyzed through the job prepared by
CSM, Rome; cf "110SSG: Status dell'attivita'di sviluppo" CSM, Rome,
January 2000, private report.
Likewise, there is a document of a Mexican Patent Publication No.
MX9708775A, where Kondo et al disclose a process to produce a seamless
steel tube resistant to sulfide stress cracking by controlling the chemical
composition in a fairly wide range and applying a rolling process and a
specific heat treatment.
This document discloses the effect of chemical elements such as C, P, S, Mo,
Cr, Mn, 0, N, Al, Ti, Ni, Si, Va, Ca, W, B and Zr establishing content ranges
technologically unfeasible even in the
CA 02462320 2004-03-29
most restricted ranges.
When the carbon effect is mentioned, a range of 0.15% to 0.50% of
the total weight of the heat is mentioned.. As the shortest interval
5 a range of 0.20% to 0.35% is considered. It
has been
demonstrated that heats containing 0.35% carbon, quenched with
water, have shown cracks, even before being in contact with media
containing sulphydric acid.
The different metallurgical factors influencing the capacity of
steels to resist to SSC are well known. The mechanical resistance
level, the micro-structure, the hardness, the segregation and the
level content of non metallic inclusions are very important, cf
'Mejoramiento de la resistencia a la iniciaci6n y propagacion de
SSC de tubos pare OCTG de alta resistencia a traves del control
de la microestructura y las precipitaciones' G. Lopez Turconi et al.
NACE 2001.
However, in spite of all that information, the problem of
manufacturing seamless tubes highly resistant to SSC, has not
been fully solved and there is not enough information about the
levels of the parameters above indicated, as well as about the
qualitative and quantitative composition of said steels.
OBJECTIVE OF THE INVENTION
CA 02462320 2004-03-29
'
6
The main objective of this invention is to provide a chemical
composition of the steel used for manufacturing seamless tubes
and a manufacturing process, permitting to obtain a product with
high mechanical resistance.
Another objective of the invention is to meet the requirements of
the previous objective, in a product also presenting high resistance
to SSC.
Still another objective is to make possible a product with high
fracture toughness values.
Other objectives and advantages of the present invention will be
apparent from the analysis of the following description and the
illustrative, but no 'imitative, examples indicated in the present
description.
BRIEF DESCRIPTION OF THE INVENTION
Briefly, in one of its modalities, the present invention is related
with the qualitative and quantitative composition of steel.
Extensive studies were carried out about a manufacturing process
to obtain seamless steel tubes with high mechanical resistance and
high resistance to the SSC, which shown that compositions of the
CA 02462320 2004-03-29
. ,
7
state of the art could not solve the posed problem. By subjecting
the material to a two continuous quenching and tempering cycles or
double quenching followed by tempering, it is possible to obtain a
material with high Kissc values measured through the NACE
TM0177 Method D test.
Steel microstructure, the level of its residual stresses and its
mechanical resistance depend on its chemical composition and on
the manufacturing thermo-mechanical treatments; this leads to
consider microstructure, chemical composition and thermo-
mechanical treatments as factors depending on the material itself.
As a result of the research carried out, it has been possible to
design a new process to manufacture seamless steel tubes wherein .
such tubes are obtained from a low-alloy steel bar having the
following content of elements by weight percentage: C between
0.20 and 0.26; P a maximum of 0.02; S a maximum of 0.005; Mo
between 0.40 and 0.85; Cr between 0.90 and 1.10; Al between
0.010 and 0.050; Mn between 0.30 and 0.50 of Mn; maximum
hydrogen when casting of 2 ppm, Cu +8 Sn 5. 0.30% (The
percentage of the copper content plus eight times the tin content
must be less than or equal to 0.30%); Si + Mn 5 0.9% (The
percentage of silicon added to the percentage of manganese must
be below 0.9%).
CA 02462320 2004-03-29
8
Following is the description of the effect that each of the elements
added has on the properties of the material claimed in the present
invention.
Carbon effect
Carbon is needed to increase the hardenability of the material, and
in this way, increase the steel resistance. The content of C
(between 0.18 and 0.30%) does not modify the resistance to SSC.
This is valid in case the remaining variables are kept constant, in
particular the material's yield stress. The contents of C shall be
kept under 0.26 % to avoid cracks during water quench. And it
must be above 0.20% to obtain an acceptable martensitic
transformation.
Manganese effect
The marked increase of hardenability produced by Manganese
addition, added to its low cost, makes it a usually used alloy
element, with contents higher than 0.30%.
The increases in manganese content tend to raise the susceptibility
to SSC. Manganese reduces the cohesion of the limits of austenitic
grains and pushes P to segregate to the grain boundaries. That is
the reason why it is preferable addition of Mn to be below 0.50 %
by weight.
CA 02462320 2004-03-29
9
Silicon effect
The silicon, like manganese, promotes the segregation of P to
grain boundaries, whereby it results harmful and it must be kept at
the lowest possible level, preferably under 0.40 % by weight.
Chromium effect
Chromium produces hardening by solid solution and increases the
hardenability of the material, thus increasing the resistance to
SSC. This is the reason why a minimum content of 0.90% is
desired, but since an excess of hardenability also means cracking
problems during quenching in water, it is recommendable to
maintain a maximum value of 1.10% by weight.
As it is well know, quenching in other type of refrigerating medium
will mean complications in the equipment to recover released gases
or in the formation of inert atmospheres.
Molybdenum effect
The molybdenum, increasing the hardenability of steel, allows to
increase the tempering temperature, thus preventing the
segregation of brittleness generating elements to the austenitic
grain boundaries. Therefore, its addition improves the SSC
resistance. The percentage to be added ranges between 0.40 and
0.85, below this level its effect is not enough and moreover it
CA 02462320 2004-03-29
would unnecessarily increase the price of the process.
Phosphorus effect
Phosphorus exists as an unavoidable element in steel, and a
5 content above 0.02 % produces segregation in the grain
boundaries, which reduces the SSC resistance.
Sulfur effect
Like phosphorus, sulfur is inevitably found in steel, and a content
10 of more than 0.005% reduces the toughness and hardenability of
material.
Oxygen effect
Oxygen exists inevitably in steel, as well as sulfur and phosphorus
in a quantity above 20 ppm can cause a reduction in the toughness
of the material and also generate an inclusionary content
worsening the steel resistance to SSC.
Hydrogen effect
The content of hydrogen in the steel has to be kept below 2 ppm, in
order to avoid the formation of cracks at the time of steel's
solidification.
Nitrogen effect
The nitrogen exists necessarily in the steel as well as phosphorus,
CA 02462320 2004-03-29
11
sulfur and oxygen. It must be kept below 80 ppm to obtain a steel
containing precipitate that do not reduces material toughness and
do not generate ageing efects during the tempering process.
Studies on the state of the art allow to find out that the
segregation of P on grain boundary has a strong influence on SSC
resistance.
It is also known that the direct measurement of this segregation
presents difficulties, making necessary indirect measurement in the
chemical composition of the elements originating such segregation.
Therefore, as a parameter of this measurement the following
expression has been used:
A-value = ( Mn + 4.3 P + 17.0 (Mn * P) )
Where Mn and P correspond to the Manganese and Phosphorus
content respectively expressed in % by weight. It has been found,
and this makes part of the state of the art, that the lower the
parameter A-value, the better the performance of the material is to
SSC.
After multiple tests carried out in our laboratories we found out
that an optimum value is 0.6 or lower. A lower parameter level is
CA 02462320 2004-03-29
12
difficult since Mn is an element necessary in our steel and is
practically impossible to reduce the content of P to zero.
Meeting this requirement , it is found out that the Kissc value of
In another of its aspects, the invention consists in a steel
manufacturing process that is carried out according to a clean
These inclusion levels are guaranteed by controlling total oxygen,
limiting it to a maximum of 20 ppm.
The clean steel manufacturing process is mainly characterized by
The practice of secondary metallurgy, assuring the floatation of
inclusions and impurities in the ladle furnace or LF, by means of an
adequate inert gases bubbling process. The generation of fluid slag
CA 02462320 2004-03-29
13
cleaned steel, as well the modification of inclusions by means of
the addition of Si Ca.
As a step of the present process, a solid steed bar is obtained,
preferably of a circular section, by using a continuous casting
machine.
Another important aspect of the present invention is the heat
treatment to be applied to the steel to obtain the desired
properties.
It is considered that steels with a tempered martensite type
structure are the most resistant to SSC.
The heat treatment procedure of this invention is characterized by
the performance of a process that permits to widely improve the
properties of steel to resist SSC.
When a steel tube with the specified chemical composition is
subjected to the heat treatment described in this invention, a
structure is obtained that contains a fine dispersion of cementite
particles, which improves the work of the material in media
containing hydrogen sulfurs.
It is known that smaller prior austenitic grain size improves the
CA 02462320 2004-03-29
. ,
14
resistance to SSC. It is also known that this grain size is modified
when changing the temperature and / or time of the austenitisation
process, which imposes the performance of a strict control during
the process.
An adequate tempering process, preferably carried out by means of
simultaneous cooling in the inside and in the outside of the steel
tube, preferably with water, is necessary to obtain a high cooling
speed and to promote the martensite formation.
We have surprisingly discovered that there is a beneficial effect of
carrying out a multiple heat treatment of austenitisation and
quenching for the refinement of the prior austenitic grain. The
application of double quenching is effective for the grain
refinement, and particularly effective is the refinement obtained
thanks to the second quenching.
The tempering temperature has an important influence on the
steel's mechanical resistance, as well as on the final
microstructure and segregation.
Besides, high tempering temperatures will be convenient to achieve
an adequate recovery and recristalization, which improves the
resistance to SSC.
CA 02462320 2004-03-29
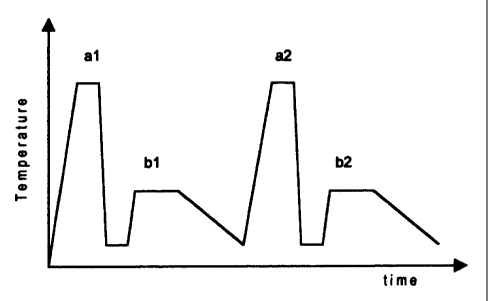
BRIEF DESCRIPTION OF THE DRAWINGS
Figure 1 corresponds to a Cartesian axis diagram, in which the axis
5 of the ordinates corresponds to the temperature and the axis of the
abscissa corresponds to the time, thus showing the cycle of heat
treatment of the seamless steel tube, with a heat treatment
consisting in a double quenching and tempering process.
10 Figure 2 corresponds to a Cartesian axis diagram, in which the axis
of the ordinates corresponds to the temperature and the axis of the
abscissa corresponds to the time, thus showing the cycle of heat
treatment of the seamless steel tube, consisting in a double
continuous quenching followed by the tempering process.
DETAILED DESCRIPTION OF THE INVENTION
In the aspect of the invention related to the heat treatment, said
treatment is made up of a double cycle of quenching and tempering
or continuous double quenching followed by the tempering and can
be described through figures 1 and 2.
Both heat treatments described in the present invention allow to
obtain a fine dispersion of the cementite particles and permit the
CA 02462320 2004-03-29
,
16
grain size refinement, which together with the achievement of a
minimum content of 95% of martensitic structure, can assure a
better performance of the material SSC.
Since tempering is an important process affecting the
characteristics of the final product, it is necessary to determine an
optimal tempering temperature according to the desired
characteristics of the final product. The steel tube must be kept at
a uniform temperature so determined. The differences in the
tempering temperatures can be in the utmost of 20 C, preferably
10 C.
The best way to carry out the invention is described in the
following steps:
Selecting a steel which adequate chemical composition above
described, in order to assure a structure with a minimum
martensite content of 95 %.
Keep the carbon content in the interval of 0.20-0.26 % by weight
since lower values generate the formation of softer martensites and
higher carbon contents can generate cracks during quenching in
water.
Reducing as much as possible content of impurities such as P and
CA 02462320 2004-03-29
17
S and residual elements such as 0, N and H (See "Brief description
of the invention")
Carry out a multiple heat treatment in order to homogenize and
refine the austenitic structure.
Precisely controlling the effective temperature of material in the
tempering phase, preferably within the interval of tempering
temperature 100 C, in order to constrain hardness variability.
EXAMPLES
In this section , application examples of the present invention,
related to the chemical composition aspect of the invention are
presented, organized in tables.
Table 1 determines the different compositions and their effects in
the A-value, table 2 establishes the effect of such composition with
the two heat treatments indicated, in the mechanical properties of
the product, and table 3 shows the effect of such compositions also
with the heat treatment indicated, on the resistance to SSC.
The effect of the chemical composition and the examples developed
are included in Table 1 and the heat treatments selected assure
the properties to resist SSC claimed in the present invention. This
can be observed in the examples mentioned in tables 1, 2 and 3.
CA 02462320 2004-03-29
. .
18
All cases referred by the same letter: "A", "B", etc. have the same
composition and the same heat treatment.
The invention has been sufficiently described such that any
person with an average level of knowledge about the subject can
reproduce it and obtain the results mentioned herein. However,
any knowledgeable person in the art of the present invention can
be capable of making modifications not described in the present
application, but if for the application of those modifications in a
given material or in the manufacturing process to obtain it, the
subjects claimed in the following claim are applied, such material
and the process shall be comprised within the scope of the present
invention.
20
19
Table 1. Chemical composition
O N N
Material C: Mn Si P S Mo Cr Cu Al
Sn Cu + 8Sn Si + Mn A-value
(PPm) (PM) (PPm)
_____________________________________________________________
A 0.23 0.42 0.26
0.009 0.001 0.54 1.07 0.14 0.018 0.009 17 79 1 0.21 0.68 0.52
B 0.26 0.38 0.25 0.011 0.001 0.49 1
0.09 0.02 0.008 12 75 1 0.15 0.63 0.50
C 0.21 0.48 0.25
0.007 0.002 0.48 1.02 0.15 0.019 0.008 19 71 1 0.21 0.73 0.57
D 0.25 0.37 0.25
0.008 0.002 0.75 0.96 0.08 0.017 0.004 18 68 1 0.11 0.62 0.45
E 0.25 0.41 0.25
0.005 0.002 0.76 0.97 0.085 0.027 0.006 17 59 1 0.13 0.66 0.47
L73
F 0.25 0.38 0.22
0.008 0.002 0.76 0.95 0.092 0.016 0.006 18 61 1 0.14 0.60 0.47
G 0.23 0.38 0.19
0.009 0.002 0.72 0.98 0.13 0.0160.009 17 72 1 0.20 0.57 0.48
H 0.24 0.36 0.22 0.009 0.001 0.73 1
0.17 0.023 0.012 18 70 1 0.27 0.58 0.45
l 0.23 0.49 0.23
0.008 0.003 0.77 1.02 0.1 0.02 0.007 16 72 1 0.16 0.72 0.59
CA 02462320 2004-03-29
Table 2. Mechanical properties
Yield Stress Tensile Stress
Material Heat treatment
MPa ksi MPa ksi
A TRTR 599 86.9
736 106.7
TTR 605 87.7 701
101.7
TRTR 647 93.8 758
109.9
TTR 728 105.7 810
117.7
TTR 736
106.9 819 118.9
TRTR 755 109.7 793
115.2
TRTR 70 111.7 862
124.9
TRTR 800
116.0 890 129.0
TTR 815 118.3 879
127.5
TRTR: Double cycle of quenching and tempering.
TTR: Double cycle of quenching followed by one tempering.
5
Table 3. Fracture toughness (Kissc measured by Method D)
Material Kissc (Mpa mv2)
A 48.1
44.0
43.4
36.6
35.2
36.4
CA 02462320 2004-03-29
21
29.9
30.2
31.1